Abstract
Over the recent years there has been a gradual rise in the use of pharmaceuticals during pregnancy. Knowledge on placental drug transfer and metabolism has increased during the past decades as well. Investigation of the transplacental transfer of any therapeutically useful drug is essential to the understanding of its metabolic processes and is a prerequisite for its use during pregnancy. The purpose of this review is to give insight on the various techniques that have been developed to evaluate transplacental transfer of drugs and xenobiotics.
Keywords
Introduction
During pregnancy, the placenta, which is a highly complex organ, serves several critical physiological functions. Among these is an important barrier role to minimize fetal exposure to drugs taken by the mother and other chemical in the maternal environment. The protective role of the placenta as a barrier and for removal of end products of metabolism is vital as the fetal hepatic and renal systems have immature and insufficient metabolic and excretory capacity (Stevens, 2006). Drug transplacental passage can occur by several mechanisms which include passive transfer, facilitated diffusion and active transport. There are specific transport mechanisms assisting in this function that are localized in the maternal-facing apical (brush border) and fetal-facing plasma membrane of the syncytiotrophoblast (Syme et al. 2004). Among them are the ATP binding cassettes (ABC) family proteins that include P-glycoprotein (Pgp), multidrug resistance proteins 1-3 (MRP1-3), and breast cancer resistance proteins (BCRP). They can efflux drugs and toxic metabolites out of the feto-placental compartment into maternal circulation. Additional transporters including the organic anion transporters (OATP), serotonin transporters (SERT), norepinephrine transporter (NET) and several organic cationic transporters (OCTs) are also expressed in the placental tissue (St-Pierre et al. 2002).
Over recent years, there has been a gradual rise in the use of pharmaceuticals during pregnancy (Bonati et al. 1990; Sabo et al. 2001). The scientific community and public now pay more attention to the potential teratogenic and fetotoxic effects with increased focus on fetal exposure. Due to obvious ethical reasons, evaluation of fetotoxicity from maternal exposure to chemical and drugs is not performed in humans. Investigation of the transplacental transfer of any therapeutically useful drug is essential to the understanding of its metabolic processes and is a prerequisite for its use during pregnancy. Some mammalian animal models have been used to assess transplacental passage of drugs and chemicals, but species differences in placental anatomy and physiology have prevented general acceptance of animal studies as they relate to humans (Faber JJ, 1983; Hamshaw-Thomas and Reynolds, 1985; Kennedy et al. 1986). Thus, although animal studies continue to be necessary for the teratogenic and fetotoxic potential compounds, transplacental transfer, metabolism, placental toxicity, and molecular mechanisms functional in the placenta are best studied in placental models of human origin (Myllynen et al. 2005). The following review presents various in vitro methods that are used today to investigate the transfer across the human placenta, with an emphasis on the placental perfusion system, as it is an interesting tool for current studies of drug transfer.
Placental tissue culture
Placental tissue can be cultured as tissue explants and used to study many facets of the maternal-fetal interface. Transport, enzyme function, nutrient and xenobiotic metabolism were evaluated in human placental explants derived from early and near term gestation (Miller et al. 2005). Explants cultures of first trimester anchoring vili have been provided an in vitro model to study post implantation events, early stages of placentation and cytotrophoblast invasion (Genbacev et al. 1992; Miller et al. 2005; Vicovac et al. 1995). Placental explants can be used in two types of experiments. The first is when the tissue obtained from uncomplicated pregnancies and can be used to study the effects of experimental condition on tissue survival and function. For instance, placental explants have been used to investigate uptake and efflux of amino acids, vitamins and sugars (Dancis et al. 1968; Miller and Berndt, 1974; Ng et al. 1981), and to identify receptors, binding sits, and factors regulating transport processes. In addition, placenta explants have been used to study how does the mode of labor (cesarean section vs. vaginal delivery) affect the sensitivity of placental tissue to cytokines as tumor necrosis factor α (TNF α), prostaglandin (PGE2), prostacyclin and non steroidal antiinflammatory drugs(Turner et al. 2002; Turner et al. 2004). Another type of experiment is when the placental tissue is obtained from patients with clinically known pathology as intrauterine growth restriction (IUGR) or preeclampsia, and its function is compared in vitro with normal, gestational age matched controls (Miller et al. 2005). For instance, Merchant et al. studied matrix metallo-proteinase in explants from normal and IUGR pregnancies (Merchant et al. 2004).
