Abstract
Background
Metronomic chemotherapy is aimed at lessening the adverse effects of treatment while rendering cancer cells cytostatic. The oral 5-fluorouracil prodrug “5′-DFUR” has been shown to inhibit angiogenesis and is regarded as a good candidate agent for metronomic chemotherapy. Moreover, cisplatin and 5′-DFUR have been shown to synergistic cytotoxic effects.
Methods
We evaluated the safety and efficacy of metronomic chemotherapy using daily oral 5′-DFUR at the dose of 600 mg/day and biweekly cisplatin infusion at the dose of 20 mg/person in 23 patients with urothelial cancer resistant to conventional platinum-based chemotherapy.
Results
Twenty-three patients were enrolled between August 2000 and December 2004. The median survival time after the initiation of metronomic chemotherapy was 15.2 months. The 1-year, 2-year and 3-year survival rates were 55.1%, 45.1% and 5.9%, respectively. Grade 3 fatigue was observed as severe toxicity in one patient. No cases showed nephrotoxicity and adverse effects necessitating medical intervention.
Conclusions
Although a large-scale prospective study would be necessary before the therapy is established as a standard, our metronomic chemotherapy regimen appears to be a potentially useful palliative treatment alternative for patients with advanced urothelial cancer resistant to conventional platinum-based chemotherapy.
Abbreviations
M-VAC: methotrexate, vinblastine, doxorubicin, and cisplatin; GC: gemcitabine and carboplatin; 5′-DFUR: 5′-deoxy-5-fluorouridine; 5-FU: 5-fluorouracil; CDDP: cisplatin; TCC: transitional cell carcinoma; ECOG: Eastern Cooperative Oncology Group; PS: performance status; UICC: Union International Contre le Cancer; WHO: World Health Organization; NCI-CTC: National Cancer Institute Common Toxicity Criteria; CI: confidence interval; PR: partial response; NC: no change; PD: progressive disease; TP: thymidine phosphorylase; AUC: areas under the curve.
Keywords
Introduction
Urothelial cancer (including cancer of the urinary bladder, ureters and the renal pelvis), which accounts for about 4% of all cancers, is one of the most common genitourinary tumours, associated with significant morbidity and mortality, in both western countries and Japan (Walsh, 2002) (Matsumoto et al. 2004). Although radical surgery may be curative, about 50% of patients with muscle-invasive urothelial carcinoma develop metastases within 2 years of surgery and subsequently die of the disease (Walsh, 2002). Since urothelial carcinoma is relatively chemosensitive, anticancer chemotherapies have the potential to improve the outcome in patients with advanced urothelial cancer. The current standard chemotherapy for advanced disease is a platinum-based combination regimen, namely, M-VAC (methotrexate, vinblastine, doxorubicin and cisplatin) (Bajorin et al. 1999), or GC (cisplatin and gemcitabine) (von der Maase et al. 2005). Median survival times for advanced urothelial cancer treated with M-VAC has been reported to be 9.3 to 33.0 months (Bajorin et al. 1999). GC has been demonstrated to exhibit similar efficacy to M-VAC, but with a significantly more favorable side effect profile (von der Maase et al. 2005); nonetheless, in absolute terms, the impact of theses chemotherapies remains quite modest. No third-line chemotherapy has been established yet, therefore, new strategies need to be developed for such cases.
Conventional platinum-based chemotherapy using the maximum tolerated dose delivers the highest dose intensity for killing cancer cells (Roth and Bajorin, 1995). “Metronomic chemotherapy”, which refers to low-dose, uninterrupted and prolonged administration of chemotherapeutic agents, is aimed at lessening the adverse effects of treatment while exerting a cytostatic effect against the cancer cells (Browder et al. 2000) (Kerbel and Kamen, 2004). Metronomic chemotherapy often exerts antitumor effect via anti-angiogenesis activity, by direct targeting of the activation, growth and proliferation of vascular endothelial cells (Browder et al. 2000) (Kerbel and Kamen, 2004).
