Abstract
Objective
This study compared relative effects of whole green coffee powder (WGCP) on cognitive functioning in neurotypical adults under three treatment conditions: placebo (A), low-dose 889.9 mg WGCP (B1), and moderate-dose 1334.4 mg WGCP (B2).
Method
Fourteen adults aged 18–25 years, acted as their own controls in three treatment conditions within a seven-session withdrawal design. Participants completed the Cambridge Neuropsychological Test Automated Battery for attention-deficit/hyperactivity disorder (ADHD) at each session. The Side Effects Behavior Monitoring Scale (SEBMS), used to assess stimulant effects in individuals with ADHD, was a secondary outcome measure to assess adverse events associated with caffeine intake delivered by capsule. Self-report of qualitative effects was collected.
Results
Results indicated that moderate doses of WGCP significantly improved sustained attention (vs placebo and low dose) and working memory (vs low dose only) but had no effect on response inhibition. Low doses of WGCP showed decreased sustained attention. Fifty percent of subjects reported positive subjective improvement in well-being. No side effects were reported.
Conclusion
Commercially available WGCP (ie, sold as GoBean®) in moderate doses improved executive functioning for sustained attention and working memory but had no effect on response inhibition. Implications for individuals with attention difficulties are discussed.
Introduction
Individuals progressing through academic study require concerted focused attention in order to successfully master the tasks posed by school. Control of attention is managed by executive functions that help to prioritize, organize, and complete work in a timely way. Russell Barkley describes executive functions as the actions people use to control personal behavior, direct behavior toward a goal, and improve outcomes for behavior in the future. 1 The role of attention in executive functions is critical in most facets of organized daily life.2–4
Attention wandering compromises executive functions and results in cognitive difficulties in learning.1,5 Attention problems are certainly evident, for example, in attention-deficit/hyperactivity disorder (ADHD) which is a developmental disorder in children, and, to varying degrees in adults.6–8 The etiology and impact of poor attention is multifactorial and impacts school performance of children, adolescents, and young adults. Alternative and complementary treatments may be helpful in managing behaviors associated with attention to school tasks, thus it is useful to investigate them.
Worldwide, caffeine is the most widely consumed substance having psychoactive effects. 9 It is the neuroactive agent in coffee and tea, and it is a nonselective antagonist of the neuromodulator adenosine; if applied in commonly consumed doses, it generates stimulating effects by blockading adenosine receptors. Cognitive performance generally is positively influenced by caffeine ingestion, and the influence of caffeine on cognitive performance is well documented.10–13 Although some studies show limited benefit to performance,14–16 caffeinated coffee is the most common form of caffeine intake, increasing alertness and lowering fatigue. Caffeine is now readily available in a variety of liquid (ie, energy drinks) and capsule forms.
Whole green coffee powder (WGCP) is a fibrous, naturally occurring endogenous substance and is a nonesterified solid source of caffeine. It is processed directly from the whole green coffee bean and contains chlorogenic acid in its natural form. It is distinct from green coffee extract because it is made from the whole bean in a specified process (current patent pending), delivers a solid (not from extract) form of caffeine in capsules, and is sold commercially as GoBean®. The presence of naturally occurring green coffee bean nutrients is not available in coffee extract, and the granularity of green coffee powder releases caffeine and cholinergic acid in an extended delivery. These unique features may impact attention, arousal, and executive functions in individuals who use it.
This study investigated the effects of commercially available dietary caffeine supplement (WGCP) on the ability of neurotypical individuals (ie, without diagnosed ADHD) to exercise executive functions associated with sustained attention, spatial working memory, and response inhibition (ie, impulsivity). These assessed executive functions promote cognitive activity in a way similar to academic study. To measure the effects of WGCP on core executive functions used in standard academic study, we used the ADHD Core Battery of the Cambridge Neuropsychological Test Automated Battery (CANTAB). This battery includes several modules: motor screening (data from this module were not used in analysis as it tests fine motor speed and is an introductory exercise to the test battery); rapid visual information processing (RVP, sustained attention); stop signal task (SST, response inhibition); and spatial working memory (SWM, working memory). We also investigated the qualitative effects of WGCP via participant self-report.
