Abstract
Small-cell lung cancer (SCLC) is a highly aggressive and rapidly proliferative malignancy that has historically had limited therapeutic advancements. Recent advancements in the understanding of SCLC have led to attempts at subtyping the disease based on transcription factor characteristics, offering new insights into its biology and potential therapeutic targets. In addition, significant progress has been made in developing treatment regimens, providing new hope for improved patient outcomes. The introduction of immune checkpoint inhibitors, such as atezolizumab and durvalumab, in combination with traditional chemotherapy, has marked a significant advancement, demonstrating improved overall survival and progression-free survival compared to chemotherapy alone. Despite these advancements, the prognosis for extensive-stage SCLC (ES-SCLC), the more advanced form of SCLC, remains poor, highlighting the critical need for ongoing research and the development of novel therapeutic strategies. New treatment modalities, such as lurbinectedin and anti-Delta-like Canonical Notch Ligand 3 antibodies, are now included in the treatment options for refractory SCLC, and many more treatment strategies involving combination therapies are being studied. Advances in molecular profiling and the identification of biomarkers are aiding in the development of personalized treatment approaches. This review focuses on these recent advancements and emerging strategies in the treatment of ES-SCLC.
Plain language summary
Small cell lung cancer (SCLC) is a fast-growing and aggressive type of lung cancer that has had very few effective treatment options for many years. Recently, researchers have made progress in understanding this type of cancer better, including trying to classify it into different subtypes based on specific biological markers. The introduction of new drugs, like immune checkpoint inhibitors (e.g., atezolizumab and durvalumab), combined with traditional chemotherapy, has shown promising results by helping patients live longer compared to chemotherapy alone. Despite these advances, extensive-stage SCLC (ES-SCLC) still has a poor prognosis, which means more research is needed to find better treatments. New therapies, such as lurbinectedin and anti-DLL3 antibodies, have been added to the list of options for patients whose cancer has not responded to previous treatments. Researchers are also studying many new treatment combinations to improve outcomes. Advances in understanding the biology of ES-SCLC and identifying specific markers in patients are aiding in the personalization of treatment strategies to better address individual needs. This summary highlights the latest developments and ongoing research efforts aimed at improving outcomes for people with ES-SCLC.
Introduction
Small-cell lung cancer (SCLC) is an aggressive and rapidly proliferating, high-grade neuroendocrine carcinoma accounting for 15% of all lung cancers.1,2 SCLC patients commonly exhibit a history of heavy smoking, rapid tumor growth, early metastasis, and frequent symptom presentation. 3 For decades, molecular and genetic subtyping has not been feasible unlike non-small-cell lung cancer (NSCLC). 4 However, with the recent inclusion of immunotherapy in mainstay treatment modalities, the need for personalized therapy and biomarkers has significantly increased in extensive-stage SCLC (ES-SCLC). The inclusion of anti-PD-L1 antibodies in the treatment modalities for ES-SCLC has improved survival rates. However, nearly all patients experience a relapse, underscoring the need for the development of alternative treatment modalities. 5
Recently, there were important breakthroughs in treatment modalities in ES-SCLC. Lurbinectedin has been approved to be used as a second or more line ES-SCLC. 6 Tarlatamab, a bispecific antibody targeting Delta-like ligand 3 (DLL3), has been reported to show improved objective response rate (ORR) in previously treated ES-SCLC patients. 7 Combinatorial managements including lurbinectedin, antibody–drug conjugates (ADC), immunotherapy, and traditional chemotherapy are under study. Molecular subtyping based on transcriptional proteins related to SCLC proliferations 4 is expected to be used to categorize patients and contribute to more personalized treatment in SCLC which was not possible before.
The treatment landscape in SCLC is evolving rapidly, necessitating ongoing updates to reflect recent advancements in treatment modalities. This review highlights newly reported findings on therapeutic approaches in SCLC (Figure 1).
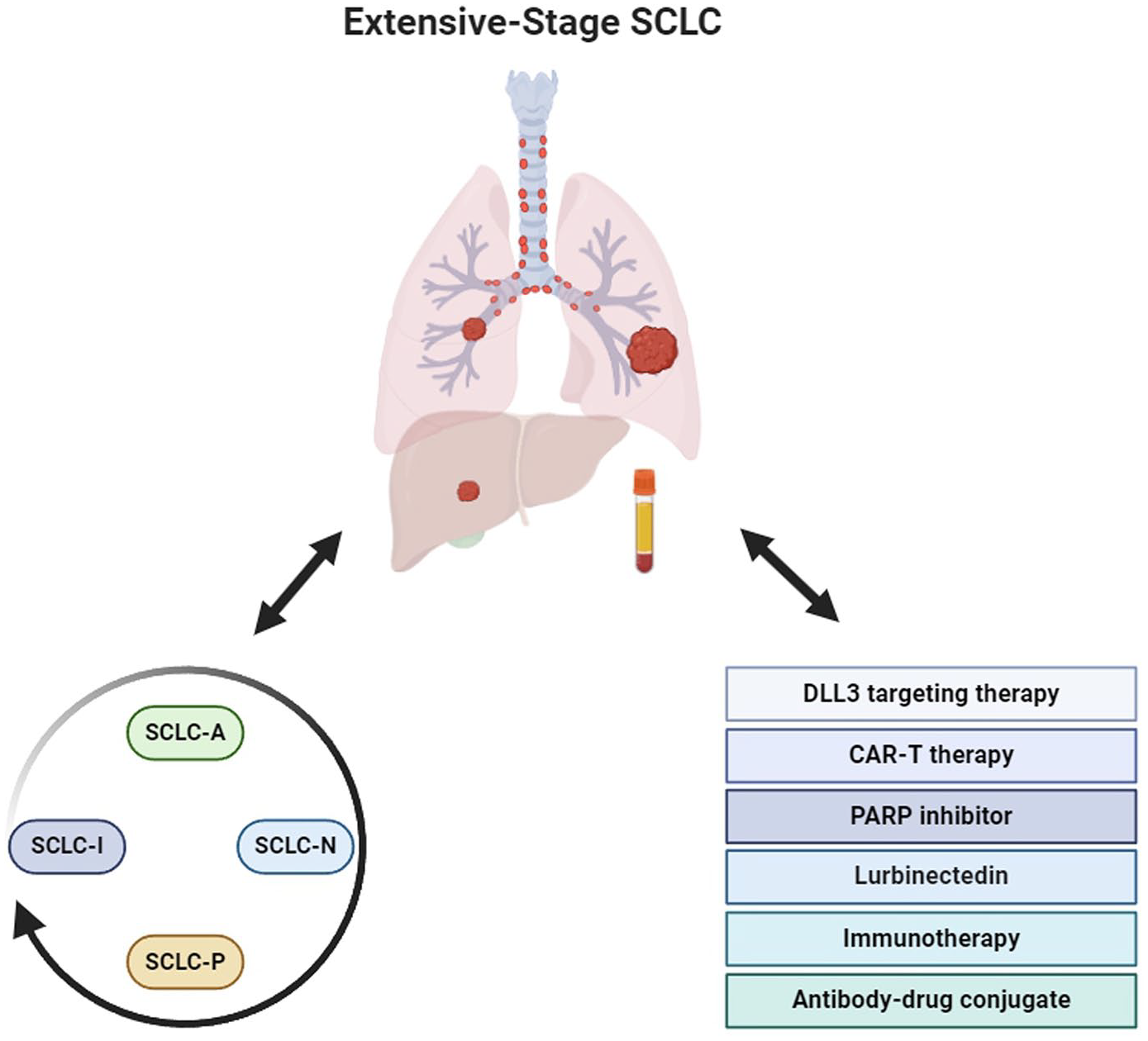
Therapeutic strategies for extensive-stage small-cell lung cancer based on molecular subtypes. Tissue and blood samples are used to determine SCLC subtypes guiding treatment selection, including DLL3-targeting therapy, CAR-T therapy, PARP inhibitors, lurbinectedin, immunotherapy, and antibody–drug conjugates. Subtype changes during treatment may require ongoing monitoring through blood samples to optimize therapy.
