Abstract
Cluster headache is a primary headache syndrome characterized by attacks of severe unilateral headache typically lasting 30 to 180 minutes without treatment and prominent autonomic symptoms on the affected side. Often attacks occur in cycles lasting weeks to months with up to 8 attacks per day, and a minority of individuals continue to experience attacks throughout the year. Persons with cluster headache usually require both acute medication for attacks and preventive treatment to keep the headaches from occurring. Subcutaneous sumatriptan is the most effective medication for acute cluster attacks, but other triptans such as zolmitriptan nasal spray are also effective. inhaling 100% oxygen is also effective and is a useful treatment for those with frequent attacks or contraindications to triptans. Corticosteroids are among the most effective transitional treatments, typically used at the start of a cycle. Dihydroergotamine is an effective treatment for refractory or severe cluster headache with multiple attacks requiring large triptan doses. Verapamil and lithium are among the most effective preventive medications with good evidence of effectiveness, but other studies support the use of gabapentin, topiramate, diavalproex sodium, and methysergide, to name a few. Each of these medications requires monitoring for adverse events and can be discontinued within a few weeks of a cluster headache cycle.
Introduction
Cluster headache (CH) is a primary headache disorder. It is the most common of the trigeminal autonomic cephalalgias. CH is characterized by severe, unilateral headache that usually lasts 15 to 180 minutes and is associated with autonomic symptoms. The 2nd edition of The International Headache Classification (ICHD-2) criteria 1 defines CH as shown in Table 1.
ICHD-2 diagnostic criteria for CH.

CH is further subdivided into episodic and chronic type patterns. Episodic cluster is defined as headache meeting the ICHD-2 criteria for CH, with attacks lasting 7 days to 1 year and temporal separation of greater than 1 month between attacks. Patients with chronic cluster have attacks that occur for more than a year without remission of greater than 1 month. 1 The majority of CH falls within the episodic definition, but many patients have headaches that transform between the two subtypes. As with other headache disorders, individual attacks can occur spontaneously or with associated triggers. Common triggers identified in CH include alcohol, lack of sleep, histamine, REM sleep, nitroglycerin usage, elevated temperature, exercise, or strong odors.
CH is one of the most painful headache or pain disorders seen in clinical practice, and aggressive treatment with acute and preventive medication is indicated in most cases. This paper provides an overview of CH as it is currently understood and a detailed explanation of its pharmacologic treatment.
Epidemiology
CH is estimated to have prevalence of 0.3% in the general population. Headaches typically occur within the second to fourth decades of life, with a 2.5:1 male to female predominance, 2 but they can start at any age. CH is more common in smokers, and though the majority of cases are sporadic, there does seem to be a genetic factor, with CH approximately 5 to 18 times more common in first-degree relatives than in non–-first-degree relatives.
Pathophysiology
The pathophysiology of CH is incompletely understood, but given its epidemiology and characteristic headache patterns, it is likely consequent to a combination of genetic, environmental, and chemical components. Specific anatomical areas within the nervous system have been implicated in CH, with notable involvement of the hypothalamus and trigeminovascular system. Positron emission tomography (PET) imaging has shown ipsilateral activation of the posterior hypothalamus during CH attacks, and other studies have demonstrated its modulation of the trigeminal nucleus caudalis. 3 On voxel-based morphometry, CH patients have increased hypothalamic volume compared with controls. 4 Other areas activated include the areas involved in pain matrix such as the limbic system. Chronic activation of the anterior cingulate cortex is more common in those with chronic CH. 5
Circadian rhythm abnormalities, melatonin, and perhaps testosterone are also implicated within cluster pathology, thus giving further support to hypothalamic and neuroendocrine system involvement. Studies have shown decreased levels of melatonin during cluster attack periods, 6 and overall lower melatonin levels in cluster patients compared with controls. 7 The common occurrence of CH within the first few hours of sleep would seem to be congruent with close association to circadian rhythm. Testosterone levels are low in some CH patients during cycles, 8 and treatment of low testosterone may improve CH attack frequency. 9 CH attacks are frequently nocturnal, and patients may awaken from sleep with attacks. Contrary to previous belief, there is no association with CH and any particular stage of sleep such as rapid-eye movement sleep. 10
Vasoactive neuropeptides have a key role in the pathogenesis of CH, with known involvement of these peptides within the trigeminovascular system. 11 A study of CH patients that sampled ipsilateral external jugular blood levels of neuropeptides during a cluster attack found increased levels of calcitonin gene related peptide (CGRP) and vasoactive intestinal polypeptide (VIP) during acute attacks. 12 Both substance P and CGRP have been shown to be present in trigeminal sensory fibers, with evidence that they are released at peripheral sites. 13 This may explain the cerebral blood vessel dilation that is seen during attacks. Both parasympathetic and sympathetic activation activity are noted during CH attacks, explaining autonomic symptoms such as congestion, swelling, ptosis, and lacrimation. 14 Modulation of the sphenopalantine ganglion is an emerging treatment for refractory CH. 15
Pharmacological Agents
Trials investigating pharmacotherapeutic options for CH have been relatively few compared to those investigating medication treatment for other neurologic disorders and have often been limited to open-label pilot studies with relatively small numbers of participants. Many studies took place prior to adoption of standardized classification for CH, and outcome measures vary from study to study. Despite this limitation, pharmacologic treatment of CH has continued to build, with evidence for specific therapeutic agents. Pharmacologic treatment can be divided into acute treatment, that is, treatment used to abort a specific headache and preventive treatment, which is usually given at the onset of the cluster cycle to decrease the number or severity of attacks.
Abortive Pharmacologic Agents
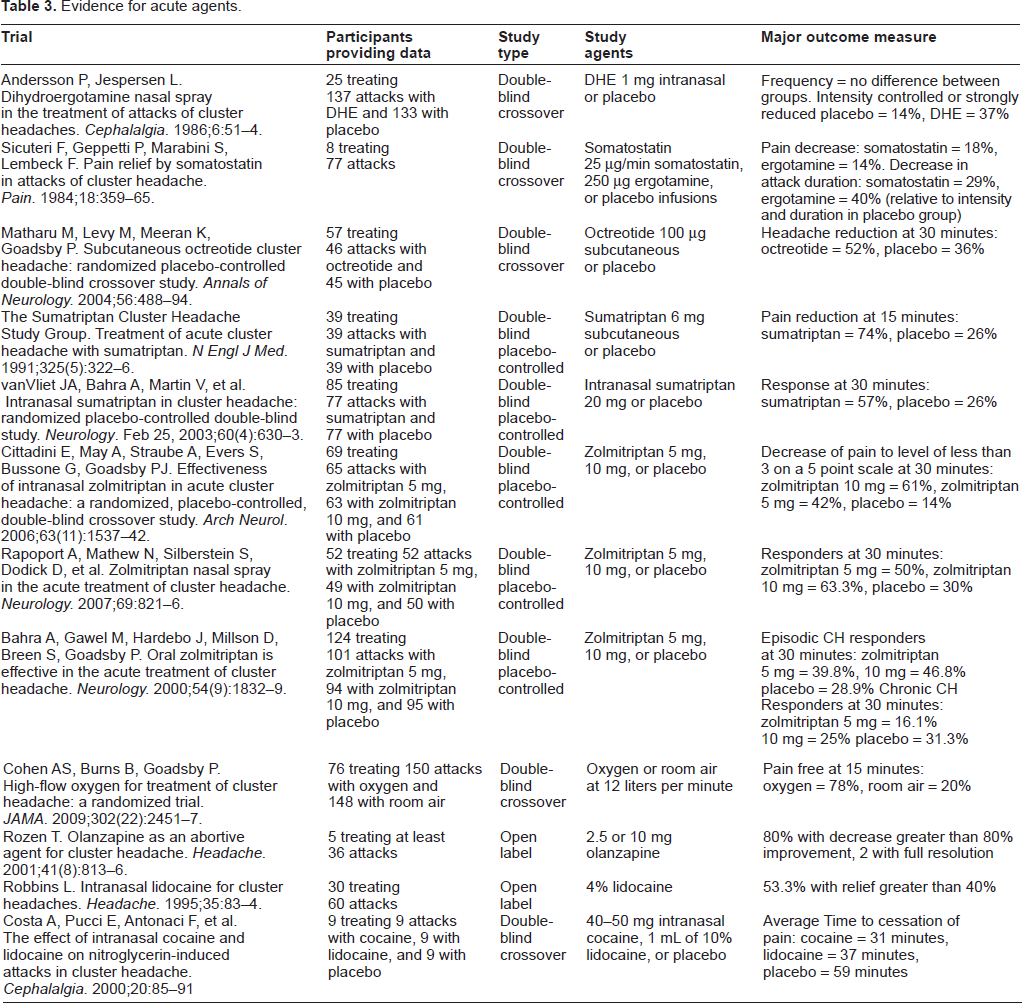
Acute CH medication should generally be rapid-acting, given the relatively sudden onset of headache pain and the short attack duration. Many acute migraine medications, such as nonsteroidal antiinflammatory drugs or neuroleptics, have limited use in CH treatment. Oral medications, including triptans, may be ineffective for patients with short-lasting CH. Sumatriptan injection is the only medication with US Food and Drug Administration approval for CH treatment. An overview of acute treatments useful in CH is provided in Table 2, and an overview of trials included in analysis is available in Table 3.