The study explored the following primary research questions:
What are the effects of WGCP compared with placebo on sustained attention, response inhibition, and spatial working memory?
How do subjects qualitatively describe the effects of WGCP on affective presentation in daily activity?
Method
This study used a small N approach to acquire preliminary information on the effects of WGCP. Small N studies are limited in controlling variability, but repeated measures allow them to be useful especially when studying novel treatments. The power of well-designed repeated measures designs is evident in that with 10 participants, receiving only five measurements across the study, power to detect significant differences within subjects across conditions is quite good (power = 0.89 using Cohen's f) when a large effect size (f = 0.40; ~d = 0.80), moderate test-retest reliability (correlation) between repeated measurements (r = 0.60), and a typical Type 1 error rate (0.05) are assumed. A large effect size is entirely reasonable to expect in repeated measures designs and the test-retest correlation is likely to actually be larger, possibly as high as the reported test-retest reliability of the test (>0.80) which would drive power even higher (>0.99). Readers interested in statistical analyses for this study may contact the first author for syntax.
Assumptions of Study Design
Referenced in previous studies with caffeine delivery,11–14 the following documented characteristics of caffeine are assumed for the use of WGCP in this study: (1) washout of WGCP effect will occur rapidly similar to caffeine washout in other delivery systems (ie, over the period of several hours); (2) dosing may be abruptly terminated without adverse side effects; (3) WGCP effects at moderate dose are not dependent on gradual increase from low dose, that is, subjects do not need gradual exposure to caffeine from low dose (in all cases, nevertheless, low dose preceded moderate dose); (4) onset of WGCP effect is established within one hour as is typical of caffeine products used in previous studies. Essentially, given no evidence to the contrary, WGCP will produce caffeine effects similar to other delivery systems.
Procedures
We used a repeated measures withdrawal of treatment design to examine the differential effects of a commercially available dietary supplement (GoBean®) and placebo in neurotypical college-age adults, aged 18 to 25 years. The design removes variability through improved experimental control of treatment conditions. 17 A withdrawal of treatment design allowed multiple observations of a small number of subjects (compared to randomized clinical trial designs that use few observations of many subjects). The design alternated treatment and no-treatment conditions across days within single subjects to provide sensitive examination of dose effects. Collection of time-series data permitted the assessment of ongoing treatment-related changes across each presentation of the dependent variable.
The trial is initiated in the baseline (BL) phase of an experimental manipulation of variables. The placebo (A) phase is alternated with the experimental phases. In this study, B1 was the first dose of the active compound (ie, WGCP), and B2 the second dose of the active compound. The Phase A is an intermediary phase between BL and active compound (B1 and B2) and controls for an expectancy of improvement associated with mere ingestion of a capsule as part of a trial (ie, placebo effect). We maintained the rule central to experimental manipulation of variables—only one variable was changed at a time. This allowed for opportunity to distinguish between expectancy (ie, placebo) and WGCP effects.
After a verbal inquiry to confirm overnight caffeine abstinence, 13 subjects received three identical capsules at each session with varying number of capsules containing WGCP. Each capsule contained placebo or 444.8 mg WGCP proprietary blend (55 mg caffeine as per GoBean® package label). Capsules were administered orally once each day in the presence of the study coordinator and one hour prior to CANTAB. Supplements were supplied in labeled plastic containers with study and subject randomization information (ie, study session number and subject coded identification), and sponsor on the label. In Phase A (placebo), all the three capsules contained an inert substance (ie, corn starch); in Phase B1, subjects received one placebo capsule and two WGCP capsules (889.6 mg); and in Phase B2, subjects received three capsules each with the same equivalent dose of WGCP (1334.4 mg). Package label instructions for using WGCP include a three-capsule dose. The study ran seven months (February–August 2013). Study visits were separated by at least one day with an average 3.66 days between each dose administration, and a range of 1–15 days. Six subjects were administered GoBean and the CANTAB before noon, and eight subjects were administered GoBean and the CANTAB between noon and 6:00 pm.
