Abstract
Objective
Oral metronomic cyclophosphamide has been used as a single agent or in combination with other drugs for several solid tumors with interesting results in disease palliation and mild to moderate toxicity, notably in patients with recurrent epithelial ovarian cancer (EOC) progressing after systemic chemotherapy. In this paper, we report a review and a metanalysis of heterogeneous data published up to date.
Data sources
The literature search was restricted to single-agent MOC. The analysis was conducted through March 2023 by consulting PubMed, Embase, Google Scholar, and The Cochrane Library databases. Research string and Medical Subject Headings included “ovarian tumor,” “ovarian carcinoma,” or “ovarian cancer,” “fallopian tube cancer,” “primary peritoneal cancer,” “oral chemotherapy,” and “metronomic cyclophosphamide.” All articles were assessed for quality by at least two investigators independently, and a < 18 patients sample size cutoff was chosen as a lower limit with a Cohen's kappa statistical coefficient for accuracy and reliability. Metanalysis of selected papers was carried out according to a fixed model.
Data summary
The percentage of agreement between investigators on literature study selection was very high, reaching 96.9% with a Cohen's k of 0.929. MOC pooled objective response rate (ORR) and disease control rate for recurrent or platinum-refractory ovarian cancer were 18.8% (range 4–44%) and 36.2% (range 16–58.8%), respectively. The mean progressive-free survival and overall survival were 3.16 months (range 1.9 to 5.0 months) and 8.7 months (range 8 to 13 months), respectively. The fixed model metanalysis of selected studies showed a 16% median ORR (12–20% CI, p < 0.001).
Conclusions
Single-agent oral cyclophosphamide in EOC holds promise as a treatment option, even in the era of precision medicine. Genetic factors, such as DNA repair gene polymorphisms, may influence treatment response. Combining cyclophosphamide with biological agents such as PARP inhibitors or immunotherapy agents is an area of active investigation.
Introduction
Metronomic or low-dose chemotherapy (MC) treatment refers to the chronic administration of low doses of antiproliferative agents that can sustain prolonged and active plasma levels of drugs with a favorable tolerability profile and promising efficacy in solid tumors. 1 The USA National Cancer Institute defined MC as a “treatment in which low doses of anticancer drugs are given on a continuous or frequent, regular schedule - such as daily or weekly - usually over a long time.” 2 It is not a new therapeutic strategy, as it has been widely used in the last two decades and is still prescribed for the palliation of incurable solid tumors.3–6
In addition to the intrinsic direct cytotoxic activity, MC acts through various mechanisms, such as inhibition of angiogenesis and immune response modulation, influencing the tumor microenvironment.7–9 Experimental data in rodents showed that MC causes DNA damage, a consistent decrease in circulating endothelial progenitors’ numbers and viability, and more durable tumor growth inhibition.10–13 MC caused tumor growth inhibition without increased toxicity in a preclinical orthotopic ovarian cancer model. MC increases the glycoprotein thrombospondin-1 which exerts an anti-angiogenetic effect thought the expression of FAS ligand and the consequent activation of caspases leading to apoptosis and decreased cancer stem cells.11,13 Prolonged in vitro exposure to chemotherapeutic agents causes induction of gene and protein expression thrombospondin-1 chi has been suggested to be a mediator of low-dose MC anti-angiogenetic effect.12,13 MC can also deplete immunosuppressive regulatory T cells and restore T and NK cell function in patients with advanced tumors, favoring a better control of tumor progression.14,15
The alkylator agent cyclophosphamide (CTX) is one of the most studied drugs employed in an oral metronomic schedule as a single agent or combined with other drugs, including other chemotherapeutics, anti-angiogenetic agents, and tyrosine kinase inhibitors.6,16 MOC has been used as a single agent or in combination with other agents for several solid tumors, including sarcoma and ovarian, lung, and prostate cancers, with interesting results in disease palliation, particularly in metastatic breast carcinoma, with only a slight increase of toxicity.17–20
In real life, MOC has demonstrated promising antitumor and antiangiogenic activity that may achieve prolonged progression-free survival (PFS) and improved objective response rates (ORRs) in patients with recurrent epithelial ovarian cancer (EOC) with progressive disease (PD) after intravenous systemic chemotherapy. 21 The co-administration of antiangiogenic agents may enhance these effects. Several trials, primarily retrospective, have reported the activity and safety of MOC in different patient populations whose results might have been influenced by small sample sizes and different inclusion or exclusion criteria. Another source of variability comes from using various combination regimens and schedules of MC. 21 Therefore, results may be difficult to interpret and draw clear conclusions. In this review, we report a meta-analysis of published data on MOC as a single agent in recurrent or residual EOC, fallopian tube, or primary peritoneal cancer.