Acute medications in cluster headache.
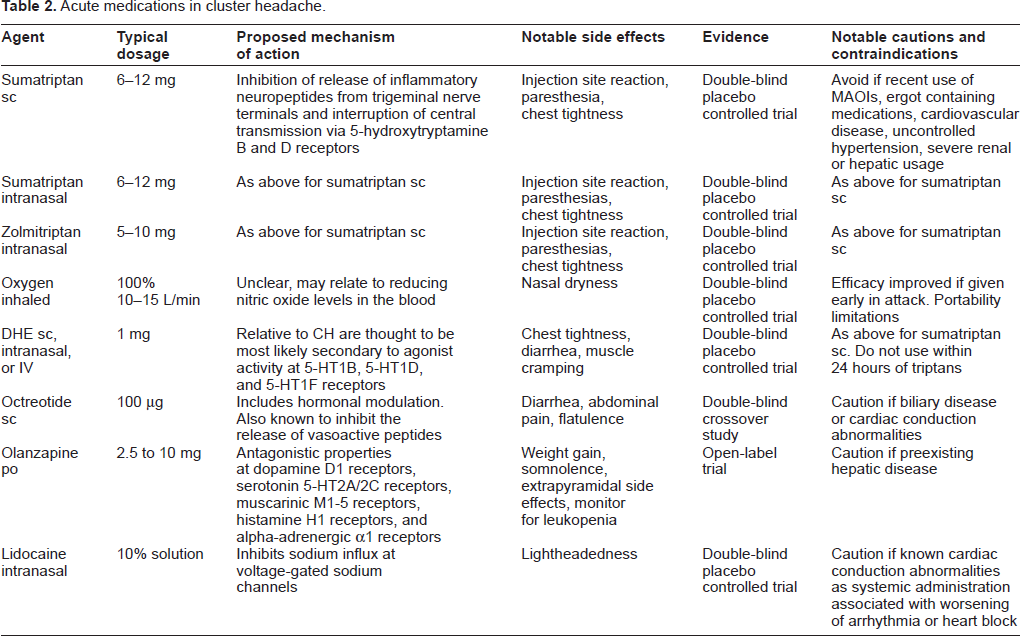
Evidence for acute agents.
Sumatriptan and zolmitriptan
The 5-hydroxytryptamine B and D (5-HT1B/D) agonists, known as triptans, have been shown to be effective in treatment of both migraine and CH. The entire mechanism by which triptans reduce headache pain likely involves both central and peripheral mechanisms of action. 16 Peripheral effects include inhibition of release of inflammatory neuropeptides such as CGRP from trigeminal nerve terminals, while central mechanisms include interruption of transmission from the trigeminal nerve to afferent areas such as the trigeminal nucleus caudalis via 5-hydroxytryptamine B and D receptors. Several trials have shown effectiveness of specific triptans for treatment of CH.
Sumatriptan
A randomized, double-blind, placebo-controlled study involving 49 participants investigating the efficacy of 6 mg subcutaneous injections in CH patients was published in 1991. 17 Participants rated initial headache pain on a scale of 0 to 4 and were instructed to treat headaches for which the pain score was greater than 2. Participants alternated treatment between sumatriptan and placebo. The primary outcome measure was pain reduction 15 minutes after injection. Seventy-four percent of sumatriptan-treated attacks were reduced from a pain level between 2 and 4 to a pain level between 0 and 1 within 15 minutes, with placebo reducing pain to these levels in only 26% of attacks. Though not the primary outcome measure, it was noted that 46% of sumatriptan-treated attacks were resolved at 15 minutes compared with 10% of placebo-treated attacks. Intranasal administration of sumatriptan also has demonstrated efficacy for CH in a randomized, double-blind, placebo-controlled trial that built upon prior open-label studies. In a trial published in 2003, van Vliet et al treated 154 cluster attacks in 85 patients with either placebo or intranasal sumatriptan 20 mg to evaluate headache response at 30 minutes. 18 Secondary outcome measures of pain-free rates, relief from associated symptoms, and adverse events were also considered. Attacks were treated at least 24 hours apart, alternating between triptan and placebo, and pain assessment was ascertained at 30 minutes from administration. Equal numbers of attacks were treated with sumatriptan and placebo; pain relief was obtained in 57% of triptan-treated attacks compared with 26% in placebo-treated attacks. The percent of attacks rendered pain-free at 30 minutes was 47% for triptan and 18% for placebo.
Zolmitriptan
Zolmitriptan nasal spray was evaluated in two randomized, placebo-controlled, double-blind, crossover studies published in 2006 and 2007. In 2006, Cittadini et al presented data from a trial of 69 participants with treatments of zolmitriptan 10 mg, zolmitriptan 5 mg, or placebo. 19 Participants rated pain on a 5-point scale prior to treatment and at 5, 10, 15, and 30 minutes after treatment. The primary outcome measure was headache response at 30 minutes, which was defined as a decrease from a pain scale greater than or equal to 3 to a level of less than 3. A total of 65 attacks were treated with 5 mg spray, 63 with 10 mg spray, and 61 with placebo. Sixty-one percent of patients reported headache relief at 30 minutes with 10 mg spray compared with 42% treated with 5 mg spray, and 14% treated with placebo. Subsequently, in 2007, Rapoport et al published a study with 52 patients treating 151 cluster attacks. 20 Similar to the 2006 trial, participants were administered zolmitriptan 10 mg, zolmitriptan 5 mg, or placebo spray, with primary outcome of headache response at 30 minutes. A secondary outcome measure was resolution of pain at 30 minutes. Of attacks treated with zolmitriptan 10 mg, 63.3% were reduced at 30 minutes, and 46.9% fully relieved, zolmitriptan 5 mg provided response in 50% of attacks and resolution in 38.5%. Placebo gave either partial or full relief in 30% of attacks, with full resolution in only 20% of attacks at 30 minutes.
