Abstract
Denosumab is a human monoclonal antibody against RANKL. This antibody decreases bone turnover markers and increases bone mineral density (BMD) in postmenopausal women. In phase 3 studies including more than 1100 women, denosumab achieved greater increases in lumbar spine, total hip, distal 1/3 radius, and total BMD than alendronate 70 mg weekly. Recent data suggest that denosumab also decreases vertebral and non-vertebral fractures. This drug seems to be safe, although the most frequent side effects are arthralgia, back pain, and nasopharyngitis. No increased incidence of neoplasia has been found compared to placebo or alendronate. However, infections requiring inpatient treatment were more frequent in study groups treated with denosumab. These were common community acquired infections and were treated with standard antibiotics. No opportunistic infections were reported. Denosumab is a very promising new drug for the treatment of osteopenia and osteoporosis, and hopefully more long-term safety information and further fracture data will support its commercial use in the near future.
Keywords
Introduction
Osteoporosis is present in more than 200 million women worldwide. Osteoporosis related fractures are more common than heart attacks, stroke, or breast cancer, rendering this disease an important global public health problem. The clinical consequences of osteoporosis related fractures can be devastating and include pain, kyphosis, depression, restrictivelungdisease, constipation, prolonged immobility, and death. As the population ages, osteoporosis-related fractures will become more frequent with an estimation of more than 6.26 million hip fractures by 2050. 1 As the treatment and devices for fractures become more expensive, osteoporosis also becomes an economic problem. The annual cost of all osteoporotic fractures has been estimated at US $20 billion in the USA and US $30 billion in the European Union. 2 A conservative estimate of the worldwide annual cost for hip fractures is US $131.5 billion. 2
Although effective therapies have been developed for the prevention and treatment of osteoporosis, these therapies are expensive. There is also an increased awareness of the low adherence of patients to drug therapies for osteoporosis. 3 In a recent review of publications describing observational or retrospective analyses of adherence, compliance, and persistence with osteoporosis therapies, the authors estimated about a 47% compliance rate with osteoporosis therapies after one year. There was a significant increase in fracture rates as compliance and persistence worsened. 4 In addition, long term safety of certain treatments, such as bisphosphonates, is unknown. All of these issues call for new strategies in treatment.
Denosumab, a new fully human monoclonal antibody against the Receptor Activator of Nuclear Factor, kappa β ligand (RANKL), seems a promising treatment for osteoporosis, targeting a central pathway in the activation, survival and action of the osteoclast. This review focuses on the latest data on denosumab for postmenopausal osteoporosis, addressing the safety and present concerns with these therapies, as well as new trials forthcoming.
The RANK/OPG/RANKL Pathway
RANKL is expressed on the surface of the osteoblast and is a member of the tumor necrosis factor (TNF) family.5,6 It was discovered soon after osteoprotegerin (OPG), a TNF receptor (TNFR)-related protein, secreted by the osteoblast. 5 RANKL was then identified as a key regulator of osteoclastogenesis and bone resorption.5,7 Its effects are mediated through the binding and activation of a TNFR-family transmembrane protein called RANK on the surface of the osteoclast. This action promotes osteoclast formation, function and survival. 6 Hormones and factors stimulating bone resorption, such as 1,25 hydroxyvitamin D3, parathyroid hormone, and interleukin 1 induce the expression of RANKL. Osteoclast-mediated bone resorption is activated by RANKL expression, which in turn activates bone formation by the osteoblast, a process called coupling.6,8 OPG acts in this pathway as a regulator, inhibiting the binding of RANKL to RANK. A scheme of the RANKL/RANK/OPG pathway is presented in Figure 1.
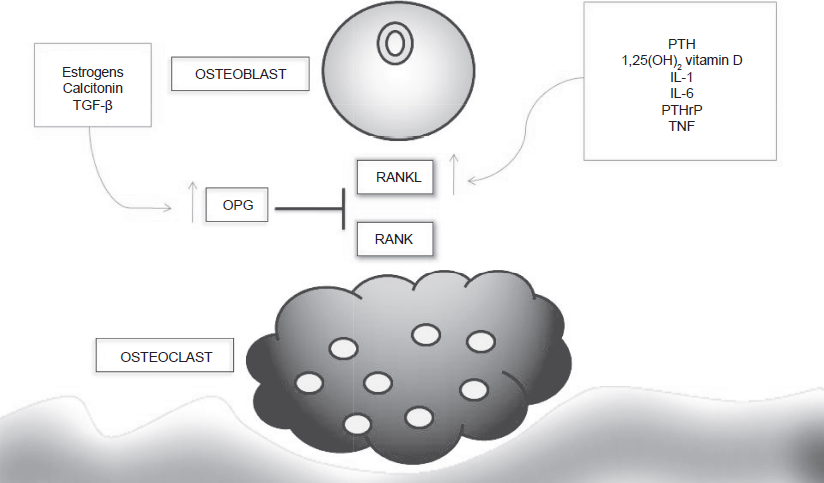
The RANKL/RANK/OPG pathway.
