Abstract
The cancer prevalence in the Malwa region of Punjab (1089/million/year) is much higher than the national average cancer prevalence in India (800/million/year). The participants in the present study were 50 healthy individuals and 49 cancer patients all living in the Malwa region of Punjab, with the healthy people being selected from the same household as the cancer patients. High concentrations of several potentially toxic elements were found in hair samples from people living in Punjab. Compared to standard reference ranges, the metals in excess in both the control and patient groups were aluminium (Al), barium (Ba), manganese (Mn), strontium (Sr) and uranium (U). The most significant findings were high lead (Pb), U and Ba concentrations. The maximum values for Ba, Mn, Pb and U were found in hair from breast cancer patients. The mean concentration of U in hair from the breast cancer patients was 0.63 μg U/g, which is more than double the value found in the control group and over six times higher than the reference range of 0.1 μg U/g. Water, soil, and phosphate fertilizers all seem to play a potential role, causing an increased metal burden in Punjabi people living in the Malwa region. The present study indicates that metals, and especially U, may be a factor in the development of breast cancer among Punjabi women.
Introduction
Punjab is one of the leading food grain producing states in India. It is also the region with the highest cancer rate. The cancer prevalence (per million per year) in the Malwa region is indicated to be 1089 (DHFW, 2013). 1 This is much higher than in two other regions of Punjab, Majha (647/million/year) and Doaba (881/million/year). The national average cancer prevalence in India is 800/million/year. 1 Four of the 11 districts in the Malwa region are most afflicted by various cancers: Muktsar, followed in order by Mansa, Faridkot, and Bathinda. 1
Breast cancer is widely feared, and studies indicate that excessive use of mineral fertilizers (Table 1) and pesticides has led to water and soil contamination, affecting farming communities. In the Malwa region, cancer mortality is directly correlated with gender and farming (Singh, 2008). 2 The indiscriminate use of pesticides and fertilizers, as well as poor groundwater quality, are considered to be among the main reasons for the high incidence of several diseases in the Malwa belt. 3 Farming communities have higher cancer rates. 4
Consumption of fertilizers in Punjab (000, nutrient tonnes).

Among non-genetic factors, toxic metals are considered to be contributory causes for a variety of cancers.5–8 Exposure to cadmium (Cd) and other heavy metals is a well-known risk factor for cancer, and the effect of Cd on estrogen receptor levels and estrogen-induced responses in human breast cancer cells has been studied. 9 Cadmium and other heavy metals inhibit enzyme function, DNA synthesis and repair. 10
Metals such as Cd and Pb have been found in fruits and vegetables grown on contaminated soil. 11 Finnish studies have shown that more than 90% of the total dietary intake of nutrient elements and potentially toxic metals comes from plant foods, spices and plant-derived beverages. 12 Studies conducted by various reputable institutions have indicated that drinking water, particularly in the Malwa belt, can be a source of heavy metals, including fluoride and pesticides.2,3,13,14 This relatively high intake of contaminated food and water may explain the higher incidence and prevalence of cancer as found in the Malwa region of Punjab.
The Bhabha Atomic Research Centre has tested groundwater samples from four districts of the Malwa region of Punjab (Bathinda, Mansa, Faridkot and Ferozpur) in collaboration with Guru Nanak Dev University of Amritsar, and the results indicated that about 42% of total samples have uranium concentration above the Atomic Energy Regulatory Board (AERB) permissible limit of 60 μg/L for drinking water. 15 The World Health Organization has set the recommended level for uranium in drinking water to, 15 μg/L. 16 The Federal Environment Agency (Umweltbundesamt) in Germany has lowered the acceptable level for U in drinking water to <10 μg/L. 17 The debate about the source of the apparent uranium contamination continues. Some authors speculate that industries like thermal power plants, fertilizer factories, chemical factories, cement factories etc. have an impact. Others predict that uranium could have originated from the Tosham hills in Haryana state of India, where granite rocks are found. Geochemical studies suggest that high salinity and total dissolved solids (TDS) influence the mobility of uranium in the groundwater of these districts. 18
The first purpose of this study was to assess the levels of toxic and essential elements in hair from a group of cancer patients living in the ‘cancer belt’ of the Malwa region and a healthy control group, living in the same area. Hair is a body tissue, and hair metal analysis is a diagnostic test to evaluate chronic metal exposure. Although hair metal analysis is often dismissed as unreliable, a critical evaluation concludes that it is a reliable test for detecting environmental and occupational exposure. 19 In Labor and Diagnose, a German textbook for laboratory medicine, Tomas demonstrates that the concentration of metals such as lead in hair tissue represents that of bone. Lesser concentrations are found in liver, heart and brain tissue. 20 Meißner determined that hair analysis is a diagnostic means to determine chronic exposure, 21 while Hac and Krechniak 22 demonstrated via animal experiments that circulating blood metals such as lead (Pb) actually feed the hair root, leading to elevated tissue concentrations. The uptake of lead, for example, suggests a three compartmental tool for lead metabolism, namely circulation in blood, accumulation in soft tissue (with keratin and collagen being the target proteins), and accumulation in hair and the skeleton. 23 Thus, hair can be used as an indicator of long term exposure to lead and other metals. 24
Our next purpose was to investigate if U is a metal of concern and what other toxic metals may possibly be epidemiologically linked to cancer, regardless of whether the association is a causal one or not. While uranium is a naturally-occurring radioactive element, commonly found in very small amounts in water, rock, soil or human tissue, it contributes to low-level background radiation and various cancers. Long term chronic intakes of uranium isotopes in food, water, or air can lead to internal irradiation and/or chemical toxicity. Intakes exceeding Environmental Protection Agency (EPA) standards increase cancer risks, liver damage, or both. 25 A study by the Department of Environmental Health, Cincinnati, Ohio concluded that ‘for women living within five miles of a uranium processing plant, the degree of exposure to uranium particulates was related to risk of incident breast cancer’. 26
Thirdly, we wanted to evaluate if those metals found at elevated concentrations can be linked to contaminated groundwater or the use of chemicals in agriculture. Indeed, metal intake, especially of U from food and/or drinking water, is unusually high in areas such as Punjab where oxidizing soil conditions exist and where U-containing phosphate fertilizers are commonly, if not excessively, used.
