Abstract
The primary objective of this ongoing study is to examine the long-term safety and tolerability of olanzapine long-acting injection (LAI). Current results are from a 190-week interim analysis. Patients were 18–75 years of age with schizophrenia (N = 909) or schizoaffective disorder (N = 22) previously enrolled in 1 of 3 randomized clinical trials of olanzapine LAI. In this open-label extension study, all patients received flexibly-dosed olanzapine LAI every 2–4 weeks. At time of analysis, rate of study discontinuation was 46.3%. The 18-month discontinuation rate was 34.2%. Adverse events in ≥5% of patients were increased weight, insomnia, anxiety somnolence, headache, and nasopharyngitis. There were 26 occurrences of post-injection delirium/sedation syndrome which all fully resolved within 72 hours. Mean weight change was +1.81 kg, with 32.1% of patients experiencing ≥7% weight gain. Mean Clinical Global Impressions-Severity scores remained stable throughout (2.9 at baseline to 2.8 at endpoint). Pharmacokinetic analyses indicated consistent olanzapine plasma concentrations over time, with no evidence of long-term accumulation. Safety profile was consistent with that of oral olanzapine, with the exception of findings specific to intramuscular injection. During the study period, there were 16 (1.7%) occurrences of treatment-emergent diabetes and 1 occurrence of treatment-emergent diabetic ketoacidosis. Percentages of patients with EPS scale-defined treatment-emergent akathisia, parkinsonism, and dyskinesia were 3.3%, 6.6%, and 3.0%, respectively.
Introduction
Recently introduced long-acting formulations of second-generation antipsychotics offer the advantages of atypical agents, such as a lower risk of extrapyramidal symptoms (EPS), while also offering the prospect of greater adherence due to its less frequent dosing schedule. The use of antipsychotics in depot form has been limited in the United States (approximately 15% of patients with schizophrenia on maintenance treatment),1,2 but the use of depots may increase as new atypical depot formulations are becoming available. Because depot antipsychotics are intended for long-term use, there is a critical need for long-term safety data. However, few studies of depot antipsychotics have examined use beyond 1 year of treatment.
Safety results for long-acting risperidone, the first long-acting second generation antipsychotic introduced to the market, have been reported in a 12-month open-label study (N = 615) 3 and a long-term open-label extension study (N = 371) of at least 12 months in duration. 4 The most commonly reported adverse events in these studies were psychosis, headache, insomnia, agitation, and anxiety. Rates of extrapyramidal adverse events were 25% for Fleischhacker et al and 33% and 22% for the 2 study cohorts in Lindenmayer et al. Reported 12-month discontinuation rates were 35% for Fleischhacker et al and 45% and 48% for Lindenmayer et al. A more recent subgroup analysis 5 of a larger study of risperidone long-acting injection 6 followed 529 patients out to 18 months and showed an 18-month discontinuation rate of 55.7% based on Kaplan-Meier estimates.
Two previous studies have presented data on the acute efficacy and safety of olanzapine long-acting injection (LAI) in schizophrenia. In an 8-week, randomized, placebo-controlled study of olanzapine LAI in schizophrenia, 7 patients were randomly assigned to 1 of 3 dosing groups or to placebo. Supplementation with oral antipsychotics was not permitted. Mean decreases in baseline-to-endpoint Positive and Negative Syndrome Scale (PANSS) total score were significantly greater for all olanzapine LAI groups versus placebo. The acute tolerability profile for olanzapine LAI appeared consistent with that for oral olanzapine, with the exception of injection-related adverse events, and the results did not suggest any differences in efficacy between the 2-week and 4-week dosing regimens.
In a maintenance study of olanzapine LAI, 8 patients with schizophrenia who were stabilized on open-label oral olanzapine for 4 to 8 weeks were randomly assigned to 1 of 4 olanzapine LAI dosing groups (1 of which was considered subtherapeutic) or to oral olanzapine for 24 weeks of double-blind treatment. At 24 weeks, relapse rates ranged from 5% to 16% for the 3 therapeutic dose groups, and the 4-week and pooled 2-week dosing regimens demonstrated noninferiority to oral olanzapine as well as to each other. There were no clinically significant differences between olanzapine LAI and oral olanzapine with respect to laboratory measures, vital signs, electrocardiography, or EPS. Two olanzapine LAI-treated patients in this study experienced post-injection delirium/sedation syndrome following possible inadvertent intravascular injection, resulting in symptoms consistent with olanzapine overdose. 9
Although these studies have helped to establish the efficacy and safety of olanzapine long-acting injection in the acute and maintenance treatment of schizophrenia, longer-term data are helpful when evaluating a depot medication intended for chronic use. The present trial is an open-label extension designed with the primary objective of studying the long-term safety and tolerability of olanzapine LAI in patients with schizophrenia or schizoaffective disorder. This paper reports results from an interim analysis with up to 3.6 years (190 weeks) of data from patients with schizophrenia or schizoaffective disorder treated with olanzapine LAI.