5′-deoxy-5-fluorouridine (5′-DFUR; Doxifluridine or Furtulon) is a second-generation oral prodrug of 5-fluorouracil (5-FU) (Cunningham and Coleman, 2001). Although 5-FU has modest activity in cases of advanced bladder cancer, with reported response rates of between 12%–20% (Roth and Bajorin, 1995) (Logothetis, 1992), 5-FU which is the metabolite of 5′-DFUR have been shown to inhibit angiogenesis (Kerbel and Kamen, 2004) (Basaki et al. 2001) (Munoz et al. 2005). Moreover, since 5′-DFUR can be given orally in the outpatient setting (Cunningham and Coleman, 2001), it appears to be a good candidate agent for metronomic chemotherapy. Furthermore, cisplatin (CDDP) and 5-FU have been demonstrated to exert synergistic cytotoxic effects against urothelial and other cancers in many experimental and clinical studies (Matsuyama, Yamakawa, and Hirata, 2002) (Kemeny et al. 1990). During subsequent studies to improve upon the combination of 5-FU and CDDP, consecutive low-dose CDDP with 5-FU was also reported to elicit high response rates without severe toxicity in several cancer patients (Matsuyama, Yamakawa and Hirata, 2002) (Nishiyama et al. 1999).
Therefore, as a clinical practice, we conducted metronomic chemotherapy using oral 5′-DFUR administered daily with biweekly (once every two weeks) low-dose CDDP for the control of advanced urothelial cancer resistant to conventional platinum-based chemotherapy and evaluate the efficacy and safety in 23 cases.
Patients and Methods
Patients
The ethical approval was obtained for this study. Tumor assessment was performed in all cases by computed tomographic scanning after the conventional platinum-based chemotherapy. Patients whose bidimensionally measurable cancer showed the progression disease were eligible in this therapy. Twenty-three consecutive advanced urothelial cancer patients who received at least one cycle of 5′-DFUR + low-dose CDDP chemotherapy between August 2000 and December 2004 were evaluated. All 23 cases were resistant to prior conventional platinum-based systematic chemotherapies. The main regimes used for the prior systemic platinum-based chemotherapy were conventional M-VAC regimens and/or CDDP (CDDP: 70 mg/mm2) monotherapy (Table 1). The other eligible criteria for analysis in this study were: age ≥20 years old, histologically proven transitional cell carcinoma (TCC) of the urothelial tract (urinary bladder, ureter or the renal pelvis), Eastern Cooperative Oncology Group (ECOG) performance status (PS) ≤3. Patients were eligible irrespective of whether they had been previously treated by radiotherapy or surgery. Informed consent was obtained from all of the patients prior to the start of this therapy. For tumour restaging at the beginning of this study, the patients underwent chest X-ray, computed tomographic scanning of the chest, abdomen and pelvis, and radioisotopic bone scanning. The tumour stage was evaluated according to the 2002 Union International Contre le Cancer (UICC) cancer staging system (Sobin L, 2002).
Characteristics of the patients with urothelial cancer enrolled in this study.

“Both” refers to Bladder cancer with Ureteral or Renal pelvic cancer.
MVAC denotes the chemotherapeutic regimen, consisting of methotrexate (MTX), vinblastine (VBL), doxorubicin (ADM) and cisplatin (CDDP).
Treatment Schedule
This therapy was conducted at the Yamaguchi university hospital, Ube, Japan. 5′-DFUR was administered orally in the form of 200-mg capsules in 2 or 3 divided doses daily (400–800 mg/person/day). Because of the patient's heterogeneous clinical backgrounds, dose adjustments of the chemotherapeutic drugs were necessary, and were determined individually by the attending physician based on the patient's body weight, ECOG PS, age and laboratory data by the discretion of the patient's attending physician. The majority of the patients received 600 mg of 5′-DFUR per day. We planned for CDDP infusion at the outpatient clinic to be repeated every 2 weeks in each cycle (weekly in some cases). CDDP (20 mg/person) was administered by intravenous infusion on day 1 after infusion of 500 mL of saline, along with administration of a 5-HT3 serotonin receptor antagonist. According to our study protocol, the metronomic outpatient-based chemotherapy was to be continued unless any severe complications occurred, the general condition of the patient worsened or the patient refused to comply with continued chemotherapy.
Outcome measures (efficacy and toxicity analysis)
The overall survival was calculated from the database. Our database was updated in August 2006. Tumour assessment was performed every three months by clinical evaluation, chest X-ray, computed tomographic scanning of the chest, abdomen and pelvis, and bone scintigraphy. Patients with bidimensionally measurable disease and had at least one follow-up tumour assessment were eligible for the response evaluation. The WHO (World Health Organization) Standard criteria (Miller et al. 1981) were used for classifying the responses. Toxicity evaluation, including by complete blood count, serum electrolyte evaluation and serum biochemical analysis, was conducted every two weeks, in accordance with the National Cancer Institute Common Toxicity Criteria (NCI-CTC), Version 2.0.