RANKL is not only present in the osteoblast, but is also expressed in T cells and dendritic cells. RANKL knockout mice have osteopetrosis along with deficient lymph node formation and poor lymphocytic differentiation, suggesting a role of RANKL not only in bone metabolism, but also in the immune system.9–11
Pharmacology of Denosumab
The immunization of XenoMouse with full-length human RANKL protein produced a human IgG1 monoclonal antibody against RANKL. 12 This antibody recognized an epitope with a single amino acid difference between human and mouse. 12 However, IgG1 antibodies can activate complement and antibody-dependent cell cytotoxicity (ADCC). Thus this antibody was converted to IgG2, which is non-cytotoxic. This is the current form of denosumab. It has a high affinity for human RANKL and does not recognize rodent RANKL. 12
Denosumab is a highly specific anti-RANKL antibody. In escalating dose, randomized, placebo-controlled clinical trials, the effective dose defined by the producing company AMGEN was 60 mg. 13 The solution of denosumab is 1 mL in 5% sorbitol with 10 mM sodium acetate in water for injection as a subcutaneous drug every 6 months. 14 The effect of one 60 mg dose of denosumab, as determined by levels of bone turnover markers, is approximately 6 months, at which point the levels of serum C-telopeptide (sCTX) return toward baseline.15–17
Pharmacokinetics
The pharmacokinetics of denosumab were studied in 49 postmenopausal women 17 who each received a single subcutaneous injection of 0.01, 0.03, 0.1, 0.3, 1.0, or 3.0 mg/kg. This study found that the kinetics were nonlinear and three phases were detected. The first one was a prolonged absorption phase (5 to 21 days), although the effect of denosumab was detected as early as 12 hours, determined by decreases in urinary N-telopeptide (uNTX).17,18 Serum concentrations of denosumab increased disproportionately greater than the increases in dose. The second phase was a prolonged β-phase with half lives ranging from 20 to 32 days, increasing with dose. The third phase was a faster terminal phase detected at concentrations <1000 ng/ml with a half-life of 5 to 10 days depending on the dose. The drug's mean serum residence time (MRT) was up to 46 days. 17
Why Denosumab and Not OPG
