Abstract
We have performed a proteomic analysis of peripheral blood monocytes from ACS patients in comparison with healthy subjects and stable coronary patients in order to search novel biomarkers of ACS in circulating monocytes. Monocytes were isolated from blood of patients with non-ST elevation ACS (n = 27) at day 0, 2 and 6 months, and from patients with stable coronary disease (n = 10) and matched healthy controls (n = 11). The proteomic analysis of monocytes from ACS patients at day 0 showed that cathepsin D is differentially expressed compared to healthy subjects and stable coronary patients. Western blot analysis indicated that the mature form of cathepsin D at day 0 was overexpressed in monocytes of ACS patients in relation to healthy subjects. In contrast, the precursor of this enzyme, absent at day 0 in ACS patients, was highly expressed in monocytes of healthy subjects. Furthermore, the upregulation of the mature form of cathepsin D diminished along the time, while the expression of the precursor increased. ACS patients also showed significantly increased plasma cathepsin D levels on admission compared to healthy subjects and stable patients. Cathepsin D plasma levels diminished at 2 and 6 months to control values. Finally, cathepsin D levels were independent of the existence of coronary risk factors and CRP levels, correlating only with CD40L. Since this protease participates in the genesis and rupture of atherosclerotic plaques, it could represent a potential marker of ACS.
Introduction
Over the last decades, the risk of patients which had suffered an acute coronary syndrome (ACS) has been reliable assessed by evaluating the release of troponins (1,2) which are surrogate markers of myocardial cell necrosis, but are not directly involved in the pathophysiology of ACS. The most extensively potential biomarker studied to date has been C-reactive protein (CRP). Although its use could be advised in some risk situation, it seems evident that it has only a moderate predictive value (3). Some other proteins are being studied in the last years as potential candidates for risk prediction (4). Among these are CD40L(5), MCP-1 (monocyte chemoattractant protein-1) (6), adhesion molecules (7), myeloperoxidase (8) and several interleukins (9), which in the majority of the cases were identified in blood. Nevertheless, none of them has consistently demonstrated to add predictive value to the clinical variables used in the clinical practice and, in most cases, there are not commercially available standardised assays (10). As an alternative, we have searched for novel sources of markers (11) and thus, we have examined whether circulating monocytes from ACS patients express and/or secrete in plasma, specific proteins that could serve as individual markers or define a characteristic profile (12). Among the proteins specifically expressed by monocytes from ACS patients we identified Cathepsin D. Since this protease could play an important role in the pathophysiology of ACS (13), we have measured plasma levels of Cathepsin D from patients with ACS compared with stable coronary disease and/or healthy subjects. Moreover, we have compared the concentrations of Cathepsin D with other known biomarkers (CD40L and CRP) of cardiovascular disease.
Methods
Patients
Thirty-four patients 40 to 80 years old admitted into the Fundació Hospital de Alcorcó with a non-ST elevation acute coronary syndrome (ACS) entered the study. ACS was defined as GIII-IV angina (CCS) with transitory ST depression (> 0.05 mV) and/or new T wave inversion (>0.02 mV) on the ECG and/or positive troponin I. The exclusion criteria were: inflammatory or neoplasic disease, coagulation disorders, other significant heart disease excepting left ventricular hypertrophy secondary to hypertension, chronic treatment except for preexisting clinical atherosclerosis or its risk factors, normal coronary angiograms, ejection fraction less than 0.45, and having suffered surgical procedures, major traumatisms, thromboembolic events or revascularization procedures in the previous six months.
Age and sex distribution of ACS patients (60 [56 to 67] years, 78% male) were not significantly different from those of stable patients and healthy volunteers (58.6 [53.7 to 62.6] years, 90% male and 61.7 [55.0 to 70.0] years, 64% male respectively). Table 1 shows the main clinical characteristics of ACS patients. Troponin I was elevated in 89% of cases (levels above 0.2 μl were considered abnormal). The ejection fraction was 0.45 in one case and normal in all other patients. All patients were treated with enoxaparin and aspirin and/or clopidogrel, and 7% received also a IIb/IIIa inhibitor. The final treatment was percutaneous revascularization in 52%, coronary artery by-pass graft in 18% and medical therapy in 30%. On discharge, 96% received aspirin and/or clopidogrel and 4% anticoagulants, 92% statins, 44% renin-angiotensin system inhibitors, 80% beta-blockers, 36% nitroglycerin patches, 20% calcium channel blockers, 4% insulin and 16% oral anti-diabetic drugs.
