Abstract
The first case of noncompaction was described in 1932 after an autopsy performed on a newborn infant with aortic atresia/coronary–ventricular fistula. Isolated noncompaction cardiomyopathy was first described in 1984. A review on selected/relevant medical literature was conducted using Pubmed from 1984 to 2013 and the pathogenesis, clinical features, and management are discussed. Left ventricular noncompaction (LVNC) is a relatively rare congenital condition that results from arrest of the normal compaction process of the myocardium during fetal development. LVNC shows variability in its genetic pattern, pathophysiologic findings, and clinical presentations. The genetic heterogeneity, phenotypical overlap, and variety in clinical presentation raised the suspicion that LVNC might just be a morphological variant of other cardiomyopathies, but the American Heart Association classifies LVNC as a primary genetic cardiomyopathy. The familiar type is common and follows a X-linked, autosomal-dominant, or mitochondrial-inheritance pattern (in children). LVNC can occur in isolation or coexist with other cardiac and/or systemic anomalies. The clinical presentations are variable ranging from asymptomatic patients to patients who develop ventricular arrhythmias, thromboembolism, heart failure, and sudden cardiac death. Increased awareness over the last 25 years and improvements in technology have increased the identification of this illness and improved the clinical outcome and prognosis. LVNC is commonly diagnosed by echocardiography. Other useful diagnostic techniques for LVNC include cardiac magnetic resonance imaging, computerized tomography, and left ventriculography. Management is symptom based and patients with symptoms have a poorer prognosis. LVNC is a genetically heterogeneous disorder which can be associated with other anomalies. Making the correct diagnosis is important because of the possible associations and the need for long-term management and screening of living relatives.
Introduction
Left ventricular noncompaction (LVNC) also known as ‘spongy myocardium’ is a rare abnormality of the left ventricular (LV) wall that results from intrauterine developmental arrest of the normal compaction process of the myocardium during the first trimester leading to the formation of two layers of the myocardium: the compacted and the noncompacted layer [Benjamin et al. 2012]. The first case of noncompaction was described in by Bellet and Gouley (1932) after an autopsy performed on a newborn infant who also had aortic atresia and coronary–ventricular fistula [Engberding and Bender, 1984]. LVNC without other cardiac abnormalities (isolated noncompaction cardiomyopathy) was first described by an echocardiogram performed by Engberding and Bender. In 1990, Chin and colleagues proposed the term ‘isolated noncompaction of left ventricular myocardium’ [Chin et al. 1990] which is LVNC without other anomalies. Noncompaction was first described in children and has recently become increasingly seen in the adult. In the subsequent sections, the pathogenesis, histology, epidemiology, clinical features, current diagnosis, recommendations, differential diagnosis, management, screening, and prognosis will be discussed.
Pathogenesis
During normal embryology, the heart consists of a spongy meshwork of muscle fibers and trabeculae that are separated by recesses that link the myocardium to the LV cavity. Blood is supplied to the myocardium through the intertrabecular spaces. Typically, during 5–8 weeks of normal human fetal development, the ventricular myocardium undergoes compaction with transformation of the intertrabecular spaces into capillaries, and the residual spaces within the trabecular meshwork then disappear [Taylor, 1992]. The process starts from the epicardium to the endocardium and from the base of the heart toward the apex. In LVNC, there is persistence of trabeculation and deep recesses communicating with the ventricular cavity due to noncompaction. The exact cause for the persistence of these deep trabeculations is unknown but it is believed that myocardial ischemia or pressure overload could be responsible for the regression of embryonic sinusoids [Rosa et al. 2011].
LVNC can be genetically sporadic or familial. The American Heart Association classifies LVNC as a primary genetic cardiomyopathy (Table 1) [Maron et al. 2006]. Gene defects identified in patients with LVNC have been limited and infrequent, suggesting genetic heterogeneity [Ichida et al. 2001]. Mutations have been found in several of these genes, including tafazzin (TAZ-G4.5), alpha-dystrobrevin (DTNA), LIM domain binding protein 3 (ZASP/LDP3), and lamin A/C (LMNA).
Clinical manifestations of patients with left ventricular noncompaction.

The first gene found to be responsible for LVNC was G4.5 which encodes for tafazzin, located on the Xq28 chromosome region, and is mostly expressed in skeletal and cardiac muscles [Bissler et al. 2002]. The action of the tafazzin protein takes place in the mitochondria [Chen et al. 2002]. The X-linked inheritance of this gene was seen in a family of 6 children in a study conducted by [Bleyl et al. 1997]. In this family, the mutation of the G4.5 gene was seen in the children. This gene is also responsible for other systemic myopathies including Barth syndrome, Emery–Dreifuss muscular dystrophy, and myotubular myopathy. The gene was initially described in patients with the Barth syndrome (a complex X-linked disorder in infant males which is characterized by dilated cardiomyopathy (DCM), skeletal myopathy, and neutropenia) [Bleyl et al. 1997].
Another gene mutation found in LVNC is a transition C to T mutation which was identified in the alpha-dystrobrevin (DTNA) gene. This gene was localized to the 18q12 chromosome region and is associated with muscular dystrophy [Metzinger et al. 1997]. This autosomal-dominant gene was identified in a Japanese family in which four generations were affected with LVNC [Ichida et al. 2001].