Materials and Methods
Participants
The participants in the present study were 50 healthy individuals and 49 cancer patients all living in the Malwa region of Punjab, India. Most of them live in the Bathinda District. Ethical approval for this research has been received. The mean age of the total 99 individuals was 41 years. For the 50 individuals of the healthy control group, the mean age was 25.1 years of age, for the 49 patients of the cancer group it was 56.5 years. The age difference is due to the fact that the healthy person selected was chosen from the same household as the selected cancer patient. For most of the cancer patients, the only healthy family members living in the same household were of a younger age. The reason behind this selection is to identify if living conditions influence the toxic metal burden of the healthy vs the cancer group.
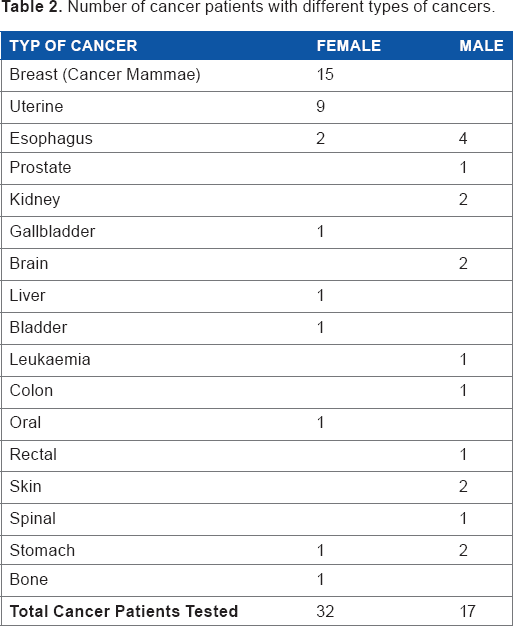
The control group consisted of 27 males and 23 females. The cancer patient group consisted of 17 male and 32 female patients. The cancer group included 15 breast cancer patients (all females), nine females with cancer of the uterus, six patients with esophagus cancer (four males and two females) and patients with a variety of other cancers as shown in Table 2. For the 15 breast cancer patients, the mean age was 56.5 years; for the nine patients diagnosed with uterine cancer the mean age was 60.6 years; and the mean age for the six patients with cancer of the esophagus was 57.5 years. All participants gave their written, informed consent to be part of this study.
Number of cancer patients with different types of cancers.
Analysis of hair samples
Hair samples were collected by surveyors (PhD candidates) of Punjab Technical University during the period of December 2012 to January 2013. The surveyors took detailed patient information about health issues and lifestyle. Surveyors were instructed to reject chemically treated hair.
The mean hair sample weight for the control group was 103.7 mg. For the patient group, the mean hair sample weight was 101.6 mg. The hair samples were shipped to the laboratory Micro Trace Minerals/Laboratory Friedle in Germany, where the analysis was performed.
Before testing, samples were repeatedly washed in the laboratory with a metal-free detergent, rinsed three times with ultrapure water and dried in a special drying oven before weighing. For sample digestion, certified ultrapure nitric acid was used. Digestion took place in a closed-vessel microwave digestion system. Ultrapure water was used for final sample dilution and the elemental analysis was performed via inductively coupled plasma mass spectrometry (ICP-MS) utilizing collision/reaction cell methods coupled with ion-molecule chemistry, a reliable new method for interference reduction.
In a total of 99 hair samples, we analyzed the elements aluminium (Al), arsenic (As), barium (Ba), cadmium (Cd), manganese (Mn), lead (Pb), strontium (Sr) and uranium (U). For Sr, the naturally occurring isotope 88 Sr was measured. It is non-radioactive and considered non-toxic. For U, the isotope 238U was measured, the most common uranium isotope found in nature.