The order of the dose was not randomized since the concern was not if dose improves performance but only whether WGCP improved performance. To varying degrees across subjects, this also permitted us to detect residual effects of withdrawal. Because the safety of subjects is always paramount and despite the assumption that WGCP does not require gradual introduction, we moved from low to moderate dose to ensure that the subjects did not start with a dose to which they may be overly sensitive.
Because the order of presentation of treatment was defined a priori, placebo was counterbalanced across two orders of treatment to maintain the double-blind requirement; experimenters were unaware of the order of treatments. The counterbalanced treatment orders are indicated below:
BL − A − B1 − A − B2 − A − B2
BL − B1 − A − B2 − A − B2 − A
We assessed for subjects' medications, recreational drugs, or caffeine consumed through diet or other supplements prior to each administration and relied on subjects' accurate representation through verbal query.
The study focused on acute administration of WGCP, that is, subjects were provided doses of caffeine within a short period. This was practiced because similar studies with chronic caffeine use showed diminished sustained attention and working memory compared to those who abruptly terminated chronic caffeine use.13,14,18
Eligible subjects were randomly assigned to orders I and II; eight subjects received presentation I and six subjects received presentation II. Each subject arrived at the clinic at the same time in each experimental phase and the assigned times did not differ across placebo and active WGCP days. As an example, if Subject 1 arrived for her BL visit at 8 am, she came to subsequent WGCP active and placebo visits at 8 am. After arrival, subjects were given the randomized dose of WGCP and/or placebo. In one hour, subjects were presented the CANTAB which took approximately 30 minutes to complete and was administered at the same time of day, replicating BL conditions. The CANTAB was presented in a quiet, moderately lit room located in the library of a local university campus or in a similar room in a hospital setting; each subject completed the CANTAB in the same room in which it was initiated. Sessions were separated by at least 1 day to completely eliminate carryover effects as WGCP has duration of action of 4–6 hours (as per package label). After CANTAB administration, subjects verbally completed the Side Effects Behavior Monitoring Scale (SEBMS) 19 with the study coordinator.
Statistical Analyses of CANTAB Subtests
Sustained attention was measured using the discriminability parameter of the Rapid Visual Processing subtest. Response inhibition was measured using stop signal reaction time from the Stop Signal subtest. Spatial working memory was measured using total errors from the Spatial Working Memory subtest. For each dependent variable, a generalized estimating equations (GEE) model was computed with Treatment (placebo, low dose, and moderate dose) as fixed-factor predictors and the dependent variable as the response variable. GEE is advantageous in that it flexibly accounts for repeated measurements with each participant permitting missing data and explicitly modeling relationships between repeated measures conditions. Alternative covariance structures were examined. Results are based on autoregressive structure. GEE analysis assumed a Poisson distribution with log-linear link for ordinal/count data and a normal distribution with linear link for continuous data. The analyses estimate treatment condition effects using all available conditions. They are not technically averaged but rather using GEE models with all available data, the estimate is generated based on data from each instance of each treatment condition. Preliminary analyses examined order effects. However, there were no significant main effects or interactions with order. Therefore, results are presented without order included as an independent variable. Furthermore, given that the low- and moderate-dose conditions were in opposite directions relative to placebo, it is highly unlikely that any order effects influence the pattern of results.
Measures
The CANTAB ADHD Battery was the primary outcome measure. It has been demonstrated to detect neuropsychological effects with selectivity and sensitivity, allows ready interpretation of the effects, and it has a variety of applications in psychology, neuropsychology, and medicine. 20
The Motor Screening Task is administered at the beginning of the CANTAB and assesses whether a subject can respond to the requirements of the other tasks in the battery; it confirms appropriate visual, movement, and comprehension abilities. Rapid Visual Processing of Information (RVP) is a test of sustained attention and is similar to the commonly used Continuous Performance Test; it is a sensitive measure of general cognitive performance. The Stop Signal Task (SST) is a common assessment task used to assess response inhibition; it estimates an individual's reaction time and gives a measure of how well an individual can inhibit responses and resist the tendency to respond automatically. Spatial Working Memory (SWM) is a test of the participant's ability to use working memory by retaining spatial information, remembering items, and manipulated them in space; this test measures global executive dysfunction.