Clinical data of patients with ACS.

At the moment the diagnosis was made, the patients were asked to participate in the study. In case of acceptance, they signed the informed consent and 30 cc of blood were withdrawn for analysis and introduced in EDTA-treated collection tubes (Venoject, Termo Europe). Management included coronary angiography in all cases. Two and six months later, blood was extracted again for analysis. Seven patients did not complete the protocol: 4 were excluded because of normal coronary angiogram, 1 died during the admission event and 2 patients were withdrawn from the study before the two-months visit. Finally, 27 patients were studied.
Ten stable patients with coronary artery disease (CAD) from Hospital de Móstoles, with the same exclusion criteria than for the ACS patients, and 11 healthy volunteers were also studied. Both groups were not significantly different from ACS patients in age and sex distribution. Stable CAD patients must have been diagnosed by the existence of a previous myocardial infarction or by coronary angiography and could not have a history of acute cardiovascular event in the previous year to the inclusion in the study. All patients signed the informed consent and the study was approved by the local Ethical Committees in accordance with institutional guidelines.
Isolation of peripheral blood mononuclear cells
Thirty ml of blood were obtained and centrifuged at 750 g for ten minutes at room temperature to obtain the plasma, and then kept under −80 °C. Then, PBMC were obtained, lysed and kept under −80 °C until protein analysis, as described previously (14).
Proteomic Analysis
The procedure for the 2-DE has been described previously (14, 15). Cells (107) were solubilized in 200 l of lysis buffer containing 7.0 M urea, 2 M thiourea, 4% CHAPS, 2% carrier ampholytes and 1% DTT and frozen at −80 °C until use. Proteins were precipitated with five volumes of cold acetone for 15 min and centrifuged at 1.500 g for 5 min. Protein content was quantified by Bradford method. Protein samples (200 to 250 μl) were diluted with rehydration solution (350 μl final volume) containing 8M urea, 0.5% CHAPS, 10 mM DTT and 0.2% pharmalyte pH 3-10, 4-7 or 6-9, prior to rehydration in a reswelling tray (Pharmacia Biotec). The IEF were performed in an IPGphor (Pharmacia Biotec) unit at 10 °C following the protocol previously described (14). After IEF the strips were equilibrated in 1.5 M Tris, pH 8.8 buffer containing 6 M urea, 30% glycerol, 2% SDS and bromophenol blue, with the addition of DTT 1% for 20 min, followed by the same buffer with the addition of 4.8% iodoacetamide for 20 min. SDS-PAGE was performed according to Laemmli (16), as previously described (17), using a Protean II system (Bio-Rad), at 25 mA/gel at 4 °C. Gels (17 × 20 cm) were fixed overnight (5% acetic acid, 30% ethanol) and stained with a silver staining kit (Pharmacia). Evaluation and processing of the 2-D gels were performed by PDQuest (BioRad) gel analysis software version 6.2. Protein patterns in the gels were recorded as digitalized images using a desktop scanner (DuoScan HiD, Agfa-Gevaert) and imported to PDQuest (15, 18). Comparison between patient samples and controls was performed using the replicate group option and the statistic part of the software package of PDQuest. To compensate for any variation in protein loading and development of silver stain, spots quantity was normalized using the normalization option total density in gel image. Spots of interest were in-gel digested with trypsin as previously described (15, 18), for subsequent mass spectrometry analysis.
Western Blot Analysis
Equal amounts of proteins were loaded into 12% acrylamide gels and electrophoresed. The resolved proteins were transferred onto nitrocelulose membranes (Biorad). The non-specific sites of the membranes were blocked by incubation 1 h at room temperature in 7.5% nonfat dry milk powder in PBS containing 0.1% Tween 20 (PBST). The membranes were incubated overnight at 4 °C with goat anticathepsin D polyclonal antibody (ab-6313, abcam) in PBST containing 5% nonfat dry milk. It is important to note that this antibody specifically recognizes cathepsin D isoforms of 34 kDa (mature) and 52 kDa (precursor). The membranes were washed with PBST, and incubated 1 h at room temperature with horseradish peroxidase-conjugated secondary antibody (Amersham) in PBST containing 5% non-fat dry milk. After washed with PBST, detection was performed by enhanced chemiluminescence (ECL kit, Amersham). The membranes were stained with Ponceau S to confirm equal loading and transfer of samples.
