Abstract
Vascular insulin resistance often precedes endothelial dysfunction in type 1 diabetes mellitus. Strategies to limit vascular dysfunction include intensive insulin therapy (4–9 mM) and aerobic training. To avoid the risk of hypoglycaemia, individuals often prescribed conventional insulin therapy (9–15 mM) and participate in resistance training. In a model of type 1 diabetes mellitus, this study examined insulin-induced vasomotor function in the aorta and femoral artery to determine (1) whether resistance training with conventional insulin therapy provides the same benefits as aerobic training with conventional insulin therapy, (2) whether aerobic training or resistance training, when paired with conventional insulin therapy, results in superior vasomotor function compared to intensive insulin therapy alone and (3) whether vessel-specific adaptations exist. Groups consisted of conventional insulin therapy, intensive insulin therapy, aerobic training with conventional insulin therapy and resistance training with conventional insulin therapy. Following multiple low doses of streptozotocin, male Sprague-Dawley rats were supplemented with insulin to maintain blood glucose concentrations (9–15 mM: conventional insulin therapy, aerobic training and resistance training; 4–9 mM: intensive insulin therapy) for 12 weeks. Aerobic training performed treadmill exercise and resistance training consisted of weighted climbing. Coinciding with increased Akt signalling, aerobic training resulted in enhanced insulin-induced vasorelaxation in the femoral artery. Intensive insulin therapy displayed increased mitogen-activated protein kinase signalling and no improvement in insulin-stimulated vasorelaxation compared to all other groups. These data suggest that aerobic training may be more beneficial for limiting the pathogenesis of vascular disease in type 1 diabetes mellitus than merely intensive insulin therapy.
Keywords
Introduction
Type 1 diabetes mellitus (T1DM) is associated with an increased susceptibility to cardiovascular diseases. 1 Both clinical and experimental T1DM are associated with impaired endothelial function due to reduced production and bioavailability of nitric oxide (NO), which is often a precursor to overt atherosclerosis.2–5 Vascular insulin resistance, identified in patients with T1DM,6–8 has been associated with increased adhesion molecules (ICAM-1, VCAM-1 and E-selectin), 9 accelerated development of atherosclerosis 10 and increased vasoconstriction.11–13 Strategies to improve vascular insulin sensitivity, such as exercise training, may provide an important means to mitigate the increased risk of peripheral arterial disease (PAD) in populations with T1DM. Importantly, the exercise-induced benefits of both aerobic and resistance exercise to the vasculature can be both modality- and vessel-specific.14–16
Insulin stimulates both endothelium-dependent vasodilation and vasoconstriction, which is believed to be an important means to regulate the delivery of insulin and glucose to skeletal muscle.11,17 Briefly, insulin-induced endothelium-dependent dilation occurs following the activation of the phosphatidylinositol 3-kinase (PI3K)-Akt pathway, which phosphorylates endothelial nitric oxide synthase (eNOS) and increases the production of NO. Insulin can also lead to endothelium-dependent vasoconstriction by activating the Ras/mitogen-activated protein kinase (MAPK) pathway, subsequently releasing endothelin-1 (ET-1). During states of vascular insulin resistance, available evidence indicates that there is a pathological imbalance in endothelial insulin signalling, reflected by decreased PI3K-Akt signalling and increased MAPK signalling. The net result is an attenuation or loss of insulin-induced vasorelaxation.11–13
Regular exercise has been shown to improve the vascular actions of insulin.13,18–20 However, patients with T1DM who adhere to a physically active lifestyle are often prescribed a more conventional insulin therapy (CIT) to mitigate the risk of hypoglycaemia that can accompany exercise. 21 In this regard, due to the risk of hypoglycaemia associated with aerobic exercise training (AT) in patients with T1DM, the use of other forms of exercise such as resistance exercise training (RT) has gained attention.22–24 Although a relationship exists between glycaemic control (HbA1c) and the degree of endothelial dysfunction in T1DM, 25 physical activity may be a more effective means to improve the vasoreactivity to insulin, conceivably mediated by shear stress.13,19,26,27 Indeed, it has been reported in type 2 diabetes mellitus (T2DM) that AT improves the vascular actions of insulin,19,26 while metformin treatment alone does not enhance insulin-stimulated vasodilation, despite lowered HbA1c, and body mass. 27 These findings stress the importance of regular exercise for the improvement of vascular insulin sensitivity and suggest that a reduction in HbA1c through intensive insulin therapy (IIT) alone may not be sufficient to improve the vasoreactivity to insulin.
The purpose of this study was to (1) determine which modality of exercise (AT or RT), when paired with CIT, results in improved vasomotor function and particularly insulin-induced vasorelaxation; (2) examine whether long-term AT or RT, when paired with CIT, results in superior vasomotor function compared to IIT alone, the standard treatment regime for T1DM; and (3) establish possible vessel-specific adaptations to exercise between either central (aorta) or peripheral (femoral) macrovasculature. We hypothesized that both AT and RT (paired with CIT) would result in improved vasomotor function compared to IIT alone; however, the magnitude of improvement would be greater in AT. Second, enhanced insulin-stimulated vasorelaxation would be larger in the femoral artery due to its closer proximity to the skeletal muscles engaged during the exercise and our past work showing greater rate of relaxation and eNOS content in the femoral artery compared to more proximal arteries.14–16
Methods
All procedures complied with the Canadian Council on Animal Care and were approved by the Research Ethics Board of the University of Western Ontario. Male Sprague-Dawley rats (8 weeks old) were obtained from Charles River Laboratories (Saint-Constant, Quebec, Canada). Rats were housed in pairs, at a constant humidity (50%) and temperature (21.5°C), and were provided water and standard rat chow ad libitum.