Methods
Studies selection
A literature search was restricted to single-agent MOC. The search was conducted through March 2023 by consulting PubMed, Embase, Google Scholar, and The Cochrane Library databases. Research string and Medical Subject Headings included “ovarian tumor,” “ovarian carcinoma,” or “ovarian cancer,” “fallopian tube cancer,” “primary peritoneal cancer,” “oral chemotherapy,” and “metronomic cyclophosphamide.” The analysis included only fully published, prospective or retrospective studies and single-arm or randomized trials reporting overall ORR, disease control rate (DCR), complete or partial response (CR, PR), stable disease (SD), PD, PFS, overall survival (OS), and rate of treatment-related adverse events (SAEs). Literature analysis excluded abstracts, preclinical research, case reports, conference papers, and phase II or retrospective studies with MOC in combination with other agents or with a small sample size. All articles were assessed for quality by two investigators independently, and a < 18 patients sample size cutoff was chosen as a lower limit.
Agreement between investigators in choosing scientific papers to be included in the review was calculated by Cohen's kappa statistical coefficient, which represents the degree of accuracy and reliability. The whole team decided on the document retrieval methods. One researcher conducted a literature search. Two investigators cross-checked the relevant studies independently, selected the eligible articles, and extracted the information. The whole team solved problems through discussion and negotiation. The extracted data included first author, year of publication, country of origin, clinical trial design, types of cancer, drug schedules, median age, number of participants, median prior chemotherapy lines, ORR, DCR, median follow-up time, criteria for assessing effectiveness and toxicity, PFS, OS, SAEs.
Eligible patients had to be ≥ 18 years old with histologically confirmed recurrent or residual epithelial ovarian, fallopian tube, or primary peritoneal cancer. The patient's disease was then sub-classified as platinum-refractory or resistant cancer. Patients with platinum-free interval (PFI) of 6 months or longer were considered platinum-sensitive disease. In contrast, those with less than six months of PFI were considered to have a platinum-resistant disease. Patients who experienced disease progression during first-line platinum therapy were considered a platinum-refractory disease.
Statistics
Investigators employed Cohen's kappa coefficient to measure the inter-rater degree of agreement on selected papers. 22 Objective responses were reported as percentages rounded up to the nearest unit. Classical fixed model metanalysis employed a GraphPad statistical software (Boston, MA, USA). Data were reported both as forest and funnel plots.
