Abstract
Lisdexamfetamine (LDX) is the only available stimulant prodrug formulation that has been approved by the Food and Drug Administration (FDA) for treatment of ADHD in children and adults. It is enzymatically hydrolyzed to the active d-amphetamine and essential amino acid L-lysine. This rate limited enzyme mediated step underlies the consistent, predictable levels throughout the day and limits its potential for misuse. It is effective, safe and well tolerated and has an adverse effect profile comparable to other extended release stimulant formulations. The d-amphetamine levels from LDX are more consistent than that from extended release mixed amphetamine salts. LDX has shown efficacy for ADHD in clinical trials involving children and adults. Ongoing research studies are attempting to establish its long term efficacy and tolerability in patients with ADHD and also its use for some novel applications in patients with depression and other psychiatric disorders.
Keywords
Introduction
Attention deficit hyperactivity disorder (ADHD) is one of the most common childhood disorders with a prevalence ranging anywhere from 4%–7%.1,2,3 ADHD is being increasingly recognized as a chronic disorder that may significantly persist through adolescence into adulthood and cause functional impairment.4,5 ADHD impairs social, academic and occupational functioning and is often associated with comorbidities like oppositional defiant disorder (ODD), conduct disorder (CD), substance use disorders, anxiety and mood disorders. Multi-modal interventions, led by pharmacological measures have the best evidence for treating ADHD. Pharmacological options for treatment of ADHD include stimulant medications such as various formulations of methylphenidate, mixed amphetamine salts and non-stimulant medications such as atomoxetine. The efficacy of stimulants for treatment of ADHD is well established 6 and there is growing evidence that early and effective treatment of ADHD with stimulant medications also protects against the subsequent risk of some comorbid psychiatric disorders such as substance abuse. Improvements in academic performance have also been reported with use of such pharmacological agents.7,8
While the available stimulant medications are effective, there are concerns about potential abuse of such prescribed preparations. 9 This has led to concerted efforts to develop alternative stimulant formulations that are effective and safe, yet possess less potential for abuse. One such potential option is lisdexamfetamine (LDX), a prodrug that was approved by the FDA for treatment of ADHD in children in February 2007 and for adults in April 2008.
Mechanism of Action
LDX is an inherently inactive prodrug that contains dextroamphetamine (d-amphetamine) covalently bound to the essential amino acid L-lysine. It is broken down to d-amphetamine and l-lysine by enzymatic hydrolysis of the covalent bond. Enzymatic hydrolysis is a rate limiting step that is believed to be responsible for the decreased abuse potential and extended duration of action (Fig. 1). While initially thought to be a predominantly gastrointestinal phenomenon, more recent studies have shown that the major site for such a hydrolysis may in fact be the red blood cells. This has further intrigued researchers in the field, who are now looking at the clinical implications of this interesting finding. Like other amphetamine molecules, this active d-amphetamine moiety mediates its therapeutic effect by blocking norepinephrine and dopamine reuptake and by stimulating the release of several biogenic amines, especially dopamine, from presynaptic terminals into the extraneuronal space.
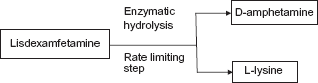
Breakdown of Lisdexamfetamine.
Efficacy, Safety and Tolerability
Currently available literature includes at least four studies in children and two trials in adults that have looked at the efficacy, tolerability of LDX in treating ADHD symptoms.