Mutations in another cytoskeletal protein, known as the Z-band alternatively spliced PDZ-motif protein (ZASP), Cypher, or LDB3 (a LIM domain binding protein) resulted in cardiomyopathy in mice [Vatta et al. 2003; Xing et al. 2006].
LVNC and other congenital anomalies (e.g. ventricular septal defects) are seen in mutations of the FKBP12 gene of mice [Shou et al. 1998]. This gene is responsible for release of calcium from the sarcoplasmic reticulum by the ryanodine receptor 2 (RyR2) [Shou et al. 1998]. Arrhythmogenic right ventricular cardiomyopathy (ARVC) has also been reported in people with the RyR2 gene mutation [Milting et al. 2006].
Lamin A/C (LMNA)-related sequence is another gene associated with LVNC and DCM. This gene is mapped to chromosome 1q22 [Hermida-Prieto et al. 2004]. Other genetic mutations associated with LVNC include transcription factors NKX2.5 and TBX5 [Ichida et al. 2001]. The locus for autosomal-dominant LVNC was mapped to human chromosome 11p15 [Sasse-Klaassen et al. 2004]. A distal chromosome 5q deletion causes a loss of the cardiac specific gene CSX. This deletion was described in a child with findings of LVNC [Pauli et al. 1999].
A study by Sasse-Klaassen and colleagues showed that mutations in sarcomere protein genes which were previously known to cause hypertrophic cardiomyopathy (HCM) and DCM were also associated with LVNC [Sasse-Klaassen et al. 2004]. There were nine distinct mutations found in the genes encoding alpha-cardiac actin (ACTC); cardiac troponin T (TNNT2), and B-myosin heavy chain (MTH7). The study results supported a concept that there may be a similar molecular etiology to various cardiomyopathic phenotypes.
The human cardiac sodium channel alpha-subunit gene (SCN5A) has been shown to be involved in cardiac arrhythmias, cardiomyopathies, and LVNC [Shan et al. 2008].
The genes responsible for some familial types have been described while those for the sporadic forms of LVNC are yet to be identified. In the study by Chin and colleagues, there was familial involvement in approximately 50% of patients which were mostly children [Chin et al. 1990]. Later, Oechslin and colleagues reported that 6 of 34 patients had a family recurrence of LVNC. These results, however, may be an underestimation since there was incomplete screening of asymptomatic relatives of affected patients [Oechslin et al. 2000]. In the familial form, the disease can be inherited by X-linked, autosomal-dominant patterns (in adults), or mitochondrial inheritance patterns (in children) [Sasse-Klaassen et al. 2003] with the autosomal dominant inheritance being more common than X-linked or autosomal recessive inheritance [Zaragoza et al. 2007].
Histology
LVNC has no specific findings and no difference has been seen between isolated and nonisolated forms. Interstitial fibrosis and endocardial fibroelastosis have been described on endomyocardial biopsy of this illness [Burke et al. 2005; Hamamichi et al. 2001]. At autopsy, a variety of gross patterns of noncompaction have been noted, including anastomosing broad trabeculae, coarse trabeculae (resembling multiple papillary muscles), and sponge-like interlacing smaller muscle bundles [Burke et al. 2005]. Perhaps the absence of well-formed papillary muscles is the best clue to the diagnosis [Burke et al. 2005].
Epidemiology
The prevalence of LVNC has been underestimated in the past because of the lack of knowledge about this previously uncommon disease and its similarity to other diseases of the myocardium and endocardium. Diagnosis of LVNC is gaining popularity over the last 25 years [Oechslin and Jenny, 2011] perhaps secondary to the availability and improvement of the diagnostic technique [Oechslin et al. 2000]. A study of children with primary cardiomyopathy demonstrated that 9.2% of the patients had LVNC [Andrews et al. 2008]. The prevalence of LVNC in the adult population ranges between 0.01% and 0.27% of all adults patients referred for echocardiography studies [Benjamin et al. 2012; Ozkutlu et al. 2002; Ritter et al. 1997; Stollberger and Finsterer, 2004; Weiford et al. 2004; Stollberger et al. 2008]. A Swiss registry recorded a total of 34 cases over 15 years among a patient population who underwent echocardiogram [Oechslin et al. 2000]. According to a French registry, the diagnosis of LVNC diagnosis was confirmed in 105 cases through the use of echocardiogram performed in a laboratory between 2004 and 2006 [Habib et al. 2011]. In this study, LVNC was first detected in 12 patients with rhythm disorders, 45 patients with heart failure (HF) symptoms, and 8 patients through familial screening. During the follow-up study, patients suffered several complications including HF occurring in 33 of the patients, ventricular arrhythmia in 7, embolic events in 9, and 9 of the patients received heart transplantation while death occurred in 12 of the patients [Habib et al. 2011].
LVNC is rare among White populations [Lachhab et al. 2012]. A study on HF patients of African/Afro-Caribbean athletes demonstrated a high prevalence of myocardial trabeculations (about 30%) among Blacks with almost 15% fulfilling echocardiographic criteria for LVNC [Kohli et al. 2008; Gati et al. 2013a,b]. It is not known whether the cardiac morphology observed in African/Afro-Caribbean origin is representative of LVNC or just secondary to increased cardiac preload [Gati et al. 2013a,b]. The incidence of this illness in a general population is between 0.05% and 0.25% per year [Engberding et al. 2010; Espinola-Zavaleta et al. 2006; Stöllberger et al. 2007]. The true prevalence is actually unknown [Weiford et al. 2004]. Both sexes are affected equally in the sporadic forms of LVNC [Zambrano et al. 2002].