Results
Literature analysis
Figure 1 shows papers retrieval and selection for single-agent, oral, metronomic cyclophosphamide. The percentage of agreement between investigators on study selection to include in the analysis was very high, reaching 96.9% with a Cohen's k of 0.929. Ferrandina et al. treated 50 heavily pretreated, relapsed EOC patients with oral MOC at the dose of 50 mg daily, continuously achieving a 20.4% ORR with eleven patients (20.4%) experiencing SD, and eight of them had an objective response duration ≥6 months. 23 Twenty patients (37.0%) had primarily platinum-refractory/resistant EOC, while 34 patients (63.0%) had platinum-sensitive disease. Seventy-nine percent of patients had received ≥2 previous lines before starting MOC. The median PFS was four months, and the 12-month PFS rate was 19.6%; the median OS was 13 months, and the 12-month OS rate was 51.5%. Responder patients had a more favorable PFS (median 17 months) compared to those with SD (median 6 months) or PD (median 3 months; p = 0.0001). The median OS of responding patients was 30 months compared to 11 months in cases with SD or PD (median 8 months; p = 0.0001). Only one patient reported grade 3 anemia. Non-hematological grade 3 toxicity was observed in two patients. In 2015, Kummar et al. reported the activity of single-agent, low-dose 50 mg orally once daily CTX showing a 23% ORR (1 CR, PR 19.4%; 95% CI: 8.2–36.0%) and good tolerability in a series of 37 patients within high-grade serous EOC, peritoneal, fallopian tube, and BRCA-mutant ovarian cancers. 13 This study randomized patients to single-agent MOC versus MOC plus the PARP inhibitor olaparib but failed to demonstrate an advantage for the combination. Four patients who responded to the CTX-only arm had BRCA-mutant ovarian cancer (including the patient with a CR), two had HGSOC, and one had fallopian tube cancer. Additionally, six patients in the MOC-only arm had SD for six or more treatment cycles. One patient on the MOC arm had prolonged SD, receiving more than 32 treatment cycles. Exome analysis of the tumor samples from this patient revealed a mutation in BRCA2. Median PFS was 2.1 months for CTX-alone, but stratification by BRCA status reveals a subset with improved PFS. Three patients experienced grade 3 lymphopenia. Mild leukopenia was recorded in 13 patients, and grade 1 diarrhea and anemia in 1 and 2 cases, respectively.
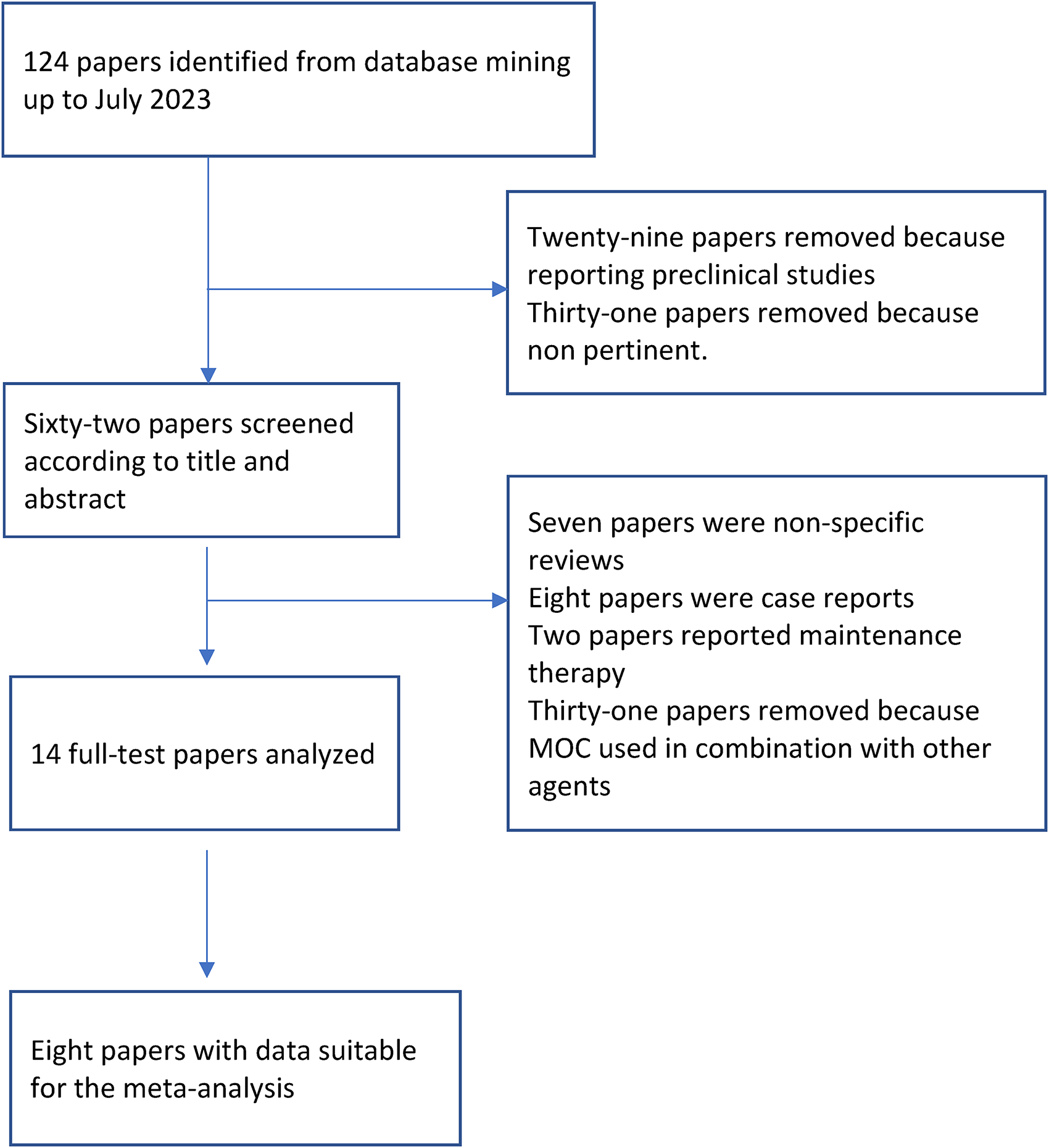
Papers retrieval and selection for single-agent, oral, metronomic cyclophosphamide.