Research in children
In a phase II double blinded crossover study, Biederman et al 10 compared LDX with extended-release mixed amphetamine salts (MAS XR) and placebo in a laboratory classroom environment. This study involved 52 children aged between 6–12 years with diagnosis of ADHD hyperactive, impulsive or combined type. Following a 3 day washout period, subjects initially received MAS XR for 3 weeks, with doses titrated based on response from 10–30 mg/day over a three week period. After this, these subjects entered three cohorts for the blinded, crossover phase of the study. In each cohort, subjects received MAS XR at their effective dose, an equivalent dose of LDX or placebo for 1 week before switching to the next therapy. The cohorts differed in the doses of MAS XR (10, 20 and 30 mg/day) and LDX (30, 50 and 70 mg/day) administered. Both LDX and MAS XR demonstrated statistically significant improvements (P < 0.0001) compared to placebo in a number of outcome measures including the Swanson, Kotkin, Agler, M-Flynn, and Pelham (SKAMP) deportment rating (SKAMP-DR), attention scale (SKAMP-AS), Permanent product measure of performance (PERMP) attempted (PERMP-A) and correct (PERMP-C) and clinical global impression (CGI) scalesSKAMP-DR was the primary outcome measure and the rest were secondary measures. Adverse events reported with LDX included insomnia (8%), decreased appetite (6%) and anorexia (4%). Both LDX and MAS XR groups had a small increase diastolic blood pressure (3–5 mmHg) and pulse rate (5–7 beats/min), as compared with placebo group, but was deemed clinically insignificant. There was no incidence of prolonged QTc interval in the LDX group. Several important limitations of the study were reported by the authors including short duration, exclusion of children with psychiatric comorbidities, improved tolerability resulting from the initial titration with MAS XR and lack of provision to do a head to head trial of MAS XR and LDX.
A phase III placebo-controlled, randomized double blinded study using “forced-dose” escalation of LDX was conducted by Biederman et al to assess the efficacy and safety in a sample of 290 children with predominantly ADHD combined type, aged 6–12 years, across 40 centers. 11 After a 1-week washout period, subjects received LDX or placebo for 4 weeks. The study subjects were randomized to receive LDX 30 mg/day, 50 mg/day, 70 mg/day, or placebo. During the first week of treatment, all children who were randomized to receive Lisdexamfetamine started at 30 mg/day and those randomized to receive higher dosages were titrated up to 50 or 70 mg/day with 20-mg increments every week. Over a period of 4 weeks, the primary outcome was assessed using the ADHD Rating Scale Version IV (ADHD-RS-IV) scores. Statistically significant reductions in ADHD-RS-IV scores in the LDX group (P < 0.001) were reported when compared to the placebo group at all dosages. The decrease in ADHD-RS scores with 70 mg of Lisdexamfetamine was greater compared to the 30 mg dose. This study also looked at parental perception of symptom improvement using the Conner's Parent Rating Scale-Revised Short Form (CPRS-R). Children on LDX were reported by parents to have lesser ADHD symptoms at 10 a.m., 2 p.m. and 6 p.m. compared to the placebo group, thus indicating to the extended duration of action. Statistically significant scores were seen in all LDX goups compared to placebo using the CGI scale (P < 0.001). Reported adverse effects included decreased appetite (39% for LDX vs. 4% for placebo), insomnia (19% vs. 3%), upper abdominal pain (12% vs. 6%), headache (12% vs. 10%) and irritability (10% vs. 0%). More than 95% of these adverse events were mild to moderate in intensity and began during the first week of treatment and diminished over time. However 9% of subjects did withdraw from the trial due to side effects. Treatment with LDX was not associated with any clinically significant changes in mean EKG parameters, laboratory values, and systolic and diastolic blood pressures. There was a significant increase in mean heart rate in the LDX group when compared to the placebo group. The forced dose escalation within a short period is thought to be related to the adverse events seen. Limitations of the study include short duration (4 weeks) and lack of teacher rated scales.
