Abstract
Objective
Patients with newly diagnosed cardiomyopathy frequently undergo coronary angiography to exclude significant coronary artery disease (CAD). Contrast-enhanced cardiac magnetic resonance imaging (CE-CMR) can detect myocardial scar in-vivo, and has the potential to identify patients with cardiomyopathy secondary to prior myocardial infarction.
Aim
To investigate the accuracy of contrast-enhanced cardiac magnetic resonance imaging (CE-CMR) in differentiating ischemic from non-ischemic cardiomyopathy in patients with new onset heart failure and previously undiagnosed left ventricular systolic dysfunction.
Methods
Forty eight patients (mean age 54 ± 8) were prospectively identified from symptoms and echocardiography, and underwent both CE-CMR and angiography. Patients with >70% diameter stenosis in > 1 major epicardial vessel on angi-ography were considered to have an ischemic cardiomyopathy. Myocardial scar was assessed using delayed enhancement inversion-recovery imaging after gadolinium administration. Patients with subendocardial enhancement typical of a myocardial infarction were classified as having an ischemic etiology by CE-CMR.
Results
Sixteen patients were classified as ischemic by angiography, and of these 15 had subendocardial enhancement on CE-CMR. The sensitivity and negative predictive value for CE-MRI were 94% and 97% respectively for detecting an ischemic cause. However 5 patients with subendocardial enhancement by CE-CMR had no obstructive coronary disease, suggesting misclassification by angiography, and reducing the specificity (82%) of CE-CMR.
Conclusion
The lack of subendocardial scar on CE-CMR identifies patients with new-onset heart failure and cardiomyopathy who may not require angiography.
Differentiating an ischemic from a non-ischemic etiology is a key step in the investigation of patients with left ventricular systolic dysfunction. Patients with significant coronary artery disease (CAD) and systolic dysfunction may benefit from revascularization or ventricular reduction procedures, and should receive secondary preventive therapy including statin drugs. The presence of significant CAD on coronary angiography is often used to confirm the diagnosis of ischemic cardiomyopathy. Contrast-enhanced cardiac magnetic resonance imaging (CE-CMR) can detect myocardial scar in vivo with high sensitivity, specificity and spatial resolution. (Kim et al. 2003) McCrohon and colleagues reported three distinct patterns of enhancement in patients with cardiomyopathy of known etiology. (McCrohon et al. 2003) Subendocardial enhancement was seen in all patients with known ischemic cardiomyopathy. Mid-myocardial enhancement or no enhancement was typical of patients with non-ischemic cardiomyopathy. The accuracy of CE-CMR in prospectively distinguishing ischemic from non-ischemic etiologies in patients presenting acutely with heart failure and a newly diagnosed cardiomyopathy is uncertain. We hypothesized that the presence of any subendocardial scar on CE-CMR would accurately identify patients in this group who had important CAD.
Methods
Patients
Patients presenting during a two-year period (January 2003-2005) with a new diagnosis of heart failure were prospectively identified. Patients were included if they had: (a) a clinical presentation of new onset acute heart failure, (b) an echocardiogram demonstrating systolic dysfunction (ejection fraction <50%) (c) undergone coronary angiography and (d) successfully completed a CE-CMR at least 2 weeks, but no more than three months, after the index clinical visit. Patients were excluded from the analysis if: (a) they had a documented history of myocardial infarction or coronary artery disease (b) their primary presentation was a tachyarrhythmia, or if (c) significant valvular disease was present. Forty-eight patients fulfilled these criteria and were included. The institutional Ethics Committee approved this analysis. Coronary angiography was analyzed qualitatively, and patients were considered to have an ischemic cardiomyopathy if they >70% diameter stenosis in a major epicardial coronary artery. The reporting Cardiologist or Cardiac Radiologist was blinded to the clinical information and CE-CMR results.