Handolias et al. carried out a retrospective study on 23 patients from three centers in Australia who had received oral CTX 150 mg day 1–14 for recurrent EOC. 24 Patients received a median number of previous chemotherapy regimens of three (range 1–6). The response rate was 44%, with 10 of the 23 patients achieving a PR according to the GCIG (CA125) criteria. The median number of cycles received was three (range 1–16). MOC showed activity in patients with platinum-sensitive (seven of 13 PR) and resistant or refractory disease (three of 10 PR). There was no grade 3–4 toxicity. However, two patients withdrew CTX due to less severe non-hematological toxicity.
Wong et al. reported a retrospective review of 20 mostly pretreated patients with a median number of previous chemotherapy regimens of 4 (range 1–8). 25 All patients had received platinum plus paclitaxel as the front-line chemotherapy. Patients received oral MOC 50 or 100 mg daily. CR and PR were obtained in 3 and 2 patients, respectively. Five patients were reported as having SD. The median PFS was 3.75 months (range 1.25–15 months). Grade 2–3 myelosuppression was the only side effect.
Ramanan et al. reported 71 patients with EOC treated with MOC 50 mg for 10 days, recycled every 28 days without significant toxicity. 26 Forty-four patients (62%) were platinum-refractory/resistant, while 26 patients (36.6%) were platinum-sensitive; 60 patients (84%) had received at least one previous chemotherapy before starting MOC. The ORR was 20%, lasting a median of ≥ 6 months; eight patients had continued response for over 1 year. Twenty-two patients (31%) had SD. Median PFS was 5 months (range 2–25 months), the 12-month PFS was 11%, and the 12-month OS was 26%.
Gupta et al. published a randomized phase II clinical trial comparing the therapeutic benefit of single-agent MOC at 50 mg oral daily dose with or without the antiangiogenic drug celecoxib. 27 In the single-agent MC arm, 4 out of 26 patients had a PR, and three patients (12%) had SD >6 months. There was no significant difference between groups in OS (9.69 vs. 12.55 months or in median time to treatment failure (1.84 vs. 1.92 months). The most common grade 2 side effects were fatigue (27%), abdominal pain (23%), nausea/vomiting (27%), diarrhea (19%), hyperglycemia (15%), hypoalbuminemia (12%), anemia (15%), and sensory neuropathy (4%). Treatment was well tolerated overall with minimal grade 3–4 toxicities. In Arm A, one patient had grade 4 hypercalcemia, and Grade 3 toxicities included fatigue in 8% of cases. Four percent of patients reported infection, muscle weakness, sensory neuropathy, and dyspnea.