Placental explants offer the advantage of intact microarchitecture and maintenance of cell-cell interactions and paracrine communications; hence contribution of mesenchymal and endothelial cells to any metabolic process can be taken into account. In addition, placental tissue explants have a lifetime of up to 11 days though the syncyitiotro-phoblast must be closely monitored for damage to microvilli, mitochondria, and basement membrane (Miller et al. 2005; Syme et al. 2004).
Placental microsomes
Microsomes are small vesicles that are derived from fragmented smooth endoplasmic reticulum (SER) produced when tissues are mechanically broken (homogenized). Microsomes contain the cell's cytochrome P450 (CYP) enzymes, involved in oxidative metabolism. The placenta is known to possess relatively extensive enzymatic activity. It contains multiple cytochrome P450 enzymes in the mitochondria and endoplasmic reticulum of the trophoblastic cells (Syme et al. 2004). The placenta metabolizing enzymes are already present in early pregnancy (Myllynen et al. 2005). During pregnancy the placenta acts as a functional barrier by virtue of its metabolic enzymes and efflux transporters, thus protecting the fetus from drugs and environmental toxins. However, there is also a possibility of production of active metabolites close to the fetus when the precursor is present in significant amounts. Microsomal preparations of human placenta have given valuable information of the various enzyme activities metabolizing drugs in the placenta. Van Patten et al. (1968) applied this procedure on various therapeutic agents. In these experiments, it was shown that the placenta could oxidize penobarbital while pethidine and amino-phenazone are only slightly demethylated by placental proteins (Van Petten et al. 1968). Others studied the effect of chronic maternal drug addiction on the metabolic pathways in the placenta by preparing microsomes from term placentas of drug-dependent vs. normal (control) mothers. Using this model it was revealed that chronic maternal addiction does not induce metabolic pathways in the placenta for the biotransformation of drugs of abuse (Ostrea et al. 1989). Environmental and occupational factors can affect the activity of xenobiotic-metabolizing enzyme in the placenta. The induction of CYP1A1 by maternal cigarette smoking or drug abuse and the effect of steroids has been amply demonstrated (Paakki et al. 2000a; Paakki et al. 2000b). Recently, metabolism of the progesterone, 17 hydroxyprogeteron caproate, (17-HPC) was studied on placental microsomes revealing that the extent of 17-HPC metabolism was much lower than that by the liver (Yan et al. 2008).
Techniques for the preparation of placental microsomes are similar to those used for the preparation of microsomes from other organs. Briefly, the placenta is collected immediately after delivery; a small fragment is taken, washed, dried and crushed, using a suitable apparatus. Following grinding the tissue undergo differential centrifugation to discard deposit, following another centrifugation of the supernatant, at very high speed to collect the deposit which contain the mitochondria. The deposit is rapidly frozen in liquid nitrogen while waiting to subsequent procedures (Bourget et al. 1995). The protein content in the microsomes, CYT P450 and aromatase activities are determined using various assays (Lowry et al. 1951; Omura and Sato, 1964a; Omura and Sato, 1964b). The placental microsome model is an experimental model derived from tissue fractions that provide us mostly with information concerning metabolism of drugs by the placenta.
Plasma membrane vesicles
Plasma membrane vesicles preparation is another efficient in vitro model to study placenta transport. This preparation is useful for studying the effects of drugs or chemicals on transport mechanisms across the plasma membrane. The membrane vesicles are prepared from the surface microvilli of the human term placenta that are exfoliated from the syncytiotrophoblast cells and are further purified by differential centrifugation and washing (Smith et al. 1974). Plasma membrane vesicle preparation can be isolated from the brush border and the basal surface of the trophoblast enabling us to separately investigate their function; thus, making it possible to individually investigate the function of each distinct transporter. For instance, Ushigome et al. studied the function of the physiologically expressed Pgp, demonstrating inhibition of digoxin and vinblastin uptake into the placental membrane vesicle using the Pgp inhibitors, verapamil, cyclosporine A, progesterone or C129 anti-p-glycoprotein monoclonal antibody (Ushigome et al. 2003). The main disadvantage of this method is that exploration of membrane transporters activity is done in the absence of regulatory factors; thus, it does not exactly represent the in vivo conditions.