Biochemical Analysis
Determination of lipid levels (total cholesterol, low-density lipoproteins, triglycerides and high-density lipoproteins) were measured by enzymatic assays (Sigma Diagnostics, Madrid, Spain). Tro-ponin was determined by standard techniques (Dadhe-Behring).
Cathepsin D plasma concentrations were determined in duplicate with commercially available enzyme-linked immunosorbent assay kits (Calbio-chem). Each assay was calibrated using a cathepsin D standard curve following manufacturer's instruction. It is important to note that the monoclonal antibody used for cathepsin D detection in this kit was raised against the mature form of the protein. The method used for the high sensitivity CRP measurement was the particle-enhanced immunonephelometric assay on a BN II analyzer (Dade Behring) (19). CD40L plasma levels were assessed by ELISA (R&D Systems) (5).
Statistical Analysis
Statistical analysis was performed with SPSS 8.0 and Instat (Graphpad sowtware). Cathepsin D, CD40L and CRP data are expressed as median (interquartile range). The comparisons among every parameter in the different populations studied were performed by a Kruskal-Wallis test followed by a Dunn's test. Finally, the correlations between the values of the different parameters were carried out by the Pearson's “r”. Differences were considered significant at p < 0.05 (two-tailed).
Results
Cathepsin D expression in PBMC of patients with ACS
The 2-DE analysis of the proteomes of PBMC of patients with ACS versus healthy controls led us to the identification, by mass spectrometry analysis, of cathepsin D which was detected in Two spots (Fig. 1): the one corresponding to the precursor of cathepsin D was present in the monocytes of healthy subjects, but not in ACS patients the day of admission; in contrast, the spot corresponding to the mature form of this enzyme was upregulated in monocytes from patients on the day of admission as compared to healthy subjects.

In addition, when monocytes from patients with ACS where compared to those obtained from patients with stable coronary disease, a similar behaviour to that observed in healthy subjects was found (not shown). Finally, when the spots corresponding to cathepsin D were studied along the time, the intensity of the spot corresponding to the mature form of cathepsin D diminished at 2 and 6 months, as compared to day 0; in contrast, the spot corresponding to the precursor of cathepsin D began to appear as observed in the monocytes of healthy subjects (not shown).
Cathepsin D isoforms are differentially expressed by PBMC of patients with ACS
To further confirm the proteomic results, cathepsin D expression was analysed by Western blot, showing two different bands of 52 kDa (precursor) and 32 kDa (mature). The day of admission, PBMC from ACS patients expressed the mature form of the enzyme in higher amount than healthy controls and stable patients. When studied along the time, the expression of the mature form of cathepsin D diminished at 2 and 6 months. On the other hand, the expression of the precursor of cathepsin D was enhanced in the monocytes of healthy subjects but was absent in ACS patients the day of admission, appearing again after 2 and 6 months (Fig. 2).

Cathepsin D levels in plasma of patients with ACS
Since cathepsin D was differentially expressed by PBMC the day of admission and PBMC in culture secrete lysosomal enzymes (20, 21), we analysed plasma levels of soluble cathepsin D in patients with ACS by ELISA and western blot.
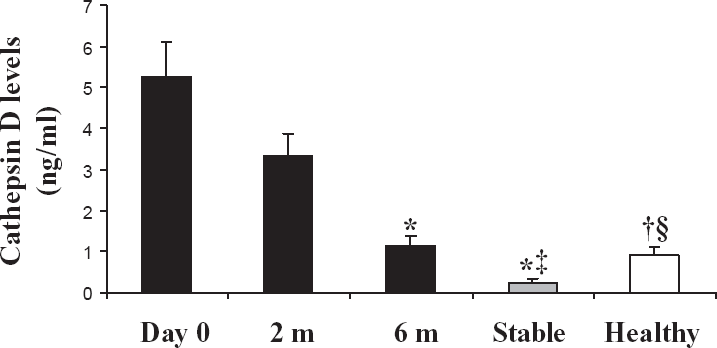
Cathepsin levels on admission (3.73 [2.25 to 6.83] ng/ml) were significantly elevated as compared to healthy subjects (0.73 [0.53 to 1.41] ng/ml, p < 0.01) and stable patients with CAD (0.00 [0.00 to 0.70] ng/ml, p < 0.001) (Fig. 3). At two months they showed a non-significant reduction, and were still higher (2.31 [1.73 to 4.46] ng/ml) than those of healthy and stable coronary patients (p < 0.05 and p < 0.001, respectively). At six months, they displayed a significant reduction as compared to admission and two months values (0.80 [0.31 to 1.58] ng/ml, p < 0.001 and p < 0.05 respectively) and were similar to those of healthy subjects and stable patients (p = NS in both cases). These data were confirmed by western blot (Fig. 2).