Clinical features
The clinical findings (Table 2) are variable, ranging from patients’ with asymptomatic disease to symptomatic patients who develop congestive HF, arrhythmias, thromboembolic events, and sudden cardiac death [Duru and Candinas, 2000; Pignatelli et al. 2003]. The average time from the onset of symptoms to the correct diagnosis has been approximately 3.5 years due to the similarities with other frequently diagnosed conditions [Ritter et al. 1997; Nikolić et al. 2011].
Classification of primary cardiomyopathy by the American Heart Association: types of primary cardiomyopathy.

HCM, hypertrophy cardiomyopathy; ARVD, arrhythmogenic right ventricular cardiomyopathy; LVNC, left ventricular noncompaction; DCM, dilated cardiomyopathy; RCM, restrictive cardiomyopathy.
Congestive HF symptoms are among the most frequent symptoms reported by patients with LVNC. It is associated with LV systolic and diastolic dysfunction. The diastolic dysfunction may be due to abnormal relaxation and restrictive filling caused by the numerous prominent trabeculae [Agmon et al. 1999] while the systolic dysfunction may result from subendocardial hypoperfusion and microcirculatory dysfunction as seen in a positron emission tomography (PET) scan [Soler et al. 2002; Jenni et al. 2002]. The mechanical dys-synchrony between the noncompacted and compacted myocardium can cause global LV dysfunction. In a study, symptoms of HF such as tachypnea appeared to be the most common presenting symptom [Pignatelli et al. 2003]. In a case series of 34 patients, 27 patients complained of dyspnea, 9 patients reported chest pain, 9 patients had chronic atrial fibrillation, and 12 patients had HF functional class III/IV [Oechslin et al. 2000].
Arrhythmias have been frequently described in patients with LVNC. Electrocardiogram is usually abnormal. The frequency and type of arrhythmia may vary by age. Wolff–Parkinson–White (WPW) syndrome [Nihei et al. 2004] and ventricular tachycardia are more commonly seen in children, while atrial fibrillation and ventricular arrhythmias were reported in 25% and 47% of adults with LVNC respectively [Ritter et al. 1997]. Other rhythm abnormalities that have been reported in electrocardiogram (EKG) are paroxysmal supraventricular tachycardia, left or right bundle branch block, fascicular block, biventricular hypertrophy, T-wave inversions, AV block and complete heart block [Ichida et al. 1999; Reynen et al. 1997].
Thromboembolic events associated with LVNC can be secondary to the extensive trabeculated ventricle, atrial fibrillation, and decreased ventricular function [Agmon et al. 1999]. These events can subsequently lead to stroke, transient ischemic attack, pulmonary embolism, systemic emboli, and mesenteric infarction if not recognized. Prevention of thromboembolic complications in patients with LVNC is still a subject of controversy [Benjamin et al. 2012].
Sudden cardiac death is also one of the worst events that occurred in patients with LVNC. In a large case series, sudden cardiac death was responsible for approximately 40% of deaths with LVNC [Rigopoulos et al. 2002].
Children who are diagnosed with LVNC can have transient recovery of ventricular function and then later have deterioration in adulthood. This may explain the late presentation (‘undulating’ phenotype) found in children with LVNC [Pignatelli et al. 2003]. Symptoms found in infants and young children include cyanosis, syncope, dysmorphic features, seizures [Masihi Kocharian et al. 2010], and failure to thrive [Pignatelli et al. 2003]. There is an association between LVNC and facial dysmorphisms (prominent foreheads, low set ears, strabismus, high arching palates, and micrognathia) [Chin et al. 1990].
Noncompaction in children
The pediatric form of LVNC is usually associated with congenital abnormality but a case of newborn with isolated cardiomyopathy has been reported [Sert et al. 2012].
The prevalence of LVNC in the pediatric age group is about 0.14% [Sert et al. 2012]. Just like the adults, noncompaction affects mostly the left ventricle but the right ventricle and septum can also be affected [Sert et al. 2012]. It can manifest in different forms ranging from dilated to hypertrophic appearance [McMahon et al. 2007]. Some children can present with chest pain, heart murmur, abnormal heart sound, abnormal EKG and/or abnormal echocardiogram [Brescia et al. 2013; Pignatelli et al. 2003]. Brescia and colleagues conducted a study on 242 children (mean age of 7.2) with an echocardiogram diagnosis of LVNC, the study demonstrated the following: abnormal cardiac exams in 19%, abnormal EKG in 16%, abnormal screening echocardiogram in 14%, arrhythmia in 10%, chest pain in 9%, syncope in 5%, and heart failure occurred in 25% of the patients. The frequency of LVNC manifestations can be affected by the population under study [Brescia et al. 2013]. The incidence of embolism secondary to LVNC in pediatric patients ranges from 0% to 38% and the risk is exacerbated by severe systolic dysfunction [Chen et al. 2013]. McMahon and colleagues reported a reduction of tissue Doppler velocities in children with LVNC compared with normal controls [McMahon et al. 2007]. The diagnosis of isolated LVNC can be difficult because of the association with other congenital heart diseases [Madan et al. 2012]. Noncompaction if left undictated and/ or untreated can lead to sudden cardiac death especially in children [Madan et al. 2012].