Molecular subtyping
The majority of SCLC express insulinoma-associated protein 1 (INSM1), indicating both high levels of achaete-scute complex-like 1 (ASCL-1) and neurogenic differentiation factor 1 (NeuroD1), as seen in certain neuroendocrine subtypes. 8 However, a small proportion of SCLC tumors exhibit low levels of INSM1, ASCL1, and NeuroD1. These tumors lack neuroendocrine markers and seem to belong to distinct subtypes characterized by high levels of yes-associated protein 1 (YAP1) and POU class 2 homeobox 3 (POU2F3). Recent studies including analyses of cancer cell lines, patient-derived xenografts (PDXs), genetically engineered mouse models, and primary human tumors, suggested distinct SCLC subtypes based on varying levels of four key transcriptional regulators: ASCL1, NEUROD1, YAP1, and POU2F39–11 (Table 1).
Summary of key findings and clinical implications of studies on SCLC subtypes.

ASCL-1, achaete-scute complex-like 1; BET, bromodomain and extra terminal; CP/ET, carboplatin/etoposide; IHC, immunohistochemistry; LTS, long-term survivors; NE, neuroendocrine; NEUROD1, neurogenic differentiation factor 1; NMF, non-negative matrix factorization; POU2F3, POU class 2 homeobox 3; SCLC, small-cell lung carcinoma; WTS, whole transcriptome sequencing; YAP1, yes-associated protein 1.
However, in the following studies, the reliability of YAP1 expression as a marker for the SCLC subtype has been questioned due to inconsistencies observed between immunohistochemistry (IHC) and gene expression profiling. While strong YAP1 RNA expression has been noted in some PDXs, corresponding protein expression has not been consistently observed in IHC, suggesting that YAP1 may not be a definitive marker for distinguishing SCLC subtypes. 10 Another subtype that showed low expression of all three transcription factor signatures (ASCL1, NEUROD1, and POU2F3) accompanied by an inflamed gene signature was termed SCLC-I, “I” meaning “inflammatory.”12–14 SCLC-I tumors have higher immune infiltrate and related activities when compared to other subtypes. 12 Gay et al. showed that SCLC-I was distinct from other subtypes in other features as well. Epithelial marker E-cadherin expression was extremely low in SCLC-I, while levels of mesenchymal markers vimentin and AXL were high. Based on significantly higher expression of both CD8A and CD8B, it was assumed that the SCLC-I subtype could benefit from combining immunotherapy with chemotherapy. They further demonstrated through a retrospective analysis of IMpower133 data that the SCLC-I subtype exhibited a significant overall survival (OS) benefit compared to all other subtypes in the EP + atezolizumab arm (hazard ratio (HR) = 0.566, 95% confidence interval (CI): 0.321–0.998), but not in the EP + placebo arm. This finding suggests that SCLC-I subtype classification may serve as a predictive biomarker for immunotherapy benefit. 12
Other studies have suggested that tumor-related inflammatory activity has a significant correlation with immunotherapy response. 23 In the phase Ib KEYNOTE-028 trial, which enrolled 475 patients with advanced solid tumors expressing PD-L1, the SCLC subgroup demonstrated an ORR of 33% to pembrolizumab. This trial highlighted the potential utility of pembrolizumab in a heavily pretreated population. Furthermore, gene expression profiling of tumor-infiltrating T cells revealed a T-cell-inflamed signature that was associated with favorable responses to pembrolizumab, suggesting its potential as a predictive biomarker for immunotherapy in SCLC and other malignancies. 24
Single-cell RNA sequencing (scRNAseq) analysis has demonstrated that SCLC tumors become more heterogeneous and may shift subtypes following platinum-based chemotherapy. Notably, in patients with a predominant ASCL1-expressing cell population (i.e., SCLC-A subtype), an increase in the proportion of triple-negative cells (i.e., SCLC-I subtype) was observed post-resistance to platinum-based chemotherapy. 12 Similar to past studies that have reported differences in Notch pathway activation between NE and non-NE types, the SCLC-I subtype exhibited characteristics indicative of Notch pathway activation.15,16 The SCLC-I subtype was found to be highly plastic and exhibited more aggressive traits. However, SCLC-I tumors have shown a favorable response to immune checkpoint inhibitors (ICIs) compared to other types. This suggests the importance of therapeutic strategies targeting the SCLC-I subtype induced by platinum-based chemotherapy.
Another study has shown that SCLC subtypes can be differentiated based on variations in DNA methylation. 17 In particular, utilizing the highly conserved nature of DNA methylation in plasma, a comparison was made between tissue-based subtype profiling and plasma-based classification, with remarkable concordance observed. Moreover, as previous scRNAseq-based research indicated, 12 heterogeneity within SCLC subtypes increased with treatment, and transitions from SCLC-A to SCLC-I subtype were also observed. This suggests the potential for subtype monitoring in SCLC using liquid biopsies during cancer treatment.
A study has suggested the potential role of epigenetic modulators, specifically JIB-04 and iBET-762, in altering transcription factors associated with SCLC subtypes, proposing their combination with existing therapies for SCLC. 18 JIB-04, an inhibitor of the Jumonji family of histone demethylases, was found to decrease ASCL1 expression and exert a moderate to minimal impact on YAP1 and NEUROD1 expression. Conversely, iBET-762, which inhibits bromodomain and extra terminal (BET) proteins, increased ASCL1 expression, decreased YAP1, and did not alter NEUROD1 expression. The integration of these epigenetic modifiers with conventional chemotherapy, consisting of etoposide and platinum, as neoadjuvant or adjuvant treatment strategies, was proposed. It was observed that JIB-04 as an adjuvant and iBET-762 as a neoadjuvant in combination with EP had a suppressive effect on tumor growth compared to EP alone, whereas the reverse combination seemed to promote tumor growth compared to EP alone.
