Abstract
Aims
Essential hypertension is characterized by increased reactive oxygen species (ROS) generation harmful for insulin sensitivity and nitric oxide (NO)-mediated vasomotor function, a noxious effect that paraoxonase (PON)l, an antioxidant circulating high-density lipoprotein (HDL)-bound esterase, may counteract. The PON 1 gene contains several polymorphisms including a glutamine (Q_) to arginine (R) transition at position 192 encoding circulating allozymes with higher antioxidant activity that might influence both parameters.
Methods
Q192R was determined by polymerase chain reaction in 72 never-treated, glucose-tolerant, uncomplicated essential hypertensive men. Insulin sensitivity was assessed by homeostasis model assessment (HOMA) and endothelial function by forearm vasodilation (strain-gage venous plethysmography) to intra-arterial acetylcholine (ACH) with sodium nitroprusside (NIP) as a NO-independent control. Additional evaluation variables included 24-hour blood pressure (BP), lipids, BMI, smoking status, and metabolic syndrome (MetS) by Adult Treatment Panel (ATP)-III criteria. R192 was considered as the rare allele, and its associations analyzed by dominant models (Q/Qvs. Q/R + R/R).
Results
Genotype frequencies were consistent with the Hardy-Weinberg equilibrium. HOMA was lower and insulin resistance (the upper fourth of HOMA values distribution) less prevalent in Q/R + R/R carriers in whom ACH-mediated vasodilatation was greater and endothelial dysfunction (the bottom fourth of ACHAUC values distribution) less frequent than in Q/Q homozygotes. Q192R polymorphism and MetS were unrelated parameters despite their common association with insulin resistance. 24-hour BP, BMI, lipids, and smoking habits were homogeneously distributed across genotypes.
Conclusions
Q192R polymorphism associates differentially with insulin sensitivity and endothelial function in essential hypertensive men.
Keywords
Introduction
Experimental and clinical evidence supports a pathophysiological role for oxidative stress in the development and progression of arterial hypertension.1,2 The unbalanced production of reactive oxygen species (ROS: superoxide anion, hydroxyl anion, and hydrogen peroxide), while contributing to hypertensive end-organ damage,1,2 also impairs insulin-mediated glucose uptake 3 and nitric oxide (NO)-mediated vasomotor regulation, 4 two pathophysiologically relevant and interconnected parameters 5 frequently abnormal in human essential hypertension.6,7 Paraoxonase (PON)l, a high-density lipoprotein (HDL)-associated hydrolytic and antioxidant enzyme subject to genotypic regulation, 8 tends to restore redox balance thus protecting vessels from atherosclerosis progression. 9
The PON1 gene contains several polymorphisms including a glutamine (Q to arginine (R) transition at position 192 (Q192R) 8 encoding circulating allozymes with higher antioxidant activity. 10 Therefore, the Q192R PON1 genotypic variants may contribute to the modulation of insulin sensitivity and NO-mediated vasomotion in essential hypertensive patients, a plausible but by and large untested possibility.
The purpose of this study was to test the hypothesis of an association between the PON1 Q192R polymorphic variants and insulin sensitivity, as estimated by the homeostasis model assessment (HOMA), 11 a robust and validated clinical tool for the assessment of insulin sensitivity, 12 and the vasodilatory response to intra-brachial acetylcholine (ACH), a NO-releasing muscarinic agonist. 13 The study was carried out in a group of carefully screened, glucose-tolerant men with untreated, uncomplicated mild-to-moderate essential hypertension.
Methods
Subjects
Seventy-two never-treated sedentary, Caucasian, genetically unrelated hypertensive men [casual blood pressure (BP), greater than 140/90 mmHg on at least three occasions as outpatients] participated in this study (see Table 1, left-hand column, for the overall characteristics of the sample) as part of a preventive program for the identification and treatment of hypertension and associated risk factors.
Age and clinical characteristics of the sample overall and by Q192R PQN1 polymorphism.