Statistical analysis
All the analyses were performed using the R statistical software (The R Foundation for Statistical Computing Version 2.2.1 for Windows). The preparation of the overall survival curves and determination of the 95% confidence intervals (CI) were conducted by the Kaplan–-Meier method. Univariate analysis on survival was performed using the log-rank test.
Results
Patient population
A total of 23 patients comprising seventeen male and six female patients with a median age of 75 years were evaluated in the study. The patient characteristics are shown in Table 1. All the patients had histologically proven primary urothelial TCC. In patients with both bladder and ureteral/renal pelvic cancer, the tumour stage was represented by the stage of the more advanced of the two cancers, and all cases had advanced cancer (stage III, 3 cases; stage IV, 20 cases). The metastatic lesions consisted of 13 cases each with visceral and/or lymph node metastases and 6 cases had both. The major sites of visceral metastasis were the lung, liver and bone, in 8, 4 and 5 cases, respectively. The ECOG PS of the patients was 0, 1, 2 and 3 in 4, 13, 3 and 3 cases, respectively. The number of patients with a previous history of radiotherapy and surgical treatment at the start of the study were 18 and 7, respectively.
Duration of therapy and toxicity
A total of 497 cycles were given to the 23 patients. The median number of chemotherapeutic cycles per person was 15 (CDDP: 300 mg) (range, 2 to 63 cycles). Including the previously administered regimen, the median total CDDP dose was 615 mg (range, 67.5 to 1730 mg). Nine patients could be administered a total CDDP dose of over 900 mg. The toxicities observed throughout the treatment in all the 23 patients are shown in Table 2. Eighteen patients (78.0%) had at least one toxicity. As Grade 3/4 toxicity, one patient (4.3%) had grade 3 fatigue, and none had grade 4 toxicity. Other grade 2/1 toxicities were well tolerated, with no necessity of medical interventions. Notably, no nephrotoxicity and ototoxicity occurred in any of the cases, despite the higher total dose of CDDP administered.
Toxicity of 5′-DFUR + low-dose CDDP chemotherapy.

Grade of toxicity according to NCI-CTC Version 2.0.
Response and survival
Among the 20 evaluable patients, two showed partial response, with an overall response rate of 10.0% at the first 3 months’ evaluation. Stable disease was noted in four patients (20.0%). Overall, 30% of the cases showed no progression at the first evaluation. The details of individual patient demographics and treatment course are shown in Table 3. The median and mean follow-up period were 9.3 and 15.9 months, respectively (range: 1.2–38.4 months). Figure 1A shows the overall survival curves after the initiation of 5′-DFUR + low-dose CDDP therapy. The median survival was 15.2 months (95% CI: 7.4–32.5 months), and the 1-year, 2-year and 3-year survival rates were 55.1% (95% CI: 37.1%–81.9%), 45.1% (95% CI: 27.8%–73.2%) and 5.9% (95% CI: 0.9%–38.6%), respectively. Figure 1B shows the overall survival curves after the start of the previous conventional chemotherapy; the median survival was 32.0 months (95% CI: 9.8–43.1 months).

Overall survival curves. Overall survival curves of the 23 patients after the beginning of 5′-DFUR + low-dose CDDP (
Individual patient characteristics and prior therapy in relation to the response to 5′-DFUR + low-dose CDDP therapy.
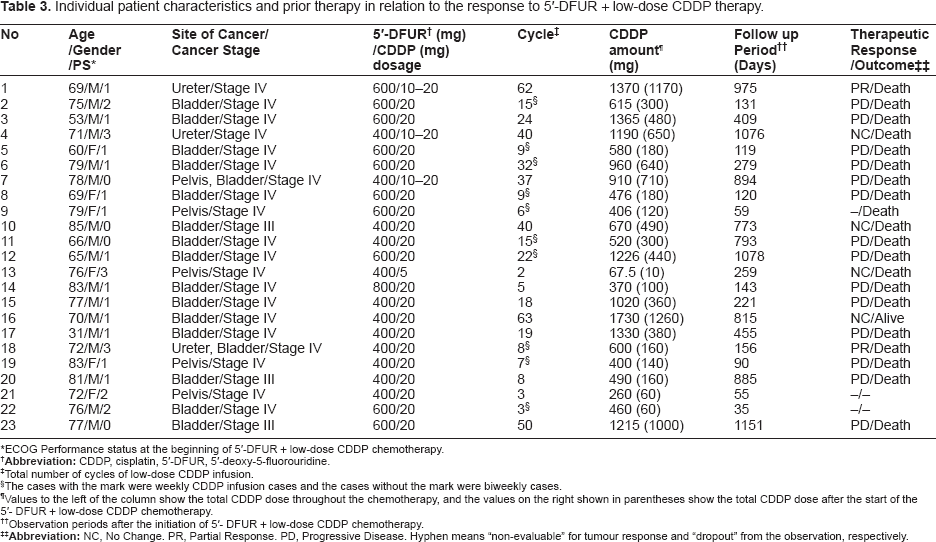
ECOG Performance status at the beginning of 5′-DFUR + low-dose CDDP chemotherapy.
