Abstract
The first selective If current inhibitor, ivabradine, lowers heart rate (HR) at rest and during exercise with no vasomotor, negative inotropic, or negative lusitropic effects. Given that elevated resting HR is a key factor in the onset of myocardial ischemia and a strong independent predictor of cardiovascular outcomes, ivabradine provides new therapeutic prospects in coronary artery disease (CAD). Its selective HR-lowering action has proven anti-ischemic and anti-anginal efficacy, and ivabradine is currently indicated for the symptomatic treatment of stable angina pectoris. Ivabradine can also be safely combined with other anti-anginal agents, and addition of ivabradine to beta-blocker therapy further improves anti-ischemic efficacy and exercise capacity of patients with stable angina. The recent BEAUTIFUL trial demonstrated that although the primary endpoint was not met in the overall population, addition of ivabradine on top of standard preventive treatments significantly reduced the risk of coronary events in stable CAD patients with left ventricular systolic dysfunction among the subgroup of patients with a resting HR ≥ 70 bpm. This is in accordance with pre-clinical data showing that long-term HR reduction improves endothelial function and reduces the progression of atherosclerosis. A significant proportion of patients with stable angina have elevated resting HR and ivabradine should therefore be considered as an important therapy in these cases. In combination with other standard treatments, ivabradine can improve angina and could potentially improve coronary outcomes. Ongoing and future clinical studies will evaluate the presence and magnitude of the cardioprotective benefits of HR lowering with ivabradine in patients with cardiovascular diseases.
Keywords
Introduction
Coronary artery disease (CAD) is currently the leading cause of death and disability worldwide. 1 The presenting symptom in half of patients with CAD is stable angina pectoris, which significantly impairs daily activities in most patients and often leads to premature retirement.2,3 Formal investigations using validated questionnaires show that even mild angina symptoms seriously reduce the quality of life (QoL) of patients in terms of physical functioning, sense of well-being, and perceived health. 4 In agreement with these results, reduction in angina attacks through medical treatment or coronary revascularization is associated with marked improvement in QoL.5–8 In addition to the impact on QoL, the frequency of anginal symptoms is also positively associated with higher rates of hospital admissions for coronary events. 9 Furthermore, recent data from a community cohort study indicate that CAD patients with anginal symptoms alone have a similar prognosis to that of patients with a history of myocardial infarction (MI) or revascularization. 10 These findings indicate that the importance of stable angina pectoris cannot be underestimated, and it is crucial for the patient that this condition be optimally managed.
Although much progress has been made in the management of CAD and angina, many patients remain poorly treated and there is a clear need for novel means of management. One established management strategy is heart rate (HR) reduction, which reduces myocardial oxygen demand and increases diastolic perfusion time. 11 This strategy is well-recognized as a beneficial therapeutic intervention for the prevention of myocardial ischemia and angina. In addition, elevated resting HR is independently associated with an increased long-term risk of cardiovascular outcomes, particularly among CAD patients as demonstrated by retrospective clinical and epidemiological observations.12,13 Control of HR should therefore be regarded as a key therapeutic target in CAD to improve symptoms and potentially long-term cardiovascular outcomes.
Consistent with this understanding of the central importance of HR, ivabradine, the first clinically available selective If current inhibitor, provides new opportunities in the management of stable angina pectoris. If is an inward mixed current of Na+ and K+ ions carried through hyperpolarizationactivated cyclic nucleotide–activated channels (f channels) and is crucial for regulating cardiac pacemaker activity in the sinoatrial node. Ivabradine acts by selective and specific binding to f channels to inhibit If in a dose-dependent manner, with no effects on other cardiac channels. 14 Inhibition of If reduces the slope of diastolic depolarization in sinoatrial node cells, decreases the frequency of action potential initiation (Fig. 1), and thereby lowers HR at rest and during exercise.15,16 Unlike other pharmacological agents used to lower HR, ivabradine can be considered as a selective HR-reducing agent with no vasomotor, negative inotropic, or negative lusitropic effects. In addition to lowering myocardial oxygen demand, selective HR reduction with ivabradine increases diastolic perfusion time and improves oxygen supply, while preserving cardiac stroke volume at rest and during exercise.17–19 In patients with CAD, these actions have proven anti-ischemic and antianginal efficacy, 20 and ivabradine is currently indicated for the symptomatic relief of stable angina pectoris. In this article, we review preclinical and clinical studies that demonstrate the anti-ischemic and anti-anginal effects of ivabradine and that give further insights into its potential to modify disease progression in patients with stable angina.