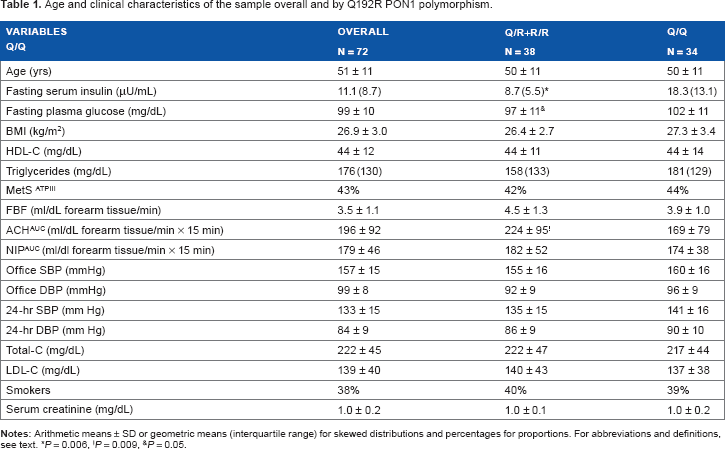
P = 0.006,
P = 0.009,
P = 0.05.
Exclusion criteria were glucose intolerance (two-hour plasma glucose ≥ 140 mg/dL after a 75 g oral glucose load), renal insufficiency (serum creatinine > 1.2 mg/dL), proteinuria at the dipstick test, and impaired cardiac function (ejection fraction < 50%). Coexisting coronary and vascular diseases were excluded based on medical history, physical examination, routine blood chemistry, baseline electrocardiogram or, when indicated, treadmill test, stress echocardiography, carotid, and lower limb echo-Doppler sonography. In all, renal ultrasound scans showed normal-sized kidneys and no evidence of cortical scarring or obstructive uropathy. Angiograms, when indicated, had excluded renal artery stenosis, whereas routine clinical and biochemical evaluation excluded other secondary forms of hypertension. Screening was completed in two weeks period.
The protocol was approved by the local ethics committee, and all patients signed the required consent form before the study.
Genotype Determination
Genomic DNA was isolated from whole blood cells as described by Blin and Stafford 14 and stored at 4 °C till assessment of the Q192R polymorphism by polymerase chain reaction (PCR) and restriction digestion methods. Amplifications were performed in a final volume of 20 μL containing 100 ng of genomic DNA, 0.5 μM of appropriate primers (MWG Biotech Ebers, Germany), and 1 × Mastermix Cat.# M7505 (Promega). Briefly, DNA was denaturated at 94 °C for five minutes, and the reaction mixture was subjected to thermocycling on GeneAmp PCR System 2700 (Applied Biosystem) by means of 35 denaturation cycles (60 seconds each) at 94 °C with 60 seconds of annealing at 61 °C and 60 seconds of extension at 72 °C, and final extension at 72 °C for 10 minutes. Sense primer 5′TATTGTTGCTGT GGGACCTGAG-3′ (22-mer) and antisense primer 5′-CACGCTAAACCCAAATACATCTC-3′ (23-mer) encompassing the 192 polymorphic region of the human PON1 gene were used to identify Q192R polymorphic variants. 15 The 99 bp PCR product was digested overnight at 37 °C with 2 U of Alwl restriction endonuclease (New England BioLabs, Cambridge, MA, USA). The R allele, but not the Q allele, contains a unique AlwI restriction site that results in the production of two fragments of 68 and 31 bp, respectively. Digestion products were separated by 12% polyacrylamide gel electrophoresis at 150 mA for two hours and visualized by silver staining. To minimize source of bias, two independent observers, blinded to the pertinent characteristics, assigned genotypes.
Glucose and Insulin Determinations
Plasma glucose and insulin were measured by the glucooxidase method (Glucose Analyzer II, Beckman Instruments, Fullerton, CA, USA) and immunoradiometry (INSI-CTK Irma, DiaSorin S.p.A., Vercelli, Italy; no cross-reactivity with human pro-insulin; interassay variation coefficient < 5%), respectively.
