Abstract
Small cell lung cancer (SCLC) is a very aggressive cancer with poor outcome if left untreated, but it is also one of the most chemotherapy responsive cancers. Overall it has a very poor prognosis especially if it is chemotherapy resistant to first line treatment. Second line chemotherapy has not been very beneficial in SCLC as opposed to breast cancer and lymphoma. In the last few years topotecan is the only drug that has been approved by the food and drug administration (FDA) for the second line treatment of SCLC but in Japan another drug, amrubicin is approved. There are many combinations of different chemotherapies available in moderate to high intensity, in this difficult to treat patient to overcome the chemo resistance, but many of these studies are small or phase II trials. In this article we have reviewed single agent and multidrug regimens that were studied in both chemo sensitive and refractory setting, including the most recent clinical trials.
Introduction and Background
Lung cancer is the leading cause of cancer related mortality in US. 1 Small cell lung cancer (SCLC) which is the second most common type after non-small cell lung cancer (NSCLC), is almost always seen in patients with history of smoking and is generally very chemotherapy sensitive. Most SCLC patients are present with systemic disease with both micro and macro metastatic deposits. It is one of the most common cancers than can cross the blood brain barrier with frequent metastasis to the brain. In one report, approximately 18% of patients were found to have brain metastasis at time of diagnosis and the 2-year cumulative risk of brain metastasis reached 49% for patients with limited disease and 65% for patients with extensive disease, this can go up to 80% after two years of survival. 2 For this reason we screen all patients with brain imaging at the time of diagnosis and use prophylactic cranial irradiation (PCI) as part of their treatment, for those without brain metastasis.
In recent years, incidence of this disease has dropped dramatically as shown by SEER data mainly because of drop in tobacco use in the last few decades or perhaps because of the change in cigarette composition. 3 Proportion of SCLC of all lung cancers decreased from 17.26% in 1986 to 12.95% in 2002. Of all patients with SCLC, the proportion of women with SCLC has increased from 28% in 1973 to 50% in 2002. 3 SCLC is staged either as limited stage or extensive stage. Limited disease (LD) is confined to one hemi-thorax (but may include contra lateral mediastinal lymph node), without pleural or pericardial effusion which can be encompassed in one radiation port. Disease not meeting these criteria is called extensive disease (ED). About 30% of SCLC patients are present with LD and 80%–90% of these respond to combination chemotherapy with radiation. In turn 40%–70% achieve complete remission, with median survival of 12–20 months, and two and five year survival of 20%–40% and 5%–10%, respectively. Extensive stage is present at diagnosis in more than 60%–70% of cases, and median survival for such patients is about 9.3 months, while 5-yr survival is 2% and 1% at ten years. 4
Current standard of care for limited stage is platinum based chemotherapy with radiation and for extensive stage is platinum based chemotherapy alone. In both stages if patient has had at least a partial response they should be offered PCI, as it has shown to prolong overall survival. 5 Unfortunately, even though we can get complete response (CR) or partial response (PR) in most patients, majority of these will relapse and die of their disease. Options for refractory or relapsed disease depend on the patient's overall general condition, sensitivity to first line chemotherapy and duration of remission.
Relapsed SCLC
Although initially SCLC is very chemotherapy sensitive, many patients tend to relapse. Majority of the patients relapse (about 80% with LD and almost all with ED). 6 If they relapse within 60–90 days of treatment, they are called chemotherapy refractory as opposed to chemotherapy sensitive patients who relapse after this time. Patients who are chemotherapy refractory tend to have a poor overall survival, with response rate to further chemotherapy being around 10% or less, but for chemo sensitive patients it can be up to 25%. 7 Studies have shown that 2nd line treatment can give significant palliation in these patients.8,9
Treatment Options for Relapsed SCLC
We have very few treatment options to improve survival over best supportive care.8,9 Topotecan and amrubicin are two agents currently in use for relapsed SCLC. Topotecan is approved in US and amrubicin is approved only in Japan.