Reference range values used for comparison with the hair sample data from Punjab
Certified hair standards and in-house standards were used as part of the laboratory quality control and for the validation of results. Reference range values had been developed from apparently healthy people. For the most common metals in blood and urine, governmental agencies provide guidelines. For hair, no guidelines are given at this time and institutions have to set up their own reference ranges based on the particular populations they serve. Reference intervals serve as the basis of laboratory testing and aid the physician in differentiating between the healthy and diseased patient. Standard methods for determining the reference interval are to define and obtain a healthy population of at least 120 individuals and use nonparametric estimates of the 95% reference interval. 27 The hair reference ranges used in the present study were developed in 1984 by Micro Trace Minerals (MTM), Germany and Trace Minerals International (TMI), USA and are based on a healthy, mostly Western population (Europe and US) of about 500 people. In 2006, a statistical re-evaluation was made based on 674 people. The last re-evaluation (N = 525) and adjustment was made in 2010. The reference ranges used here are largely in agreement with those used by other US and German laboratories.
Statistical Analysis
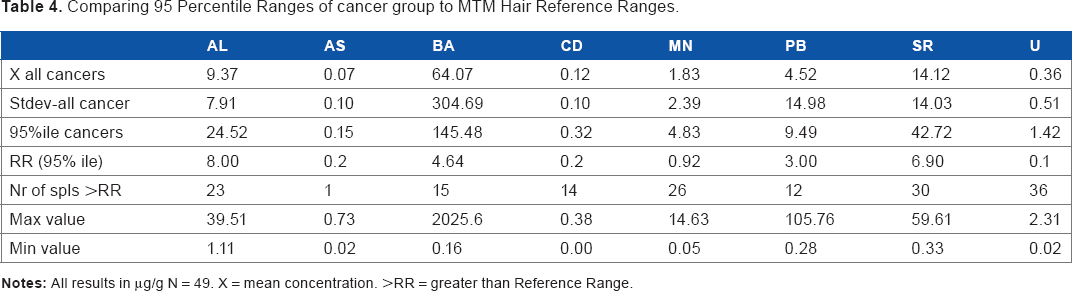
The mean concentrations of Al, As, Ba, Cd, Mn, Pb, Sr and U were calculated for the control group and the total patient group. The values of the total patient group were compared with the individual cancer groups (breast cancer, uterine cancer and esophagus cancer), and mean values were compared with the standard 95 percentile reference ranges of hair as developed by MTM/TMI (Table 3). In addition, standard deviations and the 95 percentiles were calculated and compared to existing hair reference ranges (Table 4). Minimum and maximum levels were also calculated (Table 5).
Comparing the mean metal concentration of the healthy group vs. the cancer groups.

Comparing 95 Percentile Ranges of cancer group to MTM Hair Reference Ranges.
Maximum value concentration (in μg/g).

Results
High concentrations of several elements were found in the hair samples from people living in Punjab. Compared to standard reference ranges, the mean concentration of Al, Ba, Mn, Sr and U exceeded reference ranges in both the healthy as well as the cancer group, which can be seen in Table 3 and in Figure 1 and Figure 2. For U, the highest mean concentration was found in the breast cancer group, exceeding the reference sixfold. All other groups showed mean concentration higher than the reference range. For Ba, the breast cancer group showed the highest mean concentration of all groups. For Pb, the reference range of 3.00 μg/g was only exceeded by the total cancer group and the breast cancer group, with the breast cancer group showing the highest mean of 11.42 μgPb/g (Table 3). The healthy group showed a mean concentration of 2.15 μgPb/g, while the total cancer group showed a mean concentration of 4.52 μgPb/g.
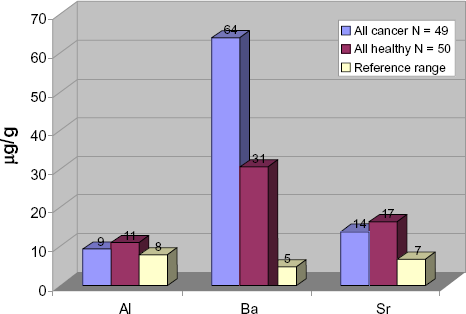
Comparing mean values of aluminium, barium and strontium in hair of healthy control Punjabis vs. Punjabi cancer patients.
The mean concentration of As and Cd did not exceed the reference ranges in any of the test groups, but the mean Cd concentration was highest in breast cancer patients. (Table 3).
While Table 3 indicates that the breast cancer group has an overall higher metal burden than the control group and the other cancer categories tested, Table 5 lists the maximum metal concentrations as seen in the control and the cancer group. The healthy groups showed the highest concentrations detected for Al, As, Cd and Sr, while the highest levels of Ba, Mn, Pb and U were found in the breast cancer group. The authors believe that this is an indication that the health of this control group is endangered; careful monitoring and routine health checks are recommended.
For Ba, we noticed extreme deviations from the reference range values (Table 4). In the total patient group (N = 49), four hair samples showed extreme Ba concentrations >100 μg/g. For the control group, the mean Ba concentration of 30.64 μg/g significantly exceeded the reference range of 4.64 μg/g. For the total cancer group, the mean concentration was even higher (213.36 μg/g). The highest maximum value of 2026 μg/g was detected in the hair of a breast cancer patient.