A study analyzed the changes in SCLC subtype classifications before and after treatment within tissue samples. 19 Based on previous findings that IHC classifications of SCLC subtypes show a strong correlation with RNA-based results, the study utilized IHC for subtype classification. The results suggested that in most patients,10,20 SCLC subtypes remained unchanged pre- and post-treatment, implying that re-biopsy for the investigation of subtype changes due to therapy might not be necessary. This conclusion contrasts with the outcomes of prior studies and warrants cautious interpretation due to the small sample size and reliance solely on IHC analysis. Further investigation with larger sample sizes and integrated analytic methods could provide more definitive insights.
The proportions of each subtype vary based on the platform used (IHC, RNA-based analysis), the heterogeneity of the SCLC population, and the institutions involved. In a study by Park et al., which included 90 limited-disease and 162 extensive-disease cases, molecular subtyping using IHC revealed that the ASCL1 subtype (IHC-A) was the most prevalent (56.3%), followed by co-expression of ASCL1/NEUROD1 (IHC-AN, 17.9%), NEUROD1 (IHC-N, 12.3%), and POU2F3 (IHC-P, 9.1%). The authors also demonstrated a high level of concordance between IHC-based subtypes and subtyping results obtained through whole transcriptome sequencing and non-negative matrix factorization clustering methods. 21 In another study involving patients enrolled in the IMpower 133 trial, the proportions of SCLC-A, SCLC-NA, SCLC-P, and SCLC-I were 51%, 18%, 2%, and 28%, respectively, among patients who survived more than 18 months, compared to 60%, 20%, 8%, and 11% in non-long-term survivors, based on RNA-seq analysis. 22
Current and ongoing treatment strategies
Tables 2 and 3 show the results of key trials on ES-SCLC.
Results of the clinical trials for patients with ES-SCLC.

CR, complete response; CTFI, chemotherapy-free interval; DLL3, Delta-like Canonical Notch Ligand 3; DOR, duration of response; ES, extensive stage; LS, limited stage; NA, not applicable; NE, not evaluable; NR, not reached; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; PS, performance status; SCLC, small-cell lung carcinoma; SD, stable disease.
Ongoing clinical trials for patients with ES-SCLC.
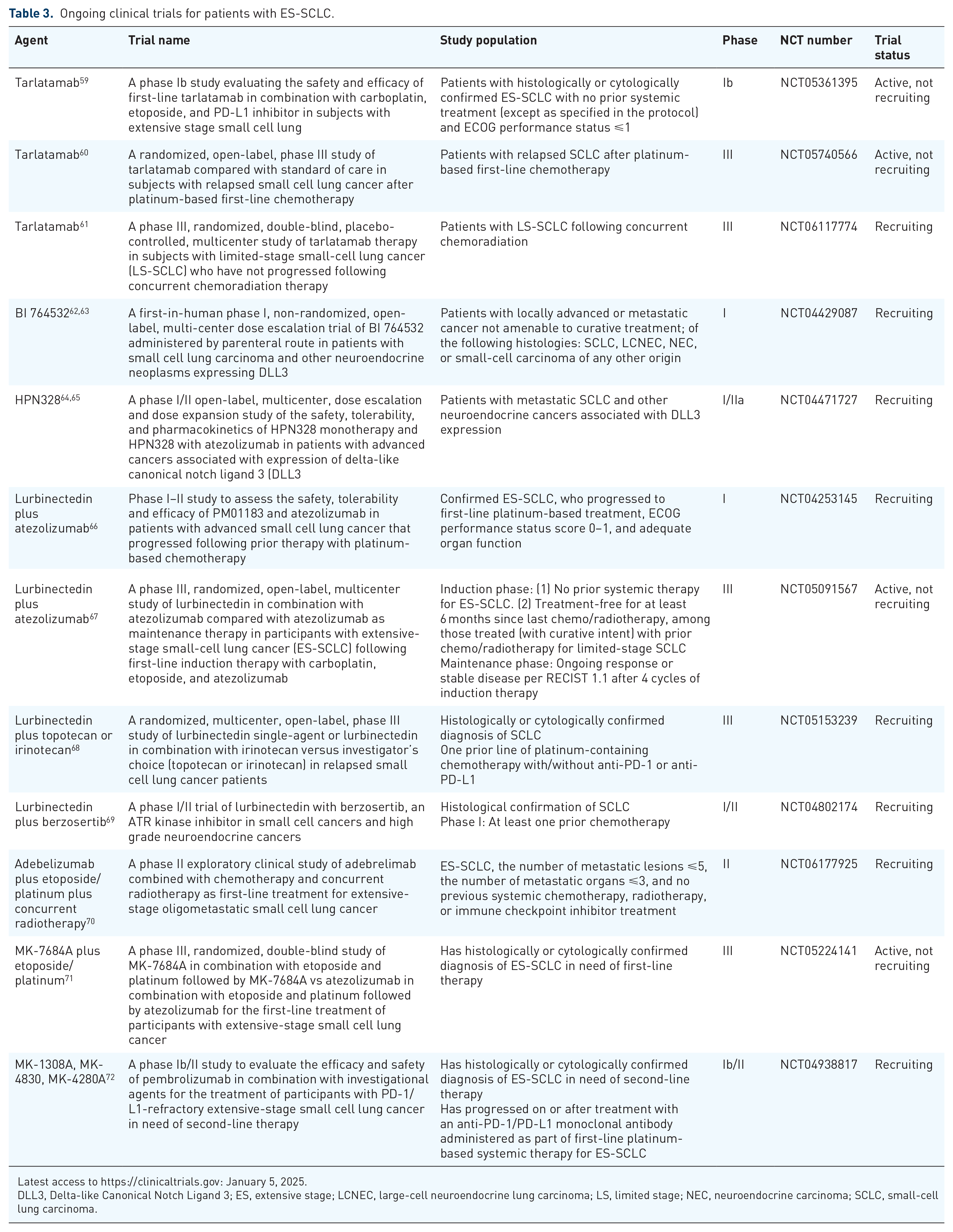
Latest access to https://clinicaltrials.gov: January 5, 2025.
DLL3, Delta-like Canonical Notch Ligand 3; ES, extensive stage; LCNEC, large-cell neuroendocrine lung carcinoma; LS, limited stage; NEC, neuroendocrine carcinoma; SCLC, small-cell lung carcinoma.
DLL3 targeting therapy
Underlying molecular and pathological pathophysiology
DLL3 is an atypical Notch ligand whose overexpression contributes to the growth of tumor cells and their migratory and invasive capacity in SCLC, 73 and DLL3 expression is associated with a neuroendocrine cancer phenotype.74,75 More than 80% of SCLC tumors demonstrate DLL3 mRNA and protein expression. IHC has shown consistent cytoplasmic and membranous DLL3 staining, exhibiting a high degree of uniformity across cancerous cells. On the other hand, in non-malignant tissues, DLL3 expression is limited to a few normal cell types, such as neurons, pancreatic islet cells, and pituitary cells, where it is exclusively localized to the cytoplasm.76,77 DLL3 showed potential advantage as a therapeutic target because its overexpression on the surface of SCLC cells with minimal to no expression on normal cells allows selective targeting of tumor cells. 78 Various drugs are developed and are being developed to target DLL3 including antibody–drug conjugates, bi-specific T-cell engagers, and CAR-T therapy.
