Abstract
Introduction
Intramuscular (IM) olanzapine has been approved for the management of acute agitation/psychosis secondary to schizophrenia, bipolar disorder, and dementia, in a dose not to exceed 10 mg per injection and 30 mg per day. Many other clinical situations exist in which patients are acutely agitated and in whom IM olanzapine might be warranted at recommended or higher doses. We systematically reviewed data on 8 such patients, during 10 inpatient admissions, to assess the overall safety and tolerability of this approach.
Methods
Using a standardized data recording sheet, we retrospectively collected information on 10 inpatient admissions between 2004 and 2007, in which IM olanzapine was administered outside of product dosing and/or indication guidelines. We present composite data and the details of each case individually.
Results
Records of eight patients (5 males, 3 females; mean age/age range = 43.9/18–77 years) were reviewed over 10 inpatient admissions, 5 to Psychiatry, 4 to Surgery, and one to Internal Medicine. Indications for admission were mania (2), schizophrenia (1), schizoaffective disorder (2), brain injury (2), sepsis (1), sepsis with respiratory failure (1), and cancer (1). Five patients were intoxicated with or experienced withdrawal from alcohol and/or drugs, likely adding to their agitation. Only one symptomatic episode of orthostatic hypotension occurred, in a patient with comorbid C. difficile colitis, which resolved when antibiotics were initiated and olanzapine discontinued; olanzapine IM was successfully reinstituted 2 days later, without incident. No other significant adverse events or side effects were ascribed to IM olanzapine.
Conclusions
Intramuscular olanzapine may be safe to use outside of product dosing and indication guidelines. Randomized clinical trials are warranted to study such off-label use further.
Introduction
The management of patients with acute psychosis is one of the most challenging responsibilities of emergency room personnel, and of all others who must care for these patients during the acute period. 1 Besides often being non-cooperative, patients in the midst of an acute psychotic episode can be violent, posing a danger to themselves and to others, and most especially to those who are trying assist. 1 8 A significant proportion are put in seclusion or physical restraints for their protection and the protection of others.9,10
Pharmacological management has long been the main-stay of management, but it is not without challenges of its own. To begin with, oral preparations cannot be administered to a large portion secondary to agitation, so that the intramuscular route often is required. 11 Perhaps a more significant problem is that, until recently, anti-psychotic medications have been associated with a variety of significant side effects. Prolongation of the cardiac QT(C) interval caused virtual cessation of the use of droperidol. 12 Haloperidol, a commonly used drug, and for years the first line of therapy, has been associated with potentially severe and certainly problematic extrapyramidal adverse effects, including tardive dyskinesia.13,14 Atypical antipsychotics seem to be better tolerated in the acute phase, especially in terms of cardiac and extra-pyramidal side effects; 15 the recent emergence of intramuscular forms of ziprasidone and olanzapine5,16,17 has led many to consider these new drugs to be the first line of therapy in the agitated patient with acute psychosis.8,18,19
Among the intramuscular preparations, intramuscular (IM) olanzapine, typically given in a dose from 2.5 to 10 mg, up to but not exceeding 30 mg in 24 hours, seems to be gaining particular favour as an effective and relatively safe drug.20,21 Some of the perceived benefits of intramuscular olanzapine are that it takes effect quickly, sometimes within 15 to 30 minutes22,23 it is more calming than sedating;4,24 it has a lower frequency of adverse events;4,6,21,25–27 and it may make for an easier transition from acute parenteral to maintenance oral (PO) therapy.25,28 Largely within the context of clinical trials, it has been shown to be effective and safe in the management of agitation secondary to acute psychosis in general, 27 as well as specifically in patients with schizophrenia and schizophrenia-related disorders,14,22,25,28–31 bipolar mania, 30 33 dementia,30,34 and borderline personality disorder.35,36
Yet problems exist with assessing the effectiveness and safety of antipsychotic medications within clinical trials. This is largely so because, within most clinical trials, written voluntary informed consent must be received prior to an individual receiving treatment; and this invariably excludes the most acutely psychotic patients, who are frequently not capable of providing such consent. 17 A second problem relating to the generalizability of results in clinical trials deals with exclusion criteria, since patients with certain medical conditions, such as cardiac disease, typically are excluded; but excluding such patients from treatment often is not a viable option in the ‘real world’. 17 A third limitation of clinical trials pertains to drug dosage, since dosage schedules usually are strictly enforced, with little laxity for higher doses, even when clinically deemed reasonable. Some patients with acute psychosis require higher doses of medication to control their agitation, such as some patients with schizophrenia.37,38 Some authors have asked whether higher doses of olanzapine might be warranted in certain clinical situations. 38 However, olanzapine is not without adverse effects and potential risks, such as transient hypotension.27,39,40 There is a widespread agreement to try not to combine IM olanzapine with any drug from the benzodiazepine class, especially because of the risks of over-sedation, cardiorespiratory depression, and drug-induced hypotension.39,41 The risk of hypotension may be especially great in the most severely psychotic patients, who often present quite dehydrated, and many of whom have been consuming alcohol. These patients may be the ones who might benefit the most from higher doses of an intramuscular anti-psychotic. However, they are also the ones least likely to be included in a formal clinical trial.
The aim of this retrospective study was to determine the safety and tolerability of IM olanzapine in patients with acute psychosis and/or agitation who might warrant a trial of olanzapine outside of the usual published guidelines (off-label), by documenting the variety, frequency and severity of adverse effects. As the study was exploratory, no hypotheses were tested. Real life prescribing was studied, such as use of the relatively contraindicated combination of olanzapine with benzodiazepines when the benefit-risk balance of this combination was considered positive. Approval of the Research Ethics Board at the University of Western Ontario was obtained.