Re-induction
As in any other tumor, if patients are sensitive to first line chemotherapy and are able to tolerate it, we can always use the same treatment with good response rate. The most important factor in predicting future response is the duration of first response. 10 A retrospective multi institutional analysis on outcomes of small cell lung cancer with second line chemotherapy involving 161 patients showed an overall response rate of 22.2%. Response rate was better for platinum sensitive patients who are re-challenged with platinum based chemotherapy as opposed to platinum refractory (34.5% vs.17.5%). OS (overall survival) was also better in these patients 9.2 vs. 5.8 months respectively. In this study, response to first line treatment and performance status (PS) were the only independent prognostic factors. 11
Topotecan Based Treatments
Topotecan, a water soluble semi synthetic derivative of camptothecin is a specific inhibitor of topoisomerase I, and is the most studied drug in relapsed SCLC. In a phase II study by Ardizzoni et al involving 92 patients reported a response rate of 6.4% in refractory and 37.8% in sensitive disease, with dose of 1.5 mg/m2 for five consecutive days every three weeks. 12 Treat et al retrospectively analyzed data from five clinical trials that included 479 patients who were treated with single agent topotecan, had ORR (overall relapse rate) of 14% with eleven complete responses among good PS patient, but it was 17% with three CR in patients with PS two. In sub analysis, chemotherapy sensitive and refractory patients responded about the same regardless of their PS, good PS had ORR of 18% and 3% but in poor PS it was 24% and 3%, respectively. Treatment benefited patients regardless of PS, with median overall survival of 36.3 weeks, 25.4 weeks, and 16 weeks for PS 0, 1, and 2 patients, respectively. Major toxicity was hematological in about 42% of patients and febrile neutropenia occurred in 3% or 4% depending on the PS and treatment related death occurred in 8% of patients. 13 This retrospective analysis suggests that topotecan has clinical benefit mainly among chemo sensitive patients even if they have poor PS. (See Table 1).
Topotecan based treatment as second line for small cell lung cancer.

Abbreviations: S, sensitive; R, refractory; BSC, best supportive care; T, topotecan; CAV, cyclophosphamide adryamicin and vincristine; W, week; M, month; ORR, over all response rate; TTP, time to progression; TRD, treatment related death; OS, overall survival.
Intravenous versus Oral Topotecan
Some patients prefer to take PO drugs especially if they can avoid IV lines and frequent clinic visits. Eckardt et al did an open label, randomized, phase III study comparing oral and IV topotecan in patients with SCLC sensitive to initial chemotherapy on 309 patients, included both chemo refractory and sensitive patients. Treatment with oral topotecan (2.3 mg/m2/d) on days one through five or IV topotecan (1.5 mg/m2/d) on days one through five every 21 days was given. Patients who received this regimen had median OS of 33.0 weeks and 35 weeks respectively. Oral topotecan had one and two year survival rates of 32.6% and 12.4% and for IV topotecan it was 29.2% and 7.1% respectively. Major toxicities were grade 4 neutropenia and thrombocytopenia in 47% and 29% in PO group and 64% and 18% in IV treatment group respectively. Other side effects included nausea (43% oral, 42% IV), fatigue (31% oral, 36% IV), and diarrhea (36% oral, 20% IV). This study showed that oral topotecan can be given to these patients with manageable toxicity instead of IV if needed. 14 A systematic review to look at economical stand point and clinical benefit by Hartwell et al showed that oral topotecan for patients with relapsed disease was associated with improved outcome and four months gain of life, but with increased cost. 15
Oral Topotecan versus Best Supportive Care
It is always important to determine if giving chemotherapy or supportive care to the patient is better due to side effects, cost, and the follow up needed for chemotherapy. This question was answered in a randomized controlled phase III trial by O’ Brien et al involving 141 patients with relapsed SCLC patients, who were not candidates for standard IV chemotherapy. They were randomized between best supportive care (BSC) or oral topotecan (2.3 mg/m2/d), days one through five, every 21 days. They reported median overall survival of 25.9 weeks and 13.9 weeks, among oral topotecan and best supportive care respectively (P = 0.01). Response rate was not unexpected, 7% had partial response and 44% had stable disease in topotecan group. Those who received topotecan had better quality of life and slower deterioration. Overall toxic deaths occurred in 6% of the patients in the topotecan arm. Mortality rate within 30 days of chemotherapy were 13% in topotecan and 7% in BSC group. This study provides evidence for use of topotecan instead of best supportive care if patient agrees and are able to afford the cost. Especially since it can help with clinical symptoms and can prolong their life by few weeks. 8
Weekly Topotecan
Many doses and schedules of topotecan have been investigated to see if they have similar clinical benefit with improved side effects profile or more convenient. Weekly treatment is an option in ovarian cancer, and this approach was investigated in SCLC as well. So far we do not have enough clinical studies to answer this question. Shipley et al did a phase II study, and presented an abstract regarding 103 patients with SCLC who had sensitive and resistant relapsed disease and received topotecan (4 mg/m2) IV over 30 minutes weekly for twelve weeks. ORR was 13 and 3% in sensitive and resistant patients, respectively. This study showed similar response rate when compared to historical data and was considerably less myelotoxic. 16 However, in another published phase II study on weekly topotecan, (4 mg/m2) IV on days 1, 8 and 15 every four weeks, patients who had one prior chemotherapy did not show any clinical benefit. None of the patients responded to topotecan and four had stable disease. 17 Since these two studies are conflicting and no phase III studies published, it is not a favorable option at this time.