Total number of cycles of low-dose CDDP infusion.
The cases with the mark were weekly CDDP infusion cases and the cases without the mark were biweekly cases.
Values to the left of the column show the total CDDP dose throughout the chemotherapy, and the values on the right shown in parentheses show the total CDDP dose after the start of the 5′- DFUR + low-dose CDDP chemotherapy.
Observation periods after the initiation of 5′- DFUR + low-dose CDDP chemotherapy.
Univariate analysis on survival was performed for each variable using the log-rank test (Table 4). The survival benefit was statistically significantly superior in the male than female patients (p < 0.001), although the number of female was very small (6 patients) and the analysis was a univariate setting. The age, clinical response to therapy, PS, clinical stage and other factors were not found to be statistically significantly related to the patient survival.
Univariate analysis of factors influencing survival using a log-rank test.

Statistically significantly superior survival benefit as compared to females (P value < 0.001) by log-rank test.
ECOG Performance status at the beginning of 5′-DFUR + low-dose CDDP chemotherapy.
According to the 2002 Union International Contre le Cancer (UICC) cancer stage classification.
Three cases was excluded from the evaluation of tumour assessment.
Discussion
Metronomic chemotherapy is a potentially useful treatment modality in patients with cancers resistant to conventional chemotherapy. While cancer cells undergo constant mutations to become resistant to chemotherapeutic drugs, endothelial cells are genetically stable (Kerbel and Kamen, 2004). Even if a drug is no longer effective against cancer cells, it could still exert antitumour activity by targeting endothelial cells (Kerbel and Kamen, 2004). In our setting, we believe that the biweekly low-dose CDDP infusion exerts antitumour effect by enhancing the cytotoxic effects of 5′-DFUR in a synergistic manner and also by exerting cytostatic effect by targeting the endothelial cells.
The mechanism of the antitumour activity of metronomic chemotherapy with 5′-DFUR + low-dose CDDP is summarized in Figure 2. The enzyme thymidine phosphorylase (TP), also known as platelet-derived endothelial cell growth factor, is required for conversion and activation of 5′-DFUR to 5-FU (Cunningham and Coleman, 2001). TP is also an angiogenic factor that promotes angiogenesis in vivo and stimulates the growth of a variety of endothelial cells in vitro (Toi et al. 2005). A high level of TP in tumour tissues reflects activation of angiogenesis; in one study, the expression of TP in invasive tumour tissues was reported to be 33-fold higher than that in superficial tumour tissues and 260-fold higher than that in normal bladder tissue (O'Brien et al. 1995). In addition, TP is expressed not only in cancer cells, but also consistently in cancer stromal cells, including tumour-associated endothelial cells (Shimabukuro et al. 2005). Therefore, it is speculated that 5′-DFUR may have cancer-specific antiangiogenic activity, because it is activated in cancer tissues where angiogenesis itself is strongly activated.

Model of the mechanism of action of metronomic chemotherapy with 5′-DFUR + low-dose CDDP. The high TP in the cancer tissues potentially activates angiogenesis
Since CDDP and 5-FU modulate each other's activity, the combination has a synergistic cytotoxic effect. In regard to the mechanism underlying the well-known synergistic cytotoxic effects of the drug combination, it is speculated that CDDP inhibits intracellular L-methionine metabolism, thereby increasing the levels of the reduced substances, 5, 10-methylenetetrahydrofolate and tetrahydrofolate, in the tumour. The increase in the intracellular reduced folate levels raises the binding level of 5-FU-derived fluorodeoxyuridine monophosphate to thymidylate synthase, resulting in the inhibition of DNA synthesis (Esaki et al. 1992) (Scanlon et al. 1986). Another mechanism is believed to be inhibition of the repair of CDDP-induced DNA interstrand cross-links by 5-FU pretreatment (Esaki et al. 1992) (Scanlon et al. 1986). Production of 5-FU–-CDDP conjugates with antitumour effects is also proposed (Wang et al. 2003).