Methods
In a retrospective review of patient charts dating from December 2003 to January 2007, records of all patients who had been admitted to adult inpatient programs at 2 academic health care organizations in London, Ontario, and who had been prescribed intramuscular olanzapine beyond the confines of published indications or doses, were identified and assessed. These patients included: 1) those who had been prescribed intramuscular olanzapine or some combination of IM plus oral olanzapine at a dose greater than 30 mg daily or 10 mg as a single IM injection; 2) patients prescribed intramuscular olanzapine for some indication other than the published product indications of schizophrenia, bipolar disorder and dementia; and 3) patients prescribed olanzapine in combination with benzodiazepines or anti-hypertensive medications.
Each chart was reviewed by a research assistant, using a pre-developed chart data abstraction form. Charts then were reviewed again by the first author to verify all collected information. Data collected included patient age, birth date and gender; date of, and indication(s) for hospitalization; admitting program; presenting symptoms; past and current illnesses; past surgeries; past psychiatric admissions, with indications/diagnoses; history of alcohol and drug use/abuse and the results of admission toxicology screens, when available; other relevant personal history; medications upon admission and drug allergies; prior use of and reactions to olanzapine, with details; date of initiation and initial dose of, and indications for olanzapine; all subsequent dose changes for olanzapine over the hospital course; all other medications used, with dates, doses and all dose changes; all potential medication-related adverse events and a rating of the likely role of olanzapine (No vs. Yes vs. Unknown); and date and disposition of discharge.
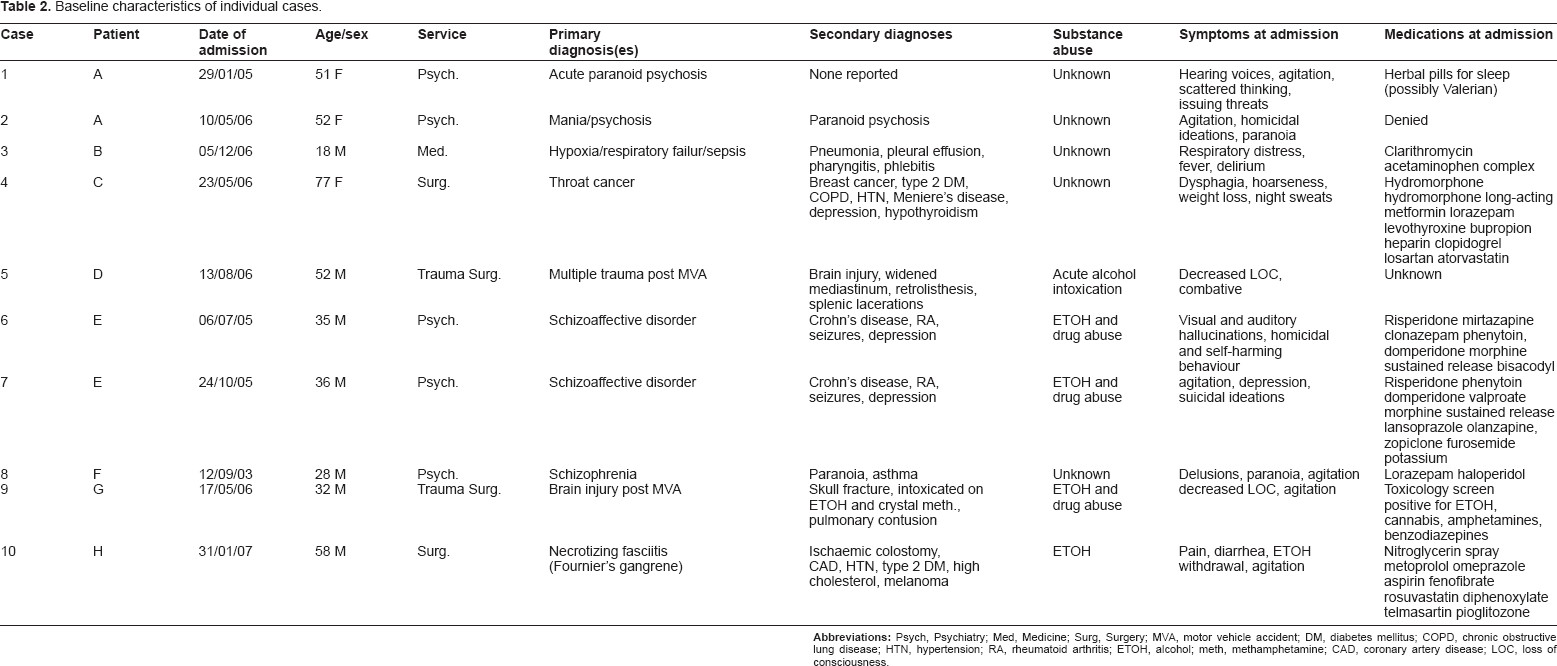
Over the 3 year period of review, 8 patients were identified who met the study criteria, accounting for 10 inpatient admissions, with 2 of the patients having received IM olanzapine beyond usual guidelines during 2 separate hospital admissions; for demographic and clinical information on these patients, see Table 1. Case-by-case details are presented in detail below and concisely in Tables 2 and 3.
Summary of baseline characteristics.

Baseline characteristics of individual cases.
Treatment characteristics of individual cases.