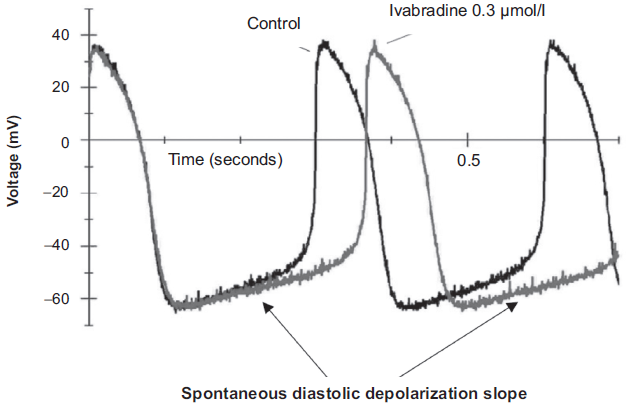
Spontaneous diastolic depolarization and action potential generation in rabbit sinoatrial node in the absence (control) and presence of 0.3 μmol/L ivabradine. Reproduced with permission from DiFrancesco. Pharmacol Res. 2006;53(5):399-406.
Preclinical Studies Demonstrating Beneficial Effects of Ivabradine on Myocardial Ischemia, Endothelial Function and Progression of Atherosclerosis
Experimental animal models have clearly demonstrated that HR lowering with ivabradine reduces myocardial oxygen consumption while improving oxygen supply. In exercising dogs, graded HR reduction with varying doses of ivabradine linearly decreased myocardial oxygen consumption. 21 In the same study, ivabradine also increased diastolic perfusion time in a dose-dependent manner. Additional animal studies showed that HR reduction with ivabradine increased coronary blood flow during exercise. 19 In contrast, for the same HR reduction, β-blockers did not increase diastolic perfusion time to the same degree and also reduced coronary blood flow during exercise.17,19 Unlike β-blockers which can lead to vasoconstriction due to unmasked alpha-adrenergic tone, ivabradine does not alter the balance between α- and β-adrenergic tone. The cardioprotective benefits of selective HR reduction with ivabradine are in part due to reduced myocardial oxygen consumption, together with improved oxygen supply, through longer diastolic perfusion time and preserved coronary artery dilation during exercise.
The anti-ischemic effects of ivabradine were initially investigated in a canine model of exercise-induced myocardial ischemia and subsequent stunning. 22 In this model, dogs underwent surgical coronary stenosis to suppress the normal increase in coronary artery blood flow during exercise. Under these experimental conditions, administration of ivabradine improved LV regional function and perfusion in the ischemic zone during exercise compared with saline. 22 The anti-ischemic effect of ivabradine also protected against myocardial stunning during the recovery period after exercise. Administration of ivabradine after exercise, when the myocardium was stunned, also improved LV regional contractility. None of these effects occurred when dogs underwent atrial pacing, indicating that the anti-ischemic effects of ivabradine are the result of selective HR reduction. In contrast to treatment with ivabradine, administration of a beta-blocker during the stunning period greatly impaired myocardial function as a result of negative inotropic effects. 23 Ivabradine also reduced regional myocardial ischemia during exercise by approximately 80% in pigs with surgical coronary artery stenosis. 24 In the same study, ivabradine preserved LV contractility at rest and during exercise to a significantly greater degree than the beta-blocker. Taken together, the findings of these studies demonstrate that, under short-term ischemic conditions, HR lowering with ivabradine maintains cardiac function by improving myocardial perfusion and its relationship to myocardial oxygen demand and sustaining myocardial contractility.