The CANTAB subtests were administered once in each session. Published studies20,21 demonstrated that parallel versions of the CANTAB allow repeated measures and that the CANTAB shows very small practice effects over repeated measures.
Qualitative descriptions and adverse events were assessed in each session. Side effects were assessed using the SEBMS adverse events checklist. 19 The SEBMS uses the Clinical Global Impressions-Severity (CGI-S) anchored scale (1 = normal, 2 = borderline, 3 = mild, 4 = moderate, 5 = marked, 6 = severe, and 7 = most extreme).22,23 Subjects completed the SEBMS at the end of each session to track change in behavior. All ratings were based on participants' subjective experience of the 1.5-hour period and on subjective reports between sessions. An adverse event was defined as any untoward medical or physical occurrence in a subject administered WGCP during the course of the study. Participants were probed as to the presence of side effects including heart function anomalies.
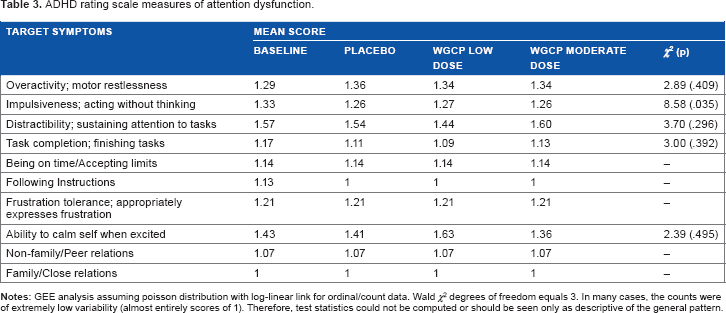
The ADHD Rating Scale (ADHD-RS) is an 18-item scale used to rule out symptomatic attentional difficulties. It was administered at BL coinciding with assessment of working memory and response inhibition as measured by the CANTAB. It was used to screen for the presence of ADHD. Scores over 32 are generally considered symptomatic threshold.
Drug screening was conducted by inviting the participants to give verbal self-report of use.
Subjects
Inclusion Criteria
To be eligible for inclusion, participants met criteria at initial screening and BL, in which (a) a written consent was signed by the participant; (b) the participant was aged 18–25 years; (c) females of childbearing age had a negative response to a verbal inquiry for pregnancy and were not at risk for becoming pregnant; (d) participants completed an ADHD rating scale; (e) participants had a minimum level of intellectual functioning (determined by the investigator, all participants were or had been enrolled in college courses); (f) symptom criteria for a comorbid mental health condition that could affect safety or tolerability of medication, or interfere with the participant's participation in the study were not in evidence; (g) blood pressure measurements were within the 95th percentile for age and gender at screening; and (h) participants were able to comply with the requirements of the study protocol.
Exclusion Criteria
At screening or BL, eligibility was declined if the participant (a) had a current, controlled, or uncontrolled comorbid psychiatric diagnosis with significant symptoms, that, in the opinion of the study investigator, contraindicated treatment, or assessment; (b) was suspected of substance abuse or dependence disorder within the past 12 months in accordance with DSM-IV-TR criteria; (c) admitted to the use of prescription or illegal substance; (d) had a history of seizures during the last two years, a severe tic disorder, and a current diagnosis or family history of Tourette's syndrome; (e) had a conduct disorder; (f) had taken an investigational product within 30 days prior to screening, or participated in any other research study during the trial; (g) had clinically significant blood pressure abnormalities at BL; (h) had a known history of structural cardiac abnormality; (i) had a concurrent chronic or acute medical illness that would prohibit the participant from completing the study or would not be in the best interest of the participant; (j) taking any medications that are excluded, have other central nervous system (CNS) dysfunction, or effect performance, such as sedating antihistamines and decongestant sympathomimetics (bronchodilators were not exclusionary); and (k) the female subject was pregnant or lactating.