Diagnosis
The most commonly used technique for diagnosis of LVNC is echocardiogram. Other imaging modalities used for the diagnosis of LVNC include cardiac magnetic resonance imaging (CMRI), computed tomography (CT) scan, and contrast left ventriculography (LVG) (Algorithm 1). LVNC is associated with reduced global LV systolic function, diastolic dysfunction, hypokinesis, LV thrombi, and abnormal papillary muscle structure [Burke et al. 2005].
Echocardiography
Two-dimensional echocardiography is abnormal in all patients with LVNC and can be used to establish the diagnosis and for follow up (Figures 1, 2, 3, 4, 5 and 6). There currently exist three non-standardized sets of criteria to diagnose LVNC by echocardiography [Chin et al. 1990; Stollberger et al. 2002; Jenni et al. 2001]. The characteristic echocardiographic findings of noncompaction are multiple, prominent myocardial trabeculations and deep intertrabecular recesses communicating with the LV cavity. Criteria by Jenni and colleagues also rely on color Doppler that shows blood flow through these deep recesses in continuity with the ventricular cavity [Jenni et al. 2001]. A diagnosis of LVNC is made if coexisting cardiac lesions are excluded. Chin and colleagues also described quantitative approach to diagnose non-compaction using a trabeculation peak to trough ratio, C/NC’ ≤ 0.5 (which is the ratio of the distance from the surface of the epicardium to the trough of the trabecular recess (x) to the distance from the epicardial surface of the peak of the trabecutions (y), with ratio x/y ≤ 0.5) [Chin et al. 1990].This criteria is not widely used in clinical practice [Chin et al. 1990].

Echocardiogram in parasternal short-axis view showing left ventricular noncompaction.
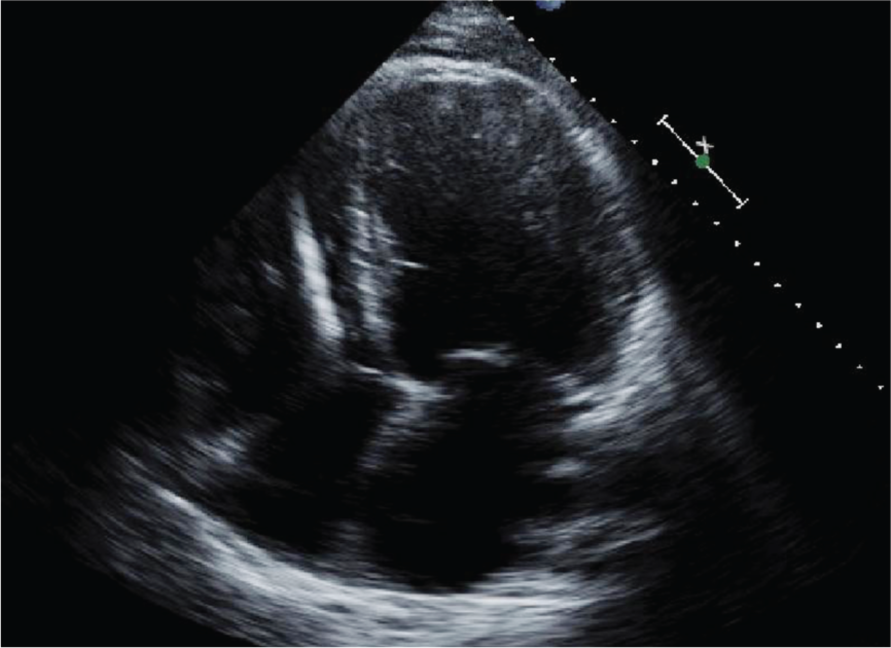
Echocardiogram in apical four-chamber view showing left ventricular noncompaction.

Two-dimensional echo apical four-chamber view and parasternal short-axis images with mild pericardial effusion. Reproduced with kind permission from BioMed Central [Espinola-Zavaleta et al. 2006].