The subjects enrolled in the above two studies were followed in an open-label, multicenter, single-arm study conducted by Findling et al. 12 This study involved 272 children, with an initial 4 week phase for dose titration based on response to 30, 50, or 70 mg of LDX over 4 weeks and continued on maintenance treatment for 11 months. From baseline to endpoint, mean ADHD RS scores improved by 27.2 points (P < 0.0001). Statistically significant decreases in ADHD RS scores were evident by the first week and improvements were maintained throughout the duration of the study. ADHD-RS-IV inattentive and hyperactivity-impulsivity subscale scores at end point changed by -13.4 ± 7.0 points (>60% change from baseline) and -13.8 ± 7.0 points (66% change from baseline), respectively (both, P < 0.001). Adverse events were reported in 13%, 23%, and 35% of those who received LDX 30 mg, 50 mg, and 70 mg, respectively. Insomnia and irritability, respectively, were the only psychiatric adverse events reported by more than 5% of patients treated with Lisdexamfetamine. Only one serious psychiatric adverse event, an episode of mania, was reported, but was deemed unrelated to LDX. No statistically or clinically significant changes in EKG or blood pressure were noted. Similar to other stimulants, LDX can slow growth rate and hence it is very important to monitor height and weight changes using growth charts. In terms of limitations, as the subjects were recruited as continuation from prior LDX studies it had the advantage of following patients with proven tolerability hence leading to decreased adverse events.
Wigal et al 13 conducted a 13 hour “laboratory” school, open-labeled study with 117 children aged 6–12, in which dose-optimization of lisdexamfetamine between 30–70 mg/day was done over a four week period followed by a randomized, placebo-controlled, two-way crossover phase of 1 week each. SKAMP-DR was the primary efficacy measure. Secondary measures included SKAMP-AS and PERMP-A/C scales, measured at pre-dose and at 1.5 (primary endpoint), 2.5, 5, 7.5, 10, 12, and 13 hours post-dose. LDX demonstrated significant improvements (P < 0.005 for all time points) on the SKAMP-DR/ AS and the PERMP scales compared with placebo, at 1.5 hours and continuing through all time points upto and including 13.0 hours post-dose (the last time point measured). Side effects were mainly seen in dose optimization phase and included decreased appetite (47%), insomnia (27%), headache (17%), irritability (16%), upper abdominal pain (16%), and affect lability (10%). The side effects decreased in frequency with the duration of treatment and in the cross over phase only 6%, 4%, 5%, 1%, 2%, and 0% respectively, reported the above adverse effects. Consistent with the earlier studies and similar to other stimulant medications a modest increase in blood pressure systolic/diastolic (2–4 mmHg) and pulse rate (3–6 beats per minute) was noted. QT/QTc changes were reported in 14 subjects but the authors state that none of them had an interval of >480 msec. Short duration, open label design, exclusion of psychiatric comorbidities are some of the limitations of this study.
Research in adults
Adler et al 14 conducted a randomized, double-blind, placebo-controlled, forced dose titration study involving 420 adult subjects (18–55 years) with ADHD. The subjects were randomized to receive LDX at doses of 30, 50, 70 mg or placebo for a total duration of four weeks. All the subjects randomized to receive LDX started at 30 mg/day for week 1, following that, those individuals in the 50 mg group received the 50 mg dose from week 2 onwards whereas the 70 mg group received 50 mg dose for week 2 and the 70 mg dose from week 3 onwards. The primary outcome measure used in the study was ADHD-RS with and CGI was the secondary outcome measure. Significant differences relative to placebo were observed in each LDX dosage group, with no statistical difference between medication dosages. Statistically significant differences (P < 0.0001) in ADHD-RS scores, relative to placebo were evident starting at week one and continued throughout the study period of four weeks. A significantly greater percentage of subjects showed improvement relative to placebo, with CGI-I score of 2 or less (1-very much improved; 2-much improved). There was no statistically significant differences in ADHD symptoms seen with the different doses of LDX at endpoint though the 70 mg did perform better than the 30 mg dose at weeks 3 and 4. Side effects were transient and mainly seen in the first week and included decreased sleep/appetite, dry mouth, insomnia and anxiety. In the LDX goup 6% discontinued the medication due to adverse effects as compared to 2% in placebo group. Insomnia was the most common reason for treatment discontinuation followed by treatment emergent cardiovascular adverse events, including palpitations, hypertension and dyspnea. However no statistically significant changes in blood pressure or EKG parameters was seen. Slight increase in pulse rate of subjects treated with LDX (2.8–5.2 beats per min) was noted but this was not clinically/statistically significant. Limitations of the study included short duration, exclusion of psychiatric and general medical comorbidities (such as Hypertension).