The mean Mn concentrations for both the control and the patient group are above the reference range (Fig. 3), but there is no considerable difference in Mn exposure between these groups. However, the breast cancer group showed the highest mean of 2.91 μgMn/g compared with a mean of 1.73 μg/g for the healthy group and a reference range of 0.92 μgMn/g.

Mean concentration of uranium (U-238) in hair of Punjabi cancer patients vs. the healthy control group.
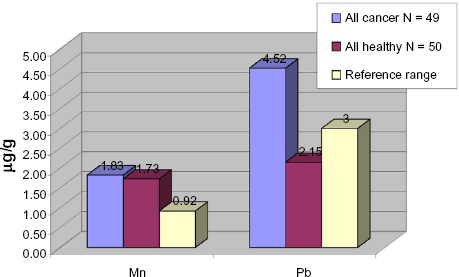
Mean concentration of manganese and lead in hair of healthy vs. cancer group.
The mean level of Pb in hair was markedly higher in the cancer group than in the control group. A mean of 4.52 μg/g was calculated for the patient group, which significantly exceeds the reference range of <3 μg/g. The control group showed a mean of 2.15 μg/g, which is within the expected reference range. The breast cancer patients showed the highest mean concentration, 11.42 μg/g.
From Table 3 it can be seen that all groups, including the healthy control group, showed mean uranium values above the reference range. Figure 2 shows that the mean concentration of 238U in the control group exceeds the reference range value of 0.1 μg/g nearly threefold (0.29 μg U/g), while the total cancer group exceeded the reference value nearly four-fold. The breast cancer group showed a mean concentration over six times higher than the reference range. This is indicative of a chronic U exposure.
Discussion
Hair as an indicator of metal accumulation
Metal intake can be via air, water and food.28,29 Hair tissue reflects long-term, chronic exposure, 30 and its validity has been confirmed. Geographical variations of hair trace element concentrations, on the whole, depend both on nutritional factors and geochemical conditions. 31 People living in geological areas where potentially toxic metals are found in excess are more prone to chronic metal overexposure. Studies by Ionescu et al demonstrate that malignant breast tissue has higher metal concentrations than healthy breast tissue. 32 While hair tissue results cannot be directly correlated with breast tissue, both are body tissue, providing an indication of metal accumulation.
Metals and cancer
Arsenic and Cadmium
Chiou et al observed a dose-response relationship between long term arsenic exposure from drinking artesian well water and the incidence of lung cancer, bladder cancer, and cancers of all sites combined after adjustment for age, sex, and cigarette smoking through Cox's proportional hazards regression analysis. 33 Chen and Ahsan demonstrated that excess arsenic in drinking water at least doubled the lifetime mortality risk from liver, bladder, and lung cancers in Bangladesh. 34 Smith et al also assessed evidence indicating that arsenic can cause liver, lung, kidney, and bladder cancer and stated that the population cancer risks due to arsenic in US water supplies may be comparable to those from environmental tobacco smoke and radon in homes. 35
The carcinogenic potential of cadmium might be affected by several factors such as smoking and hormones. 36 Åkesson's results support the hypothesis that cadmium may exert estrogenic effects and thereby increase the risk of hormone-related cancers. 37 Garcia-Morales et al suggest that the effects of cadmium are mediated by the estrogen receptor independent of estradiol. In addition to its effect on gene expression, cadmium induced the growth of MCF-7 cells 5.6–fold. 38
We could not detect an arsenic or cadmium burden in either group. The healthy group showed a mean arsenic concentration in hair similar to that of the total cancer group.

Similarly, the mean hair cadmium concentrations were below the reference range, and the healthy group showed almost the same mean value as the total cancer group.

While both of these elements are classified as human carcinogens, we do not consider the data obtained from our test groups as significant.
Aluminium
Darbre states that:
clinical studies showing a disproportionately high incidence of breast cancer in the upper outer quadrant of the breast together with reports of genomic instability in outer quadrants of the breast provide supporting evidence for a role for locally applied cosmetic chemicals in the development of breast cancer. Aluminium is known to have a genotoxic profile, capable of causing both DNA alterations and epigenetic effects, and this would be consistent with a potential role in breast cancer if such effects occurred in breast cells. Oestrogen is a well established influence in breast cancer and its action, dependent on intracellular receptors which function as ligand-activated zinc finger transcription factors, suggests one possible point of interference from aluminium. Results reported here demonstrate that aluminium in the form of aluminium chloride or aluminium chlorhydrate can interfere with the function of oestrogen receptors of MCF7 human breast cancer cells both in terms of ligand binding and in terms of oestrogen-regulated reporter gene expression. This adds aluminium to the increasing list of metals capable of interfering with oestrogen action and termed metalloestrogens. 39
Mannello et al found that breast cancer patients had significantly higher levels of aluminium in their nipple aspirate fluids when compared to a control group of healthy women without breast cancer, 40 reinforcing earlier similar findings. 41
We could not establish a clear link between aluminium and cancer. When we compared the average values of the healthy and cancer groups, the P-value between groups was 0.36.

However, the mean aluminium concentration for the control and cancer group exceeded the reference range for hair and the highest mean concentration was seen in the breast cancer group (Table 3). This shows that a low level exposure exists in the healthy and the cancer groups.