Experimental protocol
Rats were randomly divided into IIT (n = 10), conventional insulin treatment (CIT; n = 10), high intensity aerobic exercise training with conventional insulin treatment (AT; n = 8) and resistance exercise training with conventional insulin treatment (RT; n = 8). During week 1, a diabetic state was induced in all rats with multiple low doses of streptozotocin (Sigma–Aldrich; STZ; 20 mg/kg; citrate buffer 0.1 M, pH 4.5) over five consecutive days and was confirmed after two consecutive non-fasting blood glucose concentrations greater than 18 mM. This STZ protocol has been reported to result in greater immune-mediated destruction of pancreatic β-cells, which is pathologically similar to the human T1DM condition than the traditional single dose of STZ.28,29 A single rat in RT did not reach the desired blood glucose concentration and was excluded from the study. Beginning during week 2 and for the duration of the study, insulin was provided to rats via subcutaneous insulin pellets (Linshin, Toronto, Canada). Through daily blood glucose monitoring and insulin pellet adjustments, non-fasting blood glucose was maintained between 4 and 9 mM in IIT and between 9 and 15 mM in CIT, AT, and RT. From week 3 to 14 of the experimental protocol, IIT and CIT remained sedentary, while AT and RT underwent their designated 12-week exercise training programmes.
Exercise protocol
AT rats exercised on a motorized treadmill at 27 m/min at a 6% grade (1 h, 5 days/week) and RT rats climbed a vertical ladder as previously described (~30 min, 5 days/week).22,30 Briefly, rats were familiarized to the ladder during experimental week 2 by climbing the ladder with incremental weights secured to the proximal portion of their tail (5%, 15%, 20% and 35% of body mass). Regular RT sessions (week 3–14) consisted of climbing the vertical ladder at 50%, 75%, 90% and 100% of maximal carrying capacity. RT continued at 100% of maximal carrying capacity until rats reached exhaustion (~8 to 12 climbs). Every fourth training day maximal carrying capacity was re-evaluated. Maximal carrying capacity was established by having rats climb the ladder (beginning at 75% of their body mass) with incremental increases in weight (30 g) until rats reached exhaustion. Exhaustion was defined as failure to climb despite tactile stimulation to the haunches of the rat. While attempts were made to match the duration of AT and RT exercise bouts, this was deemed unmanageable. The exercise regimes were based on Diabetes Canada exercise guidelines, which recommend that individuals with T1DM engage in aerobic exercise at moderate-to-vigorous intensity, with no progression of the intensity. 31 In contrast, for RT, it is suggested that subjects progressively increase intensity.
Tissue collection
Three days after the last exercise bout, rats were anaesthetized with isoflurane and euthanized via heart excision. Aorta and femoral arteries were carefully dissected and placed in cold Krebs–Henseleit buffer (118.1 mM NaCl, 4.7 mM KCl, 1.5 mM CaCl2, 1.2 mM KH2PO4, 1.2 mM MgSO4, 11.1 mM glucose and 25 mM NaHCO3, pH 7.4). Vessels were cleaned of any connective or adipose tissue and were partitioned into 2 mm rings for in vitro isometric tension analysis. The remaining portions of the arteries were flash frozen in liquid nitrogen for protein quantification.
Blood analysis
Blood glucose concentrations were measured and recorded weekly using a One Touch Ultra 2 Blood Glucose Monitoring System (Lifescan Canada Ltd, Burnaby, BC, Canada) and One Touch test strips (Lifescan Canada Ltd). Serum was collected at the completion of the study prior to dissection via heart puncture; ET-1 (ADI-900-020A; Enzo Life Sciences, Farmingdale, NY, USA) and insulin concentrations (Catalogue no. 80-INSHU-E01.1; ALPCO, Salem, NH, USA) were determined via enzyme-linked immunosorbent assay (ELISA).
Isometric vascular myography
Vessel rings were carefully mounted on wire brackets connected to an isometric force transducer (GlobalTown; Microtech, Sarasota, FL, USA) and were submerged in 5 mL of Krebs–Henseleit buffer at 37°C. The relaxed external diameter of the aorta and femoral arteries prior to mounting were ~1.8 and ~0.55 mm, respectively. Vessel rings were washed every 15 min and gradually equilibrated over 1 h to a baseline tension of ~2 g (aorta) or ~1 g (femoral), based on previous work (and pilot testing) from our laboratory.14,15 Arterial rings were pre-contracted with phenylephrine (PE; 3e−7 M) and following a steady-state level of contraction, endothelial-dependent [concentration-response curve for acetylcholine (ACh); half-log concentrations; 1e−8.5 to 1e−5 M] and independent [concentration response curve for sodium nitroprusside (SNP); whole-log concentrations; 1e−11 to 1e−6 M] vasorelaxation were examined. Also, insulin-stimulated vasorelaxation was assessed (concentration response curve for insulin; whole-log concentrations; 1e0 to 1e5 uIU/mL). To evaluate the contribution of eNOS on endothelial-dependent dilators, arterial rings were also pre-treated with NOS inhibitor NG-Monomethyl-L-arginine (L-NMMA; 1e−5 M) for 10 min, 14 pre-contracted with PE (3e−7 M) and following a basal steady-state level of contraction, were treated with either ACh (1e−6.5 M) or insulin (1e 3 uIU/mL). L-NMMA experiments were conducted in the same arterial rings as ACh and insulin experiments to verify that the endothelium was fully functional. As such, all concentration–relaxation curves were conducted in the same arterial rings. Data were excluded if the response of three consecutive concentrations were ±3 standard deviations from the mean (n = 1 IIT for insulin-induced vasorelaxation experiment in the aorta). One AT rat was also not included after being unresponsive after dissection and isolation.