Hall et al. investigated the safety and efficacy of a combination of oral MOC 100 mg once daily in patients randomized to MOC with placebo or the oral anti-angiogenetic tyrosine kinase inhibitor, nintedanib in patients with relapsed ovarian, fallopian tube, or primary peritoneal cancer. 28 Patients were heavily pretreated since 39% and 30% had received more than five lines of chemotherapy and bevacizumab, respectively. In the single-agent MOC arm, the 6-month PFS rate was 22.8%, and the median OS was 6.4 months. The SAE rate was 54%, but mostly fatigue (9.1%) and vomiting (7.3%), being non-symptomatic lymphopenia, was the most frequent toxicity (16.4%). Diarrhea and neutropenia were absent. Overall, 26 patients (23%) took oral cyclophosphamide for ≥6 months. These data may be linked to a higher rate of patients with more indolent disease or a higher dose of cyclophosphamide.
Recently, Spiliopoulou et al. published a retrospective analysis of 50 patients treated with oral MOC for relapsed EOC, reporting a PR rate of 32% (combined Gynecologic Cancer Inter-Group criteria), SD rate of 16%, PFS, and OS of 2.6 months and 6 months, respectively. 29 A gBRCA1/2 mutation reduced the risk of radiologically detected disease progression (OR 0.07, p = 0.02). Patients with gBRCA1/2 mutations had improved PFS (7.9 vs. 2.5 months, p = 0.003) and OS (15.5 vs. 6 months, HR 0.49, 95% CI 0.28 to 0.85, p = 0.02) with MOC when compared with patient without gBRCA1/2 mutations (or unknown gBRCA1/2 status). Overall, gBRCA1/2 status can be an independent predictor of response.
