Abstract
Background
Cancer patients often experience preventable infections, including influenza A and B. These infections can be a cause of significant morbidity and mortality. The increased risk of infection may be because of either cancer itself or treatment-induced immunosuppression. 1 Influenza immunization has been shown to decrease the risk of influenza infection in patients with intact immunity. 2 In cancer patients, active immunization has been shown to confer protective immunity against several infections at similar rates to healthy individuals, which has translated into decreased duration and severity of infection and potentially improved morbidity and mortality. 3
Objectives
To assess the efficacy of influenza vaccination in stimulating immunological response in patients with cancer during chemotherapy compared to control groups.
To assess the efficacy of influenza vaccination in preventing confirmed influenza and influenza-like illness and/or stimulating immunological response in children with cancer treated with chemotherapy, compared to placebo, no intervention, or different dosage schedules.
To determine the adverse effects associated with influenza vaccination in patients with cancer.
Search Methods
We searched MEDLINE/PubMed database for articles published from 1964 to 2013 using the search terms “cancer,” “adult,” “influenza vaccination,” and “chemotherapy.”
Selection Criteria
We included studies based on systematic sampling with defined clinical criteria irrespective of the vaccination status of cancer patients. Studies measure the serological response or clinical response to compare between the study group and the control group. Studies assessed the inactivated influenza vaccines and live attenuated influenza vaccine (LAIV) protective serological reaction and the clinical outcomes after vaccination.
Data Collection and Analysis
Two independent authors assessed the methodological quality of included studies and extracted data.
Main Results
We included 16 studies (total number of participants = 1,076). None of the included studies reported clinical outcomes. All included studies reported on influenza immunity and adverse reaction on vaccination. We included 6 solid tumor studies and 10 hematological studies. In 12 studies, the serological response to influenza vaccine was compared in patients receiving chemotherapy (n = 425) versus those not receiving chemotherapy (n = 376). In three studies, the serological responses to influenza vaccination in patients receiving chemotherapy are compared to that in healthy adult. Measures used to assess the serological responses included a four-fold rise increase in antibody titer development of hemagglutination inhibition (HI) titer >40, and pre- and post-vaccination geometric mean titers (GMTs). Immune responses in patients receiving chemotherapy were consistently weaker (four-fold rise of 17–52%) than in those who had completed chemotherapy (50–83%) and healthy patients (67–100%). Concerning adverse effects, oncology patients received influenza vaccine, and the side effects described were mild local reactions and low-grade fever. No life-threatening or persistent adverse effects were reported.
Authors’ Conclusion
Patients with solid and some of hematological tumors are able to mount a serological response to influenza vaccine, but it remains unclear how much this response protects them from influenza infection or its complications. Meanwhile, influenza vaccine appears to be safe in these patients. While waiting results of randomized controlled trials to give us more details about the clinical benefits of the influenza vaccination, the clinicians should consider the currently proved benefits of influenza vaccination on management of the cancer patients undergoing systematic chemotherapy such as decrease in the duration and severity of the of the disease, and significant decrease in influenza-associated morbidity and mortality in these high-risk patients. 3
Background
Cancer patients often experience preventable infections, including influenza A and B. These infections can be a cause of significant morbidity and mortality. The increased risk of infection may be because of either cancer itself or treatment-induced immunosuppression. 1
Infection often delays the anticancer therapy, worsening the oncologic outcome. Each year, influenza infectious adverse events contribute to up to 36,000 deaths and 226,000 hospitalizations in general population. 2 Influenza virus-related mortality can reach up to 9% in cancer patients undergoing active therapy. 3
Influenza immunization has been shown to decrease the risk of influenza infection in patients with intact immunity. 4 In cancer patients, active immunization has been shown to confer protective immunity against several infections at similar rates to healthy individuals, which has translated into decreased duration and severity of infection and potentially improved morbidity and mortality. 5
However, it is currently unclear whether patients undergoing systemic chemotherapy can achieve ideal serologic responses to vaccines. Knowledge regarding vaccine efficacy, safety, and ideal timing in this patient population is still limited.