Western blotting
Since differences in insulin-stimulated vasorelaxation were only evident in the femoral artery, molecular analysis was focused to this artery. Triton X-100 femoral artery lysates were used to produce western blot-ready Laemmli samples. Protein samples (10 μg/lane) were separated by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride membranes. Membranes were probed with the following primary antibodies: eNOS (610296; 1:500) and phospho-specific eNOS at Ser1177 (61239; 1:250) from BD Biosciences; Akt (4691; 1:500), phospho-specific Akt at Ser473 (4061; 1:250), p44/42-MAPK (4695; 1:500), phospho-specific p44/42-MAPK at Thr202/Tyr/204 (4370; 1:250) and GAPDH (5174, 1:1000) from Cell Signalling; and ET-1 (117757; 1:2000) from Abcam. Intensity of individual protein bands was quantified using FluoroChem HD2 (AlphaView, version 3.4.0.0) and expressed as a ratio to control band of GAPDH. Levels of GAPDH were also quantified and were determined to be statistically similar across experimental groups (see Supplementary Data).
Statistical analysis
Weekly body mass, blood glucose concentrations and concentration–response curves were compared with two-way, repeated-measures analysis of variance (ANOVA). All other data were compared using a one-way ANOVA. When a significant difference was detected, a least-squares difference post hoc test was performed with a significance level set at p < 0.05. All data are presented as a mean ± standard error, and all statistical analysis was completed using GraphPad Prism 6.
Results
Animal descriptives
An interaction was detected between experimental week and body mass in that IIT were significantly heavier than AT after week 9 and heavier than RT after week 10 (p < 0.05; Figure 1(a)). Furthermore, CIT were significantly heavier than AT at weeks 13 and 14, as well as RT at week 14 (p < 0.05). An interaction was also detected between experimental week and non-fasted blood glucose concentrations in that IIT had lower blood glucose concentrations than CIT at every week besides 3, 5 and 10, while IIT exhibited lower blood glucose concentrations than AT at weeks 4 and 14 (p < 0.05; Figure 1(b)). Furthermore, blood glucose concentrations in CIT were elevated in comparison with RT at weeks 4, 5 and 12, as well as AT at weeks 5, 6 and 9 (p < 0.05). Serum insulin concentrations were lower in AT and RT compared to IIT (p < 0.05; Table 1), while no differences in serum ET-1 were evident between groups (p > 0.05; Table 1).
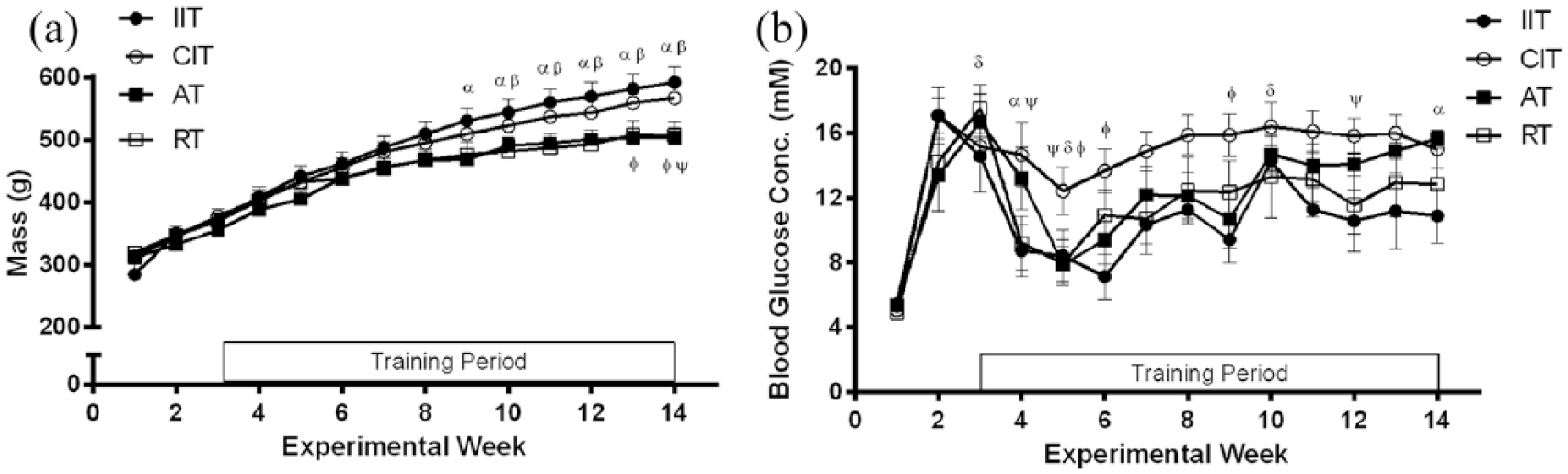
(a) Weekly body mass and (b) non-fasting blood glucose concentrations, (α); IIT different from AT (β); IIT different from RT (Φ); AT different from CIT (ψ); and RT different from CIT; p < 0.05. All data are represented as mean ± standard error.
Blood analysis.

CIT: conventional insulin therapy; IIT: intensive insulin therapy; AT: aerobic training; RT: resistance training.