Rovalpituzumab tesirine
Rovalpituzumab tesirine (Rova-T) is an ADC comprised of a DLL3-targeting immunoglobulin G1 monoclonal antibody linked with pyrrolobenzodiazepine.79,80 The phase II TRINITY study showed modest activity of confirmed ORR of 12.4% in pretreated DLL3-expressing recurred SCLC. 25 The phase III TAHOE study was conducted to compare the efficacy of topotecan and Rova-T as second-line therapy in DLL3-high SCLC. However, enrollment was discontinued due to the shortened OS observed in the Rova-T group. 26 In addition, the phase III MERU study aimed to assess the efficacy of Rova-T compared to placebo as maintenance therapy in ES-SCLC following platinum-based chemotherapy. Due to the absence of a survival benefit in the Rova-T group, the study failed to achieve its primary endpoint and was prematurely terminated. 27
Tarlatamab
Tarlatamab is a notable bispecific T-cell engager immunotherapy. It functions by directing a patient’s T cells to target cancer cells that express DLL3, independently from the major histocompatibility complex (MHC) class I. Tarlatamab has a dual binding ability to DLL3 on cancer cells and CD3 on T cells, resulting in T-cell-mediated lysis of the cancerous cells.7,28
In the phase I study focusing on dose exploration and expansion for tarlatamab in relapsed/refractory SCLC, the primary objective was to evaluate its safety across a dosage range from 0.003 to 100 mg. The study showed a manageable safety profile, with cytokine release syndrome (CRS) being the most common but generally controllable. Notably, tarlatamab achieved an ORR of 23.4%, including cases of complete responses. 28
In a phase II trial (DeLLphi-301 ClinicalTrials.gov number, NCT05060016), tarlatamab was evaluated for its effectiveness and safety in patients with previously treated SCLC. Administered intravenously every 2 weeks at doses of either 10 or 100 mg, tarlatamab achieved an objective response in 40% of the patients in the 10-mg group and 32% in the 100-mg group, with a median progression-free survival (PFS) of 4.9 and 3.9 months, respectively. Toxicity profiles were relatively satisfactory, and consistent with previous results, with the most common adverse events (AE) being CRS, decreased appetite, and pyrexia, mostly mild and occurring during the first treatment cycle. Due to the mechanism of action of tarlatamab, its safety profile includes risks such as CRS and immune effector cell-associated neurotoxicity syndrome (ICANS). Most CRS events were mild to moderate (grade 1 or 2), presenting with symptoms like fever, sometimes alongside hypoxia or hypotension, mostly occurred after 1st cycle, and were managed using supportive treatments such as acetaminophen, intravenous fluids, and glucocorticoids. Severe (grade 3 or higher) CRS was uncommon, seen in only 1% of patients receiving the 10-mg dose. ICANS and neurologic events were observed in 8% of patients at the 10-mg dose, with no grade 3 or higher events reported. Most cases of ICANS and CRS did not lead to discontinuation of treatment. Overall, the 10-mg dose of tarlatamab showed promising antitumor activity with durable responses and survival outcomes in previously treated SCLC patients, without any new safety concerns. 7
DeLLphi-303 (NCT05361395) investigates tarlatamab combined with PD-L1 inhibitors and first-line platinum chemotherapy, reflecting the current standard of care (SOC) in ES-SCLC. This phase Ib, multicenter, open-label trial evaluates tarlatamab in two settings: (a) combination therapy with carboplatin, etoposide, and a PD-L1 inhibitor (atezolizumab or durvalumab), followed by maintenance therapy with tarlatamab and PD-L1 inhibitor, and (b) combination with PD-L1 inhibitor followed by SOC chemo-immunotherapy as maintenance only. The primary objectives include assessing safety and tolerability and determining the phase II dose. 59 Interim results released at WCLC 2024 showed that 88 patients were enrolled, with 48 patients allocated to the atezolizumab arm and 40 patients to the durvalumab arm. Safety profiles revealed no unexpected treatment-related adverse events (TRAEs), with CRS and dysgeusia being the most common in the atezolizumab arm, and CRS and fatigue being most common in the durvalumab arm. Immune-related AEs were reported at 2.3%. The disease control rate was 62.5% for both the atezolizumab and durvalumab arms. The median PFS from the start of first-line maintenance was 5.6 months (95% CI: 3.5–8.5) for all patients, while the 9-month PFS rate was 39.3% (95% CI: 27.3–51.0). The 9-month OS rate was 89% for all patients, indicating encouraging outcomes. 81
The DeLLphi-304 trial (NCT05740566) is a randomized, open-label, phase III study evaluating tarlatamab against real-life SOC in around 700 patients with relapsed SCLC post-platinum-based therapy. Participants are assigned to either tarlatamab or SOC treatments (topotecan or amrubicin or lurbinectedin), stratified by factors like prior PD-1/PD-L1 treatment, chemotherapy-free interval (CTFI), brain metastases, and chosen SOC. The study’s main goal is to compare OS, with secondary goals including PFS and patient-reported outcomes related to symptoms, physical function, and quality of life. 60
Although not relevant to ES-SCLC, DeLLphi-306 (NCT06117774) is an ongoing phase III, multicenter, placebo-controlled study in limited-stage SCLC patients who have not progressed following concurrent chemoradiation therapy. 61
BI pipeline (BI 764532)
BI 764532 is bispecific DLL3/CD3 T-cell engager. In a human T-cell engrafted xenograft model, the DLL3/CD3 bispecific antibody upregulated infiltration of T cells into the tumor tissue leading to apoptosis of the tumor cells. 82 In a phase I multicenter, open-label, non-randomized, dose-escalation study (NCT04429087), the efficacy of BI 764532 monotherapy was assessed along with the determination of the maximum tolerated dose in patients with SCLC or other DLL3-positive neuroendocrine carcinomas. 62 As of December 28, 2022, 90 patients were treated with BI 764532, with a median treatment duration of 43 days. The most frequent treatment-related AEs included CRS (58% any grade; 2% grade ⩾3 toxicities), pyrexia (19% any grade), reduced lymphocytes (18% any grade; 14% grade ⩾3 toxicities), asthenia (17% any grade), and dysgeusia (14% any grade). CRS was managed with supportive care, corticosteroids, and anti-IL-6R antibodies. Tumor response data showed an ORR of 33% in SCLC and 22% in NEC across all regimens. The trial is ongoing, with further data updates to be presented. 63
Harpoon
HPN328 is a DLL3-targeted T-cell engaging agent with three binding domains: anti-DLL3 (for target engagement), anti-albumin (for half-life extension), and anti-CD3 (for T-cell engagement). 64