Water maybe the source of Al for the people tested. Studies of Punjab water indicated that most of the investigated samples showed Al concentrations above WHO standards of 0.05 mg/L. 42
Soil contains Al, as does food. The daily dietary intake of the Mumbai adult population is 6.4 mg Al/day, 43 which is equal to the approximate intake of 6 mg/day of an Italian population studied. 44 In North America, the median intake for adults is about 24 mg daily, but adults consuming mostly unprocessed food have a daily Al intake of <5 mg/day. Under normal physiological conditions, the usual daily dietary intake of Al (5–10 mg) is almost completely eliminated via renal excretion. 45 Al is also eliminated through feces.
While several nutritional factors influence the Al uptake, including a healthy nutritional status, the current scientific concern is not the amount of aluminium in food or water, but the availability of the aluminium and the sensitivity of some population groups to aluminium. 46
Barium
The EPA has determined that barium is not likely to be carcinogenic to humans following ingestion and that there is insufficient information to determine whether it is carcinogenic to humans following exposure. 47 However, Barium chromate(VI) is recognized as genotoxic, cytotoxic and carcinogenic. 48
High mean concentrations of barium were found in the healthy control group, and the total cancer group, but the highest mean was found in the breast cancer group (Table 3). Extreme hair values were also noted in these groups. When we compared the mean barium concentrations of the healthy and total cancer groups, the average concentration of barium in the cancer group was more than double that of the healthy group. However, in both groups extreme values were noted for which we found no explanation. In the cancer group, one patient's hair sample showed a level below detection and was omitted from the statistics, hence the total count is listed as 48 instead of 49.

Barium-containing hair dyes are used in India. On questioning, all subjectsconfirmed that no chemical hair treatment was used. Barium sulphate is permitted by the Cosmetic Directive of the European Union and may be used as a colorant in cosmetics and personal care products. 49 None of the people in any of the group claimed to use such products.
Barium belongs, like Ca and Sr, to the alkaline earth metals. It is similar to these elements in its general chemical properties with two important differences: the low solubility of Ba sulphate and the large ionic radius, which is similar to that of potassium (K+). Unlike Pb, Ba does not form heavily soluble sulphides and is not redox-active; it is only found in oxidation number +2 in living organisms and soil.
Barium is used in many industrial applications, 50 and has many uses. Barium chromate, for instance, is used as a pigment and in the removal of impurities and residual moisture from organic dry-cleaning solvents or from petroleum fuels. Barium sulphate is used in fertilizers, and other barium compounds are in pesticides. Barium manufacturers are located in the Punjab region. Barium waste may be released into the air, soil and water during industrial operations. Ba is released into the air during manufacturing operations. 47 Exposure to Ba can occur in the workplace or from drinking contaminated water, and EPA and WHO recommend Ba levels <2 μg/L. The major sources of barium in drinking water are discharge of drilling wastes, discharge from metal refineries, and erosion of natural deposits. Drinking Ba-contaminated water for relatively short periods of time can cause gastrointestinal disturbances and muscle weakness. If consumed for longer periods, kidney damage may result. 50
We could not locate data regarding barium testing in Malwa Region water, which suggests that Ba is not yet a concern to the Punjabi government. Barium is chemically very similar to radium (Ra), which belongs to the same group in the Periodic System. In an environment with much soluble U in the soil, it must be expected that Ra from radioactive decay of U will be taken up by plant roots at substantial levels. If the bioavailability of Ba for uptake in the plant roots is high, the same must also be expected for Ra in the soil. 51
Manganese
The nutritional function of this essential element is well understood.52,53 In excess, Mn is neurotoxic and affects reproduction.52–54 In mammalian cells, Mn causes DNA damage and chromosome aberration mastopathic lesions of the breast. 55 Information on manganese and cancer is scant, but Floriańczyk et al found that metallothionein and manganese concentration in breast cancer was higher in comparison with mastopathic lesions of the breast. Neoplastic cells contained more metallothioneins (by 330%) and more manganese (by 25%). 56
Our data points towards a relation. High mean concentrations were found in all cancer groups, as can be seen in Table 3, with the highest mean found in the breast cancer group (2.91 μgMn/g). The healthy control group, which are people living in the same household as the cancer patients, showed a mean concentration of Mn (1.73 μg/g) compared to the mean concentration of 1.83 μg/g for the total cancer group (Table 3). For all groups, the mean concentration was above the 95 percentile reference range of 0.92 mcg/g manganese in hair, indicating that manganese exposure affects all.