Although the Centers for Disease Control and Prevention (CDC) recommends annual influenza vaccination for high-risk population, including health care workers, the elderly, and patients who are immunosuppressed or have chronic medical conditions and malignancies, only less than 50% of patients receiving chemotherapy routinely receive the influenza vaccination.3,5,6
Influenza Virulence
In the United States, annual epidemics of influenza occur typically during the late fall through early spring. Influenza viruses can cause disease among persons in any age group, but rates of infection are the highest among children.7–9 Rates of serious illness and death are highest among persons aged ≥65 years, children aged <2 years, and persons of any age who have medical conditions that place them at increased risk for complications from influenza.7,10 Influenza may also result in secondary bacterial pneumonias, sinusitis, ear infection, or a worsening of chronic respiratory condition.11,12 There are main two types of influenza virus, type A and B, that are responsible for the majority of cases of severe disease in humans. Influenza A is further classified based on the presence of two surface antigens, hemagglutinin (HA) and neuraminidase (NA), and influenza B is separated into two genetic lineages, Yamagata and Victoria.2,12 Any changes in the amino acid sequences of HA and/or NA result in seasonal epidemics. In April 2009, a novel influenza A (H1N1) virus, often referred to as pdm 2009 A/H1N1, that is similar to but genetically and antigenically distinct from influenza A (H1N1) viruses previously identified in swine was determined to be the cause of respiratory illnesses that spread across North America and were identified in many areas of the world by May 2009.13,14 Influenza morbidity caused by the pdm 2009 A/H1N1 remained above seasonal baselines throughout spring and summer 2009 and was the cause of the first pandemic since 1968. The pdm 2009 A/H1N1 has now mostly replaced the H1N1 virus that was previously circulating in humans. 2
Cancer and immunization
Cancer increases the risk of complications from influenza, including recurrent hospitalization and death. 15 In cancer patients, altered humoral and cellular immunity has been noted.16,17 Moreover, many cancer patients are treated with cytostatic and immunosuppressive drugs, and chemotherapy has also been associated with various disorders. 17 For these reasons, patients with cancer may be considered a high-risk group who are at risk of particularly serious post-influenza complications and who should be immunized against influenza before every epidemic season. In a study of patients with solid tumors, such as lung and breast cancer, who were not undergoing systemic chemotherapy, the patients were able to mount protective antibody titers to influenza vaccination that approached the level of healthy controls. 17 Protective antibodies after vaccination in this population were significant when compared with patients receiving chemotherapy, and there were no major complications attributable to vaccination. 18 Patient with lung cancer, in particular, developed protective antibody responses to influenza vaccine, which did not appear to be affected by systemic steroid treatment, recent chemotherapy, or lung cancer histology. 5 Influenza vaccination coverage is currently low among cancer patients undergoing systemic treatment: only 18% in the 18–49-year group and 32% in the 50–64-year group. 19 CDC recommends that people who live with or care for a person at high risk for flu-related problems get the flu shot too. This means that if you are being treated for cancer, your family members, caregivers, and children at home should get the flu shot. 2
Methods
The MEDLINE/PubMed database was reviewed for articles published from 1964 to 2013 using the search terms “cancer,” “adult,” “influenza vaccination,” and “chemotherapy.” Studies included in this review meet one of these criteria:
Study based on systematic sampling with defined clinical criteria irrespective of the vaccination status of cancer patients.
Study assessed inactivated influenza vaccines and live attenuated influenza vaccine (LAIV) protective serological reaction among cancer patients.
Study assessed the clinical outcomes and immune response after vaccination among cancer patients.
Study showing the efficacy of influenza vaccination among different types of cancer.
Study assessed the potential benefits and complications of vaccination among cancer patients.
Study assessed the timing of vaccination.
Study reported overall vaccine efficacy against all circulating influenza strains.
Search Methods
A total of 251 studies were identified in initial PubMed database search of key words. In all, 52 studies were selected based on reviewing the abstracts of the 251 studies. These 52 studies were handling the same topic of interest of our review. After reviewing the 52 studies, only 16 studies met our inclusion criteria and reviewed the full articles in depth. We further divided the articles’ outcomes among the selected population having different types of underlying cancer disease, solid versus hematological tumors (Fig. 1). We focused in our analysis of the selected papers on the time vaccination in patients on chemotherapy versus patients out of therapy. We kept our attention also on the type of outcome measurement of the vaccination efficacy and protective titer used in the studies versus clinically based influenza flue-like illness disease.