In preclinical studies, HPN328 has been shown to effectively inhibit the growth of subcutaneous SCLC xenograft tumors in mice. In addition, a pilot toxicity study in cynomolgus monkeys revealed that a single dose of HPN328 was well tolerated at both 1 and 10 mg/kg doses. 83 Clinical studies also showed some promising results. NCT04471727 is an ongoing phase I/II study assessing the efficacy of HPN328 in patients with metastatic SCLC and other neuroendocrine cancers expressing DLL3. Interim results in 2022 indicate that HPN328 is well tolerated. 64 The updated results presented at ASCO 2024 revealed a confirmed ORR of 50% (12/24) in SCLC among efficacy-evaluable patients treated in dose optimization cohorts, including one complete response. 65
CAR-T therapy
CAR-T therapy has demonstrated successful efficacy primarily in hematologic malignancies, and its application is expanding to solid tumors. Decreased treatment response in solid tumors has various reasons, most notably heterogeneity in presented antigens and relatively more immuno-suppressive tumor microenvironment. 84
Recently, a preclinical study was published in Clinical Cancer Research. In the study, a diverse range of anti-DLL3 scFv-based CARs were tested for their effectiveness against SCLC in both lab and animal models, and potential toxicity to the brain and pituitary was also assessed. Results showed that certain CARs were particularly effective against low DLL3 density targets, achieving substantial tumor reduction in SCLC models without observed toxicity to the brain or pituitary, and without affecting pituitary hormone secretion. 85
In another preclinical study, a CAR targeting DLL3 demonstrated significant antitumor efficacy in xenograft and murine models of SCLC. The potency of this DLL3-targeting CAR T-cell therapy was greatly enhanced by the expression of the proinflammatory cytokine IL-18, which activated both the CAR T cells and the endogenous tumor-infiltrating lymphocytes, as well as improved the infiltration and activation of antigen-presenting cells. In addition, human IL-18-secreting anti-DLL3 CAR T cells demonstrated reduced exhaustion, an enhanced memory phenotype, and durable responses in multiple SCLC models. These effects were further amplified when combined with anti-PD-1 blockade, suggesting a promising therapeutic approach for targeting DLL3-expressing solid tumors. 86
PARP inhibitor
Like NSCLC, SCLC also has characteristic genetic alterations such as loss of function in RB1 and TP53 87 or amplification of SOX2 and MYC genes. 8 In addition, the significant overexpression and alterations of PARP enzyme and other DNA repair proteins, along with several DNA homologous recombination repair genes identified in SCLC cell lines, are involved in tumorigenesis and growth. In comparison to other neuroendocrine tumors and NSCLC, SCLC tumors express high PARP1 protein. 88 Targeting PARP may provide an additional option for chemoimmunotherapy in SCLC. 89
A double-blind, randomized phase II trial (NCT01638546) compared temozolomide (TMZ) combined with veliparib to TMZ with placebo in relapsed or refractory SCLC. While no significant difference was noted in median OS or 4-month PFS between the two groups, the ORR was significantly higher in the TMZ/veliparib arm. Notably, in patients with SLFN11-positive tumors, the veliparib combination led to significantly prolonged PFS (5.7 vs 3.6 months, p = 0.009) and OS (12.2 vs 7.5 months, p = 0.014). 58 The authors further suggested that high SLFN11 expression could serve as a potential biomarker for sensitivity to PARP inhibitors. One notable study is NCT04334941, a phase II trial comparing the combination of atezolizumab and talazoparib versus maintenance atezolizumab in patients with SLFN11-positive ES-SCLC. The median OS was 8.5 months (80% CI: 7.4–12.7) for the atezolizumab maintenance group and 9.4 months (80% CI: 8.1–14.2) for the atezolizumab plus talazoparib group. Hematological AEs occurred in 4% of patients in the atezolizumab group compared to 50% in the combination group, which was expected for talazoparib (p < 0.001). 36 Despite an attempt to demonstrate the potential clinical efficacy of combining a PARP inhibitor with a standard immunotherapy regimen, no dramatic improvement in clinical outcomes was observed.
Lurbinectedin
Lurbinectedin is a cytotoxic drug that targets guanine-rich regions in gene promoters, disrupting active transcription in tumor cells. This disruption leads to DNA breaks, both single and double-stranded, and ultimately induces apoptosis.90,91 Lurbinectedin also seems to suppress active transcription in tumor-associated macrophages, leading to remodeling of the tumor microenvironment. 92
Lurbinectedin as monotherapy
The efficacy of lurbinectedin monotherapy in previously treated SCLC was assessed in a single-arm phase II trial (NCT02454972) involving 105 patients. 6 The study subjects comprised 32 (30%) LS and 73 (70%) ES patients. Among all patients, the ORR was 35%, with a median duration of response (DOR) of 5.3 months. Among patients with a CTFI of 90 days or more, the ORR was 45%, with a median DOR of 6.2 months. Regardless of grade, neutropenia was the most commonly experienced AE. 6 It should be noted that only 4% of the study subjects had CNS involvement at the initiation of lurbinectedin, and 58% received prophylactic cranial irradiation (PCI). In a study of 20 SCLC patients pretreated with one platinum-containing line, without CNS metastases, and a CTFI of ⩾180 days, an ORR of 60% (95% CI: 36.1–86.9), a median PFS of 4.6 months (95% CI: 2.6–7.3), and a disease control rate of 95% were reported. 29 The study results were instrumental in obtaining accelerated approval of lurbinectedin (3.2 mg/m2) by the US Food and Drug Administration and its designation as an orphan drug by the European Medicines Agency for the treatment of relapsed SCLC. 93
Lurbinectedin in combination
Lurbinectedin is undergoing active evaluation with other treatments. In a phase I study, the lurbinectedin and doxorubicin combination achieved an ORR of 65%. 30 However, grade 3–4 neutropenia occurred in 88% of patients, with 26% experiencing febrile neutropenia. 30 A follow-up expansion cohort using reduced doses demonstrated improved tolerability and preserved antitumor activity but with a lower ORR of 36%. 31
Notably, the phase III ATLANTIS trial is an open-label, randomized study designed to evaluate the safety and effectiveness of lurbinectedin combined with doxorubicin in patients with SCLC who experienced relapse after platinum-based chemotherapy. Participants were randomly assigned to receive either intravenous lurbinectedin (2.0 mg/m²) with doxorubicin or the standard regimen of cyclophosphamide, doxorubicin, and vincristine. 94 Despite a favorable safety profile in the combination group, the median OS was comparable between the lurbinectedin/doxorubicin group and the control group (8.6 vs 7.6 months; HR = 0.97; 95% CI: 0.82–1.15, p = 0.70), showing no significant improvement in OS. 32