Topotecan vs. CAV
Some patients are still able to take multi drug regiments when they relapse therefore it is important to know if single agent is equal, worst or better than combination. In a large randomized controlled clinical trial with 211 patients, single agent topotecan was compared with doxorubicin, cyclophosphamide, and vincristine (CAV) in patients with chemo sensitive disease. Patients received either topotecan (1.5 mg/m2) as a 30-minute infusion daily for five days every 21 days or CAV (cyclophosphamide 1,000 mg/m2, doxorubicin 45 mg/m2, and vincristine 2 mg) infused on day one every 21 days. Response rate was 24.3% and 18.3% for topotecan and CAV, respectively (P = 0.285). Interestingly, their medial survival was very similar with 25 weeks for topotecan and 24.7 weeks for CAV (P = 0.795). Importantly, topotecan gave better symptom control compared to CAV. Major toxicities included grade 4 neutropenia seen in 37.8% of topotecan arm versus 51.4% of CAV arm (P < 0.001). But Grade 4 thrombocytopenia and grade 3 or 4 anemia occurred in 8% and 17.7% of topotecan group but among CAV ground it was only 1.4% and 7.2%. (P < 0.001). 9 This study established topotecan as the first choice in relapsed SCLC (if they are chemo sensitive to 1st line treatment) especially since it has shown to improve symptoms as compared to multi agent chemotherapy with manageable side effects.
Low dose Topotecan
Lower-dose topotecan regimens have been evaluated in an attempt to minimize hematologic toxicities and to maintain efficacy. In a phase II study by Koschel et al, low dose topotecan IV (1.25 mg/m2) on days one to five of a 21 day cycle in patients with SCLC reported overall response rate of 15% and median OS of 22.4 weeks which was similar to results reported in studies using the standard regimen. 18 Tadeka et al in another phase II trial in Japan on 53 patients who were administered much lower dose of topotecan (1.0 mg/m2/day) for five consecutive days every three weeks in relapsed SCLC patients. Notable major toxicity was grade 4 neutropenia (24%) thrombocytopenia (5%) and anemia (3%). They reported an overall response rate of 26% (26% PR and 42% SD) with median progression free survival of 4.3 and OS of 8.6 months. 19 Perez-Soler et al tried (1.25 mg/m2) IV on patients refractory to platinum based treatment and showed a response rate of 11% partial 7% minor and 17% stable disease, they included 32 patients for this study. Grade 3 and 4 neutropenia and thrombocytopenia were 70% and 31% respectively. 63 These trials were very interesting since they showed similar response rate and better side effect profile than historical data. But these doses have not been validated in phase III trials. From these three studies, we can conclude that low-dose topotecan therapy may be appropriate for patients who exhibit risk factors for treatment-related myelosuppression such as advanced age, extensive past treatment, or renal impairment. 20
Topotecan Combinations in Relapsed SCLC
To improve clinical response of topotecan it has been studied in combination with other chemotherapeutic agents in relapsed and refractory setting.
Topotecan and Platinum Agents
Historically, platinums have been key drugs for combinations. Topotecan in combination with cisplatin was studied in a phase II trial involving 110 patients with chemotherapy sensitive(s) and refractory(r) relapsed SCLC. Patients were given cisplatin (60 mg/m2) on day one and topotecan daily IV infusion at (0.75 mg/m2) from days one to five every three weeks. Chemosenstive patients had better response rate of 29.4% and surprisingly RR for chemo refractory patients was 23.8%.