Locally invasive and/or metastatic TCC represents a very aggressive tumour, with a median survival of the patients rarely exceeding 3–6 months in untreated patients. Effective combination chemotherapy has been reported to prolong survival, yielding a median survival of more than 12 months (von der Maase, 2005). Although our study may be biased, because of the heterogeneous backgrounds of the patients, including age, previous therapy and status of cancer progression as compared with other reported series (Bajorin et al. 1999) (Lin et al. 2006) (Bamias et al. 2006) (shown in Table 5), the median survival of 15.2 months after the initiation of metronomic chemotherapy may be comparable. Moreover, considering our cases as intermediate/high risk group as compared to those in Bajorin's series (Bajorin et al. 1999), a median survival period of 15.2 months is considered to represent excellent survival benefit of chemotherapy. However, the 3-year survival rate of 5.9% suggests that the survival benefit might be limited at best to 2 years. In our survival curve (Figure 1A), two subgroups were observed; intermediate survivors for up to 2 years and early death. The death rate between 1 year and 2 years after the treatment was only 10.0%. The over 2-year survival group may have benefitted from the cytostatic effect attributable to this antiangiogenic therapy.
Survival of advanced urothelial cancer patients with a history of receiving various chemotherapies.

Median Overall Survival.
The independent predictors of survival in patients with urothelial cancer have been reported to be the PS, presence/absence of visceral metastases, the serum alkaline phosphatase level, history of chemotherapy and gender (Bajorin et al. 1999) (Sengelov, Kamby, and Von der Maase, 2001) (Micheli et al. 1998). Although the males showed a statistically significantly superior improvement of the survival time in this study, it was possible that our study had some bias in cases.
Our treatment was associated with a lower incidence of toxicities than that reported with the use of other regimens (Roth and Bajorin, 1995). The majority of patients received 600 mg of 5′-DFUR per day and 20 mg of CDDP biweekly. In order to minimize treatment toxicity and maintain maximal outpatient performance, we selected fractional administration of CDDP in our study. The dose per fraction in metronomic chemotherapy is around one-tenth of that in conventional maximum-tolerated-dose therapy (Kerbel and Kamen, 2004). The recommended dose of CDDP per fraction for metronomic outpatient chemotherapy is around 15–25 mg/m2 (Shimoyama et al. 2005) (Hyodo et al. 2003). Although there is not enough evidence about the synergistic effects of CDDP and 5′-DFUR under our dose setting, 20 mg/person of CDDP may have several benefits, including feasibility of continuation of chemotherapy on an outpatient basis. Excessive attention to hydration was unnecessary with our regimen, because there was no nephrotoxicity, which makes it feasible for the regimen to be continued on an outpatient basis (administration of each fraction could be completed within 120 min). On the other hand, similar to the case for other prodrugs of 5-FU, gastrointestinal side effects were major adverse effects of 5′-DFUR. According to the pharmacological profiling of 5′-DFUR (Chugai Pharmaceutical Co. Ltd., Gotemba, Japan), the total incidence of toxicity with oral 5′-DFUR increased with increase of the dose: 10%–31% in patients receiving under 600 mg/day, 18%–41% in patients receiving 600–800 mg/day and 44%–47% in patients receiving 800– 1000 mg/day. Based on these toxicity data, the recommended dose of 5′-DFUR for outpatient-based chemotherapy in the elderly is under 800 mg/day. On account of the lower toxicity, our regimen was well tolerated in elderly patients with advanced urothelial cancer and our metronomic chemotherapy regimen could be continued on an outpatient basis.
Conclusion
Although a large-scale prospective study might be necessary before the therapy is established for standard use, our chemotherapeutic regimen of daily oral 5′-DFUR at 600 mg/day + biweekly infusion of CDDP at the dose of 20 mg given in the outpatient setting as metronomic chemotherapy for cases of advanced urothelial cancer resistant to conventional platinum-based chemotherapy yielded a comparatively sustained survival benefit and low toxicity. Thus, our preliminary data indicate the potential usefulness of this regimen as a palliative treatment alternative for patients with advanced urothelial cancer resistant to conventional platinum-based chemotherapy.
Footnotes
Acknowledgments
We would like to thank Dr. Aoki A, Dr. Suga A, Dr. Takai K, Dr. Tei Y, Dr. Tuchida M, Dr. Uchiyama K, Dr. Wada T and Dr. Yamamoto M as the medical staff of the clinic for their contribution to the clinical management of the cases.