Forearm Studies
Forearm studies were performed in the morning in a quiet, air-conditioned room as already described in detail. 16 Subjects were fasted and instructed to restrain from heavy exercise and to avoid smoking and emotional excitement from the day before the experiment. Total forearm blood flow (FBF) was measured by venous plethysmography with a strain-gage apparatus (Hokanson, EC 5R Plethysmograph). Strands made of silastic tubing of 0.4 mm ID and 0.8 mm OD filled with mercury were used. The gage was applied on the dominant arm, 5-6 cm distal to the elbow at a tension sufficient to keep the gage in the same position throughout the experiment. Patient forearm was kept on a table, slightly flexed and inclined at about 45° to the horizontal plane with the wrist and hand supported by sand bags. One minute before FBF determination, a pneumatic pediatric cuff was placed around the wrist and inflated to suprasystolic arterial BP in order to exclude the hand vascular region. A second cuff was placed proximal to the plethysmograph and automatically inflated to a pressure of 40 mmHg to allow FBF measurement according to the venous occlusion method.
Forearm infusions were carried out through a 22-gage polyethylene catheter (Angiocath, Becton Dickinson) connected to an infusion pump (Perfusor, Secura FT, Braun) after approximately 30 minutes of rest. BP was measured every five minutes throughout the study at the contralateral arm by an automated device (NIBP KO 7267.004, Kontron Instruments). Forearm infusion studies included administration of fresh solutions of ACH HCl (Farmigea, Italy; 7.5, 15, and 30 μg/minute, five minutes each), a NO-releasing compound, 13 and sodium nitroprusside (NIP, Malesci, Italy) (0.8, 1.6, and 3.2 μg/minute, five minutes each in syringes protected from light through aluminum foil), a NO-independent vasodilator, 17 as a control. Preliminary studies had shown that those infusion periods were sufficient to reach a plateau of FBF response without changes in systemic arterial pressure and contralateral FBF. Local administration of ACH and NIP was preceded and followed by saline infusion (0.6 mL/minute). The infusion sequence was randomized, and a 30 minutes interval was left between the first and second drug administration.
Other Clinical Parameters
Office BP, serum creati-nine, lipids, and anthropometric measurements (height and weight) were measured by standard methods. The 24-hour BP was measured through an oscillometric monitor (Diasys Integra, Novacor). The recording began between 8.30 and 9.00 a.m. with 15 minute interval readings until midnight, and 30 minute intervals from midnight to 8.00 a.m. (≥90% valid measurements).
Definitions and Data Processing
Q and R alleles were considered as dominant and rare alleles, respectively, and their relationship with study variables was analyzed by dominant models (Q/Qvs. Q/R + R/R).
Insulin sensitivity was assessed through HOMA 11 [fasting plasma insulin (μU/mL) x fasting plasma glucose (mMol/L) /22.5)]; increasing HOMA-IR values denote progression from normal to impaired insulin sensitivity. HOMA values above the 75th percentile (cutoff point: 4.7 U) were considered as indicative of insulin resistance.
In the absence of BP changes during the infusion, forearm studies were analyzed on FBF (mL/dL forearm tissue/minute) as relative changes from baseline and area under the curve (AUC, trapezoidal rule). ACHAUC values below the 25th percentile (cutoff point: 123 mL/dL forearm tissue/minute x 15 minutes) were considered as indicative of endothelial dysfunction.
Body Mass Index (BMI) was calculated as weight/height 2 (kg/m2). Low-density lipoprotein (LDL)-C was calculated as (total C — (HDL-C + triglycerides/5)). Smoking status was defined as active smoker versus non-smoker, without distinction between former and never smoker. Metabolic syndrome (MetS) was diagnosed according to the 2001 National Cholesterol Education Program (NCEP)-Adult Treatment Panel (ATP) III. 18 As all patients were hypertensive and, hence, fulfilled one of the criteria for MetS, diagnosis required at least two of the remaining four parameters, ie, triglycerides ≥ 150 mg/dL, HDL-C < 40 mg/dL, fasting plasma glucose ≥ 110 mg/dL, and BMI > 29.5 kg/m2, a validated surrogate cutoff for abdominal obesity. 19
Statistics
Differences between continuous variables were analyzed by proper Analysis of Variance models and categorical parameters by chi-square tests. The association of insulin resistance and endothelial dysfunction (present = 1 and absent = 0) with Q192R genotypes and other continuous and categorical covariates was analyzed by age-adjusted logistic regression (maximum-likelihood method), using a backward stepwise procedure (P-to-remove < 0.05) to identify the independent regressors. Odds ratios (ORs, ie, the exponentiated regression coefficients) were used to estimate relative risks and 95% confidence interval (CI). Descriptive statistics were means ± SD or geometric means (interquartile range) for skewed data and percentages for proportions. The level of statistical significance was set at P < 0.05.