Results
Patient A—Cases 1 and 2
A. was a 52-year old woman with history of three hospital admissions over the span of three years. First admission was 3 years ago, 2nd admission was 2 years ago and 3rd admission was 2 years later, only 2nd and 3rd admissions are pertinent for the purposes of this study. She was non-compliant with taking olanzapine that had been prescribed during her hospital admissions. She had symptoms of mania and paranoia during one prior psychiatric admission for psychosis a year before the 2nd admission with a working DSM-IV-R diagnosis of Bipolar Disorder, Single Manic Episode (Severe with Psychotic Features). At 2nd admission (Case 1) she denied being on prescribed medication of any kind, but past records indicated that she had been treated with olanzapine during the first psychiatric admission. During her 2nd admission, she was agitated with auditory hallucinations and disorganized thoughts, and she was verbally abusive and threatening. She self-reported use of an herbal sleeping medication, thought to be valerian. Intramuscular olanzapine was initiated because she refused all oral medications, starting with 10 mg IM. Her initial on-floor orders were for 10 mg IM as needed (PRN), not to exceed 20 mg daily. The patient was also given a single STAT dose of 20 mg PO olanzapine two weeks into her hospital stay in addition to the regular IM doses, which was tolerated. Throughout the 18 days, her only non-psychiatric symptoms were transient headaches (twice) and mild to moderate constipation, neither of which was felt to be sufficiently explained by medication. Medications provided to the patient through the first course of admission while receiving olanzapine 20 mg IM STAT and 10 mg IM bid prn included acetaminophen 325–650 mg po q4hprn, clonazepam 0.5 mg po bid, risperidone 0.5 mg po bid and docusate 100 mg po bid for 10 days. Medications at discharge included clonazepam 0.5 mg po bid and risperidone 0.5 mg po bid.
Patient A's 3rd admission (Case 2) to the hospital was via the emergency room after a police intervention a couple of years later. She was transferred to the inpatient Psychiatry service on the same day, and kept in hospital for 33 days prior to leaving against medical advice (AMA). In this admission, her primary reason for psychiatric care focussed around an acute psychotic episode with paranoia, violence and uttering threats. She also had disorganized thinking and auditory hallucinations. Other than her two prior psychiatric admissions and gynaecological surgery, her past medical history was minimal, except for a recent episode of colitis with Clostridium difficile, which was still being treated. The patient was not on any prescribed medications at admission, despite having been on an atypical antipsychotic at the time of previous discharge from psychiatry Case 1 (above).
Medications received through this second course of admission while using olanzapine included acetaminophen 325–650 mg po q4hprn, lorazepam 1 mg IM q4h prn (not to be given within 2 hours of IM olanzapine), loperamide 2 mg prn to a max of 16 mg/day, ciprofloxacin 500 mg po bid, metronidazole 500 mg po bid, dimenhydrinate 50 mg po q6h prn, vancomycin 500 mg po q12h, morphine 2.5 mg SC q4h prn and ibuprofen 200 mg po q6h prn for a month and a half.
Upon admission to the inpatient Psychiatry service, her olanzapine orders were for IM Q4h PRN, which, contrary to literature recommendations, was given in conjunction with a benzodiazepine, lorazepam 1 mg IM Q4h PRN to a max of 4 mg per day, because of continued threats and agitation. This was modified early on and addressed by pharmacy services that the patient would receive lorazepam SL and identified need for re-education of the unit. A note was placed on the Medication Administration Record (MAR) that stated “IM Olanzapine and Lorazepam” not to be given within 2 hours of each other. Fortunately, no untoward effects befell the patient. Olanzapine was switched to 5 mg IM Q4h PRN by the second hospital day. IM olanzapine was discontinued on the third day, and PO olanzapine was put on hold, in response to a transient hypotensive episode, during which the patient's blood pressure fell from 134/85 at admission to 85/60, associated with feelings of light-headedness. Also diagnosed at that time was a flare-up of her C. difficile colitis, for which ciprofloxacin and metronidazole were initiated. By the fourth hospital day, her lorazepam was changed from 1 mg IM to 1 mg PO Q4h PRN. By the fifth day, her olanzapine was restarted at 5 mg PO Q4h PRN, since her blood pressure had stabilized, presumably because her colitis now was being treated. Fifteen days into her hospitalization, her agitation worsened again, so that IM olanzapine (5 mg PO TID PRN) was reinstituted, along with IM lorazepam (1 mg IM Q4h PRN), without recurrence of hypotension or light-headedness or any other symptoms attributed to her olanzapine or lorazepam. Overall, her agitation and delusional thinking improved over her 33 day hospital stay.
Patient B—Case 3
B. was an 18-year old male who was admitted to the intensive care unit (ICU) with sepsis and respiratory failure/hypoxia secondary to Lemierre's disease. Lemierre's disease is an extremely rare condition (only 160 cases reported over 100 years) that develops most often after pharyngitis has resulted in a peri-tonsillar abscess.42,43 Deep in the abscess, anaerobic bacteria, classically Fusobacterium necrophorum, flourish and ultimately penetrate the neighbouring jugular vein to form an infected clot (phlebitis). In additional to causing widespread bacteraemia (sepsis), septic emboli can travel to the lungs to cause pneumonia, infected pleural effusions, etc., all of which appears to have occurred in this patient, after a brief course of pharyngitis for which he was treated as an outpatient with oral clarithromycin. His stay in the unit lasted for 38 days, with agitation first noted as a problem requiring treatment on the ninth hospital day.