Soluble CD40L levels were also higher in ACS patients at day 0 (2.53 [1.63 to 4.23] ng/ml) than in stable patients with CAD (0.10 [0.02 to 0.34], p < 0.001) and healthy volunteers (0.37 [0.25 to 1.19], p < 0.01) (Fig. 4A). At two months they displayed a light non-significant trend towards reduction (2.73 [1.84 to 4.44], p = NS), and kept significantly higher than the values in stable cases and healthy subjects (p < 0.001 and p < 0.01 respectively). At six months, they were significantly lower than at day 0 and at 2 months (1.40 [0.65 to 2.14], p < 0.05 for both comparisons), and values were not different from healthy subjects and stable patients.

CRP levels on admission were elevated as compared to healthy subjects (2.34 [1.98 to 5.34] vs. 0.69 [0.41 to 1.78] mg/L, p < 0.01) and stable patients, although in this case they did not reach statistical significance (1.58 [0.85 to 4.02], p = NS). At 2 months there was a trend towards a decrease (2.72 [1.26 to 5.54]), the values being still significantly higher than in healthy subjects (p < 0.05). At 6 months the decrease continued (2.08 [0.88 to 3.79]) and levels were not significantly different from stable and healthy subjects (Fig. 4B).
Cathepsin D and CD40L levels were not affected by age, sex, the pre-existence of coronary artery disease, previous treatments, the existence of diabetes, hypertension, smoking status or overweight, chest pain duration, ECG changes and time from the beginning of symptoms until the first blood extraction. In addition, they did not correlate with CRP, troponin I and lipid levels. Moreover, cathepsin D levels on admission were not significantly different in patients with CRP levels above 3 mg/L as compared with those below this value (4.15 [2.04 to 8.37] vs 3.31 [2.30 to 6.71] mg/L, p = 0.49). The only significant relationship displayed by Cathepsin D and CD40L was between themselves at day 0 (r = 0.71; p < 0.001), and remained significant at two (r = 0.70, p < 0.001) and six months (r = 0.63, p < 0.001) (Fig. 5).

Discussion
Despite the numerous advances in the managing of patients with ACS, there is still a significant need for the identification and development of new biomarkers that could be used for early detection and risk, previous to and independent of clinical manifestations (11, 22, 23). Searching for such biomarkers directly in plasma could be a complicated task because the plasma proteome is the most complex in the human body (24). Looking for biomarkers in tissues or circulating blood cells, by using proteomic strategies, can be a fruitful approach, provided that they shed or secrete the protein into the blood, where it can be easily detected (11, 22, 23).
Understanding the role of monocytes in atherotrombosis would be greatly improved with the identification of changes in their protein expression during this process (12). For that purpose, we have compared the pattern of protein expression of monocytes obtained from patients who have suffered an acute coronary event with that of healthy subjects. Among the identified proteins, we have found that cathepsin D, is differentially expressed by monocytes from ACS patients the day of admission as compared to healthy subjects and stable coronary patients. Interestingly, Western blot analysis indicated that the mature form of cathepsin D was overexpressed the day of admission in relation to healthy subjects. In contrast, the precursor of this enzyme was absent in the monocytes of patients the day of admission, but was highly expressed in the monocytes of healthy subjects. Furthermore, the upregulation of the mature form of cathepsin D diminished along the time while the expression of the precursor increased, showing that there is a specific activation of this protein during ACS.