More recently, the protective effects of ivabradine against irreversible myocardial injury after a prolonged period of myocardial ischemia have also been investigated. 25 While monitoring myocardial regional blood flow and function, anesthetized pigs were subjected to 90 minutes of controlled coronary hypoperfusion followed by 120 minutes of reperfusion. At the end of the experimental protocol, histological analysis was used to assess myocardial infarct size. Compared with placebo, treatment with ivabradine increased ischemic regional blood flow and contractile function, and reduced infarct size. Reduction in myocardial infarct size was observed even when ivabradine was administered only after the onset of ischemia or just before the reperfusion period. These beneficial effects on infarct size were attenuated by atrial pacing, suggesting that HR reduction per se plays an important part in the cardioprotective effects of ivabradine. 25
Additional animal studies have examined the long-term beneficial effects of HR lowering with ivabradine on endothelial function and atherosclerosis. Apolipoprotein E–deficient (apoE-/-) mice fed a high-cholesterol diet represent a well-characterized model of endothelial dysfunction. In this model, administration of ivabradine for 6 weeks significantly improved aortic endothelium–mediated vasodilation, without any effects on blood pressure or lipid levels. Despite the severe hypercholesterolemia, improvement of endothelial function with ivabradine was associated with >40% and >70% reductions in atherosclerotic plaque size in the aortic root and ascending aorta, respectively (both P < 0.05; Fig. 2). 26 Ivabradine also markedly inhibited vascular oxidative stress; NADPH oxidase activity, superoxide production, and lipid peroxidation were all reduced in the aortic wall of ApoE-/- mice receiving ivabradine. 26 The authors found no direct effects of ivabradine on the endothelium, implying that these protective effects were mediated by the observed 13.4% reduction in HR. In our study of a transgenic model of dyslipidemia and endothelial dysfunction, 3-month treatment with ivabradine preserved endothelium-mediated vasodilation in the renal and cerebral arteries of mice expressing the human apoprotein B (apoB-100). 27 In this study, ivabradine also restored the contribution of hydrogen peroxide to endothelium-dependent vasodilation in cerebral vessels, implying a reduction in vascular oxidative stress. Treatment of apoB-100 mice with the beta1 adrenoreceptor–selective antagonist metoprolol did not restore endothelial function to the same degree. 27 This was possibly because of inhibitory effects of metoprolol on beta-adrenoreceptor–mediated activation of endothelial nitric oxide synthase. Overall, the findings of these two studies show that long-term HR reduction with ivabradine improves endothelial function and reduces progression of atherosclerosis in mice models of dyslipidemia and atherosclerosis.
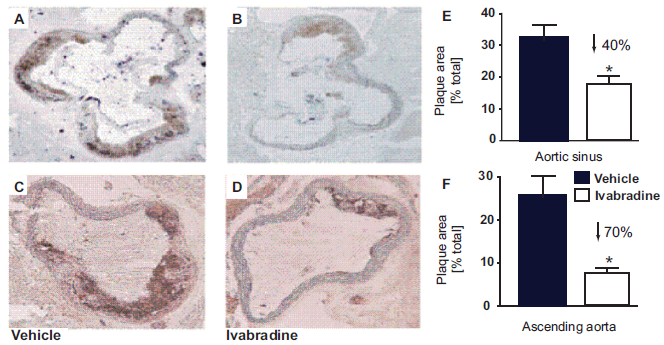
Effect of ivabradine on atherogenesis in apolipoprotein E–deficient mice. Histological samples are shown from aortic sinus (
Clinical Data Demonstrating Beneficial Effects of Ivabradine on Myocardial Ischemia, Stable Angina Pectoris and Coronary Events in CAD Patients
As in animal studies, clinical trials have demonstrated that ivabradine reduces HR at rest and during exercise in a dose-dependent manner.20,28,29 Exercise tolerance tests (ETTs) have shown the anti-ischemic and anti-anginal efficacy of ivabradine. During bicycle ETTs, time to 1-mm ST-segment depression significantly increased after 2 weeks of treatment with ivabradine 5 mg b.i.d. compared with placebo in patients with CAD and stable angina (P < 0.05). 20 In the same study, continuation of treatment with ivabradine for 3 months reduced the weekly number of angina attacks more than fourfold compared with baseline (P < 0.001).