This agitation initially was treated with haloperidol 5 mg IV Q6h and lorazepam 3 mg IV Q4h as this is standard treatment for agitation (and as full blown delirium was not diagnosed at the time). Clonidine 0.1 mg per nasogastric tube was initiated the next day, followed by a continuous infusion of profolol 25–50 mg on hospital day 13, and morphine 2–5 mg IV Q15 min on day 15. Olanzapine IM 10 mg Q6h was initiated on hospital day 24, to replace haloperidol, as the patient was noted to be excessively restless and was unable to reply to verbal commands. The patient had never been on olanzapine before. He was maintained on PRN olanzapine, clonidine, morphine and lorazepam until his discharge from the ICU on day 38. Nurses' discretion determined whether or not the patient received olanzapine 10 mg, making it more a PRN than a regular scheduled medication. However, in the early period post extubation, doses of 10 mg QID were regularly utilized, decreasing to 5 mg qid thereafter. Other than sedation, which in this instance was a desired effect, no drug-related adverse events were noted at this time. The patient ultimately was discharged home.
Patient C—Case 4
C. was a 77-year old woman admitted to the inpatient Surgery service for 17 days with a primary diagnosis of squamous cell carcinoma of the base of the tongue and throat, as well as breast cancer. Her presenting symptoms were dysphagia, hoarseness, weight loss and night sweats. Other diagnoses included depression, type 2 diabetes mellitus, hypertension, emphysema, and Ménière's disease. She was on numerous medications at admission: hydromorphone 1 mg PO PRN, slow-release hydromorphone 6 mg PO BID, methormin 500 mg BID, lorazepam 1 mg QHS, levothyroxine 0.1 mg QD, buproprion 150 mg QD, atorvastatin 10 mg QD, clopidogrel 75 mg QD, losartan 50 mg QD, and heparin 5000 units SQ BID. Among those that were potentially sedating and respiratory-depressant were hydromorphone and lorazepam. It was unknown whether or not she ever had received olanzapine before, and use of lorazepam rather than an anti-psychotic was apparently continued on admission to prevent withdrawal.
On the third hospital day, she became agitated/delirious, presumably because of ‘sun-downing’ and/or over-sedation secondary to analgesia and/or sepsis. She was started on olanzapine (which is not standardly indicate for ‘sun-downing’), 10 mg IM or 10 mg PO given as a dissolvable tablet QD, whichever was preferred by the patient, and which was continued until the day of her discharge. The patient used the PO the majority of the time, but were a number of instances, particularly in the evening where the IM was used. The PO dissolvable formulation was also given as a STAT dose on one occasion, when the patient became particularly agitated. On the day on which olanzapine was initiated and 10 mg was provided, her blood pressure fell and heart rate increased, but not outside of the normal range, and she remained asymptomatic. Otherwise, she was noted to be confused on and off throughout her hospital stay, but confusion antedated the olanzapine and did not persist despite continuation of olanzapine. Lorazepam sublingual was started on Day 2 of admission, which is potentially a contra-indication in sun-downing. However, this medication remained on the profile until the time of discharge. As the medication was written for agitation/sedation purposes, the patient received a number of doses which may have aggravated the clinical picture. She was discharged to her home on the seventeenth hospital day.
Patient D—Case 5
D. was a 52-year old man with a past history of colitis and neck pain, but generally in good health until he was involved in a motorcycle accident and was admitted to the inpatient Trauma Surgery service with multiple injuries. These injuries included acute brain injuries plus bilateral subdural haematomas, a widened mediastinum, multiple splenic lacerations, possible kidney rupture, and bilateral dependent atelectasis. On initial drug screen, he was found to have an elevated blood alcohol level of 0.12 mg/dL. He was hospitalized for 12 days because of his injuries. It was not known whether he had ever received olanzapine before.
Initial sedation was provided using titrated infusions of midazolam 100 mg/50 mL syringe and fentanyl 500 mcg/50 mL syringe, to which profolol infusion was added and titrated to sedation in line with our critical care protocols. Thiamine 100 mg po daily × 3 days was initiated on the second hospital day, because of presumed alcoholism, and evidence of alcohol withdrawal became apparent by the second day, for which sublingual (SL) lorazepam 2 mg Q6h was given. All continuous infusions were discontinued by the third hospital day, replaced by IM morphine and lorazepam. Secondary to increasing combativeness, on the fourth day intramuscular olanzapine was initiated at a dose of 10 mg Q2h PRN. Olanzapine 10 mg was provided STAT and as the patient responded to this dose, 36 hours after admission, which was in the post-operative period, olanzapine dose was changed to 10 mg IM Q2H PRN to a maximum of 30 mg per day. While haloperidol would have been the first line therapy amongst the IM agents, olanzapine was chosen as there were concerns of extrapyramidal effects that could impair recovery in this patient in the post-operative period. Olanzapine was continued onto the 5th day, at which time it was changed to sublingual dissolvable olanzapine 10 mg Q12h. Care was taken not to give olanzapine and lorazepam within 2 hours of each other. By the 6th day, olanzapine was discontinued, and not required again. Prior to the initiation of olanzapine, the patient had decreased level of consciousness and increased somnolence. After olanzapine was initiated, he experienced a sense of “woodiness” in his legs that lasted approximately 3 days, but this sensation did not start until 3 days after the last dose of olanzapine; no clear explanation was found for this sensation. A transient episode of confusion was noted on the 12th hospital day, but this was almost one full week after the last dose of olanzapine. Consequently, no adverse effects or adverse events were attributed to olanzapine over the course of his hospitalization.