Results
Genotype and allelic frequencies were consistent with the Hardy-Weinberg equilibrium (Table 2). Age, lipids, BMI, office and 24-hour BP, smoking status, and serum creatinine were homogeneously distributed across genotypes (Table 1).
Absolute and relative distribution of Q192R genotypes and R and Q allele frequencies.

Fasting insulin, glucose (Table 1), HOMA, and percentages of insulin-resistant patients (Fig. 1) were lower in Q/R + R/R than in Q/Q patients.
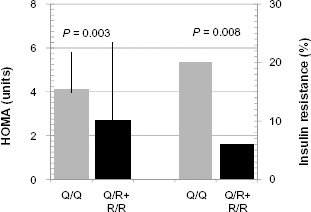
HOMA values (medians and interquartile range, left ordinate) and prevalence of insulin resistance (upper fourth of HOMA values distribution, right ordinate) by Q/R + R/R (n = 38) and Q/Q (n = 34) PON1 genotypes.
Responses to intrabrachial ACH infusion were greater (Fig. 2, left panel) and endothelial dysfunction was less frequent (16% vs. 35%, P = 0.09) in Q/R + R/R than in Q/Q patients; responses to NIP did not differ (Fig. 2, right panel), a pattern confirmed by ACH and NIP data expressed as AUC (Table 1).

Forearm response to ACH and sodium NIP infusion by Q/R + R/R (♦, n = 38) versus Q/Q (X, n = 34) PQN1 genotypes. Data are reported as fold-increase from baseline at each increasing infusion step; P value refers to the difference in forearm responsiveness to ACH between Q/R + R/R and Q/Q subjects. Means ± SD.
Age-adjusted multivariate logistic regression analyses with insulin resistance as the dependent variable showed the independent contribution of Q192R genotype (OR: 0.13, 95% CI: 0.03-0.54, P = 0.002, Q/Q as referent) and MetS (OR: 4.3, 95% CI: 1.2-15.1, P = 0.016). The prevalence of MetS did not differ by Q192R genotypes (Table 1).
When the same analysis was applied on endothelial dysfunction, only the Q192R genotype (OR: 0.34, 95%CI: 0.11-1.07, P = 0.054, Q/Q as referent) played a borderline significant role.
Discussion
Q192R PON1 SNP and Insulin Sensitivity
The main and original finding of this genetic cross-sectional study in glucose tolerant, untreated, uncomplicated mild-to-moderate hypertensive men was the greater insulin sensitivity of homo- and heterozygous carriers of the mutant R allele as compared with Q/Q homozygotes. The result, based on a well-validated index such as HOMA,11,12 is compatible with a greater antioxidant efficiency conferred by the R allele as insulin sensitivity is influenced by oxidative stress, 3 while PON1 enzymatic function, at least as assessed through paraoxonase activity, is severalfold higher in Q/R + R/R than in Q/Qsubjects. 10 Thus, the Q192R polymorphism might contribute to the maintenance of a critical metabolic parameter in essential hypertension, a clinical condition in which increased amount of ROS1,2 and oxidized LDL-C 20 synergize to impair insulin signaling. 21 Quite surprisingly in the light of those considerations but in agreement with other reports, 22 we found no relationship between Q192R PON1 polymorphisms and MetS, a well-characterized insulin-resistant state. 18 Moreover, both parameters entered in a multivariate logistic regression analysis were strong and independent predictors of insulin resistance implying that the insulin resistant states associated with the Q192R Polymorphism and Mets are pathophysiologically dissimillar phenomena. 18 Perhaps, this puzzling behavior relates to the ambiguity of HOMA, an index that does not distinguish impaired glucose disposal through skeletal muscle from uninhibited hepatic glucose output, 23 two insulin-mediated pathways 23 both sensitive to oxidative stress3,24 whose relative role might be dissected through the use of the hyperinsulinemic euglycemic glucose clamp. 25 In this regard, previous reports about the lack of association between the Q192R SNP and insulin sensitivity as assessed through that technique 26 may suggest a role for impaired regulation of hepatic glucose output that, however, remains a pathophysiologically attractive but purely speculative possibility.