As mentioned previously, OPG is a protein secreted by the osteoblast, which blocks the interaction between RANK and RANKL, impeding the activation of the osteoclast. Fc-OPG fusion protein has been developed and tested in postmenopausal women, patients with breast cancer bone metastasi s, and patients with multiple myeloma. Compared with denosumab, Fc-OPG fusion protein has less suppression of bone turnover markers and a shorter antiresorptive effect. 17 There is also evidence of development of anti-OPG antibodies with this treatment, which theoretically could bind the native OPG, blocking its action. OPG also could bind tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), a molecule implicated in tumor surveillance, complicating the safety profile with high doses or prolonged treatment with OPG. Denosumab does not resemble human OPG and antibodies against denosumab have not been report, thus far making it safer than Fc-OPG fusion protein.12,17,18
Studies in Postmenopausal Osteoporosis
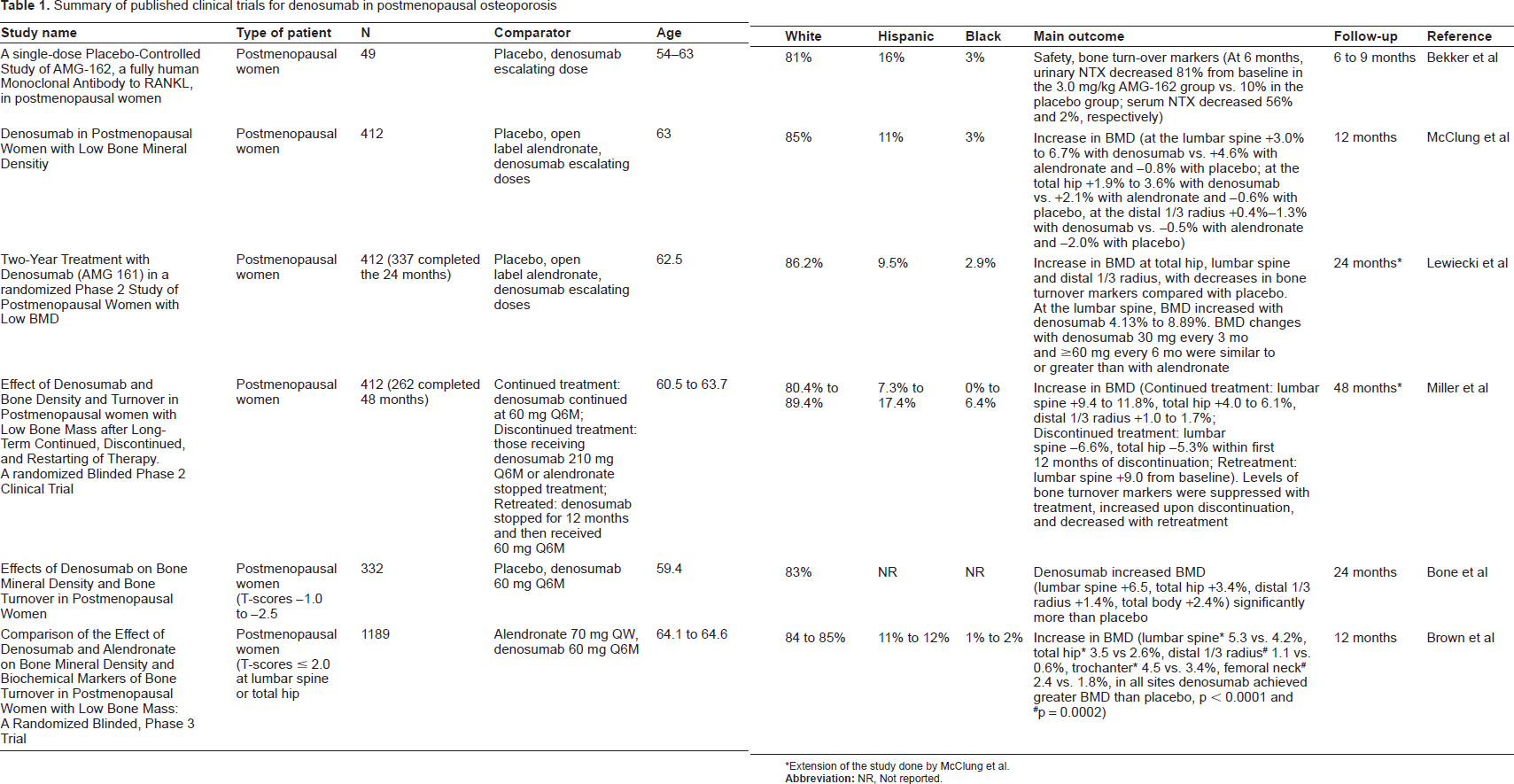
The first human study of denosumab in postmenopausal osteoporosis was a randomized, double bind, placebo-controlled clinical trial evaluating escalating doses of the study drug. 19 It included 49 postmenopausal women, 37 of whom received the drug while 12 received placebo. This study was designed to evaluate clinical parameters and biochemical markers of bone turnover as well as chemical profiles of safety including T and B cell enumeration. The study did not evaluate BMD. In all groups, dose-dependent decreases in uNTX and serum NTX (sNTX) were found. These decreases were reversible after stopping the drug, with levels returning to baseline values within a maximal time frame of 9 months. Also, a dose dependent decrease in bone-specific alkaline phosphatase (BALP) was observed. The calcium and phosphorus levels also had moderated non-clinically significant decreases. PTH increased slightly at the beginning of the therapy but the mean levels decreased during the follow-up. This profile is consistent with an antiresorptive effect with compensatory PTH secretion. In this trial no serious adverse events were reported. Side effects of the drug included pain at the injection site and site rash. 17 A summary of denosumab studies is presented at Table 1.
Summary of published clinical trials for denosumab in postmenopausal osteoporosis
Extension of the study done by McClung et al.