Patient E—Cases 6 and 7
E. was a young man with schizoaffective disorder and a history of alcohol and drug abuse, including intravenous drug abuse, who was admitted to the inpatient Psychiatry unit at age 35, and then again at the age of 36; on both occasions the indication for admission was a psychotic episode secondary to his schizoaffective disorder. On the first-mentioned admission, he was both suicidal and homicidal. He also had visual hallucinations during that time. His past medical history was otherwise significant for Crohn's disease, epileptic seizures, and rheumatoid arthritis. His pre-admission medications included risperidone 1.25 mg BID, mirtazapine 60 mg QHS, clonazepam 0.5 mg QHS, phenytoin 300 mg QD, valproate 500 mg TID, and MS Contin 60 mg BID. The phenytoin was prescribed to control his seizures and with it he had been seizure-free for years. Valproate had been added for the mood component of the patient's schizo affective disorder and was apparently not considered sufficient by itself to also control his seizures, in spite of the risk of hepatic inducing effects with phenytoin. He reported allergies to haloperidol, other ‘neuroleptics’, chlorpromazine, metoclopramide, prochlorperazine, and the artificial sweetener, aspartame. Olanzapine was initiated on the day of admission for ‘schizophrenic tendencies’ and visual hallucinations, at a dose of 5 mg PO UD (as directed), up to a maximum of 20 mg in 24 hours. It was felt that the rapid use of a maximal dose of olanzapine for his mood disorder and for acute management of agitation would hasten recovery and decrease length of stay; risperidone was not prescribed on admission, to minimize anti-psychotic adverse effects, particularly extra-pyramidal ones. While on olanzapine, the patient had decreased appetite, energy and concentration, anxiety attacks, continued hallucinations, difficulty breathing, numbness, dizziness and paranoia, but it was not felt that these were related to the olanzapine, because most had antedated initiation of olanzapine. Ultimately, olanzapine was discontinued on the 3rd hospital day, and risperidone reinstituted secondary to satisfactory control of the admitting psychiatric event. The patient was discharged home on the 6th hospital day.
The patient was readmitted to the hospital 3 months later, again for a psychotic episode secondary to schizoaffective disorder. This time, he was suicidal, depressed and agitated, but not homicidal. His pre-admission medications included, among others, olanzapine 5 mg QHS, risperidone 1 mg BID, mirtazapine 60 mg QHS, phenytoin 300 mg TID, valproate 750 mg TID, Morphine SR 60 mg Q12h, and zopiclone 7.5 mg QHS. Clonazepam was reinstituted on the day of admission, at a dose of 0.5 mg QHS PRN. Intramuscular olanzapine was initiated on the second hospital day, because the patient had become “angry, immature, offensive and aggressive” towards the treating team. His initial orders for olanzapine were for 10 mg IM Q4h PRN. Intramuscular olanzapine only was given on the second hospital day, being replaced with oral olanzapine DIS (dissolvable) 5 mg PO BID PRN, which was continued until discharge on the eighth hospital day. The patient had some nausea and vomiting on the same day as the IM olanzapine, but it was not known if these symptoms were related to the olanzapine. Later, he reported difficulty sleeping and some chest pain, which were not known to be drug-related. A cardiac cause of chest pain was ruled out, and the symptom resolved spontaneously.
Patient F—Case 8
F. was a 28-year old man who was a long-term patient diagnosed with schizophrenia. On this occasion, he was hospitalized for nearly four years, having had psychiatric admissions, but no olanzapine, in the past. His only other medical condition was asthma. The reason for this admission had been his schizophrenia, and his major symptoms included disruptive persecutory and religious delusions resulting in self-harm. According to medical records, the only medications reported at admission were being taken on a PRN basis: lorazepam 2 mg po Q4h and haloperidol 5 mg Q4h po, both for agitation. Olanzapine was started more than one year into his hospitalization, because he was agitated and non-responsive to other antipsychotics and refused to take oral medications. In addition, the patient had developed a movement disorder on flupenthixol, ruling this drug out as an option. He was started on a combination of intramuscular and dissolvable (DIS) oral olanzapine, including 10 mg IM and 20 mg DIS PO at HS, with an additional 5 mg DIS available at HS on an as needed basis. This dosing regimen was continued for 2 months (to attempt to address refractory schizophrenia with persisting non-adherence yet to minimize muscular adverse effects of IM), during which agitation decreased but psychosis persisted; he was then weaned off olanzapine, decreasing to 10 mg IM QHS 4 days after an initial decrease in dosage to 20 mg IM, discontinuing it 6 days after that, to be replaced by perphenazine 4 mg IM Q6h PRN immediately upon discontinuation of olanzapine, with clozapine up to 200 mg IM QHS added 2 weeks later. No adverse reactions to olanzapine were recorded, including no significant weight gain.