Main toxicity included myelosupression with grade 4 neutropenia in 62% and 49% along with grade 4 thrombocytopenia in 54% and 44% in sensitive and refractory patients respectively. Neutropenic fever occurred in 19% of sensitive patients and in 15% of refractory patients and five patients died from treatment related toxicity. Median OS was very similar with 6.6 months and 6.1 months in each group respectively. 21 In an another study with 34 patients who were treated with much lower dose of cisplatin (20 mg/m2) and topotecan (0.9 mg/m2) given on days 1–3 every three weeks. Overall response rate was 18% in chemo sensitive and 8% in chemo refractory patients. Median OS was 7.8 and 6.2 months for each group respectively. Major side effects were myelosupression, much lower than above study, and no toxic death related to treatment, they were grade 3–4 anemia (15%), thrombocytopenia (15%) and neutropenia (42%). 22 Carboplatin was also tried in combination with topotecan, phase I study of topotecan 0.85 mg/m2, on days one to five and carboplatin area under curve 5 every three weeks showed a partial response of 17.2% and median OS of 11 months. 23 It is important to note from these studies about the response rate on chemo refractory patients and overall survival and this combination is an option in both chemo sensitive and chemo refractory patients.
Topotecan and Vincristine
This combination was studied in eighteen patients who relapsed after carboplatin and etoposide. Topotecan (1.5 mg/m2) IV on days 1–3 and vincristine (1 mg) IV on either days 1, 3 or days 1, 4 every 21 days were given. This regimen gave an overall response rate of 19% with median OS of five months. The treatment was well tolerated. Patients had manageable toxicities including Grade 3 and 4 neutropenia (31%), anemia (38%) and thrombocytopenia (50%). 24
Weekly Topotecan and Gemcitabine
To see if there can be improvement upon single agent topotecan there was a phase II study of combinations of this drug with gemcitabine by Domine et al from Spain. Their regimen included topotecan at dose of (4 mg/m2) IV on days one and eight, and gemcitabine (1,250 mg/m2) on days one and eight every three weeks, gave a RR of 12%. They concluded that adding gemcitabine to topotecan does not improve efficacy of topotecan. 25
Overall, these studies showed that topotecan is an active agent in relapsed SCLC and better in chemo sensitive patients. Adding second chemotherapy agent to topotecan may or may not have additional benefit, but certainly gives more toxicity to these patients. It is possible that combination with platinums work due to synergism with topotecan as compared to gemcitabine.
Amrubicin Based Treatments a New Hope
Amrubicin is a synthetic anthracycline and its structure is based on doxorubicin in which the hydroxyl group at position 9 has been replaced by an amino group to enhance its efficacy. In vivo, studies showed amrubicin has more potent antitumor effects and lower toxic effects on the heart, liver and kidneys as compared to doxorubicin. This drug is schedule dependant as better antitumor effect is seen with administration over five days. It acts on topoisomerase II, stabilizing a cleavable complex. 26 It has been extensively evaluated in Japan, where it is approved for patients with relapsed SCLC. Most common side effect is myelosupression. See Table 2.
Amrubicin based treatment as second line for small cell lung cancer.
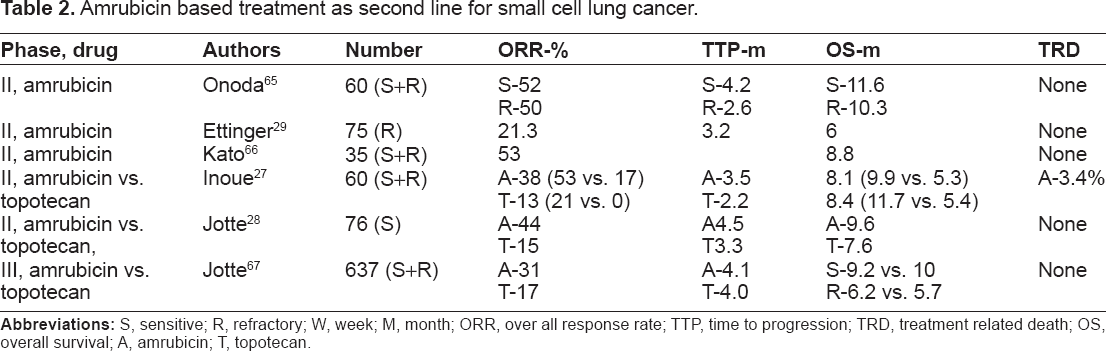
Abbreviations: S, sensitive; R, refractory; W, week; M, month; ORR, over all response rate; TTP, time to progression; TRD, treatment related death; OS, overall survival; A, amrubicin; T, topotecan.