The first phase 2 randomized clinical trial in women with low BMD was published in 2006 by McClung et al. 13 The objective of this study was the efficacy and safety of denosumab in 412 postmenopausal women with T-scores of -1.8 to -4.0 at the lumbar spine or -1.8 to -3.5 at the proximal femur. The follow-up period was 12 months. This trial compared different doses of denosumab (6, 14 or 30 mg every 3 months, 14, 60, 100 and 210 mg every 6 months), open label alendronate (70 mg weekly) and placebo. The primary endpoint was the percentage change from baseline in BMD at the lumbar spine at 12 months. The treatment with subcutaneous denosumab resulted in significant increases in BMD at the lumbar spine, total hip, distal third of radius, and total body BMD in all groups who received the different doses of denosumab. The mean change in BMD for all denosumab doses was 3.0% to 6.7% at the lumbar spine, 1.9%-3.6% at the total hip and 0.4%-1.35% at the distal 1/3 radius. Lumbar spine BMD increased 4.6 percent with alendronate 70 mg weekly and decreased 0.8 percent with placebo. Total hip BMD increased 2.1 percent with alendronate and decreased 0.6 percent with placebo. The distal 1/3 radius BMD decreased 0.5 percent with alendronate and 2.0 percent with placebo. Significant and profound decreases in sCTX and BALP were found with denosumab. No significant differences among groups were observed in the profile of adverse events, and the incidence of dyspepsia was lower in the denosumab group than in the alendronate group. 13
This randomized clinical trial 13 was continued in a study extension to complete 24 months of evaluation. 20 Out of 412 patients enrolled in the study, 337 completed the 24 month period of evaluation (81.8%). There were persistent increases in BMD at all measured sites (lumbar spine 4.13 to 8.89%) and persistent lower levels of bone turnover markers consistent with prior findings at 12 months. The increases in BMD were similar or greater than alendronate 70 mg weekly with the exception of the lower doses of denosumab (14 mg every 6 months). 20
After 24 months follow-up, the study groups were divided into a continued treatment group, discontinued treatment group, and retreatment group. 21 The continued treatment group consisted of subjects who previously received denosumab at doses of 6 mg every three months, 14 mg every three months, 14 mg every six months, 60 mg every six months, or 100 mg every six months and were then treated for the next 24 months with denosumab 60 mg every six months. Patients who had been treated with alendronate or the highest dose of denosumab (210 mg every six months) were instructed to stop the drug for 24 months (discontinued treatment group). The patients who had received denosumab 30 mg every three months discontinued the treatment for one year and were treated after this phase with denosumab 60 mg every six months (retreatment group). 262 subjects (64%) completed the 48 months of follow-up. The placebo group remained on placebo throughout the four years. 21
The continuous treatment group achieved important gains in BMD. At the lumbar spine the mean BMD increased 9.4% to 11.8% compared with a 2.4% decrease in the placebo-treated subjects (p < 0.001). The mean BMD at the total hip increased 4.0% to 6.1%, a significant change compared with placebo (-3.5%, p < 0.001). Mean BMD at the distal 1/3 radius increased 1.0% to 1.7% in the continuous treatment group compared to a decrease of 4.7% in the placebo group. Persistent low levels of sCTX, BALP and uNTX were found with continuous denosumab treatment. 21
Discontinuation of the therapy resulted in near normalization of biochemical markers of bone turnover similar to the levels of the placebo groups. It also resulted in a decrease in BMD at all measured sites. These decreases were close to baseline BMD but were greater than the BMD of placebo-treated subjects, whose BMD continued to fall throughout the follow-up period. 21 These findings suggest that denosumab effects on bone are reversible, but can delay the loss of BMD even in patients who discontinue treatment.
The retreatment with denosumab 60 mg every six months produced similar gains in mean BMD (9.0% at the lumbar spine, 3.9% at the total hip, and 1.8% at the distal 1/3 radius). Also, bone turnover markers decreased below baseline values with retreatment. 21
Phase 3 Clinical Trials
As mentioned above, previous studies suggested that denosumab 60 mg subcutaneous is an appropriate dose suitable for further evaluation. Bone et al 14 reported a phase 3 clinical trial comparing this dose of denosumab against placebo in a randomized, double blind study in postmenopausal women with low BMD (T-scores -1.0 to -2.5) with follow-up of two years. This trial studied 332 women (mean age of 59.4 years), all of whom were instructed to receive 1000 mg calcium daily and had normal levels of 25-hydroxyvitamin D3 (at baseline, or repleted prior to study entry). The primary end point was the percent change from baseline in lumbar spine at 24 months evaluated by dual-energy x-ray absorptiometry (DXA), compared with placebo. The secondary end points were the change in BMD at the total hip, femoral neck, and distal 1/3 radius. Total body BMD and trabecular, cortical and total volumetric changes were also assessed by quantitative computed tomography (QTC) at the distal radius. In addition, biochemical markers of bone turnover were assessed including tartrate-resistant acid phosphatase (TRAP-5b) and intact N-terminal propeptide of type 1 procollagen (P1NP). 14
After 24 months, significant changes were observed at the lumbar spine of those treated with denosumab compared to those receiving placebo (6.5% vs. -0.6%, p < 0.05). Also significant increases were observed at the total hip (3.4%), the distal 1/3 radius (1.4%) and the total body (2.4%). QTC analysis demonstrated that denosumab increased volumetric BMD at the distal radius. Denosumab also decreased the levels of CTX and TRAP-5b with detectable decreases after the first month of therapy. The bone formation marker P1NP also was reduced. 14 Although this study was not designed to detect any differences in fracture incidence, the denosumab treatment group experienced clinical fractures in 1% of cases (two patients) compared with 4% in the placebo group.