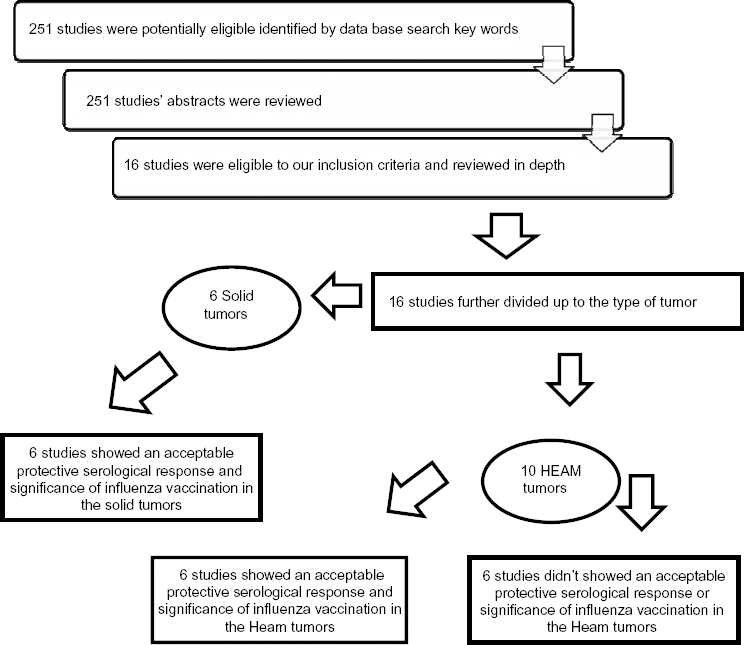
Review strategy.
Data Extraction
The two review authors independently performed data extraction using standardized forms. We extracted data on the characteristics of the participants (tumor type, anticancer treatment received, timing of vaccination, measurement of the vaccination efficacy, and series of vaccine administrations) and outcomes measures (immunological response to vaccination, laboratory response, hospitalization, length of stay in the hospital, and pneumonia). We examined and discussed the articles until consensus was made.
Assessment of risk of bias in included studies
The two review author's independently assessed trial quality. We assessed mythologies of the included study and contacted study authors for additional information where necessary. Disagreements were resolved by discussion among the review authors.
Main Results
We included 16 studies (total number of participants = 1,076). None of the included studies reported clinical outcomes. All included studies reported on influenza immunity and adverse reaction on vaccination. We included 6 solid tumor studies and 10 hematological studies. In 12 studies, the serological response to influenza vaccine was compared in patients receiving chemotherapy (n = 425) versus those not receiving chemotherapy (n = 376). In three studies, the serological responses to influenza vaccination in patients receiving chemotherapy are compared to those in healthy adult. Measures used to assess the serological responses included a four-fold rise increase in antibody titer development of hemagglutination inhibition (HI) titer >40, and pre- and post-vaccination geometric mean titers (GMTs). Immune responses in patients receiving chemotherapy were consistently weaker (four-fold rise of 17–52%) than in those who had completed chemotherapy (50–83%) and healthy patients (67–100%). Concerning adverse effects, oncology patients received influenza vaccine, and the side effects described were mild local reactions and low-grade fever. No life-threatening or persistent adverse effects were reported.
Outcomes
Influenza immunity (difference in pre- and post-influenza vaccination HI antibody titer)
All the 16 sampled studies determined the efficacy of vaccination by measuring HI titer, a serum antibody titer of 40 or four-fold rise increase in HI titer which is normally considered protective in healthy individuals. Seven studies defined as protective the development of HI titer of >40. Two studies defined as protective the development of HI titer of >20.
Laboratory-confirmed influenza infection within the epidemic period
They were not reported outcome measures. There are no stated methods used to identify influenza infection.
Influenza-like illness, pneumonia, hospitalization, length of stay, delay in chemotherapy, and mortality
They were not reported as an outcome measure in any of the included studies.
