Abstract
Calcified coronary lesions are challenging to deal with, as they require optimal lesion preparation. Direct stenting in this scenario is associated with risk of stent-underexpansion, which is related to in-stent restenosis, target lesion revascularization and stent-thrombosis. We report on the interventional management of an underexpanded bare-metal stent not amenable to high-pressure balloon dilation and cutting-balloon. By using rotablation we could abrade the underexpanded stent struts and the calcification with subsequent implantation of a drug-eluting stent. Follow-up of 6 months revealed good results without evidence of significant restenosis. Our clinical experience and case reports in the literature suggest that this strategy might be an option for underexpanded stents not amenable to conventional techniques.
Introduction
Direct coronary stent implantation is an elegant technique for coronary artery revascularization. 1 However, calcified coronary lesions, often seen in older patients suffering from diabetes mellitus, renal failure and hypertension, are challenging to deal with, as they require optimal lesion preparation prior stenting for avoiding stent underexpansion which is related to in-stent restenosis, target lesion revascularization and subsequent stent thrombosis. 2 Several strategies and technologies have been developed to address the problem of heavily calcified coronary lesions. These include simple dilatation using standard non-compliant balloon, cutting balloon and plaque modification using rotational atherectomy. We report on the management of an underexpanded bare-metal stent in a patient with heavily calcified lesion not amenable to high-pressure balloon-dilatation. Written consent was ontained from the patient.
Case
A 72-year old man suffering from progressive angina over the past 8 weeks presented to our chest pain unit. He had previously documented insulin-dependent diabetes, alimentary obesity, hyperlipidemia and arterial hypertension. An ambulatory performed myocardial perfusion scintigraphy revealed a reduced tracer-uptake in the apex, left posterior and antero-lateral wall during physical examination (100 watt-cycling). Coronary angiography, which was performed via right radial access using a 5F sheath, revealed a 50% stenosis of the left anterior descending artery (LAD) and ramus posterolateralis sinistra (RPLS) while the right coronary artery (RCA) had a critical 90% stenosis (Fig. 1A). Due to the patients' symptoms and the angiographic findings we decided to perform a percutaneous coronary intervention (PCI). The patient received 600 mg clopidogrel, 500 mg aspirin and 5000U heparin followed by primary PCI and direct stenting of a bare-metal stent (BMS) (Coroflex blue 3.5 mm/8 mm, B. Braun, Melsungen, Germany) with 18 atm for 30 sec (Fig. 1B). Post-PCI angiography revealed a 75% stenosis in the mid-portion of the stent (Fig. 1C). A subsequent dilatation with a semi-compliant balloon (Pantera 3,5/10 mm with [Biotronik, Berlin, Germany] 18 atm over 30 sec), a non-compliant balloon (Quantum 3,5/8 mm [Boston Scientific, Natick, USA] with 20 atm over 30 sec) and a cutting-balloon (3,0/10 mm [Boston Scientific, Natick, USA] with 18 atm over 30 sec) could not expand the stent further; pointing out the heavily calcified nature of this lesion. Due to the fact that an underexpanded stent is a predictor for worse clinical outcome we decided on rotablation. We puncture the right brachial artery and introduced an 8F sheath. Additionally we introduced a 5F sheath in right femoral vein and inserted a transient pacemaker lead. After passing across the stenosis with the 0.009 rotawire we ablade the heavily calcified stenosis as well as the stent struts (stentablation) (Fig. 2A). All ablations were performed with a 1.75 mm burr with at least 150,000 rpm and ablation times <30 sec without a decrease in rotational speed of >5,000 rpm. The procedure was free of complications and we continued with dilatation with a non-compliant balloon (Quantum 3,5/8 mm with 20 atm over 30 sec) and a cutting-balloon (3,0/10 mm with 16 atm over 30 sec). With complete expansion of the balloons the procedure was continued with implantation of a drug-eluting stent (Taxus Liberté 4.0/12 mm [Boston Scientific, Natick, USA] with 16 atm over 30 sec) (rotastenting) (Fig. 2B). Finally, there was TIMI 3 without evidence of dissection or residual stenosis (Fig. 2C). Following uneventful hospital stay without evidence of myocardial necrosis the patient was discharged after 3 days on 100 mg aspirin, 75 mg clopidogrel, 5 mg bisoprolol, 5 mg of ramipril and 40 mg simvastatin with a recommendation for dual antiplatelet therapy of 1 year without any change in his extra-cardiovascular medications. A routine coronary angiography performed 6 months after index-PCI revealed a good result with a mild (25%) restenosis (Fig. 2D).