Overall analysis
MOC pooled ORR and DCR for recurrent or platinum-refractory ovarian cancer were 18.8% (range 4–44%) and 36.2% (range 16–58.8%), respectively. The mean PFS and OS were 3.16 months (range 1.9 to 5.0 months) and 8.7 months (range 8 to 13 months), respectively. The fixed model metanalysis of selected studies showed a 16% median ORR (12–20% CI, p < 0.001). Figure 2 shows the forest and the funnel plots of the above-reported studies, respectively. Table 1 shows studies included in the analysis. Table 2 shows fixed and random effects as well as estimated standard error.
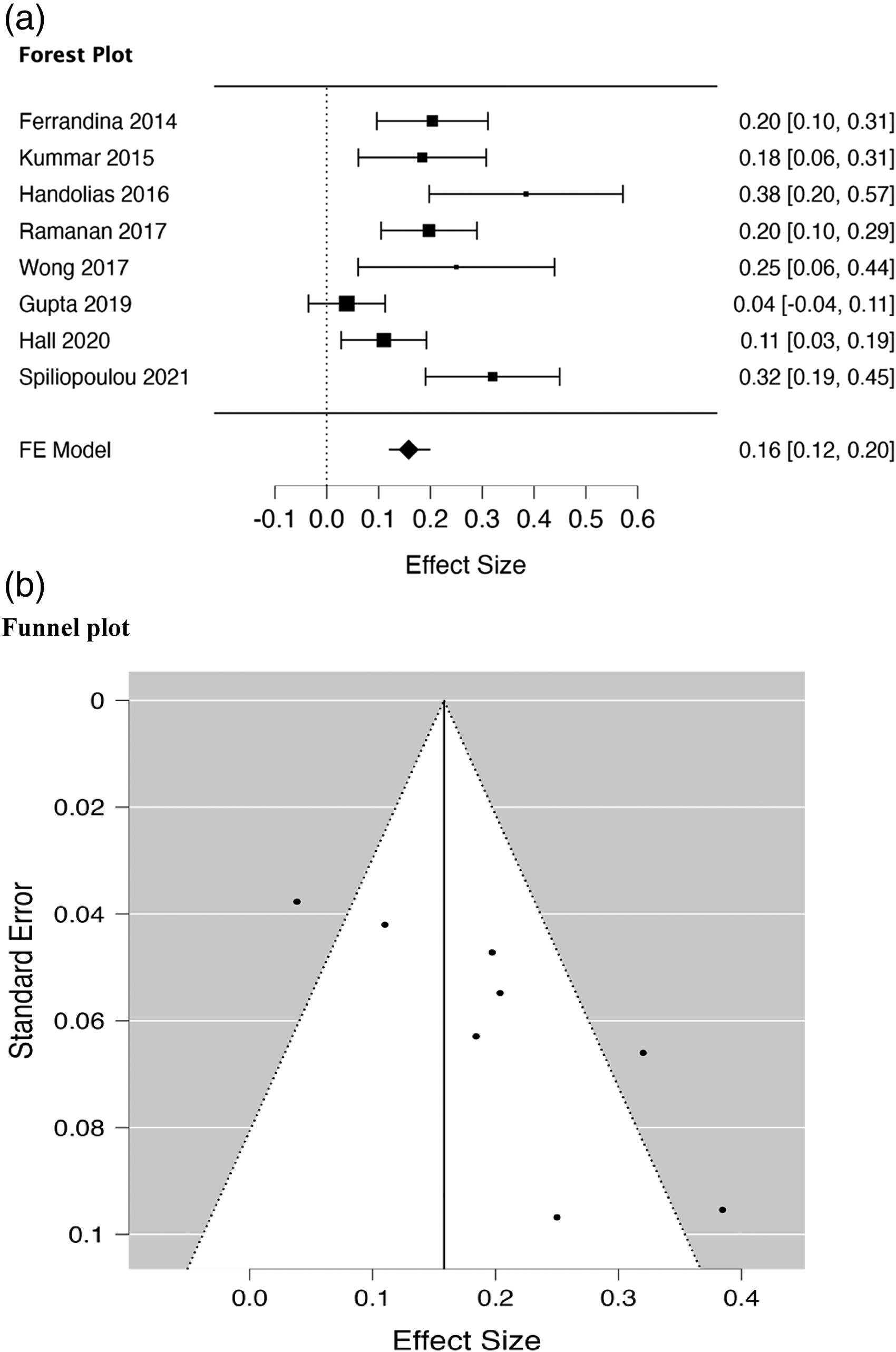
Forest (a) and funnel plots (b) of selected papers.
Studies included in the analysis.
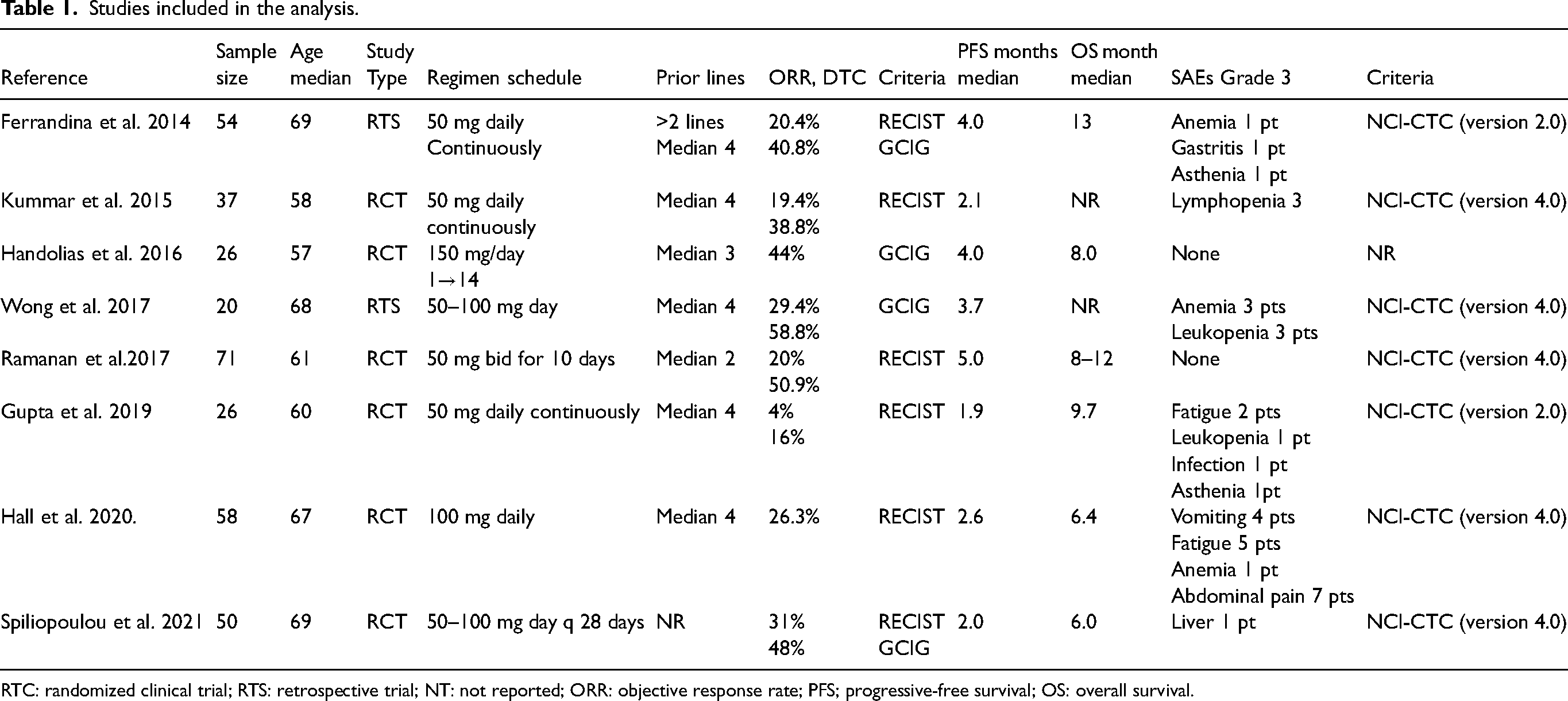
RTC: randomized clinical trial; RTS: retrospective trial; NT: not reported; ORR: objective response rate; PFS; progressive-free survival; OS: overall survival.
Metanalysis of selected studies of single-agent oral metronomic chemotherapy in advanced ovarian carcinoma.

Note. Wald test.
Discussion
Metronomic dosing of CTX, which involves chronic administration at low doses, has shown effectiveness in various tumor types, including EOC. 13 The use of oral CTX in women with EOC has been investigated in several studies, providing insights into its clinical activity and potential predictors of response. This approach induces DNA damage and may target tumor angiogenesis by inhibiting tumor endothelial cell proliferation by activating thrombospondin-1, acting as an anti-angiogenetic strategy. 