Patient G—Case 9
G. was a 32-year old man who was a known alcohol and intravenous drug abuser, which included the recent use of alcohol and ‘crystal meth’. He sustained major brain trauma in a bicycle accident, and presented to the Trauma Surgery service with a depressed right occipital skull fracture, right occipital condyle fracture, subarachnoid haemorrhage, left pulmonary contusion, right lateral chest wall fracture, left ear haemorrhage, and decreased level of consciousness. He was on no prescribed medications at admission, and there was no evidence that he had been on olanzapine before. On the admission toxicology screen, he tested positive for alcohol, cannabis, amphetamines, and benzodiazepines. Other relevant parts of his personal history include the patient being a known IV drug user who had been previously healthy medically. Upon admission, he was placed on intravenous morphine and fentanyl, as needed, for pain and agitation, and on a profolol infusion and PRN intravenous midazolam, 5 mg IV Q10 min PRN, for sedation. Despite this, he remained agitated, likely related to both his brain injury and alcohol/drug withdrawal, so that olanzapine 10 mg IM and/or per nasogastric tube (NG) BID was initiated on the second hospital day. By the ninth hospital day, olanzapine was prescribed 10 mg IM Q2h PRN, up to but not to exceed a cumulative dose of 30 mg per 24 hours. Over the same time period, haloperidol was given, as needed, either as 5 mg IM Q3h PRN; and lorazepam was given, as 2 mg IM Q6h PRN. By the ninth hospital day, methotrimeprazine also was prescribed for agitation related to his brain injury, given either IM or PO, in 10 mg doses in line with standard of care for acquired brain injury patients which present with agitation. Ultimately, all medications for agitation were discontinued on or before the 14th hospital day, except for methotrimeprazine, which was continued on a scheduled basis, 10 mg TID, with an additional 10 mg TID available PRN. The patient was discharged on the 20th hospital day. Other than confusion/sedation, no adverse effects were noted relating to any of his medications.
Patient H—Case 10
H. was a 58-year old man with numerous medical problems, which included coronary artery disease (graft × 4), hypertension, type II diabetes mellitus, hypercholesterolemia, melanoma, pulmonary bullae, past myocardial infarction, and chronic alcoholism, who was admitted to the in-patient Surgery service with an ischemic colostomy and a form of severe necrotizing fasciitis, usually involving the male genitals, called Fournier's gangrene.44,45 Pre-admission medications included nitro-glycerine spray PRN, metoprolol 50 mg BID, omeprazole 20 mg QD, fenofibrate 160 mg QD, salicylic acid 81 mg QD, rosuvastatin 10 mg QD, telmisartin 80 mg QD, pioglitazone 15 mg QD, and diphenoxylate PRN. He had not taken olanzapine before.
In addition to management of his multiple preexisting medical conditions, in-hospital treatment required aggressive debridement of his septic fasciitis, broad-spectrum antibiotics, sedation and pain control, and 6 weeks of hospitalization, including extensive time in the intensive care unit (ICU). Post-operatively, in the ICU, the patient became extremely agitated, presumably the result of ICU psychosis and alcohol withdrawal which did not respond to benzodiazepines, thiamine, and other sedation. Consequently, olanzapine was initiated at a dose of 10 mg IM daily on the 3rd hospital day and first post-operative day, in addition to him already being on a fentanyl infusion, haloperidol 5 mg IV/IM/PO Q6h PRN, and lorazepam 1 mg SL Q6h PRN. The highest dose of olanzapine administered was 10 mg PO/IM BID. After 20 days, intramuscular olanzapine was discontinued, during which time the patient had episodes of confusion presumed secondary to his alcohol withdrawal, ICU psychosis, lorazepam (given the temporal relationship between lorazepam and symptoms) or some combination of the above. Otherwise, on the 5th day on olanzapine, the patient had transient diastolic hypotension (140/42) that likely was secondary to his poor overall condition/sepsis, but could have been related to olanzapine. His blood pressure stabilized later the same day, however, and there were no further hypotensive episodes, despite continuing the olanzapine for an additional 17 days.
Discussion
Despite all of the potential problems associated with their use, even within the last decade, typical antipsychotics, such as haloperidol, have been purported to be first-line treatment in the treatment of agitation associated with various psychotic states.40,46,47 However, there has been a distinct movement away from these drugs towards newer drugs, and especially olanzapine and ziprasidone IM in the United States. The intramuscular form of olanzapine, released just in the past 7 years, has seemed like another step forward, providing an option for those patients who either refuse or are unable to take oral medication. Our study demonstrates that olanzapine IM provides some benefits and does not cause serious adverse effects or complications when cautiously used outside of indications, eg, in higher than indicated doses and in combination with benzodiazepines for patients with refractory psychosis or delirium. It is well known that the IM olanzapine and IM benzodiazepines may present a risk/safety issue. However, that is not documented when only one of the 2 agents are in an IM format, or there is greater than 2 hour spacing between the agents. The exception to this in our case series was patient A.
The fact that these patients had medical co morbidities as often the primary driver or used higher doses fits the safety and tolerability. Medical-surgical and complex care patients were not addressed in the registration trials, which was our intent. These patients are often not captured in registration trials and it befalls front line clinicians to make an assessment of medical risk-benefit assessments.