349 subjects from the above study were followed by Wiesler et al for 12 months in an open labeled multicenter study. 15 Similar to the longterm follow up results in children, improvement in mean ADHD-RS total scores which were observed at week 1 was sustained throughout the study (P < 0.0001 at all post baseline visits). At the endpoint, the mean improvement from baseline ADHD-RS total score was 24.8 (P < 0.0001). Side effects were similar to those seen in earlier study and included dry mouth, insomnia, decreased appetite and there were statistically non-significant changes in blood pressure, pulse rate. Limitations of the study include the open label design, exclusion of psychiatric and general medical comorbidities such as hypertension.
Pharmacokinetics
There are two published studies by Krishnan et al investigating the pharmacokinetics of LDX.16,17 A phase I, open label, crossover study looked at the relative bioavailability involving 18 healthy adult volunteers in whom the 70 mg dose of LDX was administered as an intact capsule on fed/ fasting state and as powder in fasting state. LDX is well absorbed after oral administration with time to reach maximal concentration (Tmax) of 1 hour whereas the Tmax of the subsequently produced d-amphetamine was 3.5 hrs indicating to additional enzymatic hydrolysis in the systemic circulation in addition to the gastrointestinal enzymatic hydrolysis. Food delayed the Tmax by 1 hour however the maximum plasma concentration (Cmax) and the plasma concentration-time curve (AUC) were similar in fed and fasting stage. Anorexia (58%), insomnia (50%), tachycardia (42%), and hyperkinesias (33%) were the most common adverse effects. No laboratory abnormalities were noted and there were no EKG abnormalities. Mean increases in systolic blood pressure ranged from 7 to 9 mmHg between 2 and 4 hours post-dose and 4 to 8 mmHg in diastolic blood pressure. 16 In another study, 70 mg of Lisdexamfetamine was administered for seven days in 12 healthy adult volunteers and blood levels were measured. It took 5 days to achieve a steady state concentration of d-amphetamine and it was not detected after 48 hours of the last dose and lisdexamfetamine was not detectable after 6 hours from the last dosing. 17 T max of LDX and the subsequently produced d-amphetamine was measured in 18 children with ADHD who received doses between 30–70 mg/day. Similar to the results in healthy adults Tmax of lisdexamfetamine was 1hour and of the d-amphetamine was 3.5 hours. 17 The pharmacokinetics of LDX 70 mg was compared to MAS XR 30 mg in children with ADHD and coefficient of variability of Tmax was 15.33% and 57.33% respectively showing the more predictable and consistent release of d-amphetamine from LDX. 18 LDX is not metabolized by the hepatic Cytochrome P450 system and hence limiting the potential for interactions with other drugs. 19 However, given that d-amphetamine is a sympathomimetic, it should not be used with other drugs with similar action and should be used carefully in patients with hypertension.
Effects on growth
In an exploratory study by Faraone et al 20 281 children being treated with LDX were followed longitudinally upto 15 months and the height, weight and Body mass index (BMI) were compared to age appropriate Center for disease control (CDC) norms. It was found that similar to other stimulants LDX can cause diminished gains in height, weight and BMI and hence it is very important to monitor these parameters using growth charts.