Subject Confidentiality and Consent
Subjects were interviewed by the study investigator or study coordinator. Subjects signed the consent form during the interview period and consents were obtained at least one week prior to the start of the study period. The hospital Institutional Review Board (IRB) approved the study protocol and informed consent procedures.
Results
A total of 16 adults not diagnosed with ADHD or other psychiatric conditions were screened for participation; 14 enrolled in the study (see Table 1). This study population was useful because of participants' ongoing engagement with academic tasks. Academic studies require sufficient working memory and the ability to delay a response to competing activities (eg, participating with friends) to complete required academic work.
Subject demographics.

The study generated information on the effects of WGCP on: a) sustained attention (RVP), b) response inhibition (SST), c) spatial working memory (SWM), and d) qualitative descriptions of the effects of WGCP among young adults.
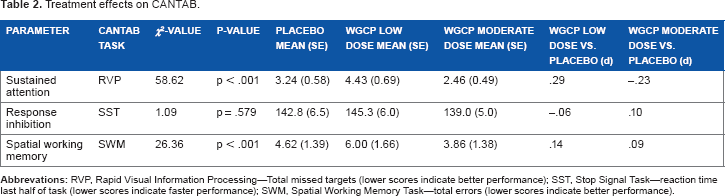
Results indicated a significant overall treatment effect for sustained attention (Rapid Visual Processing—total misses) (χ2(2) = 58.62, P < 0.001). Low-dose WGCP resulted in significantly worse sustained attention than placebo (χ2(1) = 5.56, P = 0.018), but moderate-dose WGCP resulted in significantly better sustained attention than placebo (χ2(1) = 5.22, P = 0.022). Significant differences were also noted in working memory (χ2(1) = 26.36, P = 0.001). Spatial working memory errors were highest in the low-dose WGCP and lowest in the moderate-dose WGCP. Placebo fell in-between these values but pairwise comparisons were not statistically significant (low dose vs placebo χ2(1) = 1.11, P = 0.293 and moderate dose vs placebo χ2(1) = 2.15, P = 0.142). No significant differences were observed for response inhibition (impulsivity) (overall P = .0579; see Table 2).
Treatment effects on CANTAB.
The mean ADHD rating scale score was 26.4 and nonsymptomatic for ADHD. Subjects did not differ on measures of attention dysfunction (see Table 3).
ADHD rating scale measures of attention dysfunction.
Qualitative Results
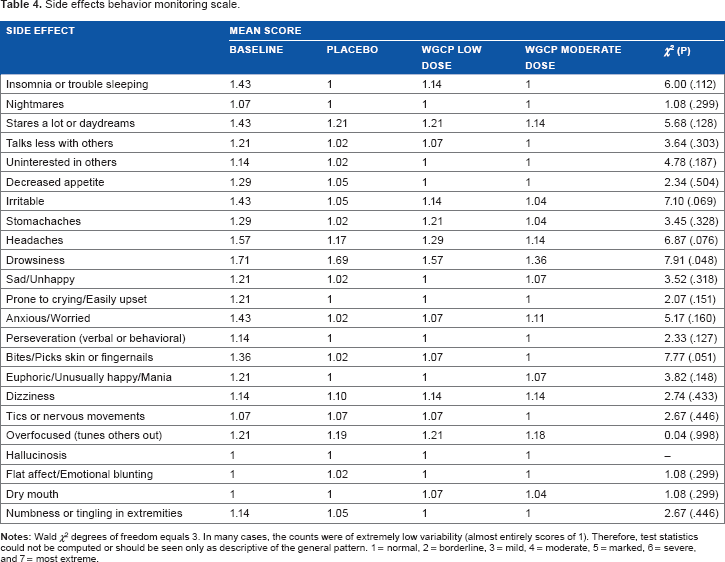
At the end of each session, participants gave subjective accounts of their experience from the time they ingested capsules until the completion of the CANTAB (approximately one and a half hours). In addition, they described reactions from the previous administration of WGCP. The SEBMS probed whether any of the 20 specific side effect reactions to stimulant medication were present. Participants showed no adverse events (Table 4). When probed whether they discerned receiving an active dose or placebo, seven participants were unable to identify whether they received active ingredient or placebo; seven participants accurately discerned that they had received WGCP moderate dose but not the low dose.