Oxygen
Oxygen therapy has long been used successfully for cluster attacks. The mechanism of action that is responsible for this is unclear, but it may relate to reducing nitric oxide levels in the blood. 21 Although it had previously been used for CH, initial trial-based evidence came from a 1981 study that showed that oxygen was an effective treatment in a comparison against ergotamine therapy. 22 Since then, two additional trials have provided evidence that support the use of oxygen for cluster attacks. A randomized, double-blind, placebo-controlled, crossover study of 19 patients treated between 1981 and 1982 compared treatment of acute attacks with 6 L oxygen to treatment with room air and reported significant decrease in pain when CH was treated with oxygen. 23 This was followed by a more recent placebo-controlled, double-blind, crossover study comparing 100% oxygen delivered to patients at 12 L/minute for 15 minutes during a cluster attack versus room air. 24 The study population was 109 patients aged 18 to 70 years with a cluster diagnosis. Exclusion criteria included chronic migraine, pregnancy or lactation, chronic obstructive pulmonary disease, inability to tolerate oxygen mask, or previous failure of oxygen usage. Of the 109 patients, 76 were treated with oxygen and 148 with room air. The primary outcome measure was freedom from pain at 15 minutes from initiation of treatment. Seventy-eight percent of patients treated with oxygen were rendered pain-free at 15 minutes compared with 20% treated with high flow room air.
Dihydroergotamine (DHE)
Also used as a preventive agent, DHE has been evaluated for treatment of acute CH attacks. In 1986, Andersson et al published a double-blind, placebo-controlled, crossover trial of 25 participants evaluating response to intranasal DHE. 25 Baseline frequency and intensity were evaluated during an 8-day run-in period, with subsequent treatment of up to eight acute attacks with either 1 mg intranasal DHE or placebo prior to crossover. One hundred thirty-seven attacks were treated with DHE and 133 with placebo. Subsequent to treatment, patients rated the attacks as controlled, strongly reduced, slightly reduced, or unchanged. Ten DHE-treated attacks were rated controlled, 41 strongly reduced, 46 slightly reduced, and 40 no change. Ratings for placebo treatments were 7, 12, 29, and 85, respectively.
Octreotide
Octreotide is an 8-peptide analogue of somatostatin. It has a longer half-life than somatostatin, which allows it to be administered subcutaneously, while somatostatin is limited to intravenous (IV) use. 26 Evidence for octreotide as a treatment for CH builds upon previous studies of somatostatin that showed clinical efficacy in both migraine and CH. Mechanisms of action for the two peptides are thought to be similar and include hormonal modulation and effects on the gastrointestinal system. Similar to triptans, they are known to inhibit the release of vasoactive peptides such as CGRP and VIP.27,28 These agents provide benefit to patients in whom triptans or DHE are contraindicated due to their vasoconstrictive effects. Efficacy for somatostatin in CH is primarily derived from a 1984 investigation of eight hospitalized CH patients. 29 This double-blind, placebo-controlled study investigated therapeutic difference in patients administered somatostatin 25 μg/minute, ergotamine 250 μg, or placebo infusions. Each participant received each treatment three times and reported pain intensity at 10-minute intervals subsequent to treatments. Pain intensity was decreased 18% with somatostatin and 14% with ergotamine. Duration of symptoms was decreased by 29% with somatostatin and 40% with ergotamine, with all measures showing significant difference compared with placebo. Twenty years subsequent to this study, in 2004, a trial was conducted in which subcutaneous octreotide 100 μg versus placebo was investigated in a randomized, placebo-controlled, double-blind, crossover study of 57 participants. 30 The primary outcome measurement was headache reduction at 30 minutes, and secondary outcome measures were percentage of headache with meaningful relief, relief of associated symptoms, time to relief of headache, and percent with headache resolution at 30 minutes. Forty-six attacks were treated with octreotide and 45 with placebo; 52% of headaches treated with octreotide and 36% of attacks treated with placebo had relief at 30 minutes. Regarding secondary outcome measures, 37% of patients treated with octreotide found meaningful relief compared with 29% treated with placebo. Octreotide-treated headaches showed improvement in associated symptoms at a higher rate than those treated with placebo, and 33% of octreotide-treated headaches had resolution at 30 minutes versus 13% of placebo-treated headaches.
Olanzapine
Olanzapine is an antipsychotic drug with antagonistic properties at multiple receptors, including dopamine D1 receptors, serotonin 5-HT2A/2C receptors, muscarinic M1-5 receptors, histamine H1 receptors, and alpha-adrenergic α1 receptors. 31 Due to its antagonistic properties on dopamine receptors and prior open-label studies showing effectiveness of chlorpromazine, olanzapine was evaluated as an abortive agent for CH in an open-label trial.32,33 Five patients with previous failure of other abortive medications were selected and instructed to take 5 mg tablets for CH with the option to increase the dosage to 10 mg if they did not obtain complete headache relief or to decrease the dosage to 2.5 mg if they experienced side effects. All achieved pain reductions of greater than 60%, and 40% experienced headache resolution after olanzapine administration.
Intranasal Lidocaine
Intranasal lidocaine has been investigated as a possible adjuvant to cluster treatment, the theory being that it could block nociceptive impulses from trigeminal nerve endings. An open-label trial involving 30 patients was performed in the 1990s to investigate this treatment for CH. 34 Study participants were administered intranasal 4% lidocaine solution ipsilateral to the headache. Of the 30 patients, 14 had no headache relief, 8 obtained mild relief, 8 described moderate relief, and none reported excellent headache relief. Mild relief was defined as headache reduction of 20% to 40%, and moderate relief was defined as a 40% to 60% reduction. Subsequent to this study, Costa et al conducted a double-blind, placebo-controlled trial that compared 40 to 50 mg of intranasal cocaine, 1 mL of 10% intranasal lidocaine solution, and saline control administered bilaterally for headaches induced by 0.9 mg of trinitrine. 35 Data were collected from 9 participants who had headaches induced to less than 2 days apart on three separate occasions, with administration of each agent on separate occasions. Primary outcome measurement was complete cessation of pain. Time to complete cessation of pain measured 31 ± 12 minutes for cocaine, 37 ± 8 minutes for lidocaine, and 59 ± 12.3 minutes for saline. There was no statistical difference between response to lidocaine and cocaine.
Transitional Therapy
Most preventive treatments require days if not weeks for therapeutic effect to fully develop. Transitional therapy can be given concomitantly with preventive therapies to decrease the severity of cluster attacks in the interim between initiation and efficacy of preventive therapy. Major transitional therapies include both systemic steroids and local infusions administered to block nociceptive impulses from the greater occipital nerve (GON).