The DECIDE trial (Determining Efficacy: Comparison of Initiating Denosumab versus Alendronate) was a recent randomized, double bind, multicenter study designed to compare denosumab versus alendronate in a non-inferiority study. 22 This trial randomized 1,189 postmenopausal women with T-scores ≤ -2.0 at the total hip or lumbar spine to receive denosumab 60 mg every six months versus alendronate 70 mg weekly. All subjects received matched placebo. The primary end-point was the percentage change from baseline in BMD at the total hip after 12 months of treatment.
Patients treated with denosumab achieved a 3.5% increase in BMD at the total hip compared with a 2.6% increase in subjects treated with alendronate. As the pre-specified non-inferiority margin was met, the secondary end-points were analyzed as well as superiority tests. Denosumab treatment produced greater increases at the total hip, trochanter (4.5% vs. 3.4% p < 0.0001)), femoral neck (2.4% vs. 1.8% p = 0.0002), lumbar spine (5.3% vs. 4.2% p < 0.0001), and the distal 1/3 radius (1.1% vs. 0.6% p = 0.0002) compared with alendronate. 22
Denosumab and alendronate produced significant decreases in sCTX and P1NP at 12 months. sCTX were lower in the denosumab-treated group with differences noted even at the first month of treatment. At 12 months, the sCTX levels were comparable in both groups. P1NP levels remained lower in the denosumab group compared to the alendronate group throughout the study. 22
Of interest, this study was the first to demonstrate greater achievements of BMD with an anti-resorptive agent (denosumab) compared to alendronate. Previous head to head studies of alendronate versus risedronate or ibandronate failed to demonstrate superiority in terms of BMD gain.6,23 The kinetics of sCTX and P1NP also suggest differences in the mechanism of action and biological effect on bone. While bisphosphonates are incorporated in the bone matrix and remain in the bone, denosumab is a monoclonal antibody that disappears from blood after approximately 6 months.
Another consistent finding in the above studies was the gain in BMD at the distal 1/3 radius. Neither alendronate nor teriparatide24,25 have demonstrated increases at this particular type of cortical bone. The increases in BMD at the distal radius with denosumab, suggest that this anti-resorptive agent has a particular effect in cortical bone. This point was evaluated in a post-hoc analysis by Beck et al 26 evaluating hip structure analysis (HSA) of phase 2 studies.13,20 HSA derivate bone geometry of the proximal femur from DXA scans of the hip. This program has been used to evaluate the effect of drugs for osteoporosis in the geometry of the hip as a surrogate marker of bone strength. Beck et al found that denosumab increased BMD (HSA-BMD) at the narrow neck, intertrochanteric area and femoral shaft (the three sites of HSA evaluation) compared with placebo. Denosumab achieved greater increases in HSA-BMD at the intertrochanteric area and femoral shaft than alendronate and had greater bone cross-sectional area, cortical thickness, and outer diameter at the shaft than alendronate. 26 This study suggested a preferential effect of denosumab in cortical bone. However, the real effect of denosumab in this type of bone needs to be validated in ongoing trials of denosumab's effects on non-vertebral fracture incidence, particularly hip and wrist fractures.