13 Angiogenesis plays a crucial role in the carcinogenesis and progression of EOC to the point that optimal medical treatment includes agents that target the VEGF/VEGFR pathway, such as bevacizumab and agents that target receptor tyrosine kinase and non-VEGF/VEGFR targets of angiogenesis. 30 Moreover, preclinical data suggest a potential additive effect of anti-angiogenetic agents and MC. 31 Two retrospective studies assessed the efficacy and safety of bevacizumab 10 mg/kg intravenously every 2 weeks plus low-dose MOC 50 mg daily in heavily pretreated patients with recurrent EOC. ORR was 42–45% with a median PFS of 4.5–5.0 months and a median OS of 9–11-7 months.32,33 Bevacizumab plus MOC is an effective, low toxic, and easily administrable regimen which may represent a good therapeutic option in patients run down from systemic intravenous chemotherapy.
Besides its direct effect on neoplastic cells, CTX has been investigated with other agents for treating OC. Poly (ADP-ribose) polymerase inhibitors (PARP) have emerged as a promising targeted therapy for EOC, particularly in patients with homologous recombination deficiency. 34 PARP inhibitors are active in cells with impaired DNA repair, such as BRCA mutations or HR deficiency. Bousssios et al. reviewed the use of PARP inhibitors in EOC and highlighted the potential for combination strategies with other anticancer therapies. 35 Combining CTX with PARP inhibitors may enhance DNA damage and HR deficiency, improving treatment outcomes at least in some BRCA-mutated patient subpopulations.