Corticosteroids
Evidence for use of systemic steroids as transitional treatment for CH has mostly been limited to open-label trials. An early study in 1952 showed that patients treated with 100 mg cortisone had little improvement. 36 Subsequent to that study, multiple additional investigations with a double-blind, controlled design have been performed. 37 The most robust studies have been limited to open-label design with control methods limited to run-in periods and retrospective comparison with previous cluster periods. The aforementioned double-blind trial took place in the 1970s and involved administering either prednisone or placebo to 19 study participants, with prednisone administered as a 30 mg dose at the first sign of a headache and as a 20 mg maintenance dose every other day. The study indicated that patients significantly improved with the prednisone, although the length of dosing intervals was unclear. Subsequently, a retrospective study reviewed cases of 19 cluster patients treated at a headache clinic in 1976 and 1977 with varying peak doses of prednisone from 10 mg to 80 mg daily tapered over 10 to 30 days. Eleven of the 19 patients had full resolution of the headache, and 14 of the 19 patients had greater than 50% improvement. 38 In 2003, methylprednisolone was evaluated for efficacy in an open-label trial of 14 episodic cluster patients. The primary outcome measure was defined as a decrease in CH frequency. Subjects were administered methylprednisolone 250 mg IV daily for 3 days, followed by 10 days of prednisone 90 mg daily tapered over 4 weeks. 39 For control data, information from previous episodes of CH in the study participants was reviewed with attention paid to number of headache days. No particular agent was used for treatment of prior episodes, although comparison could possibly be inferred as methylprednisolone versus usual treatment. In comparing headache days in the month following methylprednisolone treatment to the retrospective treatments, the treatment group had 5.86 headache days compared with 15.29 headache days during prior cluster periods. Intravenous steroid administration was investigated in a 2004 publication evaluating the efficacy of methylprednisolone with a single dose of 30 mg/kg. 40 Thirteen patients were administered the methylprednisolone following a seven-day run-in period. Headache frequency from the run-in period was compared with the 7 days following treatment. After treatment, there were no attacks for 2 days in any of the patients, with 3 of the patients experiencing no further attacks during the entire follow-up period. Of the 10 patients who did experience attacks, there was no difference compared with the run-in period in overall number of attacks despite the relief observed during the first 2 days. When patients who had full resolution of attacks were included in the analysis, there was a significant difference, with a decrease from a mean number of 35 attacks to a mean of 27 attacks. Corticosteroids are contraindicated in persons with systemic infections or significant healing wounds and should be used only with caution in those with hepatic impairment, severe depression, and medical illnesses such as diabetes, hypertension, and peptic ulcers. The use of stress ulcer prophylaxis, usually with proton pump inhibitors, and a low salt, high potassium diet may reduce adverse events.
Greater occipital nerve injections
GON block has been investigated as a therapy for CH with known input of both cervical and trigeminal neurons onto the trigeminal nucleus caudalis. Goadsby et al found that stimulation of the GON produced increased metabolism in the upper cervical horn cells at levels of C1 and C2 and in the trigeminal nucleus caudalis. 41 Malick et al demonstrated afferent projections of the trigeminal nerve to both the trigeminal nucleus caudalis and dorsal horn cells at C1, with additional effect projecting to the hypothalamus. 42 These data, along with data from clinical trials showing improvement in CH with GON block, support the theory that pain modulation in cluster headache is mediated by both trigeminal and GON nerves.
Blockade of the GON can be induced by local anesthetics exerting reversible inhibition of voltage gated sodium channels, leading to decreased conduction through nerve fibers. Blockade is most easily induced in the unmyelinated C fibers that account for nociceptive input. Longer-lasting effects of peripheral nerve injections are obtained by adding corticosteroids to the injection solution; however, this may produce sleep disturbances and other side effects consistent with systemic steroid administration. Local steroid injections may produce alopecia and thinning of skin around the injection site. 43
Efficacy for GON injections has been demonstrated in open-label studies, double-blind trials, and recently reviewed in a relatively large retrospective case series. Initial evidence for these injections was reported in a 1985 study that investigated injecting the GON ipsilateral to the site of attacks in 20 patients with 2% lidocaine and 120 mg methylprednisolone. 44 This initial trial showed significant improvement in both episodic and chronic CH, with headache-free periods ranging from 5 to 129 days following 33 of 37 injections. Subsequently, Peres et al assessed reduction in headache frequency and increase in headache-free days in 14 patients using ipsilateral GON injections with 3 mL of 1% lidocaine and triamcinolone 40 mg. 45 Headache frequency and intensity were assessed before and after injection, with response classified as good if participants were rendered headache-free for at least 14 days, moderate if headache-free for less than 2 weeks, or none if no headache-free days were observed. An average of 13.1 headache-free days were observed after injection, and a response of good to moderate was observed in 9 participants, with the remaining 5 participants demonstrating no response. In 2005, a double-blind, placebo-controlled trial by Ambrosini et al evaluated single, ipsilateral nerve injections using 13 treatment and 10 control participants. 46 Subsequent to a 1-week run-in period, participants were administered either 12.46 betamethasone dipropionate and 5.26 mg betamethasone disodium phosphate with 0.5 mL of 2% xylocaine or 2% xylocaine and saline. Participant follow-up occurred at 1 and 4 weeks, with response measured as headache resolution within 72 hours of injection, lasting until follow-up at 1 week. Eleven of the 13 participants in the treatment group were classified as responders at 1 week, and 8 of the 13 remained headache-free at the 4-week follow-up. There was no response in the placebo group. In 2006, Afridi et al published a trial of GON injections, using 3 mL of 2% lidocaine and 80 mg methyl prednisolone in primary headache patients. 47 Nineteen of the 101 patients in the trial fell within the subset of CH. Efficacy in this trial was defined as complete pain resolution, pain decreased by >30%, or no response. Of the cluster subset, 10 had complete relief of pain, 3 had partial relief, and 6 were nonresponders. Mean duration of the complete response was 17 days and of partial response 52 days. A larger double-blind, placebo-controlled trial of 43 CH patients was published by Leroux et al in 2011. 48 They investigated GON injections as transitional treatment combined with verapamil as preventive treatment. Both injection groups of cortisol and placebo were administered, with verapamil escalated to 720 mg daily as tolerated for preventive therapy. Participants in the treatment group (n = 21) were administered 3.75 mg of cortisol in each of three sequential injections spaced 48 to 72 hours apart. Of this group, 20 subjects had a mean of two or fewer mean daily attacks during follow up 2 to 4 days after the third injection, compared with the control group (n = 22), which demonstrated only 12 participants with 2 or fewer mean attacks during the follow-up period. Regarding secondary endpoints, the participants in the treatment group were observed to have fewer attacks than controls during the 15 days following initial injection, with a mean number of attacks of 10.6 (95% CI, 1.4–19.9) and 30.3 (95% CI, 21.4–39.3), respectively. Gantenbein et al conducted a retrospective analysis of 121 mixed-duration betamethasone injections in 60 CH patients seen between 2006 and 2009. 49 Overall, 54 of the injections were associated with a complete response (no further attacks), and 42 were associated with a partial response, defined as reduction in attack frequency, duration, or severity. The authors noted side effects in 18 patients, including injection site reactions, sleep disturbance, facial edema, ocular dysesthesia, acne, heartburn, esophageal candida, transient bradycardia, and 1 episode of syncope.
Dihydroergotamine (DHE)
Also discussed as both a preventative and abortive agent, DHE can also be used as a transitional treatment that can be readily administered in the inpatient setting. Evidence for this was detailed in a study published in 2011 which reviewed patients with refractory primary headaches who had undergone IV DHE treatment. 50 Within the study population of refractory headache patients, there was a subgroup of 38 CH patients. Thirty-two of the CH patients reported freedom from headache during DHE administration, but remittance was short-lived. Patients reported a mean time to return of CH of 17 days and a mean time to return to pretreatment frequency of 66 days suggesting that IV DHE treatment alone can provide brief reprieve from CH but that ongoing treatment with other agents is necessary.