Surprisingly little information is available on the concentration of manganese in Punjab groundwater. However, information on soil indicates that the amount of manganese found in soils varies greatly, and may range from 20 to 3,000 ppm (mg/kg). Manganese in locally grown foods comes almost entirely from natural sources in the soil. Manganese has an estimated average crustal abundance of 950 ppm, 57 unless it has been added as a micronutrient to fertilizers because of a problem of Mn deficiency in the plants. Common fertilizer sources of manganese are manganese sulfate and MnEDTA. 58
Divalent Mn, which is much more soluble, is taken up by plant roots with similar kinetics as for Ca++ and Mg++ ions. 59 This may explain why tea leaves contain high levels of Mn. Manganese availability increases as soil pH decreases,60,61 and Mn toxicity to plants may occur in acid soils below pH 5.5. Soils in the Malwa region are generally medium black, and have a pH in the alkaline range of 7.5 to 8.0 (Wallis, 1997). 62 In areas where Mn deficiency in soil exists as a result of high pH, the deficiency is often (and wrongly) corrected by adding Mn to fertilizers rather than attempting to acidify the soil. Just recently, the Soil Science Department of the Punjab Agricultural University (PAU) cautioned farmers about the apparent manganese deficiency in soil and advised the spraying of 0.5% manganese sulphate solution (one kg Manganese sulphate in 200 L of water per acre.
Manganese-dependent superoxide dismutase (Mn-SOD) is very differently expressed in different types of tumor cells. In some tumor cell populations, this enzyme is strongly underexpressed, compared to normal cells or to less aggressive tumor cells from the same organ,63,64 while in other tumor cell populations, it is markedly overexpressed. 65 While increased Mn-SOD expression suppresses the malignant phenotype of human breast cancer cells and suggests that the Mn-SOD gene is a tumor suppressor gene in human breast cancer, 66 a study suggests that high Mn levels of groundwater appear to be positively associated with total cancer, colon cancer and lung cancer death rates. 67
There is a strong antagonistic interaction between non-heme Fe and Mn at the level of intestinal absorption. 53 Iron (Fe) deficiency will lead to an increase in intestinal absorption of Mn, which is likely to be important for much of the population in India, since Fe deficiency is a very common problem there.68–72 There is much data demonstrating redox regulation of various intracellular signal systems containing cellular growth and apoptosis. Moderately enhanced oxidative stress can be beneficial for tumor cells, because it simultaneously causes more stimulation of cell growth and inhibits apoptosis. 73 Too much oxidative or nitrative stress is, however, lethal for tumor and normal cells.
A possible explanation for Mn-SOD overexpression in tumor cells could be that the cells concerned have expressed high levels of NO synthase-2. Superoxide anion radical reacts very fast with NO and makes peroxynitrite, which is toxic. It might thus be useful for a cell with high NO production to keep the concentration of superoxide anion radical very low in order to prevent formation of peroxynitrite at lethal levels. For cancer patients with overexpressing Mn-SOD, it is reasonable to expect that a high intake of Mn is harmful.
Lead
This element is considered mutagenic,74,75 for a number of different reasons:
It functions as a redox catalyst of the reaction between DNA and H2O2. 76
It functions as a redox-active metal catalyst of lipid peroxidation and enhances the production of lipid peroxidation.
It inhibits enzymes needed for normal scavenging of H2O2, peroxynitrite and organic hydroperoxides (all mutagenic).
Steenland et al suggest that, according to animal studies, lead is not genotoxic in vitro. 77 However, it is said to promote the mutagenicity of other mutagens, perhaps through inhibition of DNA repair. Because high lead levels have been found in the breast cancer group, a relationship between exposure and disease seems probable.
Our data shows that the highest mean concentration of 11.42 μgPb/g for the breast cancer group exceeded the reference range by nearly four times, whereas the healthy group showed a mean below the reference range of 3 μg/g (Table 3). When we compared the healthy with the total cancer group, the mean concentration for the total cancer group was more than double. The total cancer group also showed some extreme values. Overall, lead exposure does not seem a problem for the healthy group. Our data shows it is a main problem for the breast cancer group. Interestingly, patients with esophagus or uterine cancer showed lower mean hair lead levels than the healthy group.

Pollution is the most common cause of a high Pb burden. 78 The most plausible main source of Pb in Punjab is polluted rainwater, which might come from local and long-distance sources. Lead is commonly found near industrial sites and power plants. Lead arsenate pesticides can be another source for Pb in the soil, at least in other parts of the world.79,80
Lead in drinking water should be less than 0.005 mg Pb/L. Water used for irrigation may, however, be a potential source of Pb to the soil. A study of water and crops showed that the concentration of Pb, Cd and other metals in sewage-contaminated water was up to 210 times higher than levels in shallow hand pump water. 81 An animal study demonstrated the problem. Shed skins from cobras and wall lizards collected from heavily polluted urban areas contained significantly higher Pb levels than those of the same animals from less polluted rural areas of Punjab. 82
Because clean water is not easily accessible to all Punjabis, access to uncontaminated water is a pressing issue of great public health importance.