In a phase II randomized study from Japan, 60 SCLC patients previously treated with platinum-containing chemotherapy were randomly assigned to amrubicin (40 mg/m2) on days 1–3 or topotecan (1.0 mg/m2) on days 1–5. This treatment gave an overall response rate of 38% vs. 13% with amrubicin and topotecan respectively and in sensitive relapse patients it was 53% vs. 21% but it was much lower in refractory relapse with 17% vs. 0% respectively. Median PFS for amrubicin and topotecan respectively were 3.5 and 2.2 months. This study showed amrubicin improved OS in Japanese patients though it is noteworthy that dose of topotecan was lower than used in other clinical trials. 27
In an another phase II randomized study conducted in US on 76 SCLC patients who were sensitive to first line platinum based therapy were treated with amrubicin infusion (40 mg/m2/d) on days one through three, every 21 days or topotecan (1.5 mg/m2), on days one through five, every 21 days. This study also showed a higher ORR among amrubicin compared to topotecan, it was 44% vs. 15% (P = 0.021). Median survival was 9.2 months vs. 7.6 months, and median PFS was 4.5 vs. 3.3 months. Major toxicity included grade 3 or 4 neutropenia was 78% and 61% and grade 3 or 4 thrombocytopenia was 61% and 39% in topotecan and amrubicin group respectively. 28
In another phase II study recently published on 75 patients with platinum refractory SCLC who were treated with single agent amrubicin ORR was 21.3% with 1 CR and OS of 6 months with PFS of 3.2 months. Interestingly, in 43 patients who never responded to first line therapy ORR was 16.3%. Grade 3 or 4 adverse events included neutropenia (67%), thrombocytopenia (41%), and anemia (30%), with febrile neutropenia in 12% of the patients. There was no decrease in mean Left ventricular ejection fraction (LVEF) with cumulative amrubicin doses exceeding 750 mg/m2. This was a very promising study showing activity of single-agent amrubicin when used as second-line therapy in patients with platinum refractory SCLC. 29 Importantly, no cardio toxicity was seen in any of the above mentioned amrubicin trials.
Very recently, a first phase III randomized controlled study by Jotte et al was presented at ASCO 2011, comparing amrubicin and IV topotecan. This study had 637 patients including both refractory and sensitive patients to 1st line chemotherapy. ORR was 31% and 17% for amrubicin and topotecan, respectively (P-0002). OS was 7.5 and 7.8 among each group (P-0.17) and in subgroup analysis OS in sensitive patients was 9.2 vs. 10 months (P-0.62) but among refractory patients it was 6.2 vs. 5.7 (P = 0.047) with 18 month OS superior for amrubicin group with 12% and 0% for topotecan (P = 0.0006). Major toxicities were grade 3 or 4 neutropenia 41% vs. 53%, thrombocytopenia 21% vs. 54%, anemia 16% vs. 30% and febrile neutropenia 10% vs. 4%. This study definitely demonstrates the benefit of amrubicin, especially among refractory patients, with manageable toxicities. 67
Topotecan vs. Amrubicin
From all the clinical trials available, we can conclude amrubicin has clinical benefits in relapsed SCLC and is comparable to topotecan with better reported outcomes in chemotherapy refractory patients with manageable side effects. No cardio toxicity was seen in any of the clinical trials so far conducted. This drug is active in refractory patients which is a challenging group of patients with dismal prognosis. It would be interesting to know if there is additive benefit when amrubicin is combined with other chemotherapeutic agents especially platinum or topoisomerase inhibitors.
Taxanes as Single Agents
The principal mechanism of action for taxanes including paclitaxel and docetaxel is the disruption of microtubule function. These two drugs have very good response rate in many cancers even when used as second line or third line treatment. In a phase II study on 24 patients with refractory SCLC treated with paclitaxel (175 mg/m2) IV every three week had ORR of 29% with median OS of 100 days. 30 In an another study with 93 patients with refractory SCLC were treated with higher dose paclitaxel (200 mg/m2) every three weeks for up to six cycles. This regimen gave ORR of 20% and median TTP and OS of three and four months respectively with complications including febrile neutropenia (23%) and grade 3 or 4 neuropathy (8%), but no toxic deaths. This was a promising single agent study in chemo refractory patients but had a high incidence of febrile neutropenia. 31 Docetaxel has been studied in previously treated SCLC patients with a partial response of 25%, neutropenia was the significant toxicity. 32
Taxanes in Combination
Paclitaxel and Carboplatin
In a phase II study involving 35 SCLC patients refractory to cyclophosphamide, doxorubicin, and etoposide (CDE), combination of carboplatin and paclitaxel showed a response rate of 73.5% with two patients achieving CR. Paclitaxel was given at standard dose (175 mg/m2) and carboplatin at area under the curve of 7 every three weeks for five cycles. Main toxicity was hematological with grade 3 or 4 leucopenia in 33%, thrombocytopenia in 34%, and anemia was 17% with low grade neuropathy about 69%, no toxic death was reported. Median time to progression and OS were 21 and 31 weeks respectively. This study was very encouraging among CDE refractory patients with very high response rate. 33
Paclitaxel and Doxorubicin
Paclitaxel (175 mg/m2) combined with doxorubicin (40 mg/m2) was studied in 46 relapsed SCLC patients, and gave ORR of 41% with three complete remissions, of responders 14% were chemotherapy refractory patients and 52% were sensitive patients. Remarkable side effects included Grade 3 or 4 emesis and neutropenia in 11% and 80% of the patients respectively and 20% were hospitalized for neutropenic fever, one patient had grade 3 cardio toxicity and another one had grade 4 myalgia. 34 Though this combination had decent response, side effects were high, and many of our patients may not tolerate.