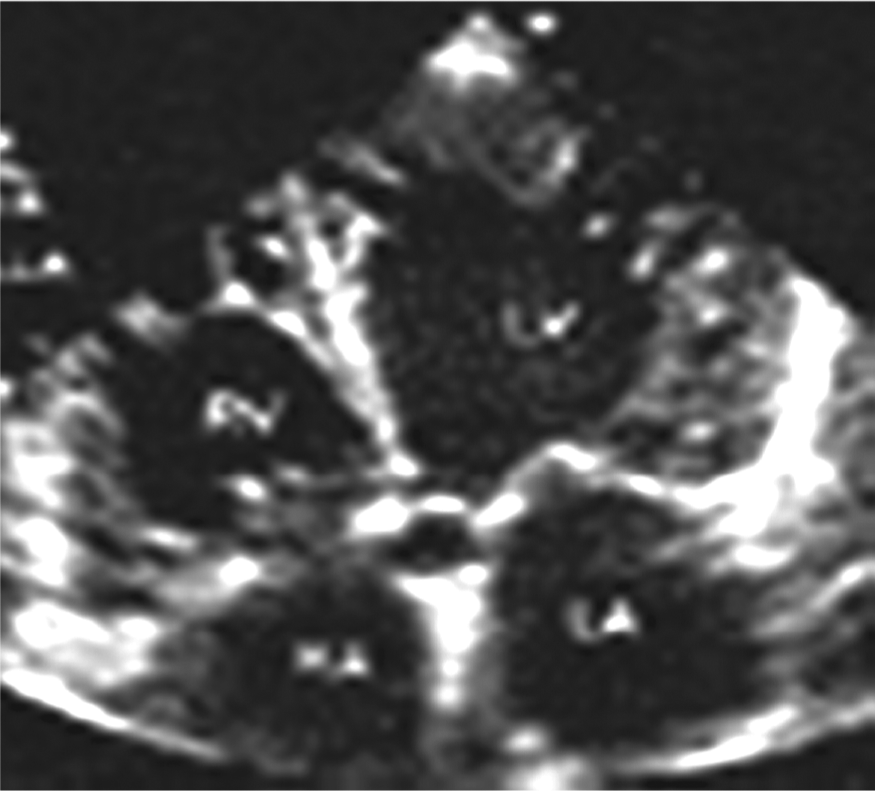
Transthoracic two-dimensional echo in apical four-chamber view. The image also illustrates evidence of noncompaction in the right ventricle. Reproduced with kind permission from BioMed Central [Espinola-Zavaleta et al. 2006].
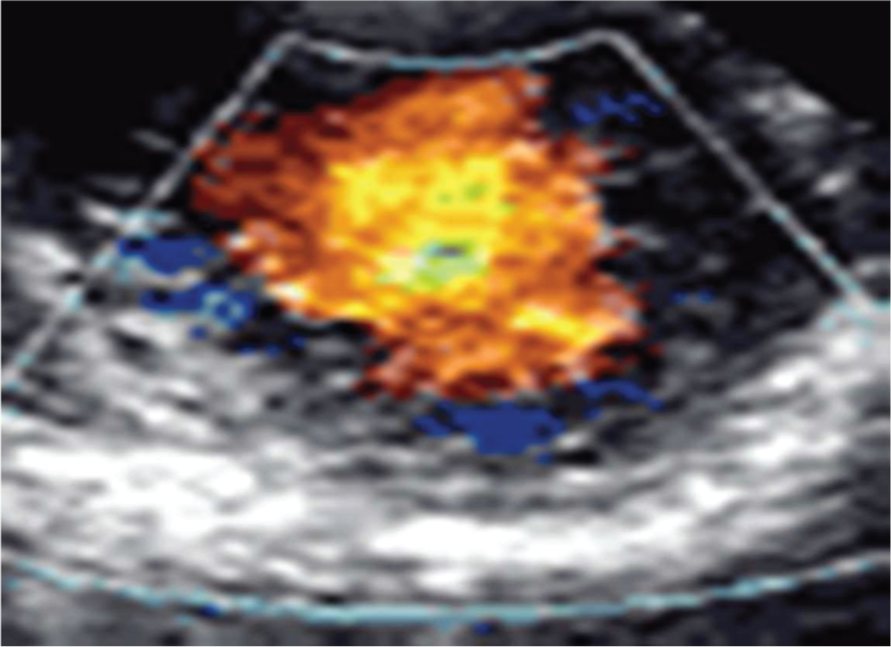
Transthoracic two-dimensional echo study with color and continuous wave Doppler shows left ventricular noncompaction associated with patent ductus arteriosus. Reproduced with kind permission from BioMed Central [Espinola-Zavaleta et al. 2006].

Two-dimensional parasternal and color Doppler images showing ‘the noncompacted:compacted wall ratio’. Reproduced with kind permission from BioMed Central [Espinola-Zavaleta et al. 2006].
Criteria by Jenni and colleagues include: a markedly thickened LV wall (which consists of two layers: a thin normally compacted epicardial layer and a markedly thickened endocardial layer with numerous prominent trabeculations and deep recesses); maximum ratio of noncompacted to compacted myocardium of > 2:1 at the end of systole; color Doppler evidence of flow within the deep intertrabecular recesses; and absence of coexisting cardiac abnormalities [Jenni et al. 2001]. These criteria were validated by blinded retrospective study of 19 patients with LVNC, 31 patients with DCM, 22 with hypertensive heart disease, and 86 with chronic severe valvular disease. Of the LVNC cases, a two-layered structure and thickened walls were present in 100% of the patients, while perfused recesses were visible in 95%, and hypokinetic segments were present in 89%. Wall thickening in LVNC was confined to the two-layered myocardial segments, whereas it was diffuse in other diseases [Frischknecht et al. 2005]. All compaction criteria were rare in diseases other than LVNC. In DCM, a perfused recesses and less often a two-layered structure were seen but without thickened walls.
Criteria by Stollberger and colleagues include: (1) more than three trabeculations protruding from the LV wall distal to the papillary muscles in the apex visible in a single image plane; (2) the perfusion of the intertrabecular spaces from the ventricular cavity as seen on color Doppler imaging; (3) the trabeculae were of the same echogenicity as the myocardium; (4) trabeculae motions synchronizing with the ventricular contraction; (5) NC/C ratio ≥ 2 in end systole; (6) focused on apical four-chamber view [Stollberger et al. 2002; Stollberger and Finsterer, 2004].