Another phase Ib–II trial is assessing lurbinectedin plus irinotecan, which has shown synergy in preclinical models.95,96 Interim results from this trial indicated an ORR of 62% and a median PFS of 6.2 months, with PFS extending to 8.1 months in patients with sensitive relapse. 33 Neutropenia was the most common grade 3–4 AE, but mostly manageable. 33 Updated results presented at ASCO 2024 for 101 patients in the phase II study (NCT02611024) reported a median PFS of 4.7 months and a median OS of 9.7 months, with neutropenia, anemia, and diarrhea as the most significant grade 3 or higher AEs. 34 An ORR of 62% and moderate hematologic toxic effects 33 formed the basis for the ongoing phase III LAGOON trial. The randomized phase III LAGOON trial focuses on relapsed SCLC patients and compares two experimental regimens: lurbinectedin alone (3.2 mg/m² on day 1 every 3 weeks) or a combination of lurbinectedin (2 mg/m² on day 1) plus irinotecan (on days 1 and 8 every 3 weeks). These are evaluated against the control arm, where patients receive either topotecan (on days 1–5 every 3 weeks) or irinotecan (on day 1 every 3 weeks), based on the investigator’s choice. 68
IMforte (NCT05091567; GO43104) is a phase III, randomized, open-label, multicenter trial evaluating atezolizumab plus lurbinectedin against atezolizumab alone as maintenance treatment in ES-SCLC patients post first-line induction with carboplatin, etoposide, and atezolizumab. Participants with an ongoing response or stable disease after completing four induction cycles are eligible for randomization in a 1:1 ratio to receive either the combination of lurbinectedin (3.2 mg/m2 IV) and atezolizumab or atezolizumab alone until disease progression. 67 Lurbinectedin is also being studied in combination with pembrolizumab and with berzosertib. NCCN guideline (Version 2.2024) recommends lurbinectedin, oral or intravenous topotecan, and irinotecan as a subsequent treatment for ES-SCLC with a performance score of 0–2 in patients with CTFI of more than 6 months. 97
Dumoulin et al. reported in the prospective cohort study on 43 SCLC patients receiving lurbinectedin as a third or later treatment, a 29% disease control rate, and a 17% objective response at 12 weeks. The median PFS was 1.5 months, with OS reaching 7 months. Despite some cases of dose adjustments for fatigue and hematologic toxicity, lurbinectedin showed good tolerance. 98
Immunotherapy
Several randomized phase III trials have shown significant benefits when adding an ICI to first-line chemo for newly diagnosed metastatic SCLC patients.37,38,42 The inclusion of anti-PD-L1 mAbs, such as atezolizumab or durvalumab, to the standard platinum–etoposide regimen, with continuation of immunotherapy as maintenance, improved both PFS and OS.37,38 Two randomized trials, IMpower133 37 and CASPIAN, 38 have led to changes in first-line treatment for ES-SCLC.
Atezolizumab
In the phase III IMpower 133 trial, 37 efficacy of the combination of atezolizumab and standard chemotherapy (carboplatin and etoposide) was assessed in 403 ES-SCLC patients. The median OS was 12.3 months in the atezolizumab arm compared to 10.3 months in the control arm (HR = 0.70, p = 0.007), while the median PFS was 5.2 versus 4.3 months, respectively (HR = 0.77, p = 0.02). 99 Notably, at nearly 2 years of follow-up, the atezolizumab arm showed that 22% of patients survived at 24 months compared to 14% in the placebo arm. 100 Based on the observed survival benefit, including 18-month OS rates of 34% versus 21%, 101 atezolizumab has become the standard first-line treatment for ES-SCLC. The FDA approval of the PD-L1 inhibitor atezolizumab in combination with first-line platinum-doublet chemotherapy in 2019 marked a significant advancement in the management of ES-SCLC.37,102
Durvalumab
CASPIAN study has shown that first-line durvalumab plus platinum-etoposide significantly improved OS in patients with ES-SCLC compared to the control group. 38 CASPIAN is an open-label trial that randomized 805 patients in a 1:1:1 ratio to receive durvalumab plus etoposide–platinum, durvalumab plus tremelimumab and etoposide–platinum, or etoposide–platinum alone for up to four cycles, followed by maintenance therapy. The addition of durvalumab significantly improved OS (HR = 0.73, p = 0.0047) compared to chemotherapy alone. The 12-month PFS rate and confirmed ORRs were higher in the durvalumab group (18% PFS; 68% ORRs) than in the chemotherapy-only group (5% PFS; 58% ORRs). 38 Furthermore, 17.9% of patients treated with durvalumab and carboplatin–etoposide were progression-free at 12 months, compared to 5.3% of those who received only chemotherapy. 103
However, the addition of tremelimumab did not improve survival outcomes, and the four-drug combination arm experienced higher rates of serious AEs (45%) and treatment-related deaths (5%) compared to the durvalumab plus carboplatin–etoposide arm (32% and 2%) and the chemotherapy-only arm (36% and 1%). 103 The FDA approved the use of durvalumab for ES-SCLC in the first-line setting based on the study results. 104 The multicenter, real-world study in China (ORIENTAL) reported positive outcomes in 166 treatment-naïve ES-SCLC patients, with a median OS of 14.8 months, a median PFS of 6.3 months, and an ORR of 76.4%. 39
Further combination treatments are also being studied. Results from the NCT04660097 trial, which enrolled 22 treatment-naïve patients with ES-SCLC, showed that the combination of durvalumab, anlotinib, and chemotherapy resulted in an ORR of 100% and a complete response (CR) rate of 27.3%. While the median OS was not reached, the median PFS was 8.51 months. The most common treatment-emergent AEs were neutropenia and leukopenia. 41
Pembrolizumab
The use of PD-L1 inhibitors has expanded across various treatments, whereas PD-1 inhibitors have encountered difficulties in demonstrating efficacy for ES-SCLC. Although some encouraging outcomes were observed in first-line therapy.48,43
As a subsequent-line treatment, the FDA has withdrawn the indication of pembrolizumab, as phase III randomized trial results did not demonstrate a significant improvement in OS.44,105 However, the NCCN guidelines suggest subsequent systemic therapy (other recommended regimens) for patients with a CTFI of less than 6 months. 106 A combined analysis of the KEYNOTE-028 and KEYNOTE-158 studies revealed a 19.3% ORR and a 7.7-month median OS among patients treated after two or more lines of therapy. 44 Pembrolizumab, when combined with amrubicin or paclitaxel in phase II trials, showed efficacy and acceptable safety in patients with ES-SCLC, including those previously treated.45,46 The KEYNOTE-158 trial, a phase II study involving 1066 patients with various solid tumors including SCLC, assesses the effectiveness of pembrolizumab in those who have not responded to at least one standard treatment. 107 Early results have highlighted a high tissue tumor mutational burden as a key indicator for durable responses to pembrolizumab. 108 This trial is slated for completion in June 2026 (ClinicalTrials.gov identifier: NCT02007070).