Paclitaxel and Gemcitabine
Results from a phase II study on 31 patients with relapsed SCLC treated with paclitaxel (175 mg/m2) on day one and gemcitabine (1250 mg/m2) on days one and eight showed ORR for chemotherapy sensitive disease was 60% and for refractory disease was 40%. Treatment was well tolerated with grade 3 or 4 neutropenia occurred in 11% of the patients. The median duration of response was five months and median time to progression seven month respectively. Non-hematological toxicity was very mild. 35 Gemcitabine and docetaxel combination was tried on 22 previously treated patients with SCLC in a phase II trial but did not have any PR or CR. 37
Paclitaxel, Ifosfamide, and Cisplatin
A phase II trial with 30 patients who had either progressed or relapsed after carboplatin and etoposide were treated with an intense regimen of paclitaxel (175 mg/m2) on day one, ifosfamide (5 g/m2) on days one and two, and cisplatin (100 mg/m2) divided over days one and two every 21 days with growth factor support. Overall response rate was 73% including 8 CR and 1-year survival rate was 12%. Grade 3 and 4 neutropenia and thrombocytopenia was seen in 73% and 27% patients respectively with 18% had febrile neutropenia 23% had grade 3 nausea and vomiting but overall no treatment-related deaths occurred. 36
Irinotecan
Irinotecan is a topoisomerase Iinhibitor and as a single agent has modest response in SCLC.38–40 It has also been studied in combination with other agents.
Irinotecan and Etoposide
Masuda et al evaluated topotecan and etoposide combination in 25 patients with refractory or relapsed SCLC. Irinotecan was given (70 mg/m2) IV on days one, eight and fifteen and etoposide (80 mg/m2) IV on days 1–3 every four weeks. Overall response rate was 71% with three patients achieving with median response duration and survival about 4.5 and 9 months respectively. Major toxicity was grade 3 and 4 neutropenia and thrombocytopenia which were seen in 56% and 20% of the patients, respectively. One patient died from treatment related death due to severe myelosuppression. 41 Interestingly, all these patients were exposed to etoposide previously and most likely a synergistic action contributed to this response.
Irinotecan and Platinum Based Combinations
Irinotecan and Cisplatin
Irinotecan and cisplatin are reported to act synergistically. Nakanishi et al conducted a phase II trial combining cisplatin and irinotecan in patients with refractory SCLC. They had 21 patients who had not responded to prior platinum based chemotherapy. Both cisplatin (30 mg/m2) and irinotecan (60 mg/m2) were administrated on days one, eight, and fifteen and the regimen was repeated every 28 days. ORR was 29% and the median survival time for all patients was 32 weeks. One and two year survival rates were 43% and 11%, respectively. Major toxicity included grade 3 or 4 leucopenia, anemia, thrombocytopenia, and diarrhea was seen in 43%, 38%, 19%, and 38% of patients, respectively. 42
This same regimen was tried in another study by Masahiro et al. But coinciding with the infusion and for four days thereafter the anti diarrheal program was given using orally administered sodium bicarbonate, magnesium oxide and basic water. Twenty-five patients who had had prior treatments with etoposide and platinum containing regimens (16 were refractory patients) ORR was 80% and the median time to progression and OS were 3.6 and 7.9 months, respectively. Main Toxicity was grades 3 or 4 neutropenia seen in 36% of the patients, with one febrile neutropenia and grade 3 diarrhea occurred in only 8% of patients, this was better due to the anti diarrheal regime. 43 These two studies provide valuable information that weekly irinotecan plus cisplatin, are well tolerated and work synergistically in combination with manageable toxicities and is active in refractory or relapsed, especially platinum refractory.
