Abstract
Left ventricular noncompaction has been classified as a primary cardiomyopathy with a genetic origin. This condition is morphologically characterized by a thickened, two-layered myocardium with numerous prominent trabeculations and deep, intertrabecular recesses. Recently, it has become clear that these pathological characteristics extend across a continuum with left ventricular hypertrabeculation at one end of the spectrum.
The histological findings include areas of interstitial fibrosis.
We present a case of left ventricular hypertrabeculation which presented as sudden infant death syndrome. Histologically areas of subendocardial fibrosis was prominent and we propose that this entity may be a hidden cause of arrhythmic death in some infants presenting as sudden infant death syndrome., with areas of subendocardial fibrosis as possible arrhythmogenic foci.
Introduction
Left ventricular noncompaction has been classified as a primary cardiomyopathy with a genetic origin. 1 Morphologically, this condition is characterized by a thickened, two-layered myocardium with numerous and prominent trabeculations and deep, intertra-becular recesses.1–3 Left ventricular noncompaction may be an isolated finding or it may be associated with a variety of other congenital heart defects. 1 Left ventricular noncompaction is associated with numerous sarcomere mutations and this fact created the view that there is a spectrum spanning hypertrophic cardiomyopathy (especially apical HCM), dilated cardiomyopathy and left ventricular noncompaction. 4
Current diagnostic criteria for left ventricular non-compaction require the visualization of two distinct myocardial layers. 5 The work of Stollberger departs from this stipulation and he coined the term “left ventricular hypertrabeculation”. 5 This entity, which can be viewed as a less severe form of noncompaction, is characterized by the presence of more than three trabeculations located apically to the papillary muscles. 5
Case Report and Discussion
We present a case report of a three month old male infant who presented with sudden infant death syndrome. The macroscopical characteristics of this case was reported before. 6 This three month old non-caucasian died suddenly and unexpectedly at his day care centre. He did not have any known medical problems and no surgical procedures were ever performed. No known allergies were present and no known family history of sudden, unexpected death were present. Unfortunately the family members were lost to follow up, before electrocardiographic and echocardiographic screening could be performed to assess their risk for sudden unexpected death.
Postmortem examination of the heart revealed numerous apical trabeculations of the left ventricle (see Fig. 1). The right ventricle appeared perfectly normal. The left ventricular wall thickness measured 1 cm. No abnormalities were detected in any other organs during the postmortem examination and no thrombi were detected in the arterial system or the left ventricle.
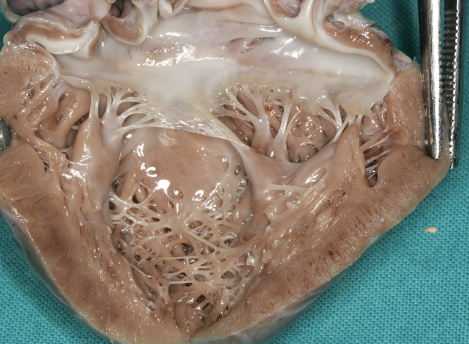
Note the numerous trabeculations in the apex of the left ventricle. More than three trabeculations distal to the level of the papillary muscles is present, thus fulfilling the criterion for the diagnosis of left ventricular hypertrabeculation.
Figure 1 clearly demonstrates more than three trabeculations in the left ventricle in a location apical to the papillary muscles. According to Stollberger7,8 this case which presented as a sudden infant death syndrome thus fulfills the criterion for left ventricular hypertrabeculation. Histological assessment of sections underlying the left ventricular trabeculae revealed prominent areas of subendocardial fibrosis (see (Fig. 2, (Fig. 3 and (Fig. 4)
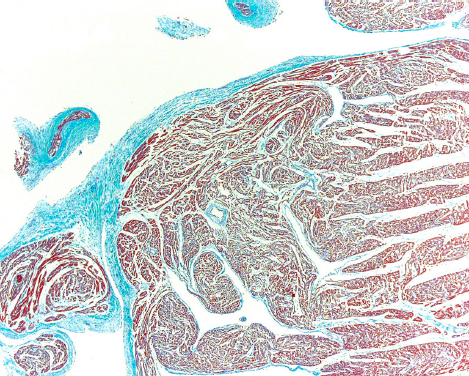
Histological sections with a Masson stain of the apex of the left ventricle. Note the areas of subendocardial and interstitial fibrosis.

Histological sections with a Masson stain of the apex of the left ventricle. Note the areas of subendocardial and interstitial fibrosis.

Histological sections with a Masson stain of the apex of the left ventricle. Note the areas of subendocardial and interstitial fibrosis.
Figure 5 is a histological section of the apex of the left ventricle of a three month old male infant who also presented as a sudden infant death syndrome, but without left ventricular hypertrabeculation to serve as a control.

Histological section with a Masson stain of the apex of the left ventricle of the control case of sudden infant death syndrome, but without left ventricular hypertrabeculation.
Endocardial fibrosis with prominent elastin deposition have been described in cases of left ventricular noncompaction.5,9,10 Currently, it is thought that ischaemia may play a major role in the pathogenesis of left ventricular noncompaction: MRI and thallium-201 scintigraphy has shown subendocardial and transmural perfusion defects which corresponds to areas of noncompacted myocardium.5,11–14 Reduced coronary flow reserve, indicating microvascular dysfunction has also been shown to be present in isolated ventricular noncompaction by PET. 12 Currently, a “chicken and egg” dilemma exists regarding the pathogenesis of isolated ventricular noncompaction: either an impairment in the development of the myocardial microcirculation impairs the normal compaction process of the myocardium or vice versa.
In this case report we describe subendocardial fibrosis in a three month old infant with left ventricular hypertrabeculation. Current literature notes the presence of fibrosis in cases of ventricular noncompaction, this is the first case describing fibrosis in ventricular hypertrabeculation—the less severe form of noncompaction. Secondly, the presence of such striking subendocardial fibrosis implies that the involved areas in the left ventricle must experience ischaemia already in utero. Lastly, we propose that these fibrotic areas may act as the foci of ventricular arrhythmia as the underlying cause of death.
Contribution from Different Authors
J Ker did the literature review and writing. L Du-Toit-Prinsloo, WFP Van Heerden and G Saayman did the post-mortem analysis and histological assessment.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material. Written consent was obtained from the patient or relative for publication of this study.