The KEYNOTE-604 trial, a phase III study, compared the efficacy of pembrolizumab + etoposide–platinum chemotherapy to chemotherapy plus placebo. 42 Pembrolizumab combination regimen significantly improved PFS (p = 0.0023; median 4.5 vs 4.3 months) and prolonged OS compared to the chemotherapy group as first-line therapy in ES-SCLC. An exploratory biomarker study from KEYNOTE-605 revealed no association between tumor mutational burden, SCLC transcriptional subtypes, and OS in the pembrolizumab ETP group with ES SCLC. However, the 18-gene T-cell-inflamed gene expression profile was positively linked with OS in both the pembrolizumab and placebo-chemotherapy groups. 109 A 2024 AACR abstract presented data from the phase II KEYNOTE-B99 study (NCT 04924101) comparing first-line pembrolizumab and EP with investigational agents (MK-4830, anti-ILT4; boserolimab, anti-CD27; or lenvatinib). At data cutoff, with a median follow-up of 14.1 months, ORR was 65%, 71%, and 74% in arms A (MK-4830), B (boserolimab), and C (lenvatinib), respectively. AEs occurred in all arms, with neutropenia and anemia being the most common. 47
Ongoing studies, which could provide potential alternatives, are evaluating combinations of lurbinectedin with immunotherapies such as pembrolizumab in the LUPER trial (NCT04358237), 110 durvalumab (ClinicalTrials.gov identifier: NCT04607954, phase II), and a triplet regimen with ipilimumab plus nivolumab (NCT04610658, phase I/II). In the phase I/II LUPER study, a total of 28 patients were enrolled, with 51.9% having a platinum-free interval of less than 90 days and all subjects experiencing progression after first-line platinum-based chemotherapy. The preliminary results showed a median follow-up of 5.7 months and an ORR of 46.4%, including two cases of CR. The median PFS was 5.3 months, and the median OS was 11.1 months. The most common treatment-related AEs (grade III and IV) were fatigue, neutropenia, nausea, and anemia. 35
Cytotoxic T lymphocyte-associated antigen 4 inhibitor
It is shown that the anti-cytotoxic T lymphocyte-associated antigen 4 blockade does not provide significant survival benefit in ES-SCLC. Combination of ipilimumab with standard chemotherapy did not yield a superior outcome compared to chemotherapy alone (HR = 0.94; 95% CI: 0.81–1.09; p = 0.38) in a phase III trial. 48
Preliminary data from the ongoing BIOLUMA study of nivolumab combined with ipilimumab in patients with relapsed SCLC (NCT03083691; n = 18) show less promising results for double ICI therapy regimen. Even though the ORR for the combination therapy was 38.8%, toxicity rates were high, and there were 2 treatment-related deaths. 51 In the updated results of 297 patients with evaluable tumor mutational burden, a total of 45 TMB-high patients were confirmed, and the disease control rate was 20.4%. 52 The CheckMate 451 trial evaluated maintenance therapy in ES-SCLC patients who had not progressed after ⩽4 cycles of initial treatment. A total of 834 patients were randomized equally into 3 groups: nivolumab plus ipilimumab, nivolumab alone, or placebo. The study found no significant OS improvement with the combination therapy compared to placebo (9.2 vs 9.6 months, HR = 0.92, p = 0.37). 50 Furthermore, the combination therapy led to a higher incidence of serious AEs and discontinuations due to AEs, compared to monotherapy or placebo (serious AEs: 37.4% vs 6.1% and 2.9%; discontinuations: 28.8% vs 7.9% and 0.4%, respectively). Treatment-related deaths were also more common in the combination therapy group.
Nivolumab
In CheckMate 032, 53 it was shown that nivolumab, alone or with ipilimumab, yielded ORRs of 11.6% and 21.9%, respectively, but combination therapy did not extend OS and led to more side effects. 111 In the ECOG-ACRIN EA5161 trial, adding nivolumab to treatment improved PFS (5.5 vs 4.6 months, p = 0.047) but not OS (11.3 vs 8.5 months, p = 0.14) for ES-SCLC patients. 49
The combination of nivolumab and gemcitabine failed to extend PFS and OS and did not enhance the radiographic response rate when compared to historical controls in patients with advanced-stage SCLC who experienced relapse or refractory disease following platinum-based chemotherapy. 112 In the phase III CheckMate 331 trial (NCT02481830; n = 569), the primary endpoint of OS with nivolumab versus chemotherapy as a second-line treatment for SCLC was not met. Nivolumab did not show a significant improvement in OS compared to chemotherapy (topotecan or amrubicin; median OS: 7.5 vs 8.4 months; HR = 0.86; 95% CI: 0.72–1.04; p = 0.11). 54
Radiotherapy with immunotherapy
The NCCN SCLC panel advises platinum-based chemotherapy with etoposide and either atezolizumab or durvalumab as the initial treatment for ES-SCLC, based on the clinical trials and FDA endorsements. Maintenance therapy with these PD-L1 inhibitors continues until progression. This shift prompts a reassessment of radiotherapy’s role, given that it had been applied after cessation of systemic chemotherapy cessation. 113 Consolidative thoracic radiotherapy (TRT) can be administered either before or during maintenance immunotherapy. Although there is a lack of robust clinical data regarding the optimal timing and safety of combining TRT with immunotherapy, the treatment may produce a synergistic effect by enhancing MHC class I expression and increasing CD8-positive T-cell activity. 114 The ongoing RAPTOR/NRG LU007 trial (NCT04402788) will show the efficacy and safety results of radiotherapy for ES-SCLC patients following immunotherapy. 115
This phase II study (NCT06177925) aims to evaluate the efficacy and safety of combining adebelizumab with carboplatin/cisplatin, etoposide, and concurrent radiotherapy as first-line treatment for oligometastatic ES-SCLC. Patients undergo an induction phase with two cycles of adebelizumab alongside chemotherapy. Subsequently, they receive concurrent chemoradiotherapy followed by one to two cycles of adebelizumab with chemotherapy. Adebelizumab maintenance follows the initial treatment and is maintained until disease progression, intolerable toxicity, or withdrawal of consent (estimated completion date September 2026; ClinicalTrials.gov identifier: NCT06177925).
Radiotherapy remains an integral component in the management of ES-SCLC. TRT is commonly used to enhance local control in patients with residual intrathoracic disease following chemotherapy and immunotherapy. Emerging evidence suggests that TRT not only improves PFS but may also contribute to OS benefits in select patient populations.116,117 PCI, on the other hand, is performed to decrease the risk of brain metastases, which are highly prevalent in ES-SCLC. While PCI has shown efficacy in reducing the incidence of brain metastases,118,119 its routine application is being reevaluated in light of recent data advocating for MRI surveillance as an alternative in specific subsets of patients.120,121 These findings highlight the need for careful patient selection when implementing radiotherapeutic strategies in ES-SCLC, prioritizing individuals most likely to benefit from improved local and systemic control while minimizing treatment-related toxicities.