Irinotecan in combination with cisplatin and etoposide
Goto et al did a multi institutional phase II study involving 40 patients with chemotherapy sensitive relapsed SCLC using irinotecan in combination with cisplatin and etoposide (PEI). All 40 patients had previously been treated using platinum based chemotherapy and eight (20%) of these patients also received thoracic radiotherapy. They received cisplatin (25 mg/m2) weekly for nine weeks, etoposide (60 mg/m2) for three days on weeks 1, 3, 5, 7, and 9 and irinotecan (90 mg/m2) on weeks 2, 4, 6, and 8 with granulocyte colony stimulating factor support and ORR was 78% including five patients with CR. The median survival time was 11.8 months, and the estimated 1-year survival rate was 49%. Grade 3 or 4 neutropenia, and thrombocytopenia were observed in 73% and 33% of the patients, respectively. 44 This was a remarkable response rate but the trial was done in chemotherapy sensitive patients and most of them had good PS whereas in our everyday practice many patients with relapsed SCLC have poor PS and may not be able to tolerate this much side effects.
Irinotecan and carboplatin
A phase II study reported by Hirose et al used irinotecan and carboplatin in 24 patients with relapsed small cell lung cancer. Patients were treated every three weeks with carboplatin (AUC 5) plus irinotecan 50 mg/m2 on days one and eight. Overall response rate of 68.2%, but most (92.3%) were chemotherapy sensitive. The median survival time was 194 days. The median survival time did not differ significantly between patients with sensitive disease (245 days) and those with refractory disease (194 days, P = 0.88). Toxicities included Grade 3–4 neutropenia (63%), thrombocytopenia (58%), anemia (67%), Grade 3 diarrheas (21%) and infections (13%). 45 Another recent phase II study by Chen et al using irinotecan and carboplatin in relapsed small cell lung cancer, had 80 patients and showed 50% response rate and OS of ten months, Major toxicities were grade 3 or 4 neutropenia (54%), thrombocytopenia (22%), anemia (13%), diarrhea (22%), and nausea or emesis (11%) and three neutropenic sepsis. 55
Other Combinations of Irinotecan
Irinotecan and gemcitabine
A phase II study evaluated the efficacy and safety of the irinotecan and gemcitabine combination in patients with relapsed and refractory small cell lung cancer. Gemcitabine (1000 mg/m2) was administered IV followed by irinotecan (100 mg/m2) IV both on days one and eight every 21 days. OS rates were 31% in sensitive and 11% in refractory patients. Median survival and progression free survival time were 7.1 versus 3.5 months, and 3.1 versus 1.6 respectively. Primary grade 3 or 4 toxic effects for chemo sensitive and refractory patients were neutropenia (36% versus 43%) and thrombocytopenia (36% versus 26%). 46
Liposomal doxorubicin in combinations with irinotecan
Liposomal doxorubicin is a novel formulation of doxorubicin, in which the drug is, encapsulated in polyethylene glycol coated liposomes which results in better uptake in cancer cells as opposed to normal cells. Xendils et al did a phase II trial using pegylated liposomal doxorubicin in combinations with irinotecan as second line therapy in refractory SCLC. 31 patients with early relapse after first-line therapy with cisplatin and etoposide were treated with pegylated liposomal doxorubicin (15 mg/m2) and irinotecan (125 mg/m2) on days one and fifteen repeated every 28 days. Main toxicities were grade 3 neutropenia and fatigue in 6.5% and 23% of patients respectively. Though it was well tolerated, activity was only modest; with ORR of 12.9% with OS of 3.16 months. 47 Another study of single agent pegylated liposomal doxorubin also did not show any response in relapsed SCLC. 48
Combination of cisplatin, ifosfamide, and irinotecan
Fujita et al studied combination of cisplatin, ifosfamide, and irinotecan with G-CSF support in eighteen patients with refractory or relapsed small-cell lung cancer. Cisplatin (20 mg/m2) and ifosfamide (1.5 g/m2) were administered on days 1–4, and irinotecan (60 mg/m2) were administered on days one, eight, and fifteen given every four weeks. Overall response rate and survival time were 94.4% and eleven months respectively. Significant toxicities including grade 4 neutropenia and thrombocytopenia seen in 61% and 33% of the patients respectively and no treatment related death occurred in this group of patients who received this strong three drug combination. 49 This study is very encouraging with very good overall response, and manageable toxicity.