Abuse Liability
Since LDX is metabolised through a rate limited step to produce d-amphetamine it is thought to have less addictive potential and scope for misuse. Jasinski et al 21 conducted a double-blind, randomised, placebo- controlled, 6-period crossover study to assess the abuse potential of oral Lisdexamfetamine 50, 100, and 150 mg (equivalent to 20-, 40-, and 60-mg d-amphetamine sulfate, respectively) compared with d-amphetamine 40 mg, diethylpropion 200 mg, and placebo. Study compared the onset and intensity of “liking effects” with LDX, dextroamphetamine and placebo. For the primary measure of subjective response on the Drug Rating Questionnaire-Subject Liking Scale compared with placebo, d-amphetamine and diethylpropion showed significant differences. At 50 and 100 mg LDX did not differ significantly from placebo. The liking effects of LDX 150 mg, however, were greater than placebo and similar to dextroamphetamine 40 mg, even though the amphetamine content in LDX 150 mg was equivalent to approximately 1.5 times the dose of dextroamphetamine used. Liking effect was significantly less for the 50- and 100-mg doses of LDX when compared to 40 mg d-amphetamine, suggesting a dose dependent abuse potential. The mean peak of drug liking scores was delayed with LDX as compared to dextroamphetamine. The attenuated liking scale scores indicate that lisdexamfetamine dimesylate may be less reinforcing when compared to an equivalent dose of dextroamphetamine and abuse liability may be dose dependent. Another randomized, double-blind study with single-dose, three-way crossover design 22 was done to evaluate the safety, tolerability and abuse liability of intravenously administered LDX in 12 healthy adult volunteers with histories of stimulant abuse. Subjects received single intravenous doses of 10 mg or 20 mg immediate-release dextroamphetamine, 25 mg or 50 mg intravenous LDX or placebo at a minimum of 48-h intervals. When compared, subjects reported euphoria within 15 minutes of dextroamphetamine, whereas subjects receiving LDX did not show a statistically significant differ- ence compared to placebo in liking effects, indicating lesser abuse potential for lisdexamfetamine. Even though theoretically LDX has an advantage over other stimulants in terms of abuse potential, this need to be validated in well designed studies and in clinical practice as the above two studies involve subjects with history of stimulant misuse.
Use in Special Populations
Like any amphetamine formulation, LDX should be avoided in patients using sympathomimetics, MAOI and in comorbid conditions such as hyperthyroidism, glaucoma and severe hypertension. It is not recommended for use in pregnancy or mothers who are breast feeding. It has been mainly studied in children6–12 and adults,18–55 hence use in geriatric population is best avoided till further data is available.
Conclusions
LDX provides a unique, effective alternative pharmacological option for management of ADHD. The rate limiting enzyme hydrolysis which breaks down this prodrug into its constituent molecules, the active d-amphetamine and the essential amino acid L-lysine is responsible for the consistent and extended duration of action, reduced potential for misuse and tampering. Side effect profile is similar to other stimulant preparations and treatment emergent adverse events such as insomnia, irritability, decreased appaetite, headache, increased anxiety, palpiatations, dizziness can occur and are usually transient in nature. Growth delay is a possibility in longterm treatment with LDX and hence the need to monitor height, weight and BMI using growth charts. Family and personal history of cardiovascular problems and seizures should be elicited prior to starting and along with monitoring of blood pressure and pulse rate, EKG may need to be performed in some cases. Though the price range is similar to other extended release stimulant medications such as Concerta, Adderall XR, at present LDX use is restricted to patients who are not responding/tolerating atleast two/three other extended release stimulants as it is still a relatively new medication, there is lack of head to head trials with these agents and also due to insurance regulations. Its role in ADHD and other psychiatric disorders will be revealed as the results of the ongoing studies are available and it is used more often in real life patient population. Currently there are 39 LDX trials listed in the FDA clinical trials website 23 ranging from treatment of ADHD from the age of 6 into adulthood, to novel applications such as smoking cessation, schizophrenia negative symptoms, chronic fatigue syndrome and as an adjunct for MDD. It is also being further tested in patients with comorbid substance misuse disorders and as a potential therapeutic option for cocaine dependence.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