All data are represented as mean ± standard error.
Different from IIT (p < 0.05).
Vascular myography
In the aorta, no differences in ACh-induced vasorelaxation were evident between groups (p > 0.05; Figure 2(a)–(c)). In the femoral artery, repeated-measures ANOVA did not detect a difference in concentration–response curves (p > 0.05; Figure 2(a)) and no differences were observed in maximal relaxation to ACh (p > 0.05; Figure 2(f)). However, EC50 values were significantly higher in IIT and AT compared to CIT and RT (p < 0.05; Figure 2(e)). No differences were found between experimental groups with regard to insulin-induced vasorelaxation in the aorta (p > 0.05; Figure 3(a)–(c)). Conversely, in the femoral artery, AT demonstrated greater vasorelaxation to insulin at every concentration during the concentration–response experiment (p < 0.05; Figure 3(d) and (e)) and showed the greatest vasorelaxation to insulin (p < 0.05; Figure 3(f)). In the aorta, SNP-induced vasorelaxation was not different between groups (p > 0.05; Figure 4(a)–(c)). In the femoral artery, repeated-measures ANOVA did not detect a difference in vasorelaxation across SNP concentrations (p > 0.05; Figure 4(a)) and no differences were observed in maximal relaxation to SNP (p > 0.05; 4 F). However, EC50 were higher in AT compared to CIT and IIT (p < 0.05; Figure 4(e)) and higher in RT compared to CIT (p < 0.05). When vessels were pre-treated with the NO inhibitor (L-NMMA), the dilatory response to ACh and insulin was attenuated (i.e. maximal vasorelaxation was ~5% for both; p > 0.05), and no group differences were observed (data not shown).
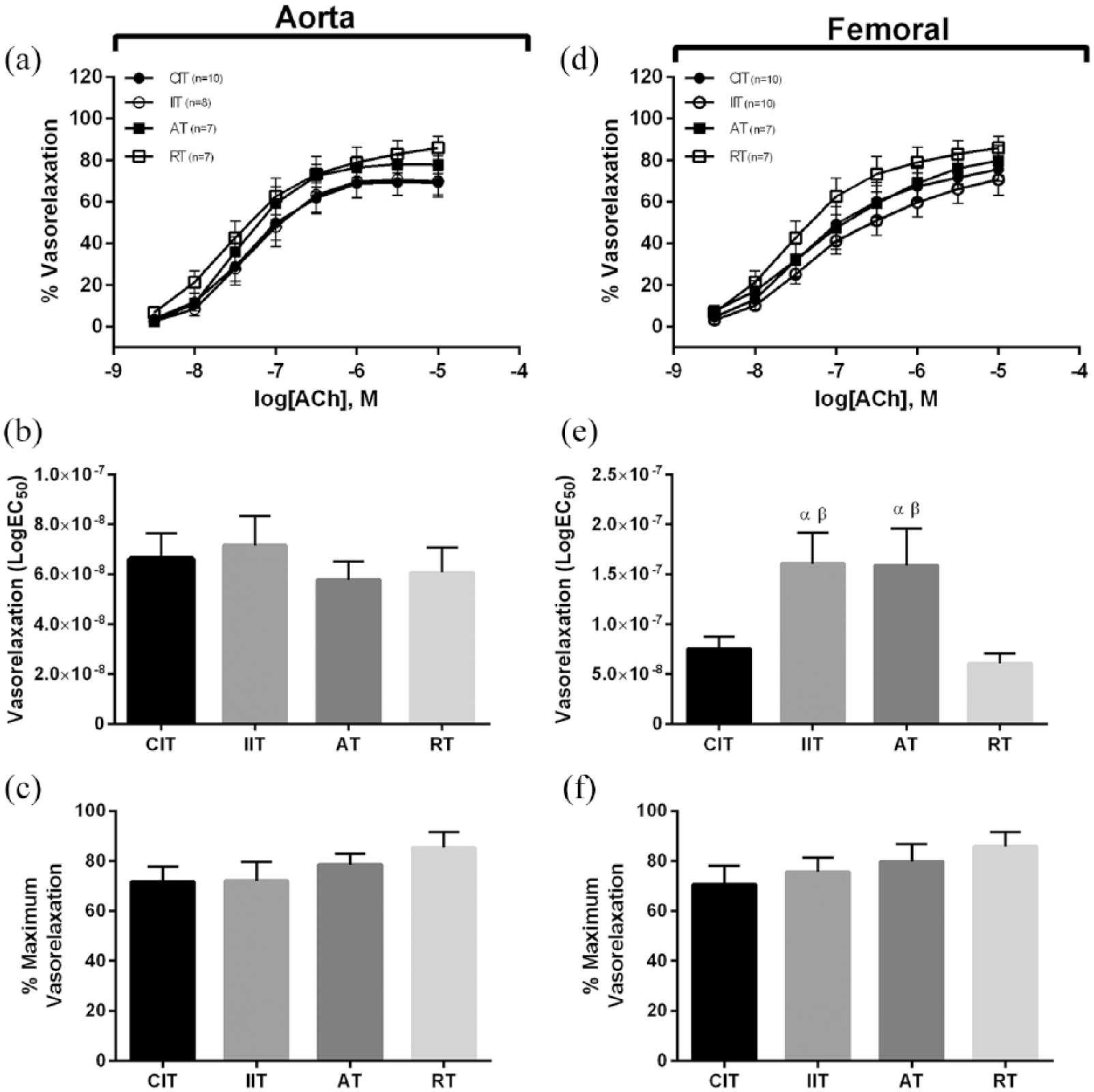
Acetylcholine-induced vasorelaxation. Aorta: (a) concentration response curves to acetylcholine, (b) EC50 for acetylcholine, (c) maximum vasorelaxation to acetylcholine; femoral: (d) concentration response curves to acetylcholine, (e) EC50 for acetylcholine, (f) maximum vasorelaxation to acetylcholine. Different from CIT (α); different from RT (β); (p < 0.05). Data are represented as mean ± standard error.