Cardiac MRI
CE-CMR was performed on a Philips Intera 1.5 Tesla magnet using a Synergy Cardiac Coil (Philips, Best). Electrocardiographic gated steady state free precession cine images were acquired in the 2 and 4 chamber, left ventricular outflow tract and short axis views. The sequence parameters were: flip angle 65°, TE = 1.64 ms, TR = 3.3 ms, slice thickness 8 mm. Delayed enhancement imaging was performed after injection of a standard dose (Kim et al. 2003) of 0.15 mmol/kg gadolinium-based contrast agent (Omniscan, Nycomed Amersham, Oslo). A three dimensional inversion-recovery segmented gradient echo sequence was used in 2 and 4 chamber, left ventricular outflow tract and short axis views. Imaging was commenced 10 minutes after contrast administration using an inversion time optimised to obtain adequate nulling of normal myocardium (260-340 ms). The imaging sequence parameters included an in-plane voxel size 1.2-1.8 × 1.2-1.8 mm2, slice thickness 6 mm, flip angle 15°, TE = 3-4 ms and TR = 8-9 ms. A Philips EzyVision workstation was used for image analysis. LV end-diastolic volume, LV end-systolic volume, LV ejection fraction were calculated from the short axis cine images. Regional wall motion and the extent of delayed enhancement were assessed using a 17-segment model. (Cerquiera et al. 2002) Segmental wall motion was visually assessed and scored as normal = 1, mildly or severely hypokinetic = 2, akinetic = 3 or dyskinetic = 4. Areas of hyperen-hancement were defined as subendocardial or mid-myocardial and the transmural extent of enhancement was graded as none = 0, 1%-25% = 1, 26%-50% = 2, 51%-75% = 3 and 76%-100% = 4. Segmental scores for both wall motion and hyperenhancement were summed, and for wall motion an index of regionality was determined using the standard deviation of the segmental wall motion scores for each patient. Analysis of the CE-CMR was undertaken by two Cardiologists with expertise in cardiac magnetic resonance imaging who were unaware of the results of prior investigations for either CAD or atherosclerosis.
Statistics
Data are presented as mean ± standard deviation (SD). Comparisons between groups were performed using Chi Square and Fisher's Exact test for discrete variables and non-paired Student's t-test for continuous variables. Differences were considered significant at p < 0.05 (2-sided).
Results
The clinical characteristics of the patients are shown in Table 1. Sixteen patients had an ischemic cardiomyopathy determined by coronary angiog-raphy, and most had three-vessel disease. The majority of patients were male, and with the exception of New York Heart Association functional class there were no significant differences between the groups. There was a trend towards a higher prevalence of standard risk factors for coronary disease in the ischemic group, but this did not reach statistical significance in this small study population.
Baseline characteristics.
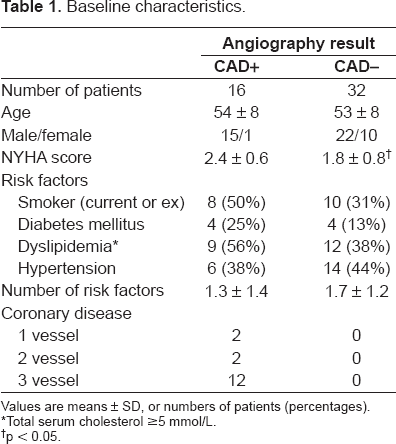
Values are means ± SD, or numbers of patients (percentages).
Total serum cholesterol ≥5 mmol/L.
p < 0.05.
CE-CMR was successfully performed in all patients (Table 2). Measurements of LV volumes were similar in both ischemic and non-ischemic groups, and the wall motion indices indicated greater regionality in the patients with an ischemic etiology. Twenty-three patients (48%) had no enhancement, and were classified by CE-CMR as having a non-ischemic cardiomyopathy. None of these patients had significant coronary artery disease. Five patients (10%) had mid-myocardial enhancement, of which 4 were correctly classified as having a non-ischemic cardiomyopathy. Fifteen (94%) of the patients with angiographically-defined ischemic cardiomyopathy had subendocardial enhancement. Six patients were incorrectly classified by CE-CMR: five with an ischemic pattern of delayed enhancement had no significant CAD, and one classified as non-ischemic had significant CAD. The sensitivity of delayed enhancement imaging for the detection of ischemic cardiomyopathy was 94%, with a specificity of 82%. The negative predictive value was 97%. MRI correctly classified 42 of 48 patients. Representative images from two patients are shown in Figure 1.
CE-CMR findings.

Values are means ± SD, or numbers of patients (percentages).
p < 0.05.