Coronary angiography reveals a 90% stenosis of the right coronary artery (RCA) (
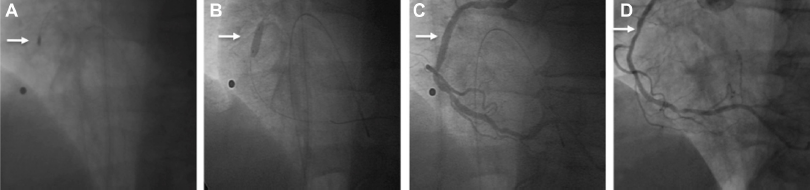
Rotational atherectomy of the remaining calcified stenosis within the stent and the underexpanded stent struts (stentablation) (
Discussion
Direct stenting is the implanation of stents in coronary lesions without predilatation. 1 From animal restenosis models, direct stenting without the need for predilatation appears to reduce vessel trauma, in particular as a result of less endothelial denudation, resulting in less neointimal hyperplasia subsequently. 1 PCI of calcified and complex lesions has been associated with lower success rates, an increased frequency of acute complications, and higher restenosis rates than PCI of simple lesions. 2 As seen in our case, delivering the stent may be difficult and stent expansion may be inadequate in heavily calcified lesions, resulting in smaller acute gain compared to non-calcified lesions. 2 It is widely accepted that achieving postprocedural residual stenosis is a major determinant of restenosis during follow-up and optimal stent expansion is a crucial factor in minimizing the risk of stent thrombosis pointed out by the fact that only 22% of patients that experienced subacute stent thrombosis have an acceptable PCI result as assessed by IVUS.2,3
A variety of strategies and technologies have been developed to address the problem of an underexpanded stent. The POSTIT trial revealed that in case of using only the stent delivery balloon over 70% of patients did not achieve optimal stent deployment. 4 Use of non-compliant balloon to achieve full distension in resitant lesions is a reasonable first-step. However, focal points of resistance within a lesion result in non-uniform balloon expansion and characteristic “dog-boning” with overexpansion in the more compliant segments. In this non-uniform expansion may cause vessel dissection and rupture acutely as well as restenosis due to deep-wall injury in the follow-up. Cutting-balloon, designed to score the vessel longitudinally rather than causing uncontrolled plaque disruption, have been used successfully in the treatment of undilatable lesions. 5 In our case, none of these techniques were successful in reducing the underexpansion, demonstrating the nature of the heavily calcification, which was not assumed on initial fluoroscopy. Thus, despite the existence of limited data, 6 we decided to rotablade the remaining calcification and the underexpanded stent struts to avoid aforementioned complications. Highspeed rotational atherectomy preferentially cuts hard plaque, increases plaque compliance and thereby renders the lesion more amenable to balloon dilatation. 7 The rotablator is able to ablate inelastic tissue selectively while maintaining the integrity of elastic tissue due to the principle of differential cutting. The abraded plaque is pulverized into microparticles, which are 5-10 μm in diameter. These particles are small enough to pass through the coronary microcirculation and ultimately undergo phagocytosis in the liver, spleen, and lung. 7
The procedure performed in our case was uneventful with no dissection, slow-flow, heamodynamic compromise or myocardial necrosis. We had applied a transient pacemaker via the right femoral vein to overcome possible conduction disturbances when handling in the right coronary artery.
Several observational studies have confirmed that rotational atherectomy prior to stent deployment in severely calcified lesions does facilitate stent delivery and expansion, but incidence of restenosis remains unsatisfactory (23%) when BMS are used. 8 There is limited information about rotational atherectomy followed by DES implantation, but initial results seem promising. 9 A comparison of BMS (n = 84) and DES (n = 213) after rotablation with cardiac death and recurrent myocardial infarction being defined as primary endpoint and binary restenosis as secondary endpoint revealed lower rates for primary endpoint in DES group (2.3% versus 7.1%; P = 0.04) during a follow-up of 1300 days. 10
Despite our procedural success and good midterm result, there are no data on long-term follow-up after stentablation and rotastening. Thus, it should be emphasized that a better lesion preparation is needed to avoid stent underexpansion in undilatable lesions.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