Methods
Study design
All procedures were conducted in compliance with the Declaration of Helsinki and any standards applicable to individual institutional review boards. Data for this analysis were collected from 30-Aug-2004 (first patient enrolled) to 30-Apr-2008, which was the interim analysis cutoff date for all analyses. The study was conducted at 128 sites in 25 countries.
Previous (feeder) study designs
Patients participated in 1 of 3 feeder studies prior to entering the present open-label study. The first was an 8-week randomized, double-blind, controlled study of olanzapine LAI (N = 306) versus placebo (N = 98) in the treatment of acutely ill patients with schizophrenia. 7 The second study was a maintenance of effect study in which patients with schizophrenia were stabilized on oral olanzapine for 4 to 8 weeks and then randomly assigned to oral olanzapine (N = 322) or olanzapine LAI (N = 743) for 24 weeks of double-blind treatment. 8 The third study was a Phase I pharmacokinetic study (olanzapine LAI, N = 134), 10 which also included patients with schizoaffective disorder.
Current study design
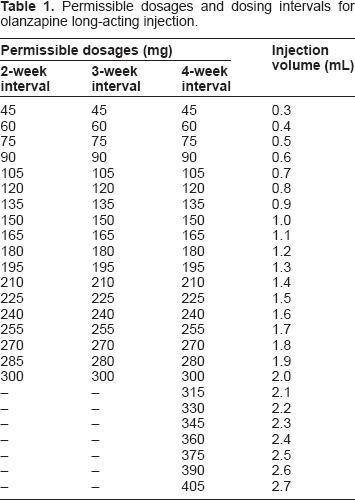
Permissible dosages and dosing intervals for olanzapine long-acting injection.
Patients
Patients were 18 to 75 years of age, had a diagnosis of schizophrenia or schizoaffective disorder according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV or DSM-IV-Text Revision), and had previously completed 1 of 3 controlled studies of olanzapine LAI. Exclusion criteria included significant suicidal or homicidal risk; pregnancy or breast feeding; acute, serious, or unstable medical conditions; or substance dependency (except nicotine or caffeine) within the past 30 days. All patients signed a written informed consent after receiving a complete description of the study and prior to participating.
Measures
Safety and tolerability
Measures of safety and tolerability included incidence of unsolicited treatment-emergent adverse events, mean changes in vital signs, treatment-emergent categorical changes in glucose and lipid measures, and mean changes and treatment-emergent categorical changes in weight and QTc.
The screening assessment, which was conducted at the first open-label visit, included a standard history and physical, psychiatric examination, laboratory profile, and electrocardiogram (ECG). Vital signs were measured at every injection visit, and otherwise were measured at Weeks 1, 4, and 8; at each quarterly or 6-month visit; and at the discontinuation visit. Laboratory assessments were conducted at the first open-label visit; at Weeks 1, 4, and 8; at each quarterly or 6-month visit; and at the discontinuation visit. Fasting status was required at each 6-month visit and at the discontinuation visit. Lipids results represent a combination of fasting and non-fasting visits but glucose was analyzed separately based on actual fasting status. An ECG was conducted at Week 1, at each 6-month visit, and at the discontinuation visit. Spontaneously reported adverse events were recorded at each visit. EPS were assessed at the first open-label visit, at each 6-month visit, and at the discontinuation visit using the Simpson-Angus Scale, 11 the Barnes Akathisia Scale, 12 and the Abnormal Involuntary Movement Scale (AIMS). 13
In addition to the safety/tolerability assessments mentioned above, beginning in August 2006, all ongoing patients were required to be observed for a minimum of 3 hours following each olanzapine LAI injection in order to assess for signs of post-injection delirium/sedation syndrome and to manage any such signs or symptoms that appeared. As part of this change in study procedures, mental status assessments were conducted on all patients immediately prior to an olanzapine LAI injection and at 1, 2, and 3 hours post injection.