Preventative Pharmacologic Agents
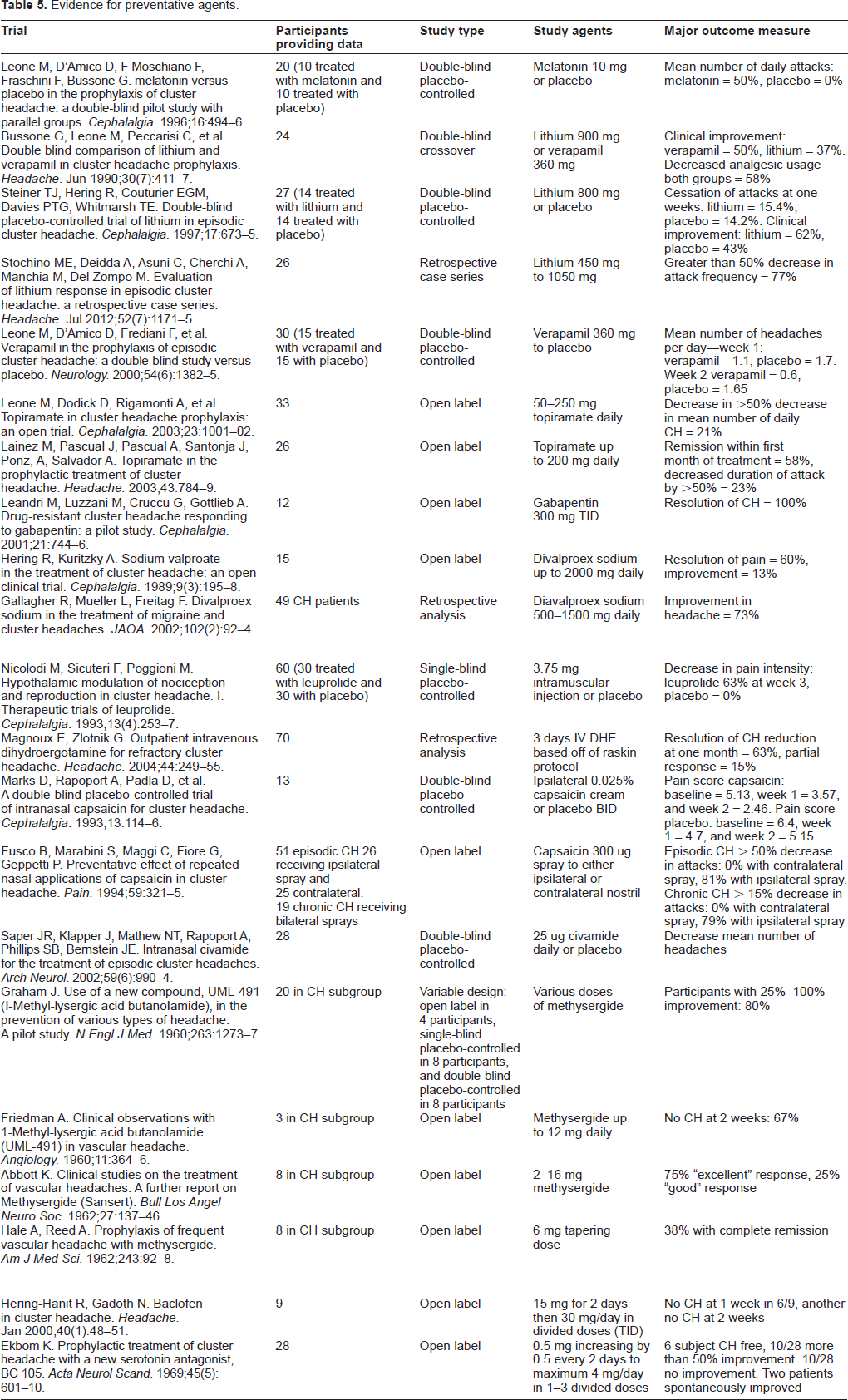
Given the severity of CH, most patients with cycles lasting more than a few weeks require preventive treatment. Episodic CH patients with predictably short-lasting cycles that respond to short-term corticosteroid use and acute medication may elect to avoid preventive treatment, but prolonged corticosteroid use may have serious adverse events. A complete list of preventive agents is provided in Table 4 and an overview of trials included in analysis is available in Table 5.
Preventive medication in cluster headache.
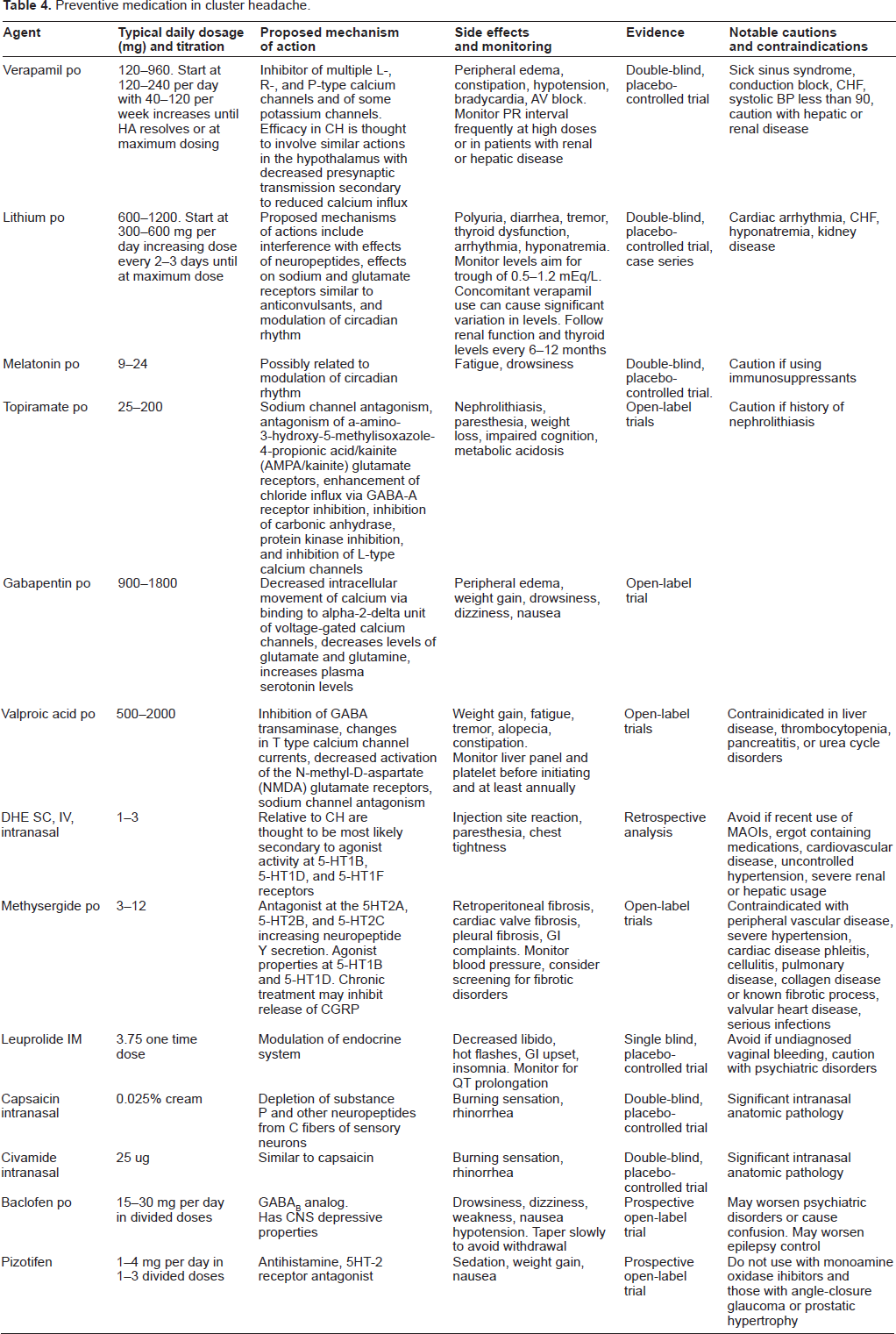
Evidence for preventative agents.