Long term exposure to low lead levels is common in countries where leaded gasoline is used, as is the case in Punjab. Exposure to Pb produces various deleterious effects on the hematopoietic, renal, reproductive, and central nervous system.83,84
Lead exposure at higher levels can induce oxidative stress,85–88 and inhibit the Ca++-ATPase and Na+/K+-ATPase,89,90 while low concentrations of Pb++ have been reported to have an opposite, stimulatory effect on the Ca++-ATPase. 91 Both forms of inhibition will tend to elevate cytosolic Ca++ concentrations and the total Ca concentration of the cell, while Ca++ accumulation in the sarcoplasmic reticulum may be inhibited because of impaired transport from cytosol into the sarcoplasmic reticulum. This may be important for all intracellular processes that are regulated by the dissolved Ca++ concentration in the cytosol or other intracellular compartments. Inorganic Pb is probably carcinogenic to humans. 92
There are two different chemical reasons for the toxic effects of Pb. One is the binding of Pb++ to thiol and selenol groups, causing inhibition of various enzymes. Lead, like uranium, can bind to the DNA molecule and catalyze the reaction between DNA and H2O2.76,93
Strontium
Lifetime cancer mortality risk coefficients have been calculated for nearly all radionuclides, including strontium-90. Most of the risk is associated with the high-energy beta particle emitted by yttrium-90. While the risk coefficient for ingestion is lower than for inhalation, ingestion is generally the most common means of entry into the body. Similar to other radionuclides, the risk coefficient for tap water is about 80% of that for dietary ingestion.
We tested 68 Sr, and in our study, the mean strontium level exceeded the Sr reference range for hair (6.9 mcg/g) in all groups (Table 3). When we compared the healthy group with the total cancer group, we found a higher average concentration in the healthy group. We also found the highest maximum level of 105 μg/g in the healthy group (Table 5). This seems to indicate long term exposure for all groups.

68 Sr is the most prevalent form, comprising about 83% of natural strontium. The other three stable isotopes and their relative abundance are strontium-84 (0.6%), strontium-86 (9.9%), and strontium-87 (7.0%). We could not locate data regarding the carcinogenicity of 68 S.
Stable Sr is ubiquitous in rocks, soil, dust, and coal, 94 and high Sr levels are found naturally in Punjabi water and soil. Governmental tests showed maximum concentrations of up to 1650 μg Sr/L (=1.650 mg Sr/L) in hand pump water of Malwa region households (Devinder Mehta, private communication). 95
Strontium is very similar to Ca in its geochemical behavior, 94 but the Sr/Ca ratio varies for different minerals and rock types, and different types of water. The Sr/Ca ratio in human tissue reflects that of the diet, and depends on the ratio of Ca from plant foods and animal foods. The geochemical situation influences the uptake and a balanced intake of Ca and Sr supports bone density. However, in the presence of a Ca deficiency, excess Sr may negatively affect bone metabolism. 96 Low Ca absorption increases Sr absorption into bone. 97
The minimal risk level (MRL) for human oral intake as set by the US Center for Disease Control is 2 mg Sr/kg/day, an appreciable amount. An MRL is an estimate of the daily human exposure to a hazardous substance that is likely to be without appreciable risk of adverse non-cancer health effects over a specified duration of exposure. EPA recommends that drinking water levels of stable Sr should not be more than 4 mg/L. 98 It is unlikely that Sr at the concentration range found in this material can have any important biological effect in cancer cells.
Uranium
After a study detected high U levels in hair and urine in a group of disabled Punjabi children, uranium became a widely discussed issue in Punjab, 99 but the source of the apparent exposure has yet to be determined.
The reference range for uranium in hair been set for 0.1 μg/g and was exceeded by the mean concentration as calculated for all groups. The healthy group exceeded the level nearly threefold, while the breast cancer group was more than six times higher. This indicates chronic exposure as a problem in all the patients we tested and supports earlier studies as outlined above that consider uranium an area of concern for the people living in the Malwa region.

Uranium radiotoxicity or chemical toxicity can be caused by breathing air containing U-rich dust particles, especially in uranium mines and coal mines, or by eating substances containing U or drinking U-containing water. Contaminated water from various areas in the Malwa region has been a source of controversy for years and since 2009, various Indian governmental agencies, including the Bhaba Atomic Research Center have researched the U concentration in water from the Malwa region. A study of water samples showed U concentrations ranging from 5.41 to 43.39 μg/l, 100 which must be considered very high. High U concentration has also been found in soil samples from the Malwa region around Faridkot. 101
Uranium is found in higher oxidation numbers in the soil and inside living organisms than Fe, since quadrivalent U is stable under anoxic conditions. Under more oxidizing conditions, even more oxidized U species can be formed, with the hexavalent being most abundant in oxidized soils and other oxidized geological environments, including seawater. 102
Salinity may increase the release of natural U. The increased input of U into the environment via phosphorus (P) mineral fertilizers might have led to a continuous accumulation of U in soils, if U had been present in poorly soluble form because of reduction by soil organic matter. With strongly oxidizing conditions, however, rainfall during the summer monsoon season promotes effective leaching of U from the topsoil. When agrochemical processes are responsible for mobilizing U that is present in soil, U contamination of ground water can be expected.
Phosphate fertilizers are a source of U, 103 and Punjab has the highest use of phosphate fertilizers in India. Use of phosphate fertilizer, a potential source of metals such as Cd and U has steadily increased since 20 08, 104 as can be seen from Table 1. The intake of U from food raises the question of U solubility in the soil. Uranium in the form of UO2 may be bound to phosphate under oxidizing conditions. It is therefore possible, when U-containing phosphate fertilizer is added, that inhibition of plant root uptake by phosphate might be more important than the enhancement of U concentration in the soil.