The diagnostic criteria for LVNC can be summarized as the appearance of multiple (at least four) trabeculations or prominent LV trabeculations, especially in the apex, inferior, central and lateral wall, LV free wall, deep intertrabecular recesses communicating with the ventricular cavity, and absence of other cardiac anomalies. Also, a two-layered structure with an end systolic ratio of greater than 2 in adults and greater than 1.4 in children, between the noncompacted and the compacted layer, are consistent with LVNC. Missed diagnoses may be due to suboptimal imaging of the lateral and apical myocardium as Oeschlin and colleagues demonstrated that apical and midventricular segments of both the inferior and lateral wall are affected in more than 80% of patients [Oechslin et al. 2000]. Diagnosis is difficult because of nonspecific manifestations and similarities with other defects. Diagnosis can be missed in 89% of children with LVNC [Ichida et al. 1999].
Real-time 3D echocardiogram is a newer technique which facilitates identifications of noncompacted/trabeculated myocardium [Engberding et al. 2010; Rajdev et al. 2007; Baker et al. 2006].
Tissue Doppler demonstrated the ventricular dys-synchronization and the strain rate (which is the velocity difference between two different points on the myocardium) in a case study of patient with suspected LVNC [Williams et al. 2003].
Speckle tracking is a diagnostic procedure that is used to recognize the characteristic pattern of myocardial texture [Van Dalen et al. 2008]. This diagnostic procedure shows the symmetrical movement pattern of apical and basal segment of myocardium in patients with LVNC [Van Dalen et al. 2008].
CMRI
The noncompacted myocardial layer in the left ventricle can be imaged by CMRI because it provides a detailed view of cardiac morphology [Thuny et al. 2010] in any image plane including the apical and lateral segments which are not well visualized by echocardiogram (Figure 7). CMRI identifies a higher rate of two-layered structures in segments such as the anterior, inferior, anterolateral, and inferolateral segments compared with echocardiogram [Thuny et al. 2010]. The efficacy of CMRI was evaluated in a report of seven patients with LVNC in whom the diagnosis was supported by other features; the results were compared with 170 healthy volunteers, athletes, or patients with DCM or HCM, aortic stenosis, and hypertensive heart disease. The most distinguishing feature was a ratio of noncompacted to compacted myocardium >2.3 during diastole (sensitivity 86% and specificity 99%) [Petersen et al. 2005].

Cardiac magnetic resonance imaging (CMRI) showing left ventricular noncompaction in four-chamber and short-axis views. Arrows point to noncompacted myocardium.
The measurement of amount of LV trabeculations using cardiac MRI was performed by Jacquier and colleagues [Jacquier et al. 2010]. They prospectively studied patients with various forms of cardiomyopathies: 16 patients with LVNC, 16 patients with HCM, 16 patients with DCM, and 16 control subjects between 2003 and 2008. LV ejection fraction, LV volumes, and trabeculated LV mass were measured in these four different populations of patients. The percentage of trabeculated LV mass measured to be about three times greater in the patients with LVNC (32 ± 10%), compared with those with HCM (12 ± 4%, p < 0.0001), DCM (11 ± 4%, p < 0.0001), and controls (12 ± 5%, p < 0.0001). Trabeculated LV mass of more than 20% of the global LV mass is highly sensitive (sensitivity = 93.7%) and specific (specificity =93.7%) for the diagnosis of LVNC [Jacquier et al. 2010]. CMRI can be used in correlation with echocardiogram to localize and quantify the extent of noncompaction. Subendocardial perfusion defects can also be described using CMRI [Soler et al. 2002]. Nucifora and colleagues demonstrated that late gadolinium enhancement (LGE) was observed in LVNC patients with LV ejection fraction of <50% compared with LVNC patients with LV ejection fraction >50%. The presence and extent of the fibrosis was independently associated with LV systolic function and clinical severity. It was concluded in this study that myocardial fibrosis is related to LV systolic dysfunction and clinical disease severity in patients with isolated LVNC [Nucifora et al. 2011].
CT scan
Contrast-enhanced CT has the capability of showing the abnormal architecture of the noncompacted LV wall as well as the qualitative and quantitative assessment of ventricular function. In addition CT has advantages over echocardiogram and CMRI in evaluating the coronary vessels to exclude coronary artery anomalies or diseases [Fazel et al. 2009]. Multidetector CT can accurately diagnose LVNC when involving more than one segment. The noncompaction/compaction cut-off ratio of 2.2 at the end of diastole can distinguish pathologic noncompaction, with a sensitivity of 100% and specificity of 95%. Involving two or more segments differentiates all patients with LVNC from those with other forms of cardiomyopathies as well as from healthy subjects [Melendez-Ramirez et al. 2012] but there are no generally accepted criteria for the use of CT [Mohrs et al. 2007; Engberding et al. 2010].