Intramuscular olanzapine is perceived to provide a faster onset of action than all oral preparations, sometimes evident within two hours14,25,29,31,33 and occasionally as rapidly as within 15 to 30 minutes.22,23 It also is believed to exert a calming effect, in contrast to the less-desired and non-specific sedating effect produced by most other neuroleptic drugs;4,24 to be associated with a lower frequency of adverse events, including extrapyramidal effects, relative to most oral and/or typical antipsychotics;4,6,21,25–27 to lack any prolongation of the cardiac QT(C) interval seen with certain other anti-psychotics;21,22,26 to be associated with potentially less cardiac risk than the other atypical anti-psychotic currently available for intramuscular administration, ziprasadone; 19 and to potentially make for an easier transition from acute parenteral to maintenance oral therapy.25,28 Largely within the context of clinical trials, it has been shown to be effective and safe in the management of agitation secondary to acute psychosis in general, 27 as well as specifically in patients with schizophrenia and schizophrenia-related disorders,14,22,25,28–31 bipolar mania 30 33 and dementia.30,34 Based upon these trials, specific indications have been published for the use of intramuscular olanzapine for the treatment of agitation secondary to schizophrenia, bipolar disorder and dementia. 48
In clinical practice, however, numerous other instances arise wherein olanzapine and, in particular, the intramuscular preparation of olanzapine might be useful. For example, olanzapine has been shown to be effective in the treatment of borderline personality disorder in two clinical trials35,36 There also are numerous other instances in which patients without schizophrenia, mania or dementia become acutely psychotic and/or agitated, to the point where they have become a hazard to themselves and/or others. Examples include patients with brain injuries, septic patients, patients in the intensive care unit, patients experiencing alcohol or drug withdrawal symptoms, and elderly hospitalized patients who are ‘sun-downing’. These are not uncommon situations. For example, depending upon how ‘agitation’ is defined, anywhere from 10% to 70% of patients with brain injuries go through a period of agitation. 49 52 Typically, this agitation is not apparent immediately upon presentation. Rather, it usually starts within the first week of hospitalization as the patient starts to be aroused. In most instances, it resolves within a week, though persistent agitation in the long-term is not all that uncommon.50,51 As a consequence, not only can this agitation be problematic and potentially dangerous to the patient him/herself and the treating staff, it also appears to have an adverse effect on short-term and possibly even long-term rehabilitation outcomes. 52 54 Despite this, few clinical trials have assessed treatments for this agitation;55,56 the empirical evidence that does exist is not that convincing;55,56 there is no consensus as to what constitutes the best therapy; 57 59 and clinical practice varies widely.57,58 Among others things, the variety of drugs used to treat the agitated traumatic brain injury patient includes propofol, lorazepam, midazolam, haloperidol, carbamazepine, tricyclic antidepressants (TCAs), trazodone, amantadine, and beta-blockers,57,58 but the only drugs among these that have clinical trial evidence supporting their use are the beta-blockers, and this evidence is limited to 2 small studies that have somewhat weak designs.55,56 The remaining evidence generally has been derived from small, uncontrolled and often retrospective case series.57,60–67 These studies include supportive case series for droperidol 64 and haloperidol; 66 but droperidol is seldom used now, because of its potential cardiac toxicity; 12 and evidence exists that haloperidol disrupts cognitive function more than olanzapine. 61
With respect to indications for olanzapine, 5 of our patients received intramuscular olanzapine for an indication consistent with product guidelines: 3 for schizophrenia-related symptoms and 2 for mania-related agitation. But intramuscular olanzapine also was prescribed for 2 patients with traumatic brain injuries, whose condition quite possibly was exacerbated by alcohol and/or drug withdrawal; for 1 elderly female with cancer who was sun-downing and possibly septic; for 1 middle-aged man with Fournier's disease, sepsis, alcohol withdrawal and ICU psychosis; and for one 18-year old male with Lemierre's disease who was both septic and hypoxic. In all instances, the patient was agitated, and agitation was the primary indication for IM olanzapine. Although not well documented in 2 of these 5 patients, overall, olanzapine seemed to exert a beneficial effect and, at least in these 5 patients, caused no major adverse events.
The guidelines published by Eli Lilly, the manufacturer of olanzapine, also recommend that the dose of oral olanzapine not exceed 20 mg per 24 hours; that the dose of IM olanzapine not exceed 30 mg per 24 hours, given in 10 mg increments no closer than 2 hours apart; and that the dose be lowered in elderly and/or otherwise debilitated patients to 2.5–5 mg per injection. Finally, it is recommended that intramuscular olanzapine be given with caution, if at all, to patients on anti-hypertensive medications, and to patients taking any benzodiazepine, in particular because of the risks of excessive sedation and cardiorespiratory depression, including hypotension. 68 In this report, in addition to prescribing IM olanzapine for indications beyond product guidelines, IM olanzapine was prescribed in conjunction with PO olanzapine in total cumulative daily doses higher than 30 mg, and in patients taking anti-hypertensive therapy and/or a concomitant benzodiazepine.
During 6 of the 10 hospitalizations, patients taking olanzapine at the same time as lorazepam was prescribed, usually both administered PRN, so that they could, theoretically, be given at the same time or very close together in time. However, we have attempted to adhere to no IM lorazepam and olanzapine co-administration within 2 hours of each other. However, no episodes of hypotension or problematic over-sedation were noted from the concomitant administration of IM olanzapine and a benzodiazepine, most often lorazepam, even though these drugs sometimes were administered within 2–4 hours of each other.
Clearly, several of our patients were quite systemically ill, due to major trauma in 2 instances, alcohol and/or drug withdrawal in 5 instances, and multiple medical problems in 6 of 8 patients. In fact, it is quite likely that only 2 of the patients we studied here would have not been excluded from a clinical trial. Two patients had extremely rare conditions, Lemierre's disease and Fournier's gangrene, that would have certainly been excluded from a clinical trial. Several patients were septic. What this demonstrates is both 1) the value of a naturalistic study, such as ours; and 2) the relative safety and tolerability of olanzapine when given intramuscularly, outside of product doses and indications.