Verapamil
Verapamil is a known inhibitor of multiple L-, R-, and P-type (and possibly other) calcium channels. 51 Additionally, it has been shown to be an inhibitor of some potassium channels. The exact mechanism accounting for efficacy in the treatment of CH is unknown, but it is thought to involve actions at these channels in the hypothalamus or elsewhere, with decreased presynaptic transmission secondary to reduced calcium influx. Pharmacologically, doses of verapamil used for treating CH are often much higher than doses used for cardiac purposes, likely due to exit from the central nervous system via efflux transporter P-glycoprotein within the blood brain barrier. Due to the high doses needed and known adverse events including atrioventricular block, an electrocardiogram (ECG) with close attention to the PR interval should be obtained both prior to treatment and with dose escalation. Bradycardia and hypotension are also common. The incidence of significant bradycardia is greatly increased in patients treated concomitantly with lithium, and routine monitoring of ECG and lithium levels is necessary in this population. Verapamil is a CYP3A4 inhibitor; it has many drug interactions. Careful attention must be paid to this, particularly with regard to statins as the combination can elevate statin levels increasing the risk of myopathy and rhabdomyolysis. Dosing for CH can start at 120 to 240 mg per day with 40 to 120 mg per week increases until at maximum dosing. Subsequent to open-label studies and a trial by Bussone et al published in 1990, which showed efficacy for both verapamil and lithium, 52 Leone et al conducted a double-blind, placebo-controlled study with lithium 360 mg daily. 53 Published in 2000, the trial compared a treatment group (n = 15) to a placebo group (n = 14) for a total of 14 days subsequent to a drug-free 5-day run-in period. During the first week of treatment, there was no significant difference between the number of headaches in the treatment and placebo groups (1.1 ± 1.02 vs. 1.7 ± 1.12, respectively); however, by the second week a significant difference was noted, with 0.6 ± 0.88 headaches in the verapamil group compared with 1.65 ±1 in the placebo group.
Lithium
Lithium's exact mechanism of action in CH is unclear. There are several proposed mechanisms of action, including possible interference with the effects of neuropeptides, such as VIP. 54 Other possibilities that may account for therapeutic benefits of lithium include effects on sodium and glutamate receptors similar to anticonvulsant-type medications and possible modulation of circadian rhythm. Current evidence for lithium's efficacy in CH follows from open-label trials, retrospective case series, and two randomized controlled trials. In the initial controlled trial involving lithium, Bussone et al compared the efficacy of lithium 900 mg daily to verapamil 360 mg daily in a double-blind, crossover study enrolling 30 participants. 50 The study protocol consisted of 2-week washout periods both initially and interspaced between crossover from 8-week treatment periods of the two different agents. Twenty-four participants completed the study, with 6 dropouts occurring at the initial washout period. Clinical efficacy for decreased headache and analgesic usage was shown in both treatment groups. Subsequently, in a 1997 publication, Steiner et al reported a double-blind, placebo-controlled study composed of 27 episodic cluster patients divided into a lithium 800 mg daily treatment group (n = 13) and a placebo group (n = 14). 55 This study's outcome measure of patients with cessation of attacks after 1 week of treatment did not show significant difference between treatment and placebo, but may have been limited by the short follow-up of the study. More recently, a retrospective case series evaluated the response to lithium 450 mg to 1050 mg daily in 26 episodic cluster patients between 2002 and 2009. 56 Analyzing 3 weeks per patient, attack frequency was evaluated 1 week pretreatment and during 2 subsequent weeks of lithium treatment. Participants were classified as either responders (n = 20) if they had a greater than 50% decrease in attack frequency or as nonresponders (n = 6) if they did not. Responders had a decrease in mean attack frequency from baseline 1.8 ± 0.9 attacks per day to 1.1 ± 1 and 0.6 ± 1 during the 2 respective weeks of treatment. Seventeen study participants established resolution of CH by the end of 2 weeks of treatment. In typical usage for CH, dosing begins at 300 to 600 mg daily with increases every 2 to 3 days until at maximum dosing. Monitoring of serum levels should be followed during dose escalation to avoid toxicity. Additionally, renal and thyroid function should be checked at initiation of therapy and at least annually thereafter.
Melatonin (N-acetyl-5-methoxytryptamine)
An indolamine derivative of the amino acid tryptophan, melatonin has a structure similar to serotonin. It is secreted from the pineal gland and is significant in maintenance of circadian rhythm. Subsequent to secretion, melatonin is capable of crossing the blood-brain barrier and is broken down in the liver into 6-hydroxymelatonin and excreted in the urine as 6-sulphaoxymelatonin. 57 Its use in CH is relative to implicated pathophysiology of the neuroendocrine system. Studies have demonstrated decreased levels of melatonin in both smokers and men, which is consistent with epidemiological characteristics of CH and gives further basis for its usage. 58 In a 1996 publication, Leone et al investigated melatonin supplementation in 20 cluster patients in a double-blind, placebo-controlled study. 59 After an initial 1-week run-in period, participants were followed over 2 weeks with either daily melatonin 10 mg or placebo administration, with primary outcome measures of mean number of daily attacks and analgesic consumption. With significance obtained in showing decrease in number of mean daily headaches, however, response was only noted in patients suffering from episodic CH.
Topiramate
Topiramate has multiple mechanisms of action, including sodium channel blockade, antagonism of a-amino-3-hydroxy-5-methylisoxazole-4-propionic acid/kainite (AMPA/kainite) glutamate receptors, enhancement of chloride influx via GABA-A receptor inhibition, inhibition of carbonic anhydrase, protein kinase inhibition, and inhibition of L-type calcium channels.60,61 The use of topiramate in CH has been investigated in several open-label trials and case series. 62 The largest open-label trial of topiramate involved 26 episodic and 10 chronic CH patients. 63 Subsequent to a 7-day run-in period, participants were administered 25 mg twice daily, increasing as tolerated, with a final dose range of 50 mg to 250 mg per day. Thirty-three patients completed 20 days of topiramate therapy, with 7 patients experiencing a greater than 50% decrease in headache by the end of the study compared with the run-in period. No significant difference was observed in mean number of headaches per day between the run-in period and the treatment period. Another open-label trial of 26 participants titrated from an initial dose of 25 mg up to a maximum dose of 200 mg per day as tolerated was performed. 64 Remission occurred in 15 patients during the first month of treatment, with mean time to remission 14 days, with other patients reporting a decrease in number of attacks. Average attack duration was also decreased.
Gabapentin
Originally developed as an analogue of gamma-aminobutyric acid (GABA), gabapentin is not believed to actually interact with neuronal GABA receptors. 65 Identified mechanisms of action include binding to alpha-2-delta unit of voltage-gated calcium channels to decrease the intracellular movement of calcium in addition to modulation of levels of neurotransmitters. 66 Specifically, gabapentin has been shown to decrease levels of the excitatory neurotransmitters glutamate and glutamine in addition to increasing plasma serotonin levels. Evidence for use of gabapentin in CH is derived from a small, open-label trial of 12 patients. 67 Participants were administered gabapentin 100 mg three times daily, with titration upward to 300 mg three times daily. Treatment duration was either 60 days in patients with episodic CH or 6 months in chronic CH patients. All 12 patients reported full headache resolution by day 8 of gabapentin treatment.