Comparisons related to the efficacy of the influenza vaccination in adult cancer patients during chemotherapy compared with that in other control groups
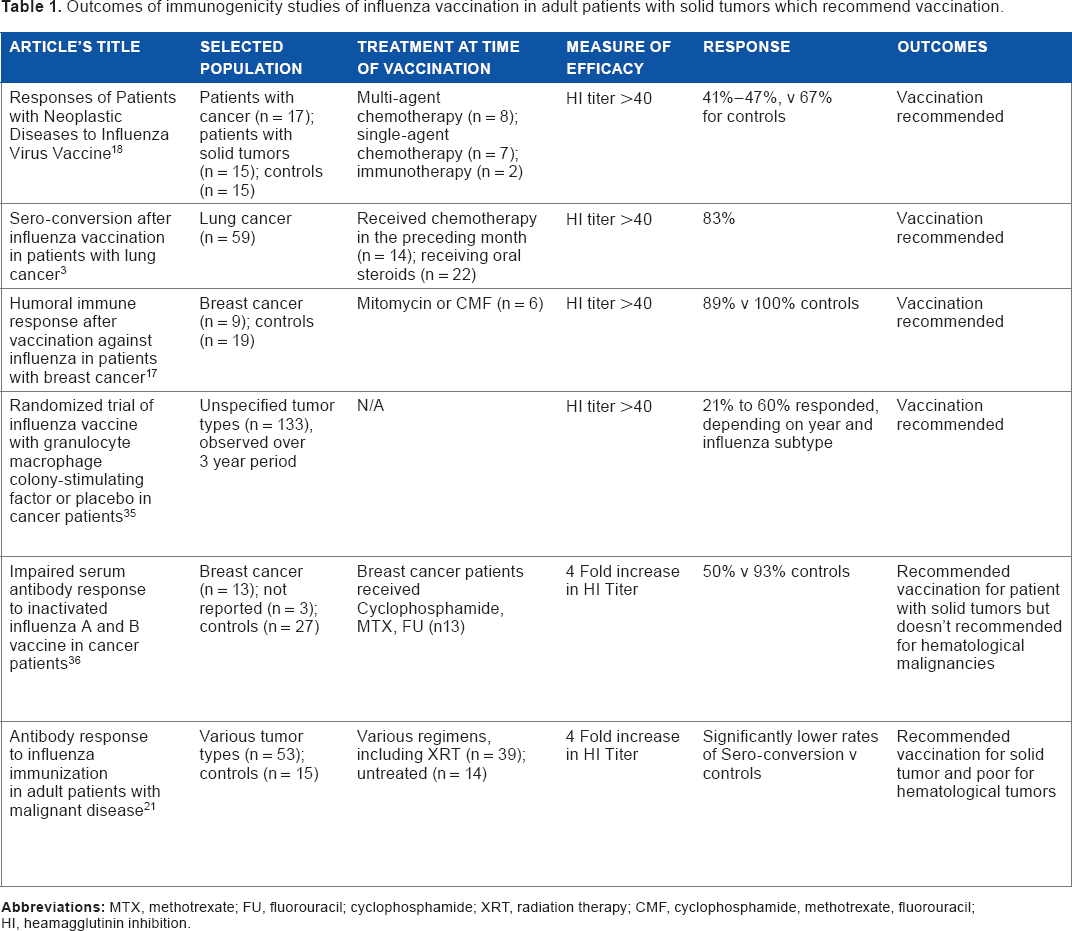
Three studies20–22 (Table 3) reported on this comparison. Result on protective HI titer, four-fold rise in antibody titer in pre and post-vaccination immune assay. The analysis shows that the serological responses to influenza vaccination in adult receiving chemotherapy were weaker than in those completed chemotherapy.
A total of 12 studies (Tables 1–3) reported on this comparison. Result on protective HI titer, four-fold rise in antibody titer in pre- and post-vaccination immune assay. The analysis shows that the serological response in patients receiving chemotherapy was weaker than those in healthy controls. Meanwhile, these patients under chemotherapy still mount a protective (HI) immune antibody titer.
In all, 16 studies (Tables 1–3) reported on this comparison. The analysis shows 6 studies (Table 1) with solid tumors mounted a protective HI titer and 12 studies with hematological tumors, and concluded with vaccination recommendation. Six studies (Table 2) with hematological tumors mounted a protective HI titer and concluded with vaccination recommendation, whereas the other six studies (Table 3) with hematological tumors did not find a significance of vaccination and they did not recommend vaccination.
Two studies23,24 reported on this comparison. One study (Table 1) reported significance of two vaccination schedules. The other study (Table 2) showed no significance of two doses of vaccination.