The clinical efficacy of ivabradine was also confirmed when compared to that of other common classes of anti-anginal agents, including beta-blockers and calcium channel blockers. The INternatIonal TrIal of the AnTianginal effects of IVabradinE compared with atenolol (INITIATIVE) compared the anti-ischemic and anti-anginal effects of ivabradine 7.5 mg and 10 mg b.i.d. and of atenolol 100 mg o.d. in 939 stable angina pectoris patients. 29 After 4 months of treatment, ivabradine had increased total treadmill exercise duration by 86.8 seconds compared with 78.8 seconds for atenolol (P < 0.001 for non-inferiority). Interestingly, HR reduction with ivabradine resulted in a greater improvement in exercise capacity than atenolol for a 1 bpm reduction in HR: 10.1 seconds with ivabradine vs. 5.6 seconds with atenolol (Fig. 3). This difference may be due in part to the absence of any negative inotropic or coronary vasoconstrictive effects of ivabradine during exercise. In another comparative study, 1195 stable angina patients received either ivabradine (7.5 mg b.i.d. or 10 mg b.i.d.) or amlodipine 10 mg o.d. for 3 months. 28 Results of ETTs showed that time to 1-mm ST-segment depression increased by 45 seconds with ivabradine versus 40 seconds with amlodipine (P < 0.001), and both treatments decreased the frequency of anginal attacks by two-thirds. Ivabradine was superior to amlodipine in reducing myocardial oxygen consumption as demonstrated by the significantly greater effect on the rate-pressure product.
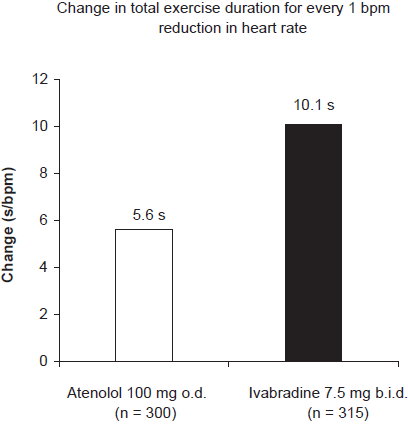
Change in total exercise duration per 1 bpm reduction in heart rate at peak exercise after 4 months of treatment with atenolol 100 mg o.d. (n = 300) or ivabradine 7.5 mg b.i.d. (n = 315) bpm, beats per minute.
Although beta-blockers are recommended as the initial therapy for stable angina pectoris, they are associated with a range of side effects and are not always well tolerated. Therefore, a large number of patients in clinical practice do not receive sufficient doses to provide optimal HR reduction and anti-ischemic protection. 30 Combination therapy is recommended to improve the symptomatic management of angina, 2 and the majority of stable angina patients receive two anti-anginal agents. However, most clinical studies demonstrate only modestly improved ETT parameters with combination therapy compared with monotherapy at the peak of drug activity, with no significant benefit 6 hours after drug intake. 31 Recently, the anti-ischemic efficacy of ivabradine in combination with beta-blocker therapy was investigated in a 4-month study of 889 stable angina patients already receiving atenolol 50 mg/day. 32 This dosage of atenolol is the most commonly prescribed one in clinical practice. 30 Patients were randomly assigned to treatment with ivabradine 5 mg b.i.d. uptitrated to 7.5 mg b.i.d. after 2 months (n = 449), or placebo (n = 440), on top of beta-blockade. During the study, the addition of ivabradine to the beta-blocker further reduced HR by 7 bpm after 2 months and by 9 bpm after 4 months compared with placebo (Fig. 4). Accompanying these HR reductions, treadmill ETT at trough of drug activity after 2 months and 4 months showed significant improvements in all test parameters for patients receiving ivabradine compared with placebo (Fig. 5). Furthermore, combination therapy of ivabradine and beta-blocker was well tolerated, with only 1% of patients receiving this treatment having to withdraw because of sinus bradycardia. 32 Altogether, the size of this study, the compliance with the regulatory recommendations, and the consistency of improvement across all ETT parameters constitute perhaps the best clinical evidence of benefit of any combination of anti-anginal drugs published to date.