Cell culture models
Primary placental cells
A wide variety of primary cell lines can be produced from human placenta. The cells derived from the dispersed placental tissue have been characterized according to their morphology, cytoskeletal protein, enzyme histochemistry, hormone production and other antigens (Kliman et al. 1986). A primary trophoblast culture cell is a useful method to study uptake and release of drugs. Human cytotrophoblasts can undergo differentiation in culture and fuse to form functional syncytiotrophoblasts (Kliman et al. 1986). To prove that cells isolated from a placenta are really trophoblast cells is very important, since contamination with different cell types may influence results and conclusions (Frank et al. 2001). Moreover, villous and extravillous cytotrophoblast differ in their function and characteristics (Ockleford et al. 2004). The selection of viable cells has been examined in a number of studies and are summarized in Workshop Reports (Frank et al. 2001; Guilbert et al. 2002; Morrish et al. 2002). The culture of mononucleated trophoblasts preparations, have been successfully used to measure uptake kinetics of the anionic steroid precursor, dehydroepiandrosterone sulphate (Ugele and Simon, 1999) and also were used to study the basal Ca (2+) uptake, and thereby, defining the membrane gates responsible for the syncytiotrophoblast Ca(2+) entry (Moreau et al. 2002).
Moreover, the endogenous expression of multiple transporters in isolated trophoblasts is an advantage to this technique, leading to a more representative view of potential interaction between drugs and natural substrates. For example, the functional expression of the efflux transporter, P-glycoprotein (P-gp), in primary cultures of human cytotrophoblasts was studied by Utoguchi et al. (Utoguchi et al. 2000); further supporting the use of this in vitro model to investigate mechanisms regulating drug distribution across the placenta. Recently, a method for preparing and maintaining tight-junctioned syncytium on a semi permeable membrane has been described, but not yet widely used (Hemmings et al. 2001).
Cell lines from human placenta
There are three main types of cell lines derived from human placenta (Sullivan, 2004):
Since cells from human choriocarcinoma display many of the biochemical and morphological characteristics reported for in utero invasive trophoblast cells (Wadsack et al. 2003), these cell lines are more commonly used as a model that mimics the placental barrier. Utoguchi et al. (2000) have been able to show that, in BeWo monolayers, the basolateral to apical transport of known p-glycoprotein substrates, vinblastin, vincristine and digoxin, is significantly greater than transport to opposite direction. Also, pharmacological inhibition of p-glycoprotein using cyclosporine A, leads to decreased basolateral-to-apical transport (Utoguchi et al. 2000). Monoamine transport has been studied in JAR and BeWo cell lines, and the transport and metabolism of opioid peptides was studied also across the BeWo cell line (Ganapathy and Leibach, 1995). The advantage of these transformed cell lines is their ability to replicate rapidly in culture and thus can be easily cloned. However, the main disadvantage of these cell lines is the ongoing debate of whether these cells express the same makers as their origin. In addition, the cellular heterogeneity of the placental trophoblast is essential to the normal physiology of this organ and cell lines reflect only part of this.
The in vitro placental perfusion model
The perfusion of isolated human placental cotyledon was first described in 1967 by Panigel et al. and later has been modified by Schnieder, Miller, Brandes and others (Panigel et al. 1967; Schneider et al. 1972; Brandes et al. 1983; Miller et al. 1985). This is the only method that simulates important in vivo features such as maintenance of the placental barrier and the separation perfusion of the maternal and fetal circuits. This model can provide information concerning three major aspects: a. placental transfer of substances; b. effects of endogenous and exogenous substances on fetal perfusion pressure and transport; and c. release of endogenous substances into maternal and fetal perfusions (Sastry, 1999).