Outcomes of immunogenicity studies of influenza vaccination in adult patients with solid tumors which recommend vaccination.
Outcomes of immunogenicity studies of influenza vaccination in adult patients with hematological tumors which recommend vaccination.

Outcomes of immunogenicity studies of influenza vaccination in adult patients with hematological tumors which didn't recommend vaccination.

Discussion
Cancer and influenza vaccination issue is still unconcluded especially in patient with cancer undergoing systemic chemotherapy. This specific group of patients seems to have an increased risk of infection for which influenza vaccine may offer additional protection and significant benefit.5,17,2 5 There are positive data in the majority of the studies reviewed showing vaccine efficacy and serological protective levels in patients with solid tumors such as breast cancer and lung cancer.5,17On the other hand, some other studies found that immunization has no benefit in providing adequate sero-conversion, especially in patients with lymphomas; hence, they did not recommend the vaccination in these patients.24,26,27
Brown et al concluded that two months are needed for influenza vaccination after completion of chemotherapy. 30 Chrisholm et al added children with acute lymphocytic leukemia (ALL), and supported the recommendation that all children receiving chemotherapy for cancer and those within six months of completing therapy should undergo annual influenza immunization.
Cost and dosage series
Molinari et al conducted a study to measure the annual impact of influenza vaccination and the cost and concluded immunization against influenza can effectively reduce the annual economic burden of influenza in the United States. 31
CDC or Advisory committee on immunization practices (ACIP) still doesn't recommend increasing in dose series in healthy adults although a study of non-Hodgkin lymphoma (NHL) receiving chemotherapy proved that 50% had a high HI titer who received two doses of influenza vaccination versus only 42% for the patients who received one dose of influenza vaccination.25,32,23 It has been proven in healthy adult population that higher dosages of influenza vaccination provide a good result and improvement in the level of HI titer. 33 However, other studies showed that high dosage influenza vaccination in NHL produces better responses and is well tolerated by the patients. 34
Debate is still ongoing on the optimal vaccination of patient with cancer undergoing chemotherapy. Many studies handled this issue in depth, but each study has its supportive and unsupportive evidences regarding the timing of vaccination. Some studies supported vaccination during chemotherapy cycle, 29 whereas other studies reveal the opposite meaning that the patients who completed the course chemotherapy have better responses than patients vaccinated at active treatment course.
Authors’ Conclusions
Implication for practices
In national guidelines, it is recommended that patients who are being treated for cancer should be vaccinated against influenza. The full picture and the clinical evidence from randomized controlled studies to support this recommendation are still lacking. We can conclude from the articles included in this review that patients with solid and hematological tumors are able to mount an immune response to influenza vaccine, but it remains unclear whether this immune response fully protects them from influenza infection or its complications. We can emphasize that influenza vaccine appears to be safe in these patients. While awaiting results of randomized controlled trials addressing the complete picture of the clinical benefits from influenza vaccination, the clinicians must consider the benefits of influenza vaccination such as decrease in the duration and severity of the disease and significant decrease in influenza-associated morbidity and mortality in these high-risk patients. 5
Implication for research
A well-designed prospective, multi-center, randomized controlled trial of influenza vaccination in patients being treated for cancer is necessary. This trial should have a low risk of bias and should measure carefully the clinical relevant outcomes, including laboratory-confirmed influenza infection, hospitalizations, hospital length of stay, and pneumonia. This study should be conducted in large-scale population. The purpose of this study is to give the clinical evidence of benefits of influenza vaccination in patients with cancer undergoing systemic chemotherapy.
Limitations
The included studies used different immunization schedules according to guidelines from different eras. These studies were published in the last 50 years. Studies’ population is from different age groups. Age is a possible confounder for immune response. The included studies had relatively small sample sizes. The results described are all based on separate small studies. Larger trials are needed to verify the result of these studies.
Author Contributions
MS, NK conceived and designed the study. MS analyzed the data. MS wrote the first draft of the manuscript. MS contributed to the writing of the manuscript. MS, NK agreed with manuscript results and conclusions. MS, NK jointly developed the structure and arguments of the paper. MS, NK made critical revisions and approved the final version. All authors reviewed and approved the final manuscript.