Side effects behavior monitoring scale.
Participants reported qualitative reactions to the moderate dose that are best defined in three areas: (1) increased efficiency on tasks, (2) enhanced ability to stay on task, and (3) a feeling of well-being. For example, Participant 14 stated that she felt “extra focused on the work I did in the morning.” Participant 10, a college student, stated that she “felt really good and focused even though I have a lot to do today.” Participant 8 reported, “I got more done in an hour today compared to yesterday.” Others reported “feeling good” and the absence of feelings of malaise or intrusive emotions.
Other qualitative reports14,18,24 showed that acute exposure to WGCP, as administered in this study, resulted in increased alertness, improved concentration, decreased fatigue, and significantly increased feelings of contentedness and satisfaction.
Side effects are summarized in Table 4. No significant side effects are reported. Side effect ratings on the CGI-S are all rated as normal or not at all present.
Discussion
This study confirmed effects of improved sustained attention and, to a lesser extent, spatial working memory with WGCP intake. Sustained attention showed improvement with moderate-dose WGCP, including separation from placebo and low-dose WGCP. The results for working memory were similar in pattern but quantitatively weaker, with moderate-dose WGCP showing improved working memory relative to low dose but not versus placebo. Results indicated a negative effect on sustained attention and working memory for low-dose WGCP (two capsules) contrasted with the strong positive effect for moderate-dose WGCP (three capsules). The positive effect of moderate-dose WCGP on sustained attention was modest in size (d = −0.23). Although not dramatic, this effect may be clinically meaningful and may be noticeable to individuals when working in contexts that require substantial sustained attention. The inhibitory effect at low dose is difficult to explain. At a low dose, WGCB may inhibit fatigue but it may not deliver enough caffeine to produce the cognitive effects that moderate doses do. If low dose was simply a non-effect, then placebo and low dose should be similar. They were not similar, however; low dose inhibited response. The inhibitory function of low dose may be evidence of a side effect, such as minor agitation, that occurs at smaller doses of WGCP. Individual variance of response may also explain these effects.
WGCP at a moderate dose tended to be associated with a qualitatively positive affective response. In interview, subjects reported a sense of well-being and an ability to initiate tasks more easily. For example, use of WGCP decreased ratings of sleepy, tired, drowsy, “half awake”, lazy, and sluggish. Subjects reported that they experienced an overall sense of contentedness and that they felt more at ease, relaxed, and satisfied. The substance induced more reportedly energetic feelings as well as heightened friendliness and sociability. In addition, evidence from previous studies 14 and common knowledge indicate a deleterious effect (eg, jitteriness or nervousness) of chronic caffeine ingestion. However, subjects in this study did not report these effects with WGCP. Additionally, inquiries regarding heart palpitations, rapid rate, or other cardiac function anomalies were not reported in probed interview.
It is an interesting finding that moderate-dose WGCP improved spatial working memory relative to low dose. Working memory is a complex function that involves the ability to manipulate and control information such that the information is both symbolically stored and processed in verbal and spatial forms. Neurologically, the information processed in working memory is stored throughout the brain depending on the nature of the eliciting information. 25 Caffeine effects, functionally altering the adenosine receptors, may impact the wide variety of neural pathways associated with working memory and may require higher WGCP concentration.
The finding that WGCP did not affect response inhibition is expected. Inhibitive functions are typically considered to be prefrontal, neurological events. Response inhibition is the ability to keep interfering information away from focused attention. It, too, is complex and may be outside the effects of neural pathways associated with caffeine effects as delivered by WGCP.