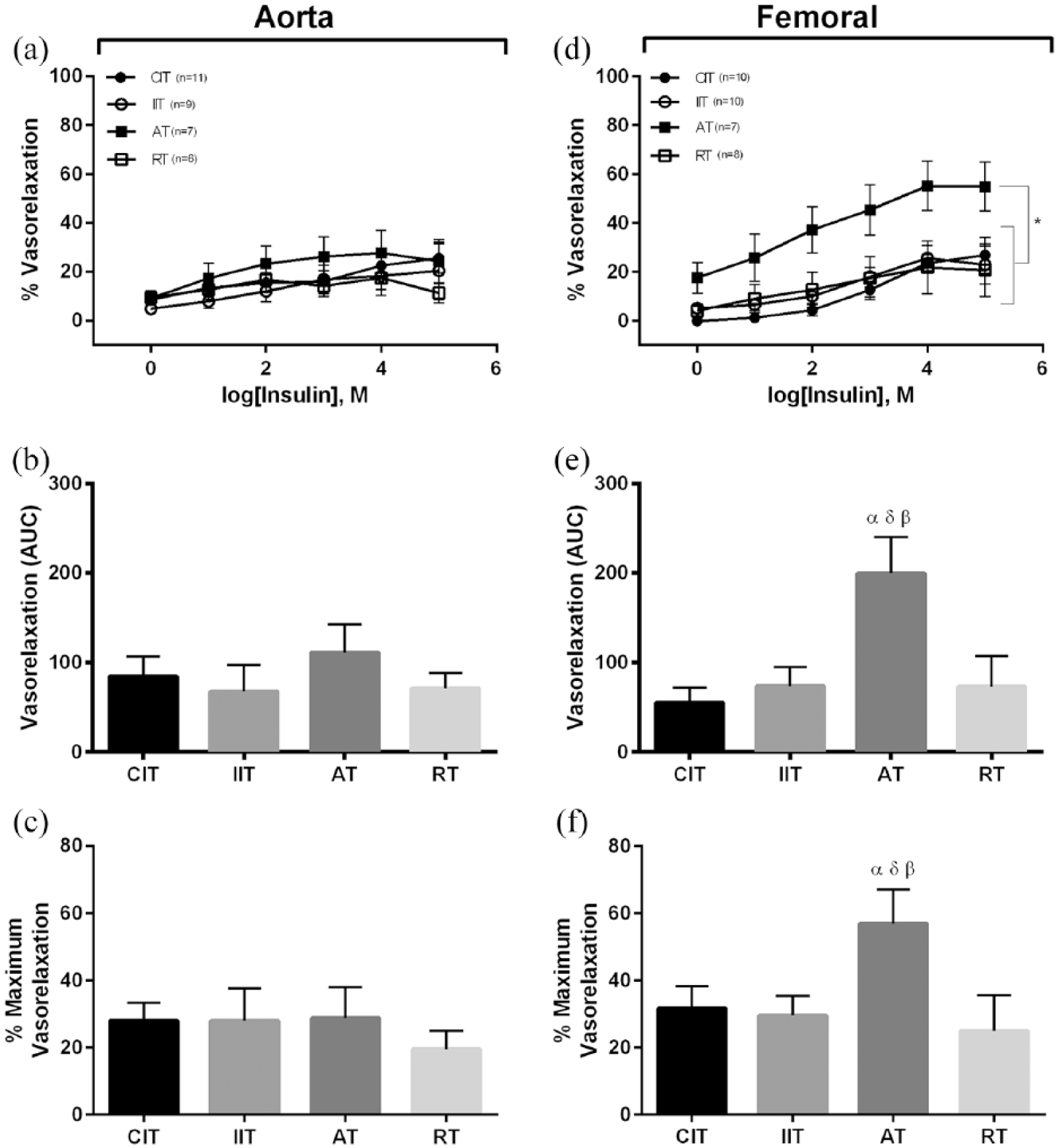
Insulin-induced vasorelaxation. Aorta: (a) concentration response curves to insulin, (b) area under the curve for insulin, (c) maximum vasorelaxation to insulin; femoral: (d) concentration response curves to insulin, (e) area under the curve for insulin, (f) maximum vasorelaxation to insulin. Different from CIT (α); different from IIT (δ); different from RT (β); *different from CIT, IIT and RT; (p < 0.05). Data are represented as mean ± standard error.