Placental transfer of substance
The in vitro perfusion model provided valuable information on transplacental transport of nutrients such as amino acids (Schneider et al. 1979; Schneider et al. 1987), fatty acids (Dancis et al. 1974; Dancis et al. 1973), hormones (insulin (Boskovic et al. 2003; Menon et al. 1990), vitamins (biotin, thiamin) (Schenker et al. 1990; Schenker et al. 1992) and various drugs, such as anesthetics drugs (Ala-Kokko et al. 1995; Giroux et al. 1997), antidiabetic related drugs (Elliott et al. 1991; Elliott et al. 1994; Holmes et al. 2006; Nanovskaya et al. 2006; Kovo et al. 2008a; Kovo et al. 2008b), HIV protease inhibitors (Olivero et al. 1999; Forestier et al. 2001), antiepileptic drugs (Pienimaki et al. 1997; Myllynen et al. 2003), abused drugs (Malek et al. 1995; Nanovskaya et al. 2008) and antibiotics (Polachek et al. 2005). Bourget et al. and Myren et al. presented lists of drugs, having therapeutic interest, that were tested in the perfused cotyledon model from 1972 to 1994 and from 1995 to 2006, respectively (Bourget et al. 1995; Myren et al. 2007). In addition, we can learn from such perfusion experiments about the effect of exogenous compounds on the placental transfer of endogenous compounds. For example, it has been demonstrated that ethanol significantly reduces the placental transfer of linoleic acid (Haggarty et al. 2002).
The information offered from these perfusion experiments should be evaluated within the boundaries imposed by this model. The model involves metabolically static system in contrast to the meta-bolically active and dynamic state of pregnancy. Some placental transporters are expressed differentially during the different stages of pregnancy. For instance, early studies suggest that the expression of the multidrug resistance gene increases dramatically during pregnancy (MacFarland et al. 1994), thus, the results from perfused term placenta can not be extrapolated to earlier stages in pregnancy. In addition, the trauma which the placenta undergoes at the time of separation may affect the validity of the results (Sastry, 1999).
Effects of endogenous and exogenous substances on fetal perfusion pressure and transport
The effect of drugs or chemical on the fetal placenta vasculature can be measured during perfusion experiments. The influence of several biogenic amines and endogenous substances such as acetylcholine, epinephrine, histamine serotonin adenosine vasopressin and prostaglandins, has been studies (Ciuchta and Gautieri, 1964; Sastry, 1991; Sastry et al. 1997). The vascular endothelium is believed to modulate the activity of many vasoactive agents, and it has been shown that these agents act directly on the fetal placental vascular smooth muscle to produce vasoconstriction. In addition, these agents may also stimulate the release of either or both NO and PGI2, thus the overall effect of such agents may be the net result of opposing vasoconstrictor and vasodilator activities (Read et al. 1995; Clifton et al. 1996). Holcberg et al. demonstrated the vasoconstrictive effect of meconium on the pla-cental-fetal vascular, and as well the opposing effect of indomethacine causing significant reduction in the basal pressure of the vasculature of isolated meconium exposed cotyledone (Holcberg et al. 2001). Using the same model other drugs, that are used during pregnancy, as hydral-azine (Magee and Bawdon, 2000), and dexa-methasone (Clifton et al. 2002), and magnesium sulphate demonstrated reduced placental perfusion pressure while labetalol did not significantly affect the hemodynamics of fetoplacental vessels (Skoczynski and Semczuk, 2001). Decreased fetal-placental vasculature perfusion pressure under acidemic conditions, was demonstrated by Pierce et al. (Pierce et al. 2002) suggesting a local physiologic adaptive response, though others showed no difference in fetal-placental vascular tone when fetal circuit perfusate was made acidotic (Hoeldtke et al. 1997).