Other Combinations
CAV regimen
Combinations of cyclophosphamide, doxorubicin, and vincristine can be used with modest activity in patients who have had platinum based treatment. This was studied by Shepherd et al in 29 patients who received CAV after their tumors failed to respond to or relapsed after etoposide and cisplatin (VPP) or etoposide and carboplatin (VPC). Thirteen patients were treated following failure to respond to VPP or VPC and sixteen at the time of relapse. There were three CR and five PR and ORR was 28%. This study demonstrates that CAV has modest activity following failure of platinum based chemotherapy. 50 As mentioned earlier, another large randomized trial has already shown equal efficacy of single agent topotecan as compared to CAV with less toxicity thus limiting use of CAV in routine clinical practice. 9
CODE regimen
This study was undertaken by Kubota et al to determine the activity and toxicity of dose intensive weekly chemotherapy (cisplatin, vincristine, doxorubicin, and etoposide (CODE regimen) in seventeen recurrent SCLC patients. All seventeen patients had been heavily pretreated with some form of cisplatin-based combination chemotherapy. This regimen gave an overall response of 88.2% including 29% CR rate. Myelosuppression was very common with grade 4 leucopenia in 76% of the patients but no treatment related deaths. 51
VIP regimen
Faylona et al did a phase II study to determine the activity and toxicity of oral etoposide, with IV ifosfamide, and cisplatin in previously treated, recurrent SCLC. 46 patients were enrolled to receive oral etoposide (37.5 mg/m2/d) for 21 days, ifosfamide (1.2 g/m2/d) for four days, and cisplatin (20 mg/m2/d) for four days, repeated every 28 days. Objective response was 55% including 14% CR. Median PFS and over all median survival were 20 and 29 weeks respectively. There were six treatment related deaths most of them due to sepsis. Though response rate was good but this regimen had significant treatment related side effects. 52
These aggressive combinations work in second line setting but at the expense of high toxicity.
Oral Etoposide
Although most patients receive IV etoposide, there is some evidence that oral etoposide can be an option as well. In a phase II study by Einhorn et al, 26 previously treated patients with refractory small cell lung cancer were given daily oral etoposide (50 mg/m2/d), 25 patients had prior exposure to cisplatin plus etoposide and fourteen had prior therapy with CAV (cyclophosphamide, doxorubicin, and vincristine). Interestingly, their overall response was 23% including one patient who had CR. 53 Johnson et al also looked into this idea as well. Twenty two patients (most of them received etoposide IV as part of induction treatment) with recurrent small-cell lung cancer were treated with single agent etoposide (50 mg/m2/d) by mouth for 21 consecutive days repeated every 28 days depending on tolerance and response. ORR was 36% and median survival was more than 3.5 months and response duration of four months. Grade 3 or 4 neutropenia and thrombocytopenia were seen 18% and 25% of the patients respectively. There were two toxic deaths due to sepsis and five patients required hospitalizations for neutropenia and fever. 54 This is an impressive response, in those patients receiving oral etoposide after previous exposure to IV etoposide, but side effects are concerning.
Experimental Treatments
As we learn more about various neoplasms at the molecular level, specific targeted therapies are being developed. These include tyrosine kinase inhibitors, apoptotic stimulants and anti angiogenesis agents alone or in combinations, but so far none of the treatments have provided clinical benefit in terms of symptom relief or survival in SCLC.56–60 Picoplatin is a new platinum compound designed to overcome platinum resistance and has shown less neurotoxicity and nephrotoxicity than other platinum agents. But a randomized control study with picoplatin versus best supportive care in chemo sensitive and refractory patients, failed to show any improvement in overall survival.61,62
Conclusion
Incidence of SCLC is certainly decreasing, due to decreasing number of smokers and there is a slight increase in overall survival over last few years as noted from most recent SEER data. Our understanding of this cancer has improved over the last few years but this understanding has not translated into improved treatment regimens or finding new drugs with better outcome. Response to 1st line chemotherapy and the duration of the response are the most important factors in overall survival and response to second line treatment. Though we have many drugs that have clinical activity not many large randomized control studies have been done except in the case of topotecan and amrubicin. Combinations of drugs have shown significant response rate but their use in previously pre-treated patients with overall poor clinical condition has not been feasible, though in selected patients combinations can be considered especially in chemo refractory patients. Topotecan is current standard of care in chemosenstive patients and its use is approved in US and other parts of the world. Amrubicin is only approved in Japan and further studies are ongoing. It has shown good response even in chemo refractory patients in phase II and III trials. It would be interesting to know if combination treatment with amrubicin gives better response and survival as compared to single agent. New clinical trials are a must, especially combinations to overcome chemo resistance of these tough to treat relapse or refractory small cell lung cancer.
Disclosure
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