At the American Society for Bone and Mineral Research (ASBMR) 30th Annual Meeting in September 2008, results were shared from a phase 3 study of the effects of denosumab on vertebral, nonvertebral, and hip fractures in women with osteoporosis, the FREEDOM study (Fracture REduction Evaluation of Denosumab in Osteoporosis every 6 Months). 27 A total of 7,868 women ages 60 to 90 years old (mean age = 72.3 years) were recruited from 214 clinical centers worldwide. Subjects had lumbar spine or total hip T-scores < -2.5 and ≥-4.0 (mean lumbar spine T-score was -2.8). Twenty-three percent of subjects had prevalent vertebral fractures at baseline. Subjects were randomly assigned to receive denosumab 60 mg every 6 months or placebo for 3 years. All participants received calcium 1000 mg and vitamin D 400 to 800 IU daily.
During three years of study, 2.3% of patients taking denosumab experienced new vertebral fractures compared to 7.2% of placebo patients, a significant 68% risk reduction (p < 0.0001). Non-vertebral fractures were reduced 20% with denosumab (8% vs. 6.5%, p = 0.011), and hip fractures reduced by 40% compared to placebo (1.2% vs. 0.7%, p = 0.036). BMD at the lumbar spine increased by 9% over the three years compared to placebo and by 6% at the total hip (both significant at p < 0.0001). The adverse event rate was similar in both the denosumab and placebo groups. 27
The double blind, randomized, controlled STAND (Study of Transitioning from Alendronate to Denosumab) trial was also presented at the 2008 ASBMR meeting. 28 504 post-menopausal women with T-scores between -4.0 and -2.0 at the lumbar spine or total hip, who previously had been treated with alendronate for a mean (±SD) 44 ± 33 months, were randomized to subcutaneous denosumab 60 mg every 6 months or oral alendronate 70 mg weekly. After one year, BMD increased significantly more at the total hip, lumbar spine, and distal third of the radius in those switched to denosumab than those who continued with alendronate. Incidence of adverse events were similar in both groups. 28
Table 1 presents summarized data from clinical trials of denosumab in osteoporosis and Table 2 presents the ongoing trials and recently completed trials reported in clinicaltrials.gov. (Accessed June 21, 2009).
Ongoing clinical trials of denosumab in osteoporosis.
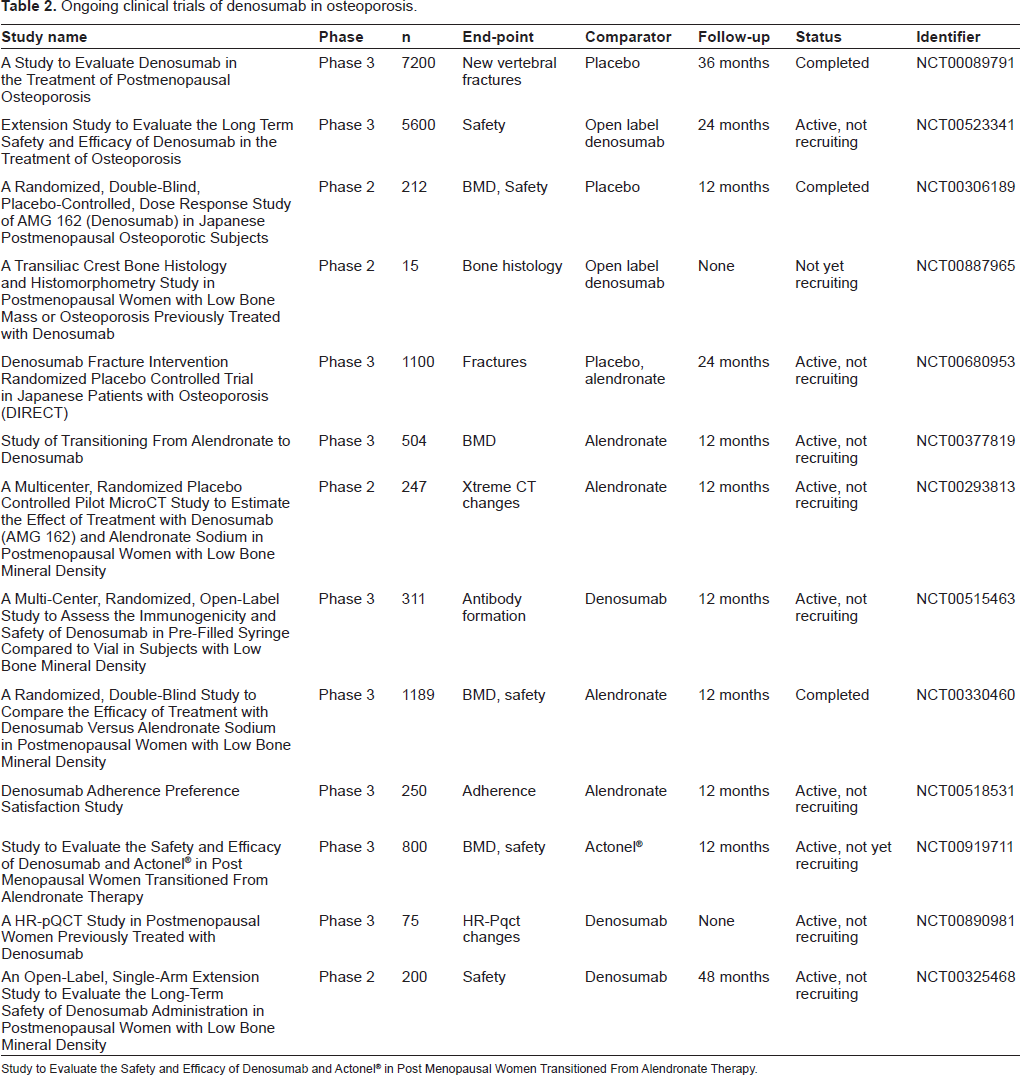
Study to Evaluate the Safety and Efficacy of Denosumab and Actonel® in Post Menopausal Women Transitioned From Alendronate Therapy.