Efficacy
Efficacy was measured using mean changes on the PANSS 14 total, positive, and negative scores and on the Clinical Global Impressions Severity of Illness scale (CGI-S). 15 The PANSS was administered at the first open-label visit, at each 6-month visit, and at the discontinuation visit. The CGI-S was administered at each visit. Measures of effectiveness were rates of and time to discontinuation due to any reason, due to adverse event, and due to lack of efficacy.
Pharmacokinetic methods
Blood samples for the assessment of olanzapine plasma concentrations were collected at quarterly study visits and at the patient's last visit. Plasma samples were measured by a validated high performance liquid chromatography/electrochemical detection (HPLC/ECD) method at BAS Analytics, Inc., West Lafayette, Indiana, US. Plasma olanzapine concentration data were analyzed graphically and descriptively. Given its open-label nature, patients in this study were on a range of dosages and permitted by protocol to change dosage at the clinician's discretion. To facilitate interpretation of pharmacokinetic data and to compare results across the range of doses studied, steady-state olanzapine concentrations were normalized by dividing an individual's measured olanzapine concentration at each visit by the total dose of olanzapine administered over the dosing regimen.
Statistical methods
Analyses were based on all the patients who received at least 1 depot injection. Except for adverse event analyses, patients were included in an analysis only if they had a baseline and at least 1 postbaseline measure. Baseline was defined as the observation at the patient's first open-label visit. All hypotheses were tested at a 2-tailed significance level of 0.05. Mean change analyses of continuous data used a la st-observation-carried-forward (LOCF) approach and were evaluated using 1-sample t-tests with a null hypothesis of mean change = 0. Incidences and rates were calculated for categorical data. Unless otherwise indicated, percentages for adverse events and reasons for discontinuation used all patients as a denominator. Time-to-event analyses were conducted using Kaplan-Meier survival analysis.
Results
Patient characteristics
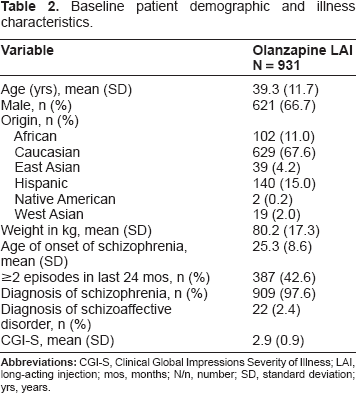
Baseline patient demographic and illness characteristics.
Patient disposition and dosing information
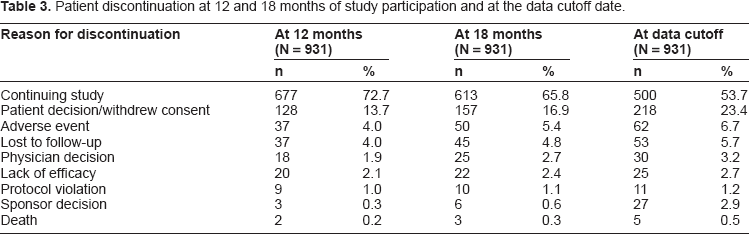
Patient discontinuation at 12 and 18 months of study participation and at the data cutoff date.
Based on injections with a quantifiable injection interval (n = 28,750), the mean prescribed dose across all olanzapine LAI injections was the equivalent of 15.5 mg/day. Dosing ranged from a minimum of 45 mg approximately every 4 weeks (1.6 mg/day) to a maximum of 405 mg approximately every 2 weeks (28.9 mg/day; use of a dose of 405 mg/2 weeks was a protocol violation). The most frequently used dosages (≥1% of total injections) were 300 mg/2 weeks (29.5% of injections), 405 mg/4 weeks (17.4%), 210 mg/2 weeks (10.3%), 300 mg/4 weeks (6.7%), 210 mg/4 weeks (5.6%), 300 mg/3 weeks (5.3%), 210 mg/3 weeks (2.9%), 150 mg/2 weeks (1.8%), and 150 mg/4 weeks (1.5%). Oral olanzapine supplementation was used at some time by 24.9% of patients and was sporadic, for a cumulative average of 133 days during the study at a mean dose of 2.6 mg/day oral olanzapine. Rate of concomitant benzodiazepine use was 21.1% for an average of 205 days at a mean dose of 4.9 mg/day (in lorazepam equivalents) (SD = 14.0). Rate of concomitant anticholinergic use was 4.9% for an average of 326 days at a mean dose of 3.0 mg/day (in biperiden equivalents) (SD = 1.5).