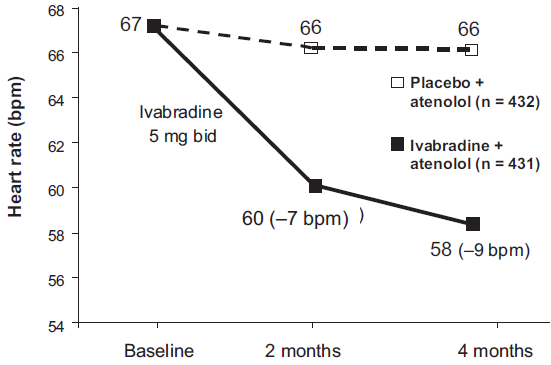
Effect of 4 months of treatment with ivabradine (5 mg b.i.d. for two months increased to 7.5 mg b.i.d. for two months) in combination with atenolol (50 mg/o.d.) on heart rate in patients with stable angina (n = 431), versus placebo and atenolol (50 mg/o.d.) (n = 432).
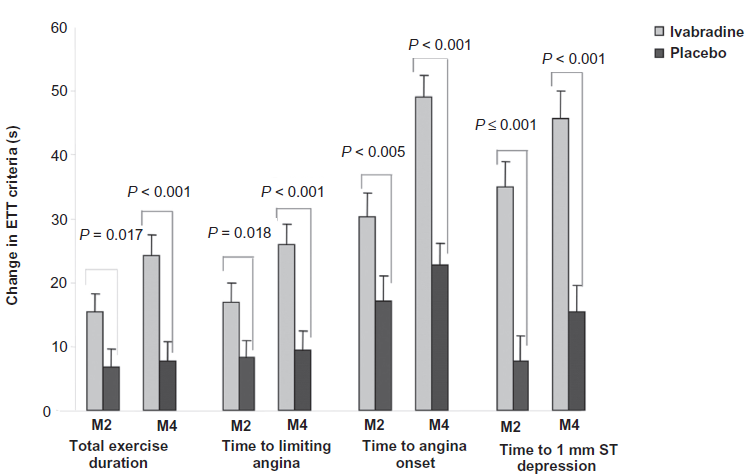
Changes in exercise tolerance test (ETT) criteria from baseline to month 2 (M2) and month 4 (M4) among stable angina patients treated with the addition of either ivabradine (5 mg b.i.d. for two months increased to 7.5 mg b.i.d. for two months) (n = 449) or placebo (n = 440) on top of beta-blocker therapy (atenolol 50 mg/o.d.). Reproduced with permission from Tardif JC, et al. Eur Heart J. 2009.
The long-term antianginal efficacy and safety of ivabradine have been investigated in a population of 386 stable angina patients in a double-blind, randomized, parallel-group study over 12 months. 33 At study entry, many of the patients were receiving concomitant therapy for cardiovascular conditions, including dihydropyridine calcium channel blockers and long-acting nitrates to manage anginal symptoms. Patients were assigned to treatment with either ivabradine 5 mg b.i.d. (n = 198) or ivabradine 7.5 mg b.i.d. (n = 188). After 12 months, mean HR was significantly reduced by 10 bpm from baseline in the ivabradine 5 mg b.i.d. group and by 12 bpm in the 7.5 mg group. These HR reductions were achieved after 1 month of treatment and remained stable thereafter, with no evidence of pharmacological tolerance. The HR-lowering efficacy of ivabradine was associated with significant anti-anginal efficacy: the mean number of angina attacks per week decreased by 50% in both treatment groups (P < 0.001). Ivabradine also significantly reduced the number of angina attacks among the 259 patients receiving concomitant anti-anginal treatment with dihydropyridine calcium channel blockers or long-acting nitrates in both treatment groups (P < 0.001 for 5 mg b.i.d.; P < 0.05 for 7.5 mg b.i.d.). Throughout the study, both doses of ivabradine were well tolerated: only 4 patients withdrew because of visual symptoms (1 in the 5 mg group and 3 in the 7.5 mg group), and only 0.8% of patients had to withdraw due to bradycardia. These findings clearly demonstrate the anti-anginal efficacy and safety of long-term treatment with ivabradine 5 mg b.i.d. and 7.5 mg b.i.d. on top of other widely used cardiovascular medications in patients with stable angina pectoris.