Safety and Tolerability
RANK protein is expressed in the osteoclast and in cells of the immune system, particularly in dendritic cells and B and T cell lines. RANKL is expressed in T cells and dendritic cells, and knockout mice for this protein have impaired lymph node formation and lymphocytic differentiation. 10 The effect of RANK-RANKL blockade by denosumab in the immune system is not known and not yet specifically studied. However, hypothetically, RANKL inhibition could decrease monocyte and dendritic cell function and survival, alter antigen presentation, disrupt CD4+ T cells activation independent of CD40L/CD40, decrease early thymocyte development, decrease isotype switch of pro-B cells and reduce B cell development. 29 This theoretically would produce an increase in infections, tumors, and hematologic or immune dysfunction. However, the immune system is characterized by redundant activation pathways which can overpass RANK-RANKL blockade. A phase 1 study with 49 subjects did not detect any changes in CD3, CD4, CD8, CD20 and CD56 positive cells, changes in levels of immunoglobulins, nor changes in routine hematological and chemistry tests. 17 But investigators must be cautious if new neoplasias or infections in study subjects develop. And clearly, longer and large studies with denosumab would be preferable before its commercial use.
On the other hand, the role of RANKL system in inflammation is not always beneficial. OPG and RANKL have been detected in atherosclerotic plaques, OPG levels are elevated in patients with coronary artery disease, and such levels have predicted cardiovascular mortality in elderly women.30–34 T cells from patients with unstable angina have increased expression of RANKL, with such expression induced by percutaneous coronary intervention (PCI), a mechanical surrogate of plaque rupture. 35 Soluble RANKL and OPG levels are independent and positive predictors of coronary heart disease risk. 36 This data suggest involvement of the RANKL/RANK/OPG pathway in the pathogenesis of atherosclerosis plaque destabilization.35,37 The question is if the blockade of this pathway in high risk patients or in acute coronary syndromes could be beneficial. 36
At the writing time of this manuscript, the longest follow-up time of patients treated with denosumab is 48 months from a phase 2 trial. 21 In this study, the most common adverse events were upper respiratory tract infections, arthralgias, and back pain. Serious adverse events were reported on 10.9% of placebo-treated patients, 17.8% in the denosumab group and 17.4% in the alendronate group. Infections requiring hospitalization were more common in the denosumab treated group, all of which were community acquired infections treated with standard antibiotics. There were 4 deaths in the denosumab-treated group (gastric cancer, adenocarcinoma, brain neoplasm and cerebrovascular accident). No deaths were reported in the other study groups. This finding is worrisome and indicates that longer follow-up studies are needed to detect this, given the fact that these deaths were not present at 12 or 24 months of follow-up.13,20 The comparison groups had fewer patients than the denosumab group in this study (46 placebo patients, 46 alendronate patients, and 314 denosumab patients). This fact therefore does not allow confident comparisons of side effects.
All the phase 2 trials of denosumab measured calcium and PTH, as well as standard chemistry and hematology tests. 13 The calcium tended to decrease slightly, with no reported hypocalcemic symptoms, but remained stable through the 48 months of evaluation. 21
Safety in Phase 3 Trials
The above studies included different doses of denosumab and defined the dose of 60 mg every 6 months as a suitable dose to continue with phase 3 trials.