Olanzapine plasma concentrations
Steady-state olanzapine plasma concentrations remained consistent over time, with no evidence of continuing accumulation over the course of 3.5 years of treatment (Fig. 3). Median dose-normalized olanzapine plasma concentrations were 2.26 (ng/mL)/(mg/day) with a range of 0.87 (ng/mL)/(mg/day) (10th percentile) to 4.73 (ng/mL)/(mg/day) (90th percentile), consistent with concentrations previously reported for olanzapine LAI 8 and oral olanzapine. 16
Safety and tolerability
All patients who entered the study (N = 931) were evaluated for safety. There were 1,676 patient-years of exposure to olanzapine LAI during the study period and a total of 30,480 injections of olanzapine LAI were administered.
Adverse events
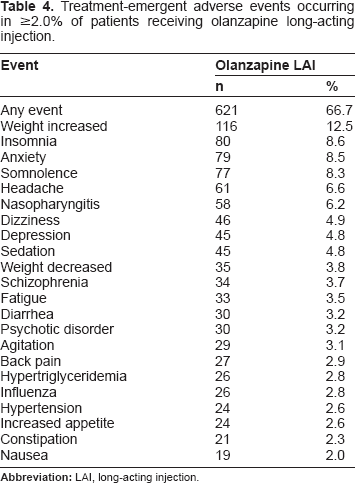
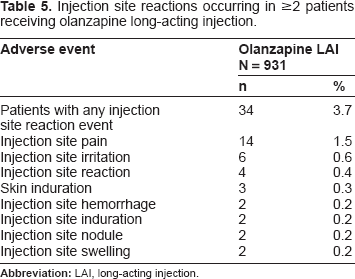
Treatment-emergent adverse events occurring in ≥2.0% of patients receiving olanzapine long-acting injection.
Injection site reactions occurring in ≥2 patients receiving olanzapine long-acting injection.
Vital signs and weight
There were small but statistically significant mean increases in blood pressure (≤3 mm Hg standing and supine systolic, standing and supine diastolic), pulse rate (approx. 2 bpm, standing and supine), and temperature (0.04 °C). These changes in vital signs were not judged to be clinically meaningful. Mean weight gain from baseline to endpoint was 1.81 kg (SD = 6.57) over an average of 642 days. During the study period, 295 patients (32.1%) experienced an increase of ≥7% of baseline body weight, whereas 157 patients (17.1%) experienced a decrease of ≥7% of baseline body weight.
Laboratory analytes
Percentages of patients with categorical glucose and lipids values at any time.

Electrocardiography
ECG analyses showed that there was a statistically significant increase in the QTc interval, Bazett: +1.2 msec (SD = 17.4), P = 0.041; however, Fridericia: +0.6 msec (SD = 15.6), P = 0.285 was not significant. Percentage of patients with baseline to endpoint changes ≥60 msec was 0.1% (Bazett) and 0.3% (Fridericia). One patient had QTc ≥ 500 msec based on Bazett; none had QTc ≥ 500 msec based on Fridericia.
Extrapyramidal symptoms
On scales measuring EPS, mean change from baseline on the Barnes Akathisia Scale was +0.00 (SD = 0.47, P = 0.774) from baseline of 0.12 (SD = 0.42). The percentage of patients with scale-defined treatment-emergent akathisia (Barnes global score of ≥2 at any postbaseline visit and baseline Barnes global score <2) was 3.3%. Mean change on the Simpson-Angus scale was –0.11 (SD = 1.57, P = 0.036) from a baseline of 0.75 (SD = 1.62). Percentage of patients with scale-defined treatment-emergent parkinsonism (total Simpson-Angus score of >3 at any postbaseline visit and a baseline total score ≤3) was 6.6%. Mean change on the AIMS was –0.06 (SD = 1.52, P = 0.256) from a baseline of 0.49 (SD = 1.40). Percentage of patients with scale-defined treatment-emergent dyskinesia (a score of ≥3 on any 1 of the AIMS items 1 through 7, or a score of 2 or more on any 2 of these items at any postbaseline visit, without either at baseline) was 3.0%.