LVG
LVG is an invasive procedure used in diagnosing LVNC in patients with nonobstructive coronary artery disease, reduced LV function, and a decreased ejection fraction [Engberding et al. 2010; Ionescu and Turcot, 2012]. This procedure is not routinely used to diagnose LVNC [Engberding et al. 2010]. It is usually indicated to be used in adult patients suspected of suffering from LVNC who also have decreased LV function. In this group of patients, LVG can be used to diagnosis LVNC and also rules out causes of LV dysfunction and/or coronary heart disease [Engberding and Bender, 1984; Engberding et al. 2010; Rosa et al. 2011]. This technique can provide more information in patients with nonobstructive coronary artery disease and decreased ejection fraction who underwent a noninvasive procedure [Ionescu and Turcot, 2012].
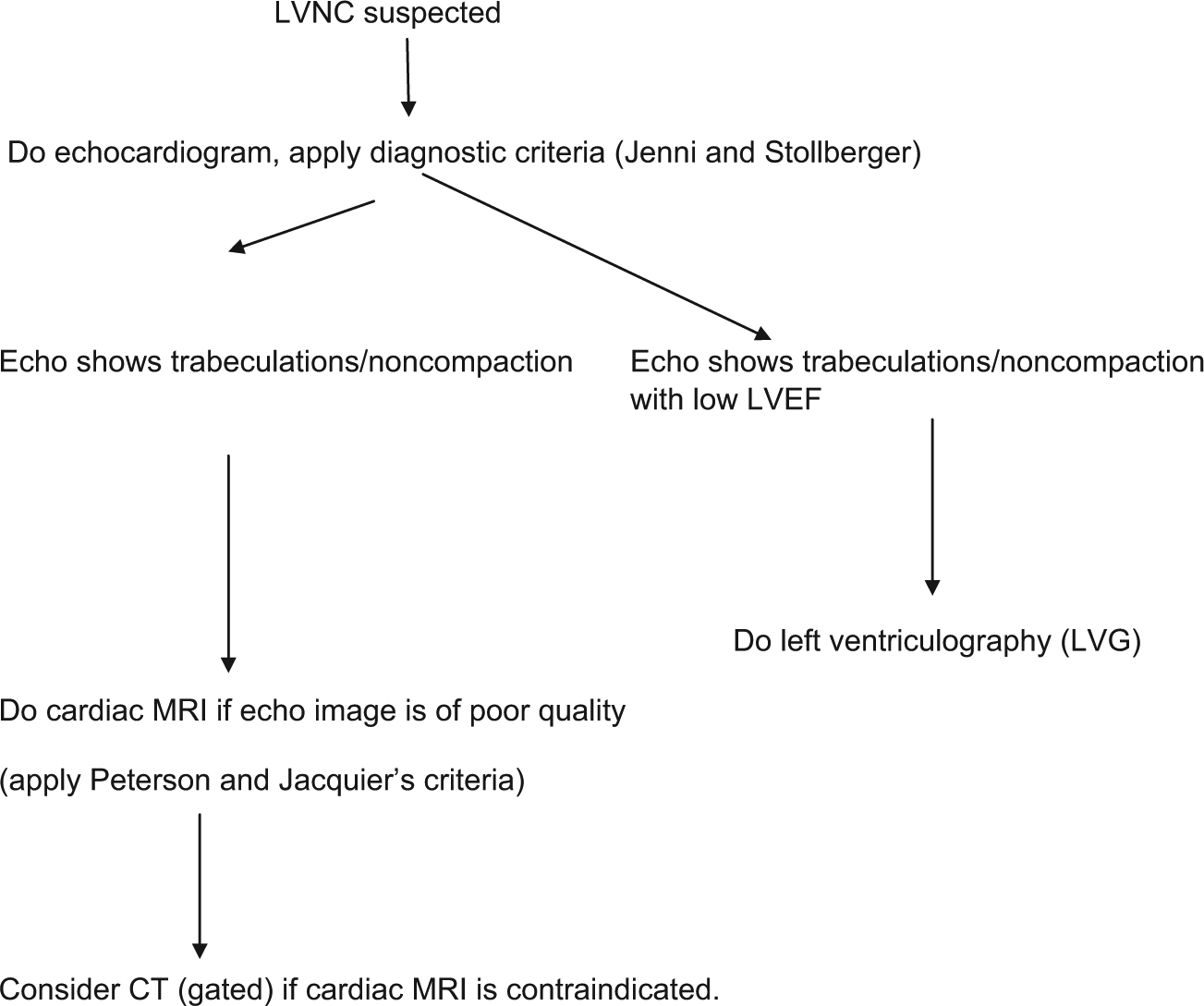
Algorithm for the diagnosis of left ventricular noncompaction (LVNC).
Differential diagnoses of LVNC
The following are differential diagnoses of LVNC: HCM, DCM, restrictive cardiomyopathy (RCM), infiltrative cardiomyopathy, hypertensive heart disease, aberrant chordate tendineae, localized left-ventricular hypertrophy, left-ventricular thrombi, intramyocardial hematoma, intramyocardial abscess, cardiac metastasis, myocarditis, pericarditis, endocardial fibroelastosis, eosinophilic endomycardial disease, and Barth syndrome [Engberding et al. 2010; Maltagliati and Pepi, 2000].