The first phase 3 trial with a complete follow-up of 24 months was in postmenopausal women with T-scores between -1.0 and -2.5% at lumbar spine. 14 This study detected a similar overall incidence of side effects between the placebo and denosumab-treated groups. In both groups, the most common side effects were arthralgias, nasopharyngitis, and back pain. Sore throat and rashes were reported more frequently in the denosumab group. Significant adverse events were noted in 5.5% of placebo-treated groups and 18% of denosumab groups. The most common significant adverse events in the denosumab groups were infections requiring inpatient treatment. The types of infections were pneumonia, diverticulitis, appendicitis, sepsis, pyelonephritis, urinary tract infection, and cellulitis with no reported opportunistic infections. The treatment consisted of standard antibiotics and had uncomplicated courses of one to six days. There were four cases of neoplasms in the denosumab group (breast cancer in situ, mycosis fungoides, ovarian cancer, and uterine cancer) and B cell lymphoma in one patient in the placebo group. No deaths were reported.
In other studies in women with high risk for infections (breast cancer and rheumatoid arthritis), the incidence of severe infection requiring hospitalization and incidence of overall infection were similar between denosumab and placebo groups.38,39
No significant differences in adverse events were found in other studies comparing alendronate versus denosumab involving 1189 subjects with follow-up for 12 months. 40 The most common side effect in both groups was arthralgia. Two deaths were reported in this study, one in each group. Neoplasms (denosumab group 6 cases: 2 breast cancer, gastric cancer with liver metastases, mycosis fungoides, renal cell carcinoma, and squamous cell carcinoma; alendronate 5 cases: breast cancer, metastatic neoplasm, ovarian cancer, vaginal cancer, small cell lung cancer metastatic) and severe infections (denosumab 9 cases and alendronate 6 cases, no opportunistic infection reported) were reported in a balanced manner in both groups.
Patient Focused Perspectives
Adherence to treatment is an important topic in the field of osteoporosis.3,41 Low adherence is associated with loss of beneficial effects of osteoporosis treatment and seems to increase the rate of vertebral fractures. Factors associated with adherence to osteoporosis treatment include the type of drug prescribed with a compliance advantage for drugs given weekly (e.g. oral alendronate) versus daily (e.g. subcutaneous injection teriparatide). 42 Side effects and low motivation are the main reasons for drug discontinuation. Effective drugs for chronic diseases with administration intervals of months to a year could increase adherence and improve quality of life. Denosumab could eventually be such a drug. Its dosing time frame could be comparable with risedronate or ibandronate once a month43–46 or zolendronic acid infusion once a year. Advantages of denosumab over other osteoporosis drugs include the biannualadministration (overalendronate, risedronate, ibandronate, and teriparatide), self-administration (over zolendronic acid), low persistence in bone (overall bisphosphonates) and potential benefits in cardiovascular disease.
The disadvantages of denosumab are patients’ fear of injections versus oral administration of other drugs, the potential of increasing infection, neoplasm or immune dysfunction, and the lack of longer and larger efficacy studies. Additionally, denosumab also must demonstrate improved quality of life and adherence, because longer interval of times of dosing does not prove improved adherence or compliance.
Conclusions
Denosumab is a new option in development for the treatment of osteoporosis. It is a human monoclonal antibody targeting RANKL. Its short half life and novel mechanism of action at bone contribute to its therapeutic potential. Efficacy phase 3 trials in postmenopausal women demonstrated increases in BMD at the lumbar spine, total hip and distal 1/3 radius. The gain in BMD is greater than alendronate, a finding only achieved by the bone forming agent teriparatide. The effect of denosumab in BMD at the total hip and the distal 1/3 radius may be predictive of decreased incidence of fractures at these sites, however, to date, the ongoing trials for fracture outcomes have been reported only in abstract and oral form. Although, targeting the RANK-RANKL pathway could affect the immune system (denosumab may prove to be contraindicated in those who are immunosuppressed), denosumab seems to be a safe drug, with similar adverse effects compared to placebo or alendronate. However, longer follow-up is needed, given the fact that deaths reported in phase 2 trials were only evident after 48 months of follow-up. With more safety and anti-fracture data being collected, denosumab may prove a very good candidate for osteopenia and osteoporosis treatment. The age of biological therapy for osteoporosis has begun.
Disclosure
The authors report no conflicts of interest.