Efficacy and effectiveness
Baseline-to-endpoint mean change was –0.26 (SD = 14.5) on the PANSS total score, –0.05 (SD = 4.3) on the PANSS positive score, and –0.13 (SD = 4.5) on the PANSS negative score. Mean baseline CGI-S total score was 2.92 (SD = 0.9), borderline to mildly ill, and this level of severity was maintained throughout the study period (see Fig. 1). There was an overall LOCF baseline-to-endpoint CGI-S mean change of-0.15 (SD = 0.9).
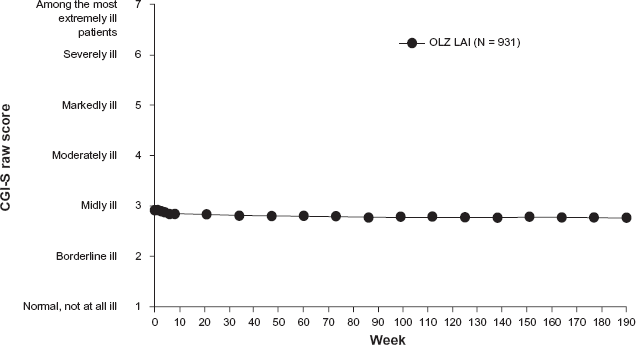
The CGI-S visit-wise mean score by week for all 931 patients enrolled in the olanzapine LAI open-label extension study (mean baseline = 2.9, mean endpoint = 2.8, P < 0.001).
During the study period, 222 patients (23.8%) were hospitalized, with the majority of hospital days attributed to psychiatric (11.13 days per patient year) rather than to general medical (1.34 days per patient year) or ICU care (0.07 days per patient year).
Times to discontinuation due to any reason, due to adverse event, and due to lack of efficacy are depicted in Figure 2. Median time to all-cause discontinuation was 1,044 days (95% C.I.: 945, 1,204).
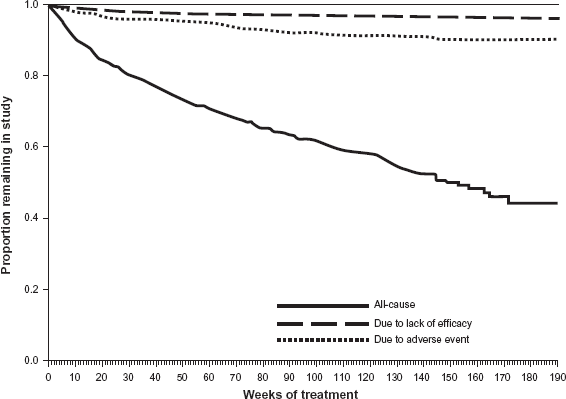
Kaplan-Meier survival curves showing time to all-cause discontinuation, time to discontinuation due to lack of efficacy, and time to discontinuation due to adverse event during the 190-week interim analysis for the 931 patients enrolled in the olanzapine LAI open-label extension study. Median time to all-cause discontinuation was 1,044 days (95% C.I.: 945, 1,204).
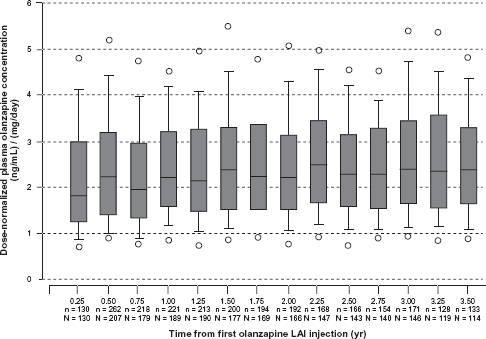
Box-and-whisker plot of olanzapine plasma concentration distributions for all doses (normalized by equivalent mg/day) through up to 3.5 years of olanzapine LAI dosing. The middle line in each box plot represents the median; the top and bottom margins of the box represent the 75th and 25th percentiles; the whiskers extend to the 90th and 10th percentiles; data points outside the whiskers represent the 5th and 95th percentiles.