The two goals of treating a patient with CAD are to relieve anginal symptoms and to prevent acute coronary complications. In addition to its anti-ischemic and anti-anginal efficacy, recent clinical trial evidence suggest that selective HR lowering with ivabradine reduces the risk of coronary events. The BEAUTIFUL (morBidity-mortality EvAlUaTion of the If inhibitor ivabradine in patients with left ventricULar dysfunction) trial investigated the effects of ivabradine on top of standard treatments in a population of 10,917 CAD patients with LV systolic dysfunction over 2 years. 34 In addition to their standard cardiovascular treatments, patients in the BEAUTIFUL trial were randomly assigned to treatment with either ivabradine 5 mg b.i.d, with uptitration to 7.5 mg b.i.d., or placebo for 24 months. The mean age of the population at baseline was 65 years, and mean resting HR was 72 bpm. The proportion of patients receiving concomitant medications for cardiovascular disease was high: 94% of patients were receiving aspirin or anticoagulants, 74% were receiving statins, 90% were receiving angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers, and 87% of patients were receiving β-blockers.
BEAUTIFUL addressed the important clinical and pathophysiological relevance of HR reduction for cardiovascular outcomes. The large placebo population of the BEAUTIFUL trial (n = 5438) provided an opportunity to examine the importance of HR for long-term coronary outcomes in a well-treated CAD population with LV systolic dysfunction. This was done by analyzing the effect of elevated resting HR at baseline on outcomes in the placebo population. 35 Analyses of the adjusted data for coronary outcomes in the placebo group showed that elevated resting HR ≥ 70 bpm was a strong risk factor for cardiovascular events in patients with stable CAD and LV dysfunction. Within the placebo group, HR ≥ 70 bpm was associated with a 34% increase in the risk of cardiovascular death (hazard ratio, 1.34; 95% confidence interval [CI], 1.10-1.36; P < 0.005) and a 53% increase in the risk of hospitalization for new or worsening heart failure (hazard ratio, 1.53; 95% CI, 1.25-1.88; P < 0.0001) compared with HR < 70 bpm. 35 Likewise, elevated HR was associated with a 46% increase in risk of fatal and nonfatal MI (hazard ratio, 1.46; 95% CI, 1.11-1.91; P < 0.01) and a 38% increase in the risk of coronary revascularization (hazard ratio, 1.38; 95% CI, 1.02-1.86; P < 0.05). These findings are the first prospective demonstration that elevated resting HR increases the risk of coronary events even in CAD patients who are well treated in accordance with current guidelines.
Ivabradine significantly improved coronary outcomes in BEAUTIFUL among the subpopulation of patients with baseline resting HR ≥ 70 bpm (n = 5392), although no effect was seen on the primary composite end point (cardiovascular death, hospitalization for acute MI, and hospitalization for new or worsening heart failure) in the total study population. 34 These differences are most likely due to the greater HR reduction with ivabradine among patients with elevated baseline resting HR. Within this subpopulation, ivabradine reduced HR by 7.9 bpm after 12 months and 6.9 bpm after 24 months versus placebo. This was associated with a relative risk reduction of 36% in hospitalization for fatal and nonfatal MI (hazard ratio, 0.64; 95% CI, 0.49-0.84; P = 0.001) and 30% in the need for coronary revascularization (hazard ratio, 0.70; 95 CI, 0.52-0.93; P < 0.05) (Fig. 6). In terms of absolute benefits for stable CAD patients with elevated HR (>70 bpm) if confirmed in ongoing and future trials, a 2-year treatment with ivabradine would prevent one MI in every 56 patients, and one revascularization in every 83 patients. These data compare favorably with the number needed to treat for other agents used to prevent coronary events in stable CAD, including statins, ACE inhibitors, and beta-blockers.36–38 Ivabradine also resulted in a 22% relative risk reduction in the composite outcome of hospitalization for fatal and nonfatal MI and unstable angina pectoris (hazard ratio, 0.78; 95% CI, 0.52-0.93; P < 0.05). It is noteworthy that 84% of the subpopulation of patients with HR ≥ 70 bpm were already receiving beta-blockers, and the addition of ivabradine was safe and well-tolerated. Taken together, the findings of the BEAUTIFUL trial reinforce the importance of addressing elevated resting HR to potentially improve cardiovascular outcomes in CAD.