Our report is not the only report of its kind for olanzapine. The previously described limitations of clinical trials have led other investigators to explore the use of anti-psychotic medications within the context of so-called ‘naturalistic studies’, usually—but not necessarily—open-label observational studies involving real patients in a real clinical setting, in the absence of exclusion criteria.9,17,23,69 In essence, if a patient required treatment, that patient received it. One such study was a prospective study in the emergency department of a hospital in Barcelona, Spain, reported in 2006, in which the first 100 patients (54% women; mean age = 36 years) with psychomotor agitation due to any cause were assessed. 9 The most prevalent diagnoses were psychotic disorder (48%) and personality disorder (24%). Physical restraint was required in 39% of patients and 52% accepted oral treatment. Haloperidol was the most frequent oral treatment and olanzapine the most frequent intramuscular treatment provided; no systematic attempt was made to assess effectiveness or safety. The authors' main conclusion was that atypical antipsychotics have become the first-line treatment for acute agitation.
In another Spanish naturalistic study conducted prior to the release of IM olanzapine, 910 inpatients with schizophrenia suffering from acute psychotic episodes and not receiving any other atypical antipsychotic were prescribed either PO olanzapine or a more traditional typical oral antipsychotic, such as haloperidol. 69 Overall, 483 (53.4%) were treated with olanzapine and 421 (46.6%) with typical antipsychotics. Three specific subpopulations of greater illness severity were defined: patients with prominent psychotic symptoms, agitated patients, and patients initially treated with intramuscular medication because of their acute clinical condition. Patients who had previously received IM drugs were not statistically different in their degree of overall symptomatic improvement with olanzapine versus other treatments, except for a greater reduction in BPRS negative symptom sub-scores (P = 0.015). The authors considered this to be evidence that olanzapine should be viewed as first line treatment for severely psychotic inpatients with schizophrenia.
A much smaller study was done in the UK, involving 7 patients (4 females; mean age = 41, range 18–65) all with bipolar disorder, schizophrenia, or schizoaffective disorder, all of whom were deemed to have a “greater severity of psychosis than clinical trial patients”. 17 All but one was given a single dose of olanzapine 10 mg IM, to which they responded quickly with decreased agitation, the one exception being an 18-year old male who required a second dose 12 hours later; he responded to both doses. Adjunct benzodiazepines were administered to one of the seven. No adverse reactions were noted, and the authors concluded that their experience in this naturalistic setting was consistent with the reports of clinical trials.
The final ‘naturalistic study’ differed from the others in that it was a prospective randomized trial. 23 In this study, 300 adults with agitated or violent behaviour presenting consecutively to a Psychiatric Emergency Centre in Southern India as a result of mental illness, were randomised either to receive a single injection of IM olanzapine or IM haloperidol plus promethazine. The primary outcome was the proportion of patients who were tranquil or asleep 15 minutes and 4 hours after receiving the IM injection. Secondary outcomes were the proportion of patients who were tranquil, asleep, restrained, absconding, or clinically improved at 15, 30, 60, 120, and 240 minutes; additional medical interventions and adverse effects over four hours; and adherence with oral drugs and adverse effects over two weeks. Follow-up data were available for 298 (99%) of the patients. The two treatments resulted in similar proportions of people being tranquil or asleep at 15 minutes (olanzapine 87%, haloperidol plus promethazine 91%; relative risk 0.96, 95% confidence interval 0.34–1.47) and 4 hours (olanzapine 96%, haloperidol plus promethazine 97%; RR 0.99, 0.95–1.03). However, more people that were given olanzapine required additional pharmacological interventions over the next four hours (43% vs. 21%; 2.07, 1.43–2.97). Adverse effects were uncommon with both treatments. The authors concluded that the two treatments were equally effective at rapidly tranquillising or sedating agitated or violent patients with mental illness, but also that the haloperidol-promethazine combination tends to result in fewer additional medical interventions over the next four hours.
Our study adds to previous publications by means of our evaluation of patients who received IM olanzapine off-label; ie, outside of product dosing and/or indication guidelines. What we have primarily demonstrated is that IM olanzapine can be used safely in most such instances. Admittedly, our study is only an exploratory evaluation of a small number of patients, and as such is limited for generalization. Yet there is promise in the study of such use of IM olanzapine, eg, as such an atypical antipsychotic may minimize the risk of extra-pyramidal and other neurological adverse effects and complications (which are likely in the patient populations requiring IM antipsychotics due to their prevalent polypharmacy and other factors), and as there are not many IM atypical antipsychotics available—and with those that are available, there is a higher risk of other adverse effects and complications, such as QTc prolongation and related cardiac arrhythmia with IM ziprasidone. Our results, despite such a small sample size, inherent with a case series suggest the need for formalized, randomized clinical trials to evaluate the safety and effectiveness of IM olanzapine in patients with traumatic brain injuries; in patients with other relatively common causes of acute agitation, such as sepsis, ICU psychosis, and sun-downing; in patients with non-adherence and non-responsive severe psychosis that may warrant multiple-drug, high-dose therapy; and in patients who have other medical conditions that otherwise would eliminate them from most clinical trials.
Disclosure
This study was supported in part by an investigator-initiated research grant from Eli Lilly Canada. Joel Lamoure discloses funding other (unrelated) projects (including speaker bureau and advisory committees) from Janssen-Ortho, Eli Lilly Canada, Pfizer, Biovail, Astra-Zeneca, Shire, and Bristol-Myers Squibb. Dr. Rudnick discloses funding for other (unrelated) projects from Janssen-Ortho, Novartis, Astra-Zeneca, and Mylan. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
The authors would like to extend thanks to Jessica Stovel for her work in data collection and manuscript review, Joelle Keypour for her data collection, and Dr. K. White for his assistance in preparation of the manuscript and tables.