Release of endogenous substances into maternal and fetal perfusions
The dually perfusion system can address also the question of relative release of placental product into the maternal and fetal circulation. This model has successfully been used to investigate the placental release of human chorionic gonadotropin (hCG), human placental lactose (hPL) and leptin which are released mostly to the maternal circulation (Linnemann et al. 2000). Further more, this model enable us to study also the release of cyto-kines by the placenta. Increase production of inflammatory cytokines IL-6 and TNF
A scheme with detailed prescription of the perfusion experiment system is presented in Figure 1. Experiments can be performed by using either closed (re-circulating) or open (non-circulating) method. In the closed model both maternal and fetal perfusate are recirculated, imitating the physiological conditions and can be used to study transplacental transfer and metabolism of the compound. In the open perfusions, maternal-fetal and fetal-maternal clearance can be studied (Sastry, 1999). The maternal to fetal transport rate (percentage) of the compounds can be calculated according to accepted formulas (Challier, 1985) while the term that enables comparing results of different perfusion experiments, and is used by various authors, is the clearance index (the ratio between maternal to fetal transport rate of the studied drug to that of the reference substance, antipyrine) (Elliott et al. 1994; Kopecky et al. 1999). Most of the perfusion experiments continue for three hours, but even are performed for more extended periods (10-12 hr) (Boal et al. 1997; Heikkila et al. 2002). Recently, ultrastructural studies demonstrated that the completeness of the placental tissue is maintained and only moderate changes occur after six hours of normoxic dual in vitro perfusion (Bachmaier et al. 2007). The integrity and viability of the placental tissue during the perfusion experiments are monitored by the lack of volume leakage from maternal to fetal perfusate or vice versa, as determined by comparison of final reservoir volumes; the ability to achieve adequate circuit perfusion rates within fetal inflow pressure ranged between 40-70 mmHg, and satisfactory antipyrine transfer, to a predetermined extent of at least 20%, by the end of perfusion experiment (Schneider et al. 1972). In addition, hCG production, lactate production and glucose consumption are also monitored to validate the reliability of the experiments (Derewlany et al. 1991). The perfusion model is technically rather complex, and there is a considerable rate of failure; most of which is due to leakage from fetal to the maternal side. However, this model allows examination of the secretory capacity, metabolism, transport and barrier function under controlled condition. In addition it allows studying the pharmaceutical influence on placental metabolism and modifi cation of placental fetal blood flow. With the advent of more precise knowledge concerning the identity, localization and regulation of drug transporting carriers in the placenta, the dually perfused placenta technique may see even wider and more resourceful use.
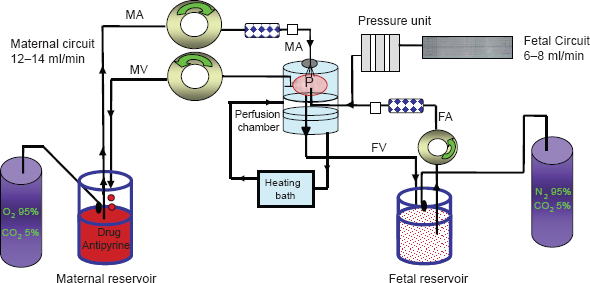
The perfusion experiment begins by obtaining placentas immediately after vaginal deliveries or cesarean sections. A suitable fetal artery
Conclusions
Understanding the degree of fetal exposure to drug/s is an important aspect of the pharmacological treatment to the pregnant women. Yet, many questions regarding the transfer of drugs, the involvement of transporters and the possibility of interactions between them remain to be answered. This review presents the different techniques that are in use today for studying the placental transport of xenobiotics (Table 1). These experiments contribute to the development of a safer and effective approach to the pregnant women. The placenta is anatomically complex, thus, experimental models retaining the tissue structure are most valuable. We consider the human placental perfusion model as the only model that fully retains the structure of the placenta, and has good viability compared to explants cultures. Therefore, it is one of the most valuable models for the investigation of transplacental transfer. Using the perfusion experiments trans-placental transfer with possible role of different transporters, xenobiotics metabolism and tissue binding of genotoxic compounds can be studied. Furthermore, by perfusion of placentas from mothers using illegal drugs or those suffering from various diseases, the impact of these factors on the fetal exposure to different xenobiotics can be studied. Drug metabolism and drug transport across the placenta should continue to be researched and guidelines need to be developed to ensure that any medications used during pregnancy are safe to both the mother and the fetus so that, in addition, successful treatment of the medical condition is carried out.
Experimental methods studying transplacental transfer.

Disclosure
The authors report no conflicts of interest.