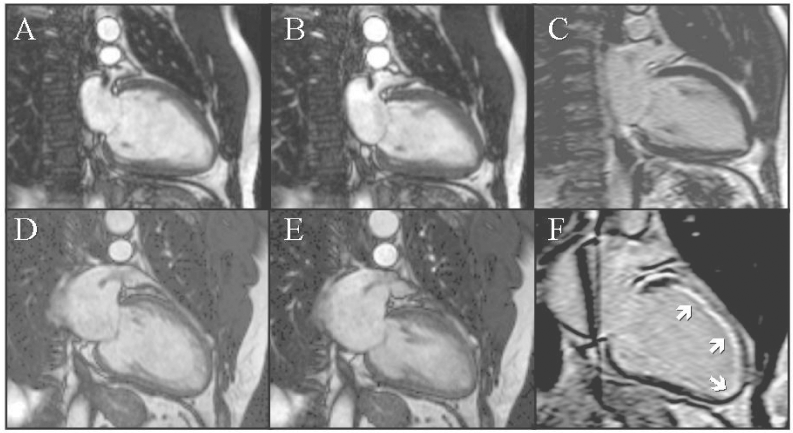
Representative two chamber images from two patients presenting with acute congestive heart failure and LV dysfunction. Patient 1 is a 42 year old smoker with dyslipidaemia, showing cine images from (
Discussion
This study demonstrates that CE-CMR can accurately determine whether LV systolic dysfunction is the result of CAD in patients presenting acutely with heart failure and a newly diagnosed cardiomyopathy of unknown etiology. In particular the negative predictive value of 97% suggests that the absence of subendocardial enhancement on CE-CMR can effectively identify patients who do not have flow-limiting CAD, potentially avoiding unnecessary diagnostic angiography.
We identified that 10% of patients with a subendocardial pattern of delayed enhancement typical of a prior myocardial infarction had no angiographic CAD, leading to a reduced specificity for CE-CMR. This finding is concordant with prior studies: both McCrohon et al. and Soriano et al. reported that 13% and 9% respectively of patients in their series were similarly classified. (McCrohon et al. 2003; Soriano et al. 2005) Myocardial infarction in patients with normal coronary arteries may result in enhancement on CE-CMR, (Christiansen et al. 2006) and the potential mechanisms include in situ thrombosis, coronary embolus or prolonged vasospasm. (Bakshi et al. 2002) However alternative causes of fibrosis and enhancement such as the sequelae of myocarditis, infiltrative processes and deposition diseases cannot be excluded from clinical data alone. (Abdel-Aty et al. 2005)
Angiography is frequently cited as the diagnostic standard for ischemic cardiomyopathy, but there is a lack of consensus regarding the angiographic threshold at which a patient should be considered as having an ischemic etiology for LV dysfunction. Felker et al. have proposed a standardized definition of ischemic cardiomyopathy that includes patients with (a) a history of myocardial infarction or revascularization, or (b) ≥75% stenosis of left main or proximal left anterior descending, or (c) ≥75% stenosis of two or more epicardial vessels. (Felker et al. 2002) For the purposes of this study we elected to use a simpler definition (>70% diameter stenosis in a major epicardial coronary artery). However our analysis is unchanged in this study population if we apply the frequently used lower threshold (>50% diameter stenosis in a major epicardial coronary artery), which lends support to the using the greater degree of coronary luminal obstruction in Felker et al.'s proposed definition.
Additionally significant proximal CAD and a non-ischemic cause for cardiomyopathy may coexist. One patient in our series with a severely dilated and impaired LV (end diastolic volume 468 mL, ejection fraction 12%) had no subendocardial scar. Angiography found an occluded proximal RCA, and stenoses of the mid-LAD (70%) and first obtuse marginal (80%). CE-CMR was therefore considered to have misclassified this patient. Although the severe myocardial dysfunction could have been attributed to resting hypoperfusion and hibernation alone, a dobutamine stress echo demonstrated no ischemia, and revascularization was not undertaken. It has been suggested that the reliance on coronary angiography to classify cardiomyopathies as ischemic or non-ischemic is flawed, (McCrohon et al. 2003) and this view is supported by autopsy studies. (Uretsky et al. 2000) Direct assessment of the degree of myocardial scar using CE-CMR may be more valid, and recently the presence of enhancement in patients without obstructive coronary disease (defined as >50% diameter luminal stenosis in any coronary artery) has been shown to have prognostic significance. (Asomull et al. 2006)
Both stress perfusion imaging and non-invasive coronary imaging are possible using CE-CMR. We did not evaluate the incremental diagnostic value of a more comprehensive evaluation including these modalities. We have already noted a high negative predictive value for CE-CMR in excluding significant obstructive CAD in patients with undifferentiated dilated cardiomyopathy, suggesting that there would be minimal further gain in using such additional imaging sequences for this purpose.
Study Limitations
This is a small study population, and these results should not be considered sufficiently robust to alter clinical practice. The interpretation and prognostic importance of delayed enhancement in patients without significant coronary disease remains an area of considerable research interest, and while the finding of fibrosis in such patients lowers the specificity of CE-CMR for diagnosis of an ischemic myopathy, it may have considerable clinical relevance. Therefore no attempt has been made to evaluate the cost-effectiveness of a diagnostic strategy involving CE-CMR.