Other immunotherapy regimens
ASTRUM-005 (NCT04063163), a randomized, double-blind phase III study, evaluated the efficacy of a combination regimen of etoposide, carboplatin, and serplulimab, another PD-1 antibody. In comparison to the group that received etoposide + carboplatin + placebo, the serplulimab group reported a median PFS of 5.7 months compared to 4.3 months (HR = 0.47, 95% CI: 0.38–0.59, p < 0.001) and a median OS of 15.4 months compared to 10.9 months (HR = 0.63, 95% CI: 0.49–0.82, p < 0.001). 55 The study results led to orphan drug designation by the FDA for the treatment of patients with SCLC. 122
At present, there is no established evidence supporting the use of ICIs in ES-SCLC patients with poor performance status (PS), as individuals with PS 2–4 were excluded from the IMpower133 and CASPIAN trials. Typically, when PS deteriorates due to cancer progression and is likely to improve with treatment, chemotherapy is considered for ES-SCLC patients with PS 2–3. A phase II, non-randomized, open-label, single-arm trial evaluates the efficacy and safety of the combination of carboplatin, etoposide, and durvalumab in ES-SCLC patients with PS 2 and 3 (NEJ045A study). 123 The recent results reported in ASCO showed that the median PFS for patients with PS 2 was 4.7 months (95% CI: 3.8–6.4), while for those with PS 3, it was 5.1 months (95% CI: 1.4–10.6). The median OS was 9.5 months (95% CI: 6.6–26.7) for patients with PS 2 and 5.1 months (95% CI: 1.4–not reached) for those with PS 3. 40
The CAPSTONE-1 trial assessed the efficacy and safety of adebrelimab (SHR-1316), a novel anti-PD-L1 antibody, with standard chemotherapy in the first-line setting of ES-SCLC. A total of 462 eligible patients were randomized to receive carboplatin and etoposide with either adebrelimab or placebo. At a median follow-up of 13.5 months, OS was significantly longer in the adebrelimab group (15.3 months) compared to the placebo group (12.8 months). Serious treatment-related AEs occurred in 39% of adebrelimab patients and 28% of placebo patients. Despite four treatment-related deaths, adebrelimab showed an acceptable safety profile and improved survival outcomes. 56
The ETER701 trial assessed whether adding the novel PD-L1 inhibitor benmelstobart and anlotinib to standard chemotherapy would result in clinical benefits in first-line ES-SCLC therapy. In the trial, one group of patients received benmelstobart and anlotinib combined with chemotherapy, another group was given anlotinib with chemotherapy, and a third group received only chemotherapy. The results demonstrated that patients treated with both benmelstobart and anlotinib alongside chemotherapy had a longer OS of 19.3 months, compared to 11.9 months in the group receiving chemotherapy alone. 57 The median PFS in the anlotinib groups was notably longer than that in the chemotherapy-only group (5.6 vs 4.2 months, p < 0.0001). Although the incidence of grade 3 or higher treatment-related AEs was high in both arms (94.3% vs 87.0%), the safety profile remained manageable. 124
Antibody–drug conjugate
Ifinatamab deruxtecan (I-DXd, DS-7300) is an ADC designed to target B7-H3. It consists of a humanized anti-B7-H3 IgG1 antibody, linked to the topoisomerase I inhibitor deruxtecan (DXd) via a cleavable tetrapeptide linker.
A phase I/II trial (NCT04145622) investigated I-DXd in patients with specific advanced solid tumors, including SCLC. As of January 31, 2023, among the 21 SCLC patients, 11 exhibited an ORR of 52%, with 1 complete response and 10 partial responses. The median DOR was 5.9 months (95% CI: 2.8–7.5). In addition, the median PFS was 5.8 months (95% CI: 3.9–8.1), and the median OS reached 9.9 months (95% CI: 5.8–not reached). However, the 16.0 mg/kg cohort had been discontinued due to safety concerns, as there were high occurrences of serious and grade ⩾3 TRAEs. Interstitial lung disease (ILD)/pneumonitis was observed in nine patients, with seven cases considered drug-related (grades 1–5).125,126 Based on the moderate ORR and PFS observed in NCT04145622, a new ongoing phase Ib/II multicenter, open-label trial, IDeate-Lung03 (NCT06362252), is recruiting approximately 149 adult patients ES-SCLC. The trial consists of two parts: Part A, which is a safety run-in, and Part B, focusing on dose optimization. Both parts are divided into two patient cohorts. The study aims to evaluate the effectiveness of I-DXd, administered intravenously every 3 weeks, in combination with atezolizumab, with or without carboplatin, as a first-line therapy for patients with ES-SCLC. 127 Another ADC worth mentioning is Trodelvy® (sacituzumab govitecan-hziy), which has received a Breakthrough Therapy Designation from the U.S. FDA for the treatment of adult patients with ES-SCLC who have progressed on or after platinum-based chemotherapy. This designation is based on results from the global phase II TROPiCS-03 study (ES-SCLC cohort), which demonstrated promising efficacy with Trodelvy as a second-line treatment for ES-SCLC.128–130
Future perspectives
It is hard to deny that patients with ES-SCLC will have more options in the near future, either in treatment-naïve or pretreated situations. Clinicians will face challenges in choosing the most optimal treatment options, similar to advanced NSCLC, and thoughtful consideration will be vital to find the most effective strategy.
Based on varying datasets and methodologies, several distinct subtypes of SCLC have been proposed.9,12 However, achieving uniform and rigorous consensus clustering of SCLC patients remains a significant challenge, and biomarkers specific to each subtype are yet to be fully validated. Comprehensive multi-omics analyses—including genomics and proteomics of large patient cohorts—will play a pivotal role in identifying novel biomarkers. Moreover, to advance personalized medicine in SCLC, it is imperative to clinically validate potential biomarkers and design prospective biomarker-driven clinical trials.
In predicting how the treatment landscape will change, drawing parallels to advanced NSCLC management strategies will help. It is conceivable that DLL-3 and other targetable proteins specific to SCLC cancer cells will become akin to targetable mutations in advanced NSCLC. 77 Patients with ES-SCLC will undergo screening for potentially targetable proteins at the initial stage. For those who test positive, treatment, whether in the first-line, second-line, or subsequent lines, will be contemplated. However, atezolizumab with platinum-based chemotherapy, based on Impower-133 results, 37 will remain a steadfast priority first-line treatment modality until other regimens with clearly superior efficacy emerge.
The final results of ongoing trials will provide a more comprehensive understanding of the treatment landscape. Ongoing treatment modalities, including combinations of recently discovered effective regimens and traditional approaches such as platinum-based chemotherapy and radiotherapy, will offer a broader array of choices for ES-SCLC patients.
Among emerging therapeutic strategies, targeting DLL3 continues to show promise. Beyond tarlatamab, other T-cell engagers targeting DLL3, such as BI 764532 and HPN328, are currently under investigation, and their clinical results are eagerly anticipated.62–65 In addition, advances in artificial intelligence (AI) are transforming drug discovery and development. AI-driven protein structure prediction tools, exemplified by AlphaFold, have already demonstrated remarkable utility in accelerating novel drug design. 131 Future research should explore how AI can be harnessed to identify and develop more effective agents with improved binding affinity to key SCLC targets.
Conclusion
ES-SCLC has not seen evident advancements in systemic therapy for decades. However, recent progress in various regimens, such as immunotherapy, DLL3-targeting therapy, and lurbinectedin, has significantly expanded treatment options. It remains the clinician’s responsibility to select the most appropriate treatment strategy to optimize outcomes for ES-SCLC patients. Ongoing trials exploring various combinatorial treatment strategies will likely provide additional treatment options.