Discussion
In this interim analysis of an open-label extension trial of olanzapine LAI in patients with schizophrenia and a small number of patients with schizoaffective disorder, the types of adverse events reported were generally consistent with the known safety profile of olanzapine, 17 with the exception of those specific to an injection. The profile of injection site-related adverse events appeared to be generally consistent with that seen with other products that involve the use of an intramuscular injection. Patients gained an average of 1.8 kg, with almost one third of patients experiencing clinically significant weight gain. Some patients experienced worsening in glucose and lipids measures (categorical increases in glucose, total cholesterol, LDL cholesterol, and triglycerides and decreases in HDL cholesterol), consistent with the safety profile for oral olanzapine. Pharmacokinetic analyses revealed no indication of long-term systemic accumulation of olanzapine, even after 3.5 years of treatment.
Post-injection delirium/sedation syndrome, which is believed to be related to inadvertent intravascular injection of a portion of the dose resulting in olanzapine overdose, 18 occurred in 0.09% of injections. All patients with post-injection delirium/sedation syndrome in this study period fully recovered without lingering or permanent sequelae. Most patients who experienced post-injection syndrome during the study (73%) chose to continue treatment with olanzapine LAI following the event. As a result of the possibility of these events, which could occur in any patient at any given injection, the study protocol was subsequently amended in August 2006 to require that patients remain at the investigator's site for 3 hours post injection. The purpose of this post-injection observation period was to ensure that the patient was free of any symptoms of olanzapine overdose prior to leaving the clinic and also to ensure that the patient was in a safe environment in the event that a post-injection syndrome event occurred.
Efficacy results evaluated maintenance of treatment effect, as the majority of patients entered the open-label extension after having been stabilized on treatment in their respective feeder studies. Patients receiving olanzapine LAI for up to 190 weeks showed improvement or no significant change from baseline to endpoint in PANSS and CGI-S scores, thus demonstrating positive clinical stability. The olanzapine LAI 18-month discontinuation rate of 34.2% was low relative to those reported for oral antipsychotic medications (eg, 74% across all antipsychotics in the CATIE trial 19 ).
Strengths of this study include the duration and ecological validity of the design. The current analysis includes patients with over 3 years of treatment with olanzapine LAI, representing the longest depot treatment study of an atypical antipsychotic to date. In addition, the study was designed to closely replicate real-world prescribing practices (eg, permitting oral supplementation, 20 use of antidepressants and mood stabilizers, flexible dosing, flexible dosing intervals, and concomitant medications). These factors enhance the generalizability of the results to clinical practice.
Limitations of this study include the open-label design and lack of comparators, making it difficult to draw firm conclusions regarding efficacy. In addition, this report is an interim analysis, and final study results are not yet available. The fact that oral antipsychotics and other medications were allowed throughout the study theoretically could have limited the detection of any potential safety signals unique to the injectable formulation. However, as stated above, these factors also increase the generalizability to actual clinical settings in which patients with schizophrenia are treated with multiple pharmacological regimens. Finally, it should be noted that the introduction of the post-injection observation period in August 2006 could have been responsible for some of the patient discontinuations, although it is not possible to determine the exact number.
In summary, these results represent some of the longest-term safety and tolerability findings yet conducted with a long-acting injectable antipsychotic and suggest good maintenance of effect and positive tolerability, as demonstrated by the low discontinuation rate over 3 years of treatment. Safety findings were consistent with the known profile observed with oral olanzapine therapy, with the exception of adverse events specific to use of an intramuscular injection. Incidence of injection site-related adverse events was low, but a number of patients did experience post-injection delirium/sedation syndrome. As a result, all patients were required to be observed at a healthcare facility for a minimum of 3 hours post injection. Patient continuation rates supported a positive benefit/risk profile of olanzapine LAI in the long term. Although health care providers should weigh the potential benefits and potential risks of treatment for each individual patient, olanzapine LAI may represent an important treatment option for patients with schizophrenia who struggle with adherence to oral antipsychotic medication.
Portions of these Data were Presented at the following Meetings
Schizophrenia International Research Society (SIRS) 1st Annual Meeting, June 2008, Venice, Italy, Collegium Internationale Neuro-Psychopharmacologicum (CINP) XXVI Congress, July 2008, Munich, Germany. Clinicaltrials.gov Identifier: NCT00088465
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. All authors are employees and/or shareholders of Eli Lilly and Company. The peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
The authors wish to acknowledge Prajakti Kothare Ph.D. and Tonya Quinlan B.A. for pharmacokinetics consultation and analysis.