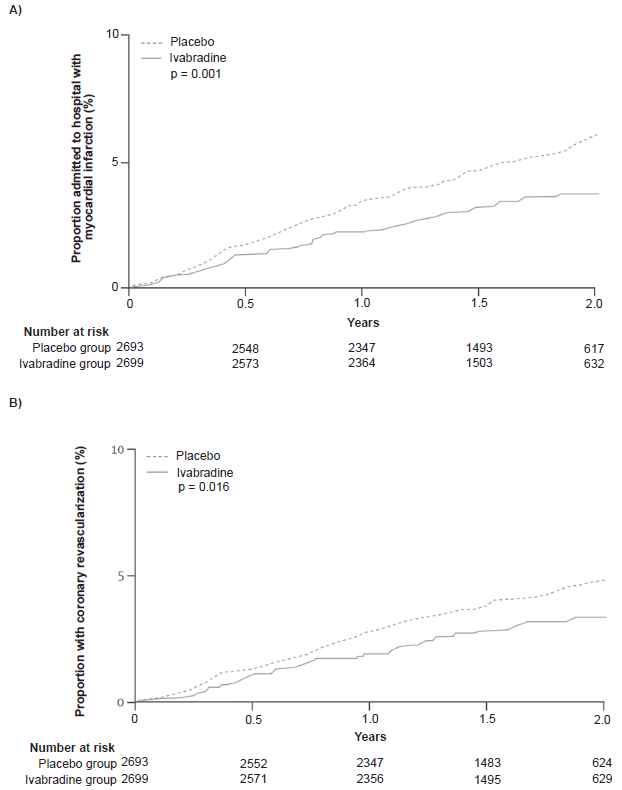
Kaplan-Meier time-to-event plots by treatment group (ivabradine or placebo) in the subgroup of stable coronary artery disease patients with an elevated resting heart rate ≥70 bpm, for hospitalization for myocardial infarction
Practical Implications in Light of Recent Clinical Trial Results
The first aspect of management of stable angina pectoris is the control of symptoms to improve exercise capacity and QoL. 2 Agents used to relieve angina include beta-blockers, calcium channel blockers, potassium channel openers, nitrates, and metabolic agents. 2 Although all these agents have documented anti-anginal and anti-ischemic efficacy, they are also associated with problems regarding tolerability and contraindications, which can considerably compromise the benefits of treatment. 39 Another option for the management of angina is surgical revascularization. However, the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial demonstrated only relatively small benefits in the QoL of patients after revascularization, which only lasted up to 3 years. 40 Furthermore, revascularization has not been shown to improve major cardiovascular outcomes in stable patients. 40 Thus, many patients with angina still have unmet clinical needs and require alternative therapies for symptomatic management.