The nature and mode of delivery of caffeine may influence its effect on executive performance. According to the packaging label of GoBean®, the delivery of caffeine using WGCP provides the “natural caffeine that is deep within the fiber of the bean”. Caffeine extracts rather than “natural caffeine” are typically used in commercially sold liquid products. The study did not compare WGCP with other caffeine delivery systems. It is possible that the effects on sustained attention and the reported positive qualitative effects may be a function of both caffeine and the nutrients that are not available in extracts. In addition, effects of WGCP may also be due to the mode of ingestion. The method of delivery—taking a capsule vs drinking a liquid—may represent a distinct difference in caffeine effects. 24 Although the features unique to WGCP are interesting, further study is required as this investigation does not provide information on the addition of nutrients in caffeine or on the delivery provided by WGCP; further investigation is required.
Research is certainly required to fully appreciate the different effects on attention and behavior associated with WGCP. Many studies of the effects of caffeine were conducted more than a decade ago, however, and these relied on extract rather than on caffeine occurring in its natural state. Given the variety of availability of caffeine products from diet supplements to energy drinks, study of new delivery systems of the raw bean is warranted especially as this relates to adverse events.
A consideration of the study is the carryover of WGCP effects from one phase of the manipulation to the other. This problem was handled in the counterbalanced repeated measures design of the study. In future studies, however, length of phases may be varied to determine WGCP latency effects (ie, onset of action of WGCP), and residual effects (ie, persisting WGCP effects during placebo phase) after active WGCP is terminated. Both latency and residual effects must be investigated to engage larger N trials to ensure safety of subjects and to determine timeliness of active WGCP exposure.
An interesting finding is that seven subjects (50%) were unable to discern between placebo and active WGCP, and the remaining subjects discerned only the moderate WGCP dose but not the low dose. Many factors impact whether a person recognizes changes in personal physiology; factors such as reaction sensitivity, ability to self-observe bodily changes, the opportunity to note changes (eg, a busy day full of attention occupying activity impairs self-monitoring vs a relatively calm, predictable day that allows it) and the same may explain this phenomenon.
In initial studies, it is often important to use specific measures to look for signal. Broader measures, while ecologically more valid, also run the risk of having loss of signal due to the complexity of measurement and the fact that the treatment effect may only tap a part of the broader measure. Thus, the next steps in examining this product will be to use broader, more ecologically valid measures. Systematic investigation with a greater number of participants is warranted given this initial finding from small N research.
Although many individuals acknowledge the beneficial effects of caffeine, further study of attention enhancers on academic tasks would be helpful. Increased availability of effective stimulant or arousal agents to students in higher education is already occurring,26–28 and understanding of proven, useful practices is helpful.
Limitations
Several limitations are extant in the study. First, it relied on subjective report of the intake of illicit substances and coffee rather than on urine drug screening. To ensure that other ingested substances were not used, future studies should include drug screening before each administration of the dependent measure. Second, while we can never completely rule out the possibility of practice effects, the pattern of findings makes it unlikely that practice effects substantively influenced the results. Two of the neurocognitive constructs showed worse performance on low dose and improved performance on moderate dose despite more exposure to the CANTAB. This pattern is inconsistent with practice effects. In addition, no studies to date confirm reliability of the CANTAB over many repetitions (in this case, six times in the treatment phase). In part, this limitation is mitigated because even if practice effects were present, differences unattributable to practice effects still emerged. A fourth consideration of the study is the varying development of frontal lobe activity in young adults. Some young adults may have more advanced executive functions than others and this variable is not discernible in a small group approach; this confound is managed in the study as subjects acted as their own controls. Also, subject variability is reflected in that only half the subjects correctly identified the presence of moderate-dose WGCP. Qualitative effects were only assessed in a rudimentary way, thus the effects reported here may vary with other factors that make an individual a good self-observer. Future studies may consider using a measure of the sensitivity of individuals to assess their own subjective reactions. Finally, future studies should be conducted to determine whether the administration time of WGCP affects results.
Author Contributions
Conceived and designed the experiments: MM. Analyzed the data: TF. Wrote the first draft of the manuscript: MM. Contributed to the writing of the manuscript: MM, TF, CS, AM. Agree with manuscript results and conclusions: MM, TF, CS, AM. Jointly developed the structure and arguments for the paper: MM, TF. Made critical revisions and approved final version: MM, TF, CS, AM. All authors reviewed and approved of the final manuscript.