Management
There is no specific therapy for LVNC. Management depends on the clinical manifestations. Patients with a reduced left ventricular ejection fraction (LVEF) are treated with standard medical therapy for heart failure. Cardiac resynchronization therapy in a small series of 4 patients with LVNC was successful in improving LV function [Oginosawa et al. 2008]. A study demonstrated that patients with isolated LVNC associated with DCM had greater LV reverse remodeling after cardiac resynchronization therapy than patients with DCM alone [Bertini et al. 2011].
According to international guidelines, symptomatic ventricular arrhythmia and impaired systolic function should be treated with antiarrhythmic agents or implantable cardiac defibrillator (ICD). WPW syndrome or other AV reentrant or AV nodal reentrant tachycardia are treated with radiofrequency ablation [Pignatelli et al. 2003; Yasukawa et al. 2001]. Holter monitoring is considered once a year to detect asymptomatic atrial and ventricular arrhythmias.
Chronic warfarin therapy is suggested in patients with history of thromboembolism, atrial fibrillation, and/or an LVEF <40% as there is a high risk of thrombus formation within intratrabecular recess. Although antithrombotics (e.g. warfarin) are commonly used to reduce the risk of systemic thrombosis in LVNC patients, Botto states that aspirin can be an alternative therapy [Botto, 2004].
LV remodeling surgery was effective in treating LVNC associated with large LV dimension and depressed LVEF by eliminating the embolic source which eventually reduces LV diameter [Shimamoto et al. 2007].
Heart transplantation should be considered in LVNC patients with end-stage HF who have failed standard medical therapy for heart failure [Stamou et al. 2004], however large-scale data in this regard is lacking.
Screening
Genetic studies are not routinely recommended but echocardiographic screening of family members is suggested [Murphy et al. 2005]. In a series of 45 patients with LVNC, 8 out of 32 asymptomatic relatives had echocardiographic abnormalities such as LVNC, LVNC with systolic dysfunction, and LV enlargement without LVNC [Murphy et al. 2005]. Results from French registry demonstrated that 8% of LVNC was detected by familial screening in asymptomatic patients [Habib et al. 2011].
Patients with LVNC are recommended to undergo neurological evaluations whether they manifest symptoms or not. In a study of 49 patients with LVNC, 82% of the patients who where neurologically evaluated had neurological disorder [Stollberger et al. 2002].
Prognosis
LVNC is associated with high rates of morbidity and mortality in adults. Much better prognosis is noted in patients with early detection of the illness and patients with few or no symptoms. The presence of symptoms such as arrhythmias, thromboembolic events, and the progression of HF worsen the prognosis of LVNC. This was demonstrated in a 46-month follow-up series of 65 patients with LVNC. Among 48 patients who presented with symptoms, 15 patients experienced cardiovascular death or heart transplantation during the period of follow up while there were no deaths or cardiac transplantations among the 17 patients who were asymptomatic at diagnosis [Lofiego et al. 2007]. In a follow-up (over a 6-year period) case series of 17 patients with LVNC, 8 (47%) patients died during the follow up while 2 (12%) patients underwent heart transplantation [Ritter et al. 1997]. In a 44-month follow-up study of 34 patients with LVNC, 53% had HF requiring hospitalization, 41% had ventricular tachyarrhythmia, 12% received an implantable cardioverter defibrillator (ICD), 24% had a thromboembolic event, 35% died, and 12% of the patients underwent heart transplantation. This study demonstrated a 58% probability of survival free of death or heart transplantation at 5 years [Oechslin et al. 2000]. However, there are conflicting reports regarding mortality because another case series of 45 adults’ demonstrated mean survival without death or transplantation to be 97% at almost 4 years and the presence of thromboembolic events were only 4% [Murphy et al. 2005]. In retrospective study of 29 children with LVNC, poorer prognosis was demonstrated in patients with early presentation age and high LV end-diastolic diameter at the diagnosis [Ozgur et al. 2011].
Conclusion
LVNC is a genetically heterogeneous congenital disorder assumed to occur as an arrest of the compaction process in early embryogenesis. LVNC can occur in isolation or in association with other cardiac or noncardiac anomalies. Children with LVNC often have an increased familial prevalence, associated facial dysmorphism, and increased concurrence of congenital arrhythmias such as WPW syndrome. LVNC in adults is usually due to mutations in the sarcomere protein. Clinical presentation ranges from patients with no symptom to patients who present with symptoms of HF, thromboembolic events, arrhythmias, and sudden death. Increased awareness of the disease and improvements in the diagnostic technique may be accountable for the high incidence of LVNC. Making this diagnosis is of major importance as it can significantly influence decisions on long-term management, prognosis, and the need to screen living relatives. Correct diagnosis of noncompaction has important implications due to the possible association with other cardiac abnormalities, neuromuscular disorders, and/or other systemic anomalies.
Echocardiography is the most common technique used in diagnosing LVNC. Nowadays, CMRI, CT scan, and LVG are other important diagnostic techniques used in diagnosing LVNC.
Currently, LVNC may be overdiagnosed because of overly sensitive diagnostic criteria. We suggest using more standardized criteria and additional imaging technique that visualizes the cardiac morphology in different planes.
There is no specific treatment for LVNC. Management involves treatment of HF, arrhythmias, and thromboembolic events. Heart transplantation is reserved for patients with end-stage HF and/or patients who failed medical treatment.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no conflicts of interest to declare.