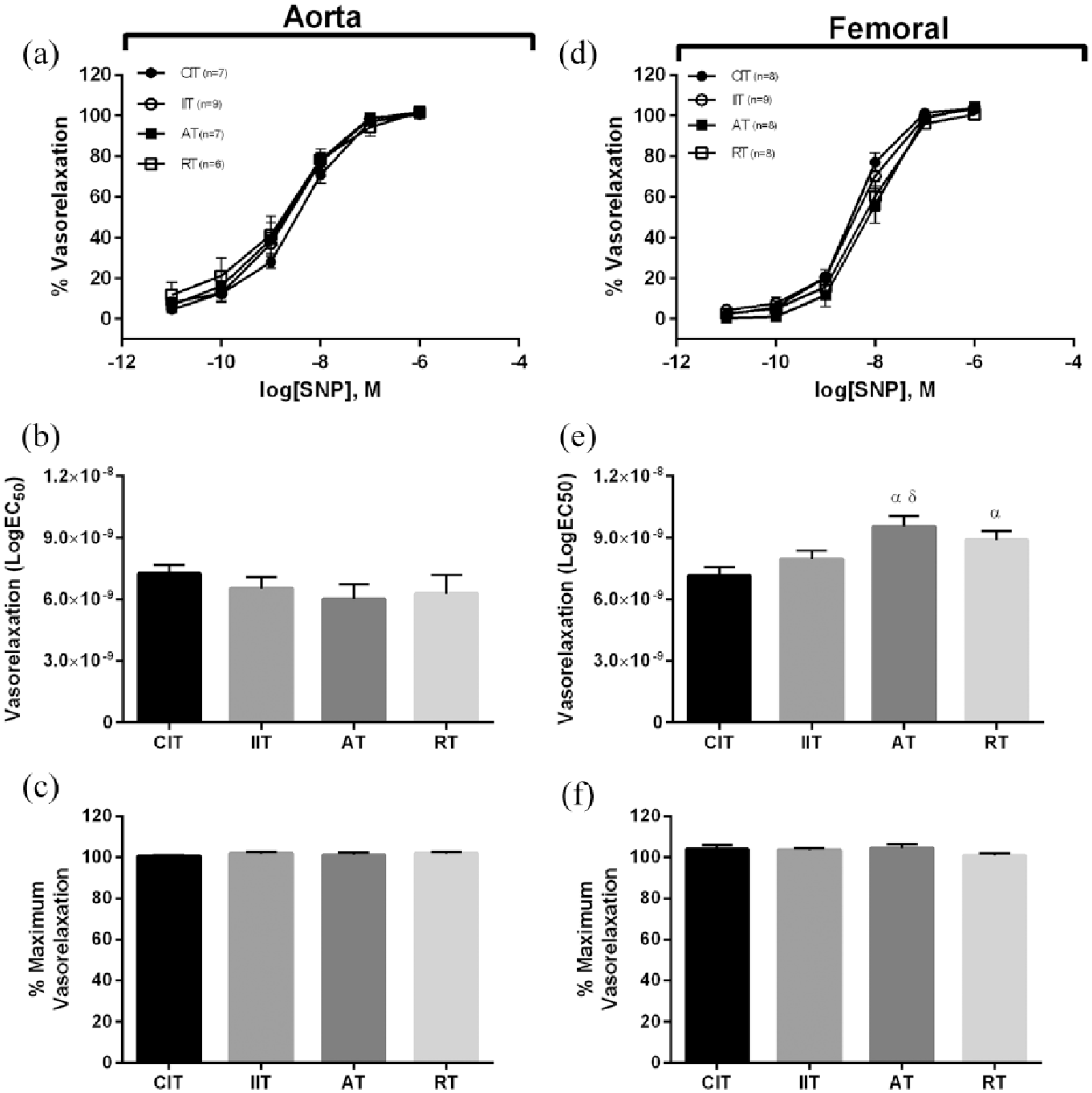
Sodium nitroprusside (SNP)-induced vasorelaxation. Aorta: (a) concentration response curves to SNP, (b) EC50 for SNP, (d) maximum vasorelaxation to SNP; femoral: (d) concentration response curves to SNP, (e) EC50 for SNP, (f) maximum vasorelaxation to SNP. Different from CIT (α); different from IIT (δ); (p < 0.05). Data are represented as mean ± standard error.
Western blotting
Molecular analysis was conducted in the femoral artery where differences in insulin-stimulated vasorelaxation were noted (Figure 3). In the femoral artery, corresponding with increased insulin-induced vasorelaxation, the ratio of basal p(Ser473)-Akt to total Akt protein was elevated in AT compared to all other groups (p < 0.05; Figure 5(a)). No difference was evident between the ratio of basal p(Ser1177)-eNOS to total eNOS protein (p > 0.05; Figure 5(b)). The ratio of basal p(p44/42)-MAPK to total MAPK protein was significantly higher in IIT and AT compared with both CIT and RT (p < 0.05; Figure 5(c)). No differences existed between groups in ET-1 protein content (p > 0.05; Figure 5(d)).