CTX has also been shown to modulate the immune response by affecting regulatory T cells and myeloid-derived suppressor cells. 36 This immunomodulatory effect of CTX has led to investigations into its combination with immunotherapy agents, such as immune-checkpoint inhibitors.36,37 Preclinical studies have demonstrated synergistic effects when combining CTX with immune-checkpoint inhibitors, leading to enhanced antitumor immune responses. Immunotherapy has shown promise in various cancers, and low-dose CTX has been shown to have immunomodulatory effects. 36 Clinical trials evaluating the combination of CTX with immune-checkpoint inhibitors, such as pembrolizumab or nivolumab, are underway in EOC patients. 37 Liu et al. assessed the combination of the immune-checkpoint inhibitor nivolumab and the antiangiogenic agent bevacizumab in recurrent OC. They reported a clinical benefit in some patients. 38 Adding CTX to immunotherapy regimens may enhance the immune response and improve treatment outcomes. Considering the potential side effects and toxicities associated with CTX treatment is essential.
Mantan et al. compared the efficacy of intravenous pulse CTX with a combination of intravenous dexamethasone and oral CTX in patients with steroid-resistant nephrotic syndrome. 39 This study highlighted the need to carefully monitor and manage adverse events associated with CTX therapy.
The present meta-analysis shows that single-agent MOC may benefit some patients with recurrent OC. The metanalysis of selected studies showed a 16% median ORR (12–20% CI, p < 0.001). These data align with those recently reported by our group in 62 patients, with exciting activity, especially in indolent ovarian cancer. 40 Interestingly, a significant percentage of patients showed a stabilization disease, even if short-lasting. A recent exhaustive review and meta-analysis on the activity and toxicity of all schedules and combinations of MOC for recurrent or platinum-refractory EOC reported an ORR and DCR of 25% (95% CI 12–41) and 61% (95% CI 43–77), respectively, and a median PFS exceeding 4 months (95% CI 2.62–5.97) and a median OS of (95% CI 2.62–5.97) and 11.26 months (95% CI 8.13–14.39). 7
Low-dose MC is reasonably well tolerated with mild grade 1–2 toxicity in most cases. Severe grade 3–4 side effects are rare and mainly represented by neutropenia or lymphopenia with an incidence lower than 7% and transaminitis more peculiar for methotrexate. 41
The toxicity of single-agent MOC was relatively low but increased if MOC was combined with bevacizumab and pazopanib.
One crucial aspect may be awareness of the potential predictive factors for response to oral CTX treatment. Several studies have investigated potential biomarkers to identify patients more likely to benefit from CTX therapy. Some studies explored the association between DNA repair gene polymorphisms and treatment response in EOC patients receiving chemotherapy.42–46 These studies found that specific polymorphisms in DNA repair genes, such as XRCC1 and XRCC3, were associated with improved ORR and survival outcomes. These findings suggest that genetic variations in DNA repair pathways may influence the efficacy of CTX treatment in EOC. In addition to genetic factors, the tumor microenvironment is crucial in determining treatment response.
Limitations
The potential limitation of this review is represented by the nature of the papers included in the analysis. Several studies are retrospective series or phase I/II trials whose results might have been influenced by small sample sizes and different inclusion or exclusion criteria.6,7 Another source of variability comes from using various combination regimens and schedules of MOC. 7 Therefore, interpreting results may be challenging and lead to more precise definitive conclusions.
Conclusions
In conclusion, oral CTX holds promise as a treatment option in largely pretreated women with EOC or if one wants to assure a systemic intravenous chemotherapy-free interval, even in the era of precision medicine. Genetic factors, such as DNA repair gene polymorphisms, may influence treatment response. Combining CTX with immunotherapy agents is an area of active investigation. Further research and clinical trials are needed to optimize the use of oral CTX in managing EOC and identify patient populations that may benefit the most from this treatment approach. Combination strategies with PARP inhibitors and immunotherapy are being explored to enhance treatment outcomes.
Footnotes
Author contributions
VG and FM designed the study. GS, MRV, MVS, and SC collected the data and performed the analysis. VG, PV, and PS interpreted the results. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