The family of lysosomal enzymes cathepsins, as well as MMPs, have been implicated in atherosclerosis (25, 26). Cathepsin D is expressed by macrophages in the intimae of human carotid atherosclerotic plaques, whereas cells in the intimae of a normal coronary artery do not display cathepsin D immunostaining (13). Interestingly, cholesterol oxidation products induce the release of cathepsin D (27). After being released by human macrophages, cathepsin D is able to modify LDL particles, favoring the formation of foam cells (13) and has been suggested to participate in the apop-tosis of macrophages/foam cells, a determinant of plaque instability (28). Although the family of cathepsins, as well as MMPs, has been shown to be secreted by cultured monocyte-macrophages, only the cathepsins were detected in the extracellular milieu as fully processed active enzymes (29). In the case of Cathepsin D, it was previously known that this protease is secreted by macrophages in vitro and it is detected in the active form in atherome plaques (25). Thus, the search for soluble Cathepsin D in plasma was clearly justified. Therefore, using an ELISA which recognizes specifically the mature form of cathepsin D, we have shown increased plasma levels of cathepsin D in ACS patients the day of admission compared to healthy subjects or stable coronary patients, consistent with the results obtained in monocytes. In addition, cathepsin D plasma levels begin to decrease at 2 months, returning to control values at 6 months, coinciding with a diminution in the expression of mature cathepsin D in monocytes. The same results were observed by Immunoblotting of ACS plasmas.
Several blood biomarkers have been involved in the pathogenesis of ACS (30), and they have also been explored as potential risk predictors. The most extensively studied has been CRP, which has been shown to participate in different biologic phenomena that contribute to the atherosclerotic process (31). However, although elevated levels of CRP seem to predict future vascular risk in a variety of clinical settings (32-34), it has recently been shown to have a limited prognostic power (3). In the present study, both cathepsin D and CRP levels were significantly higher in ACS patients on admission than in healthy subjects. However, CRP values of ACS patients did not reach a significant difference as compared to patients with stable coronary disease. Moreover, the pattern displayed by cathepsin D showed a stronger statistical significance than that of CRP. In addition, cathepsin D levels were not influenced by the pre-existence of CAD, the presence of traditional coronary risk factors or the previous use of antiatherothrombotic treatments. Furthermore, cathepsin D was not related to troponin I and CRP levels. Troponin I is used as a prognostic marker in ACS at present (35, 36), but it reflects myocardial necrosis secondary to acute coronary thrombosis and does not reflect the evolution of the atherothrombotic process. On the other hand, CRP seems to participate in the genesis of atherothrombosis and could bring important information about its evolution. However, the lack of correlation with cathepsin D levels suggests that this molecule is yielding different information.
Cathepsin D showed a significant correlation with soluble CD40L concentrations that was present from day 0 to 6-month follow-up. Increased levels of soluble CD40L have been previously reported to be present in ACS, where they may have prognostic value (5, 37). CD40-CD40L interaction contributes to the development and thrombotic complications of the atherosclerotic lesions (38). In the present study, CD40L levels were increased in ACS patients the day of admission and diminished along the time, following a similar pattern to cathepsin D and showing also independence with respect to the same clinical and biochemical variables. Nevertheless, cathepsin D seems to be involved in different mechanisms of the atherothrombotic disorder from that of the CD40-CD40L system.
In conclusion, proteomic strategies enable the identification of novel biomarkers in ACS. Using this approach we have shown that mature cathepsin D is increased both in plasma and mono-cytes of patients with ACS and thus, it could play a pathogenic role in the triggering of acute coronary events. Moreover, given its independence of clinical and biochemical variables, it should be tested whether cathepsin D plasma levels alone or in combination with different biomarkers could improve their prognostic power in subjects with an ACS.
Disclosure
The authors declare that no conflicts of interest exist.
Financial Support
Pfizer-Spain, Cardiovascular Network (03/01), Fundacion Española del Corazó, SAF, FIS (PI 02/1047), CAM 08.4/0021.1-2003, European Network (QLG1-CT-2003-01215).
Footnotes
Acknowledgements
Fondo de Investigaciones Sanitarias (FIS: PI 02/1047), CAM 08.4/0021.1-2003, European network (QLG1-CT-2003-01215), INDAS. BMC 200-02596. BFU 2005-08838. CAM (Proteomarkers) 2007.