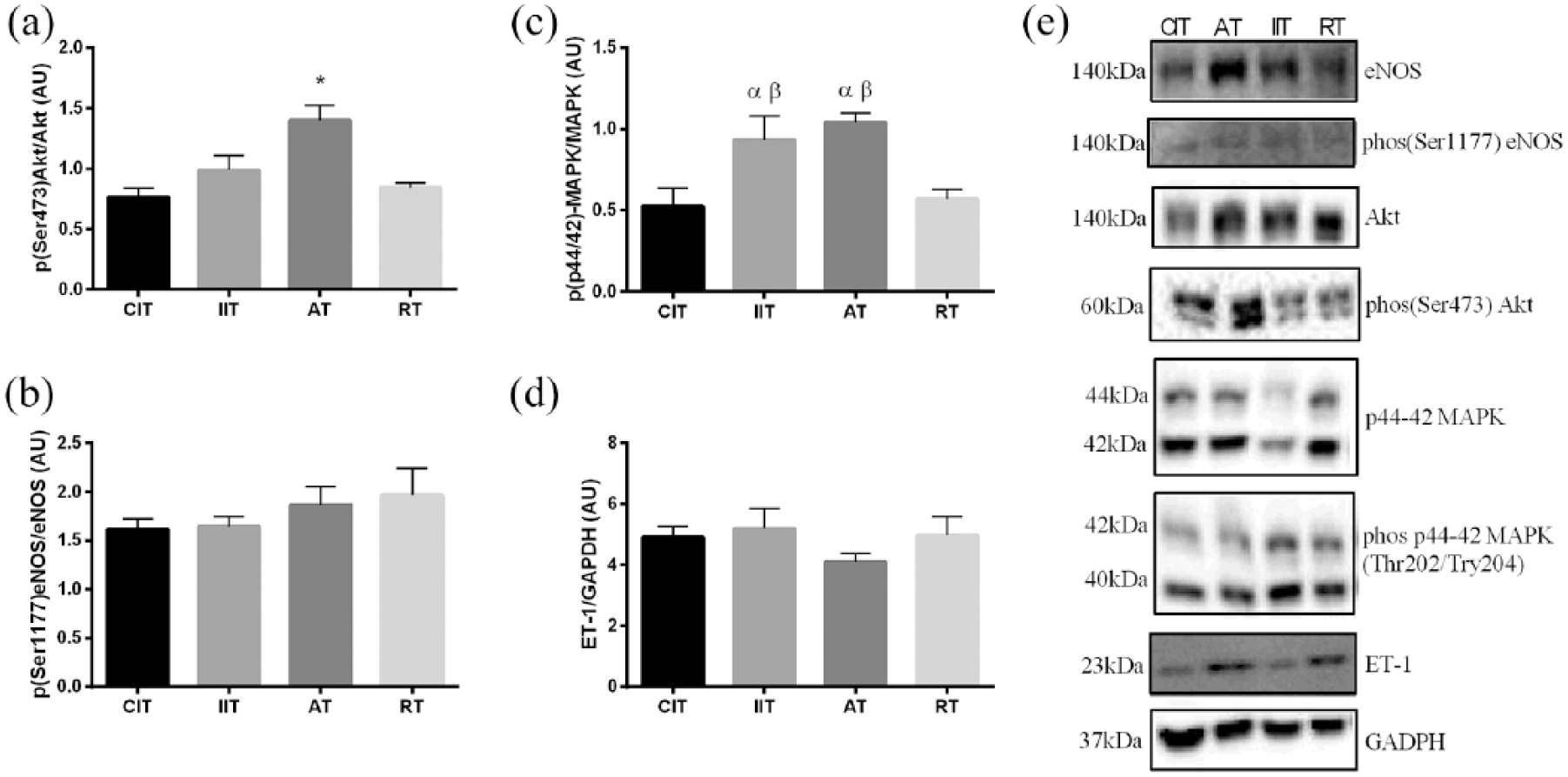
Molecular analysis of the femoral artery: (a) ratio of phosphorylated Akt to total Akt protein, (b) ratio of phosphorylated eNOS to total eNOS protein, (c) ratio of phosphorylated MAPK to total MAPK protein, (d) total ET-1 content and (e) representative images of western blots. Different from D-CIT (α); different from D-RT (β); *different from D-CIT, D-IIT and D-RT; (p < 0.05). Data are represented as mean ± standard error.
Discussion
The primary finding of this study was that long-term AT leads to significant improvements in insulin-induced vasorelaxation in the femoral artery. Furthermore, our findings suggest that this adaptation in vascular insulin sensitivity is exercise-mode and vessel-specific, since the beneficial vascular adaptations to insulin were not evident in the RT group or in the aorta of the AT group. Improvements in insulin-induced vasoreactivity in AT were also greater than IIT, suggesting that this form of treatment was a more therapeutically efficacious means to improve vascular insulin sensitivity than IIT alone. Given that AT was maintained at significantly higher levels of blood glucose concentrations than IIT, these data suggest that glycaemia is not the primary determinant of vasomotor function in peripheral arteries. Rather, this study emphasizes the importance of AT for the improvement of vascular insulin sensitivity in the femoral artery in T1DM, which may be an important factor in mitigating T1DM-related vascular disease.
Insulin treatment strategy
The T1DM model used in the current investigation has demonstrated endothelial dysfunction in both resistance and conduit arteries.14,18 Despite the maintenance of different blood glucose concentrations between CIT and IIT, no differences were evident in the aorta to either acetylcholine- or insulin-induced vasorelaxation. These comparable levels of acetylcholine-stimulated vasorelaxation are likely a result of the similar levels of insulin supplementation between groups, since short-term insulin treatment in T1DM rats has been shown to attenuate endothelial dysfunction in the aorta by inducing the expression of eNOS. 32 Indeed, eNOS expression and activation between these groups appeared similar, and others have reported similar acetylcholine-induced vasorelaxation in the aorta of STZ-T1DM rats following different insulin regimes and dramatically different blood glucose concentrations. 33 Similar insulin-stimulated vasorelaxation between CIT and IIT in both the aorta and the femoral was also not surprising, given that metformin treatment in T2DM rats fails to enhance insulin-stimulated vasodilation in skeletal muscle arterioles despite lowered HbA1c. 27 However, the finding that basal femoral artery Akt signalling appeared similar between CIT and IIT, yet MAPK signalling increased in the IIT group suggests that the IIT strategy may be detrimental and causes the beginning stages of vascular insulin resistance. Indeed, whole-body insulin resistance has been shown to exist following IIT in humans and rats,34,35 and extensive evidence from cell culture, animal and human studies has shown that increased MAPK signalling is associated with a pro-atherogenic endothelial cell phenotype.9,36–40 Additional work is warranted to discern whether the increase in the MAPK signalling in IIT suggests that this form of insulin therapy may actually be detrimental to the vasculature. 34
Aerobic versus resistance exercise
Given that the benefits of AT and RT on endothelium-dependent vasodilation in T1DM have been established both clinically 41 and experimentally,2,41–43 the inability of both exercise regimes to significantly improve acetylcholine-induced vasorelaxation in this study was unexpected. However, these discrepancies can partially be explained by the insulin supplementation provided in this study. We have previously reported that both AT and RT exercise regimes increase whole-body insulin sensitivity in T1DM rats, resulting in a reduction in the concentration of insulin required to maintain a similar blood glucose concentration range.22,30 Given that insulin treatment alone can restore endothelial-dependent vasorelaxation and increase eNOS expression in T1DM rats,3,32,33 it is conceivable that some of the benefits of exercise training on acetylcholine-induced vasorelaxation may have been masked by the fact that sedentary rats (IIT and CIT) required more insulin (through implanted insulin pellets) to maintain desired blood glucose ranges.