Q192R PON1 SNP and NO-mediated Vasomotor Function
A second interesting outcome of this study was the greater responsiveness to ACH, a NO-releasing muscarinic agonist, 13 with preserved effect of NIP, a NO-independent vasoactive drug, 17 in Q/R + R/R than in Q/Q patients. This diverging pattern, diagnostic of a differential interaction with the NO-mediated endothelial pathway, could not be ascribed to a series of potentially damaging factors for endothelial function such as BP levels, LDL-C, and smoking habits 27 or pathophysiological factors related to the MetS phenotype 28 because all those variables were evenly distributed across Q192R genotypes. Rather, responses to ACH are dependent on endothelial NO, 13 whose degradation is increased by oxidized LDL and other peroxides 29 that PON1 seems to dispose to a greater extent in the presence of the mutant R allele.30,31 This hypothesis contrasts with in vitro evidence, 32 although in vivo and in vitro regulation of LDL oxidation may differ or the Q192R PON1 polymorphism might influence forearm response to ACH in hypertension by mechanism unrelated to LDL oxidation, eg, by interacting with other functional PON gene polymorphisms in the coding or promoter regions. 8 Whatever be the case, our data are consistent with previous reports of greater flow-mediated forearm vasodilatation 33 and preserved epicardial coronary vasodilatation to NO-acting substances34,35 in Q/R + R/R patients as compared with their Q/Q counterparts. The evidence in the field is, however, controversial36–38 possibly because of heterogeneous experimental conditions, different methods to investigate endothelial function and the specific pathophysiology of the clinical conditions under investigation.
Q192R SNP, Insulin Resistance, and Endothelial Dysfunction
Some final comment deserves the weaker and borderline significant strength of the association of the Q192R polymorphism with endothelial dysfunction as compared with the highly significant and quantitatively stronger link with insulin resistance. However, forearm vasodilator responses to ACH, albeit highly specific for the L-Arginine-NO system, 13 relate poorly to the underlying tonic activity of the L-Arginine-NO system. Moreover, they are unpredictably dependent upon endogenous muscarinic receptor reserve and ACH-esterase activity 39 and mechanisms additional to NO 40 concur to impair endothelial function in hypertension. Quite importantly, the high inter-individual variability of the infused forearm model method may have hindered stronger associations emerging more clearly in larger series. 41
Limitations of the Study
Our study has some important limitations to be taken into account. First, we did not assess paraoxonase/arylesterase and other esterase activities including thiolactonase activity targeting homocysteine thiolactone, a main pro-oxidant agent, 42 an important point because measurement of one enzymatic activity alone may not provide a complete sense of PON1 metabolism. As a matter of fact, paraoxonase activity was increased in non-diabetic subjects with insulin resistance as assessed by HOMA, 43 confirming how difficult it may be to derive functional inferences based on the Q192R polymorphism. Second, cross-sectional studies such as ours do not provide causative relationship but only associations of uncertain cause-effect relevance. Third, our sample size was relatively limited although the phenotypic penetrance of the Q192R polymorphism was evidently stronger than that of other potentially relevant polymorphisms44–46 analyzed by us with negative results in this same series. Fourth, our all male cohort does not allow to extrapolate the present results to females in whom, eg, anti-oxidant effects of endogenous estrogens 47 or increased HDL concentration typical of women 48 might be conducive to different results.
Conclusions
The presence of the mutant R allele associates with greater insulin sensitivity and NO-mediated forearm vasodilatory response when compared with the wild Q/Q configuration of the Q192R polymorphism in uncomplicated essential hypertensive men. This finding has potentially relevant pathophysiological and clinical implications in need, however, of further evaluation.
Author Contributions
Conceived and designed the experiments: RP. Analyzed the data: GDO. Wrote the first draft of the manuscript: RP. Contributed to the writing of the manuscript: GP. Jointly developed the structure and arguments for the paper: LP, DL. Made critical revisions and approved final version: SDP. All authors reviewed and approved of the final manuscript.