Divalproex sodium
Divalproex sodium is a branch chain fatty acid made up of valproic acid and valproate sodium. It is known to increase GABA levels due to inhibition of GABA transaminase, resulting in decreased GABA concentrations. Other known effects include changes in T-type calcium channel currents, decreasing activation of the N-methyl-D-aspartate (NMDA) subtype of glutamate receptors, and sodium channel antagonism. 68 Measures of divalproex sodium's effectiveness in CH has been limited to open-label trials. Hering et al investigated divalproex sodium in 15 CH patients, with initial treatment of 600 to 1000 mg, with doses increased to a maximum of 2000 mg daily as tolerated. 69 Treatment duration was individualized to each patient and lasted the estimated duration of their previous CH periods. The authors report that 11 of the 15 patients had response to divalproex sodium with decreased length of cluster period and complete resolution of cluster period for 9 of the 15 patients. The decrease in duration of the cluster period ranged from 1 to 5 months. Subsequent to this initial study, Gallagher et al conducted a retrospective review of 284 patients treated for either migraine or CH. 70 Patients had been treated with divalproex sodium at daily doses ranging from 500 to 1500 mg as either monotherapy or in combination with other preventive agents. Forty-nine of the of the 284 patients were being treated for CH. Thirty-six of the CH patients showed improvement on divalproex sodium, although it should be noted that only 11 of the 36 were treated with monotherapy.
Leuprolide
CH is associated with involvement of the hypothalamus and derangement of the endocrine system. Leuprolide is a slow-release gonadotropin-releasing hormone (Gn-RH) analogue. The investigation of leuprolide for CH occurred in a single-blind, placebo-controlled trial of 60 male patients given either 3.75 mg intramuscular leuprolide or placebo injection following a two week run-in period. 71 Participants were then followed for 8 weeks, with follow-up time subdivided as 4 weeks of active treatment and 4 weeks washout. Decrease in CH attacks and pain intensity over 10-day intervals were the predefined outcome criteria, with leuprolide showing a 63% decrease in CH intensity during the third 10-day period, in addition to decreased mean number of attacks.
Dihydroergotamine (DHE)
Originally developed as an antihypertensive agent and later found effective for CH, DHE is a semisynthetic hydrogenated ergot alkaloid differing from ergotamine at a reduced, single chemical bond. 72 Compared with ergotamine, the reduced bond yields a compound with greater activity as an alpha adrenergic antagonist, less vasoconstriction, and generally fewer side effects relating to the gastrointestinal system. Both ergotamine and DHE have activity at numerous receptors inclusive of agonist activity at 5-HT1A, 5HT2A, 5-HT1B, 5-HT1D, 5HT1F receptors and dopamine D2 receptors. Gastrointestinal effects are thought to be primarily secondary to agonist activity at 5HT1A and 2A and dopamine D2 receptors, while properties reducing CH are thought to be secondary to activity at receptors 5-HT1B, 5-HT1D, and 5-HT1F. In the past, vasoconstrictive action of ergots and triptans at 5-HT1B receptors was thought to be the primary basis of therapeutic benefits. However, advances in understanding the pathophysiology of headache disorders suggest that therapeutic effects are additionally relative to reduced neurogenic inflammation at peripheral tissues and decreased nociceptive impulses within the trigeminal nucleus caudalis via 5-HT1D receptors. Administration of DHE can be achieved via intranasal, intramuscular, or intravenous routes, but oral administration is limited by first pass metabolism and poor GI absorption, giving an estimated bioavailability of approximately 1%. 73 In CH, DHE has been investigated as both preventive and abortive treatment. In a retrospective case series published in 2004, Magnoux et al reviewed charts of 74 patients who had received IV DHE treatments from 1992 through 2000 at a single headache clinic. 74 The protocol for IV DHE treatment was similar amongst the patients: 3 days of IV DHE with subsequent daily subcutaneous DHE for several weeks. Measurement of efficacy in this case series was either resolution of headache or headache reduction greater than 50% at 1 month from completion of IV therapy. The authors reported that the majority of patients were responders: 63% of patients experienced full resolution and 15% experienced partial resolution.
Capsaicin
Capsaicin is a derivative of homovanillic acid derived from red peppers of the genus Capsicum shown to cause the release and eventual depletion of substance P and other neuropeptides from C fibers of sensory neurons with decreased sensitivity with repeated administrations.75,76 Multiple studies have investigated intranasal capsaicin application in treatment of CH. Marks et al conducted a double-blind, placebo-controlled trial of intranasal capsaicin 0.025% cream and placebo camphor cream to simulate capsaicin's painful sensation. 77 A total of 13 participants were divided into treatment (n = 7) and control (n = 6) groups and respective treatment was applied via cotton tip to the nostril ipsilateral to the headache twice a day for 7 days. Participants rated their pain on a 0 to 10 scale 3 days prior to start of study, throughout the treatment week, and for 7 days subsequent to treatment. Baseline scores of headache severity were 5.13 for the capsaicin group and 6.4 for the placebo group. During the week following treatment, there was a significant difference between headache severity in the treatment group compared with the control group, with average pain scores of 2.46 and 5.15 respectively. Subsequently, Fusco et al investigated intranasal capsaicin in 52 episodic CH and 18 chronic CH patients in an open-label trial, with administration of 300 ug nasal spray to either the ipsilateral or contralateral nostril of CH patients. 78 Applications were begun 15 days after the initiation of a cluster period, and number of attacks and use of symptomatic agents was recorded at 10 days before and 60 days after treatment. Seventy percent of episodic CH patients reported improvement with ipsilateral administration compared with no relief with contralateral treatment. The study also investigated 19 chronic CH patients with a protocol in which they were initially administered capsaicin in the contralateral nostril and changed to the ipsilateral nostril at 1 month. These patients reported significant improvement 30 days after treatment in the ipsilateral nostril compared with no improvement with contralateral treatment. The study noted that patients tended to relapse following discontinuation of capsaicin treatment.
Civamide
As a synthetic isomer of capsaicin with a similar mechanism of action, civamide has been investigated as preventive treatment for CH. As a potentially less irritating treatment than capsaicin, civamide was investigated in a double-blind, placebo-controlled study involving 28 patients. 79 The patients in the treatment group (n = 18) were administered 25 micrograms of civamide to each nostril daily for 7 days and followed for an additional 20 days. The primary outcome measure was change in weekly headache frequency, with secondary outcome measures considering pain intensity, analgesic usage, and number of severe headaches per week. Most outcome measures in the study did not obtain statistical significance. The change in headache frequency from baseline to post treatment days 1 through 7 was significant, with a 55.5% decrease in the civamide group compared with a 25.9% decrease in the control group. Change in headache frequency at the other post-treatment periods did not obtain statistical significance.