The ability of regular exercise to enhance the vascular actions of insulin within resistance and conduit arteries in different populations has recently been reviewed. 13 To date, few studies have determined the effects of exercise training on the vasodilatory actions of insulin in T1DM or the spatial distribution of these adaptations.18,44 Using this insulin-treated model of T1DM, our group has shown that 10 weeks of AT can alleviate diabetes-related impairments in insulin-stimulated vasa nervorum dilation, which also coincides with improved motor nerve conduction velocity. 18 Furthermore, others have shown an increase in cutaneous blood flow in response to local microinjections of insulin in exercise-trained STZ-T1DM rats. 44 To our knowledge, this is the first study to demonstrate that AT can increase vascular insulin sensitivity in a conduit artery such as the femoral artery. Furthermore, AT was superior to IIT in terms of improving vascular insulin sensitivity in the femoral artery, indicated by improved insulin-induced vasorelaxation coupled with increased Akt signalling.
In contrast to AT, RT failed to demonstrate an improvement in insulin-induced vasorelaxation in the femoral artery. This finding does not support previous evidence indicating an increase in insulin-induced vasodilation in mesenteric arteries of healthy rats immediately following a single bout of RT. 45 These discrepancies may be explained by the fact that arteries in the present investigation were not harvested until 3 days following the last exercise bout to mitigate the acute benefits of exercise and focus on chronic adaptation to regular exercise. Furthermore, it has been hypothesized that increases in vascular insulin sensitivity following exercise training occur as a result of shear stress caused by exercise-induced hyperaemia. 13 While blood flow was not directly measured during these exercise regimes, a bout of AT was generally of longer duration than RT. It is plausible that the increase in the sensitivity to insulin in the femoral artery following AT compared to RT occurred due to the longer duration of AT. Indeed, while both AT and RT rely on hindlimb skeletal muscles supplied by the femoral artery to perform the exercise regime, the longer duration of an AT bout would result in a greater shear stress stimulus.
Vessel-specific increase in insulin sensitivity
A novel finding of this study was that the aerobic exercise-induced improvement in vasoreactivity to insulin was vessel-specific, that is, enhanced vascular insulin sensitivity was specific to the artery closest to the skeletal muscle engaged in the exercise (i.e. the femoral artery). In line with this interpretation, Dela et al. 20 reported that following one-legged ergometer bicycle training, insulin-induced increases in leg blood flow were larger in the trained leg compared with the untrained leg during an insulin clamp. More recently, similar findings were obtained following one bout of single-leg knee extension exercise. 46 Together, these findings suggest that aerobic exercise improves insulin sensitivity in the vasculature closest to the active skeletal muscle, presumably a result of its greater shear stress exposure during running.
Conclusion
T1DM rats treated with long-term AT demonstrate enhanced insulin-induced vasorelaxation in the femoral artery, likely a result of increased Akt signalling. Findings presented here suggest that the inclusion of AT into the treatment of T1DM, despite being accompanied by less reliance on glycaemic control, can provide greater improvements in vascular insulin sensitivity than IIT alone. Long-term clinical studies are warranted to investigate the importance of AT accompanied with a more CIT (i.e. 9–15 mM) in mitigating T1DM-related vascular insulin resistance. Future work should investigate the potential benefits of AT when paired with more CIT in other vascular beds. Moreover, clinical evidence is needed to determine whether the vascular benefits associated with regular aerobic exercise are greater than IIT alone, the standard treatment regime for T1DM.
Supplemental Material
dvdres-jan-2018-00002-File007 – Supplemental material for Aerobic exercise training improves insulin-induced vasorelaxation in a vessel-specific manner in rats with insulin-treated experimental diabetes
Supplemental material, dvdres-jan-2018-00002-File007 for Aerobic exercise training improves insulin-induced vasorelaxation in a vessel-specific manner in rats with insulin-treated experimental diabetes by Matthew W McDonald, T Dylan Olver, Michelle S Dotzert, Thomas J Jurrissen, Earl G Noble, Jaume Padilla and CW James Melling in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors would like to thank Michael Murray and Zach Nickels for their assistance with animal care and experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: M.W.M. and M.S.D. were supported by an Ontario Government Scholarship; C.W.J.M. was supported by a Natural Sciences and Engineering Council Discovery Grant (RGPGP-2015-00059); T.D.O. was supported by American Heart Association postdoctoral fellowship grant; and J.P. was supported by National Institutes of Health (K01 HL-125503 and R01 HL-137769).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