Excessive uranium intake and exposure affects human health. However, ingested insoluble compounds are poorly absorbed from the gastrointestinal tract and are only retained in the body for a short time, thus they generally are of low toxicity. The main chemical effect associated with exposure to high doses of U and its compounds is renal toxicity. 105
Once in the bloodstream, the U compounds are filtered by the kidneys, where they can cause damage to the kidney cells. Both functional and histological damage to the proximal tubulus has been demonstrated, but little is known about the effects of long-term environmental uranium exposure in humans. Only two small studies with 50–100 subjects have been published on the kidney toxicity of natural uranium from drinking water. They have shown an association of uranium exposure with increased urinary glucose, alkaline phosphatase, and β-2-microglobulin excretion, as well as increased urinary albumin levels. 106 None of the participants in the present study had listed kidney dysfunction as a health problem.
Uranium mining and lung cancer have been linked.107–112 Samet et al demonstrated this association among predominantly non-smoking Navajo men in a population study between 1969 and 1982, but the high rate of breast cancer among Navajo women living on reservations where mining is common has not been officially linked to U. 113 However, Al-Dujaily et al concluded that exposure to depleted U plays a role in the pathogenesis of breast cancer in Iraqi females. 114 Aside from the long-term consequence of war, the U intake from food and/or drinking water is unusually high in human populations living in areas where oxidizing soil conditions exist. This is the case in the territories inhabited by Navajo Indians in the United States, and in Iraq and Punjab.
Because uranium is an endocrine-disrupting chemical, populations exposed to environmental uranium should be followed for increased risk of fertility problems and reproductive cancers. 115
Conclusion
Water, soil, and phosphate fertilizers all seem to play a potential role in causing an increased metal burden in Punjabi people living in the Malwa region. The high concentration of several potentially toxic elements as found in the hair reflects a high toxic metal burden. Toxic metals such as Pb or U lead to oxidative stress and DNA damage, 116 thus long-term exposure to multiple toxic elements is a possible cause of the Malwa region's rising cancer rate.
While all the elements tested are potentially toxic, the present study supports previous research stating that the metals Mn, Pb and U are factors in the development of breast cancer among Punjabi women. Since the metals Al, Mn, Pb and U have carcinogenic or mutagenic potential, the combined effects of these toxins enhances carcinogenicity and mutagenicity.
The main concern of this study is the relatively high burden of multiple metals in all test groups, indicating long term exposure of the healthy as well as the cancer patients. Compared to the reference ranges of a so-called healthy Western population, our study groups all show multiple metal exposures in hair tissue. Since all test persons live under the same conditions, the results are not surprising. They are, however, a disturbing fact, because excess long term exposure to multiple toxins cannot and does not support health. Adverse health effects of exposure have been reported for many environmental toxins, 117 but only recently has attention been given to the long term effect of low level exposure. Epidemiologic evidence on the relation between exposure to metals and cancer is receiving increasing concern, 118 and the authors suggest the hypothesis that the healthy group may be affected in years to come, just like family members in the cancer group. Considering the age discrepancy between the healthy group (mean age 25.1) and the cancer group with a mean age of 56.4, the absence of cancer in the healthy group may be a factor of youth and time. It nevertheless cries for preventive measures and immediate action.
The metal burden of multiple toxic metals as found in all groups can be reduced with precautionary measures, including a change in agricultural approaches and detoxification treatments for those already burdened. A reduction in total metal burden, however achieved, can only improve health.
To further substantiate our analytical findings, a follow-up research project, including diagnosis and treatment of immediate and long term exposure, is recommended. This could include blood and urine testing. Where multiple metal burdens have already been detected through hair analysis, urine provocation tests and detoxification treatments are recommended. Furthermore, cancer patients may have compromised detoxification systems, making them more vulnerable to environmental exposures, thus phenotyping seems essential. The GSTM1 (Glutathione S-Transferase M1) enzyme is needed for the natural detoxification process and a GSTM1 deletion can be found in about 50% of today's population. 119 In patients with emphysema and lung cancer, a 65% increase in the frequency of deletion was found. 120 It may be safe to assume that other cancer patients are similarly affected and that the Punjabi's population is no exception.
The authors realize that in this study the population of breast cancer patients and other cancers is relatively small to claim clinical relevance of a high metal burden and its association with breast cancer. However, this study received no funding and was in fact a labor of love in support of people living in the cancer belt of the Malwa region.
To benefit the people of Punjab and elsewhere, we would hope that governmental agencies, medical institutions and other groups fund a larger study that will either confirm or disprove the association as suggested by our data.
Author Contributions
Conceived and designed the experiments: EBB, CP, YB. Analyzed the data: AF, EBB, HB. Wrote the first draft of the manuscript: EBB. Contributed to the writing of the manuscript: EBB, YB. Agree with manuscript results and conclusions: EBB, YB, AF, HB, CP, AK. Jointly developed the structure and arguments for the paper: EBB, YB, HB, CP. Made critical revisions and approved final version: EBB, HB, YB, CP. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