Methysergide (1-Methyl-D-lysergic acid butanolamide)
Methysergide is a semisynthetic ergot alkaloid. It was developed as a 5-HT antagonist and is known to act at the 5HT2A receptor, with additional antagonistic activity at 5-HT2B and 5-HT2C. 80 Additionally, methysergide is known to have agonist properties at some 5-HT1 receptors including 5-HT1B and 5-HT1D, though less so than ergotamine or the triptans. Chronic treatment is theorized to inhibit the release of CGRP and interfere with its downstream effects, possibly via the active metabolite methylergometrine. It is notable that 5-HT2B and 5-HT2C antagonism can yield increased neuropeptide Y secretion, thereby decreasing neurogenic inflammation. There have been multiple open-label studies and blinded clinical trials regarding methysergide use in headache, though most trials have focused on its use in migraine. Methysergide is currently unavailable in the United States due to concerns regarding adverse events and side effects, including retroperitoneal fibrosis and lower extremity vasospasm. Additional side effects are common and include angina, weight gain, abdominal pain, peripheral edema, and peripheral artery insufficiency. Since cardiac murmurs and renal insufficiency are possible, methysergide use is contraindicated in patients with renal or cardiac disease. Prolonged use may require monitoring with cardiac echocardiography, chest X-ray, and abdominal MRI. Investigation of methysergide for CH occurred primarily as subset analysis of clinical trials from the early 1960s. These trials investigated what was then termed vascular headache, which included several distinct headache disorders. Most data obtained were from migraine patients; however, select trials did provide information on smaller numbers of CH patients. Friedman et al reported on 3 CH patients in a clinical trial of 26 patients, the rest of whom suffered from migraine. 81 In this small trial, two of the CH patients obtained improvement of their headache on methysergide dosed at up to 12 mg daily. Also in 1960, Graham et al reported on 20 participants given methysergide and placebo. 82 Sixteen participants showed improvement on methysergide, and, of these, 11 rated their response to be superior to placebo treatment. In 1962, two trials were reported, one in which 3 of 8 CH participants had complete remission of their headaches on a 6 mg tapering dose of methysergide. 83 The other 1962 trails reported “good to excellent” results in all 8 CH participants on doses of methysergide ranging from 2 to 16 mg. 84
Baclofen
Baclofen acts as an analog of GABAB receptors which is primarily used for treatnment of spasticity due to CNS disorders such as spinal cord injury or multiple sclerosis. Hering-Hanit and Gadoth assessed the effective of baclofen for the prophylaxis of CH in a clinic-based study of 9 consecutive patients in cycle. 85 They prescribed 5 mg three times daily for at least 2 days then increased the dose to 10 mg per dose for a total of 30 mg/day. Patients were assessed at 7 and 14 days for subjective improvement. Six patients reported cessation of attacks within 1 week, with another improving the next week. Interestingly many of these patients had a long duration of CH (mean 14.4 years), and all patients were male.
Pizotifen
Pizotifen is an antihistamine and 5-HT2 receptor antagonist weakly related to tricyclic antidepressants. At high doses it may act as a calcium channel blocker. Pizotifen was introduced as a prophylactic agent for vascular headache including migraine. Ekbom studied pizotifen as a prophylactic drug in 28 clinic patients with CH. 86 Patients increased doses by 0.5 mg every 2 days to a maximum of 4 mg in 1 to 3 divided doses. The maintenance dose varied from 1 to 4 mg with a mean dose of 2.4 mg for a total treatment ranging 4 to 12 weeks. Of these patients, 6 reported no attacks on the medication, 10 reported at least 50% improvement, and 10 were unimproved. Two patients likely had spontaneous remission of their cycles. Adverse events included 10 patients with drowsiness, 8 with weight gain of at least 2 kg and 1 each with nausea or anxiety. Of 10 patients who had previously used ergotamine for CH prophylaxis, 6 felt that pizotifen was superior to ergotamine, 2 noted similar effectiveness, and 2 felt pizotifen was less effective.
Pharmacologic Treatment of CH: A Practical Approach
In general, persons with CH, unless attacks are unusually short lasting or mild, should receive acute treatment. The most proven treatment is subcutaneous subtriptan, but nasal spray triptans may be acceptable for those with mild attacks with a slower onset. The 4 mg dose of sumatriptan injection may be useful for patients who get more than 2 attacks per day, as the usual maximum daily dose is 12 mg/day. The 4 mg dose allows patients to treat 3 attacks per day. Oxygen is the most proven alternative for CH patients with contraindications to triptans, or with frequent attacks. Oxygen can also be given in combination with other CH treatments. Medication overuse is not as common in CH as in migraine but may occur, especially in persons with a family history of migraine. 87 Given the expense of medications and insurance limitations on quantities of triptans, some persons with episodic CH hoard acute medicaitons when they are out of cycle. Oral medications have little use for acute CH attacks, and chronic opiate or barbiturate use is never recommended. Infusion therapy, usually with intravenous dihydroergotamine, is useful for refractory CH. Refractory CH can describe a patient with very frequent attacks and excessive triptan use or considerable disability or a patient who develops continuous pain. Remember to discontinue triptans 24 hours prior to infusion with DHE.
Transitional treatment with corticosteroids is effective for the majority of persons with CH, especially episodic cluster. The intensity of treatment depends to some extent on the clinical situation. For persons with infrequent episodic CH (1–2 cycles per year or less) an aggressive treatment using high doses (80 or 100 mg prednisone, for example) with tapering doses lasting weeks may drastically improve an individual cycle. In persons with chronic CH, corticosteroid tapers may be shorter to avoid the adverse events associated with frequent use. Given the adverse events associated with long-term steroid use (adrenal suppression, weight gain and hyperglycemia, and immunesupression, to name a few) start a prophylactic drug when starting corticosteroids as a transitional treatment. Patients should be aware that the corticosteroid steroid taper is a short-term treatment only, even if effective. In general, nausea is not a common symptom in CH as in migraine, and oral corticosteroids are adequate unless patients are hospitalized for DHE or other drug infusion or cannot take oral medications.
Given the severity of cluster headache, and the frequent nature of attacks, prophylaxis is usually indicated. Verapamil is the most commonly used preventively due to its effectiveness and relatively benign adverse event profile. In persons with episodic CH, start prophylaxis at the first sign of a cycle. After 1 to 2 weeks of no attacks, it is usually acceptable to lower or stop prophylaxis. Lithium is also effective in the majority of those with CH but tends to be used as a second-line drug due to the potential for adverse events. Some persons with CH develop a sense of when they are out of cycle, with a lack of sensitivity to alcohol being a common sign that a cycle has resolved. A minority of episodic CH patients will elect to continue their prophylactic medication, especially if they have frequent cycles and a coexisting medical condition such as hypertension, epilepsy, or migraine. In chronic CH, most patients will remain on prophylaxis but may increase the dose or add a second agent during times of exacerbations. If attacks persist, use the highest tolerated dose for at least 2 weeks before discontinuation. Combination therapy may be necessary, and preventive treatment in CH is usually apparent within a few weeks of starting medication. The majority of CH prophylactic medications can be given in combination, but generally avoid using medications with similar mechanisms of action such as topiramate and valproate.
Discussion
The above review attempts to summarize the strongest clinical evidence for current pharmacologic management of CH. Most evidence for CH treatment is derived from relatively small open-label trials, although more rigorous double-blind controlled studies of sumatriptan, lithium, and verapamil have been conducted. However, even trials with stronger design can suffer from small absolute numbers, and many CH treatment trials identify trends that were not able to achieve statistical significance due to their small size. Most drugs for CH are used off-label, and, of all the mediations reviewed here, only subcutaneous sumatriptan has FDA labeling for CH. In looking toward the future of CH treatment research, goals will likely include expanding on existing studies with larger trials and identification of new therapeutic agents.
The therapeutic approach for CH should involve providing preventive treatment at the onset of a cluster period, with several abortive medications on hand for acute CH. Patients who are provided oxygen as an abortive treatment should have other treatment available for attacks that might occur outside of the home. In any case, treatment modality must focus on comorbidities and tolerance of side effects, with particular care taken in regard to cardiovascular effects, which make certain agents unsuitable for large populations or require increased monitoring.
Author Contributions
Conceived and designed the paper: JLP, MJM. Analyzed the data: JLP, MJM. Wrote the first draft of the manuscript: JLP. Contributed to the writing of the manuscript: JLP, MJM. Agree with manuscript results and conclusions: JLP, MJM. Jointly developed the structure and arguments for the paper: JLP, MJM. Made critical revisions and approved final version: JLP, MJM. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Dr. Pomeroy has nothing to disclose. Dr. Marmura has received honoraria for consultation from MAP Pharmaceuticals and a grant for clinical research from Merck.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