The clinical data that we have reviewed demonstrate that ivabradine has further applications that can considerably improve the management of stable angina pectoris. Ivabradine is currently indicated for relief of anginal symptoms in CAD. 3 The anti-anginal and anti-ischemic efficacy of ivabradine apply to a wide range of patients irrespective of their background treatment. Ivabradine can be safely added on top of anti-anginal therapy with dihydropyridine calcium channel blockers, long-acting nitrates, or beta-blockers.32,33
In addition to controlling symptoms, the management of patients with stable angina pectoris requires cardioprotective treatment to improve clinical outcomes in the long term. 2 The main classes of agents used for cardioprotection in CAD include statins, ACE inhibitors and antiplatelet agents. Despite their anti-anginal efficacy, beta-blockers have never been shown to improve long-term outcomes in patients with stable CAD. 2 In this context, ivabradine is the first anti-anginal and anti-ischemic agent that can potentially reduce the risk of MI and revascularization in a stable CAD population with LV dysfunction and resting HR ≥ 70 bpm. 34 Furthermore, the BEAUTIFUL trial showed that resting HR ≥ 70 bpm at baseline increased the risk of cardiovascular outcomes, even within a CAD population that was being well treated with a combination of cardioprotective agents, including beta-blockers. 35 A large number of stable angina patients in clinical practice have resting HR above this threshold and this ought to be considered as an important factor in risk stratification. 41 Addition of ivabradine treatment to lower HR in this population should be considered as an important part of the long-term management strategy to improve potentially cardiovascular outcomes. Together with its anti-anginal efficacy, ivabradine will potentially play an important role in improving the long-term management of patients with stable angina pectoris, although this will need to be confirmed in new randomized trials.
Ongoing and Future Clinical Studies with Ivabradine
The VIVIFY (Evaluation of Intravenous If Inhibitor Ivabradine in Acute Coronary Syndrome) study is currently investigating the safety and tolerability of intravenous ivabradine (5 mg bolus followed by an 8-hour infusion of 5 mg) versus placebo among 120 patients with acute MI and ST-segment depression on ECG. Follow-up of patients after 6 months will also evaluate the cardioprotective effects of intravenous ivabradine on infarct size as assessed by magnetic resonance imaging. Support for the VIVIFY study comes from the protective action of ivabradine against irreversible myocardial injury in pigs after prolonged ischemia. 25
In patients with congestive heart failure, HR-lowering with beta-blockade is associated with a reduction in mortality. 42 Furthermore, pilot studies in patients with moderate LV dysfunction showed that ivabradine reduced LV end-systolic and end-diastolic volumes, suggesting a preserving effect on cardiac function. 43 These results have formed the basis of the SHIFT (Systolic Heart failure with the If inhibitor ivabradine Trial) study, which is currently investigating the effects of ivabradine in congestive heart failure. 44 This large-scale, randomized, placebo-controlled trial has recruited over 7000 patients with moderate to severe heart failure and resting HR ≥ 70 bpm. Throughout the study, all patients have continued their current standard treatments for heart failure and any other cardiovascular conditions. The aim is to evaluate the advantages of adding treatment with ivabradine versus placebo in terms of reduction in cardiovascular events in patients with heart failure.
Conclusion
Management of stable angina pectoris requires symptomatic treatment to improve patients’ QoL, as well as cardioprotective treatment to reduce the risk of cardiovascular events. The clinical studies that we have reviewed show that HR lowering with the If inhibitor ivabradine can play a role in both of these management strategies. The selective HR-lowering action of ivabradine reduces myocardial oxygen demand while increasing oxygen supply by virtue of a longer diastolic perfusion time. Ivabradine has proven anti-ischemic and anti-anginal efficacy without many of the problems associated with commonly used anti-anginal agents. Ivabradine is therefore suitable for a wide range of stable angina patients irrespective of their background treatment.
The BEAUTIFUL trial has shown that ivabradine is unique among anti-anginal agents, in light of its potential effect on coronary outcomes in stable CAD patients with elevated resting HR. This is consistent with preclinical data showing that long-term HR reduction with ivabradine protects against endothelial dysfunction and progression of atherosclerosis. Interestingly, the same preclinical studies showed that HR reduction with beta-blockers provided less endothelial protection. Selective HR reduction with ivabradine should therefore be considered as an important approach in the long-term management of stable angina patients with elevated resting HR. Furthermore, data from ongoing and future clinical studies will help us to determine more precisely the role of HR lowering with ivabradine as a new therapeutic modality for the management of patients with cardiovascular diseases.
Disclosure
The author reports no conflicts of interest.
