Abstract
Objective
To investigate peripheral blood lymphocyte subpopulations, particularly helper T (Th) cells and cytokine expression before and after treatment in polymyositis (PM) and dermatomyositis (DM).
Patients and Methods
Ten patients with PM and 15 with DM were enrolled in this study. As a control we used 11 age-matched healthy subjects. We analyzed peripheral blood lymphocytes using flow cytometry. Serum cytokines were determined by enzyme-linked immunosorbent assay.
Results
Th2/Th1 and Th2/Th17 ratios were significantly higher in both PM and DM than in controls and this Th2-predominancy is marked in DM associated with interstitial pneumonia. IL-1β, IL-6 and TGF-β were significantly higher in both PM and DM and IL-4 showed a significant increase in DM when compared to controls. There was no significant correlation between either any lymphocyte subpopulations or serum cytokines and clinical severity markers such as the manual muscle strength test, serum creatine kinase, and the total CT score. Th2/Th17 in both PM and DM, as well as Th2/Th1 in the latter, significantly decreased after clinical remission compared with before treatment.
Conclusions
Th2-predominancy as shown by the increase in Th2/Th1 and Th2/Th17 ratios may suggest active disease in PM/DM but does not reflect clinical severity.
Introduction
Polymyositis (PM) and dermatomyositis (DM) are autoimmune inflammatory systemic disorders characterized clinically by involvement of skeletal muscles, sometimes with fatal interstitial pneumonia (IP).1,2 Autoimmune abnormalities are considered to underlie PM/DM, and of these abnormalities, lymphocytes play a central role in the pathogenesis. 3 CD8+ T cells directly attack muscle fibers expressing major histocompatibility complex class I antigens in PM, while in DM CD4+ T cells and B cells infiltrate around endomysial capillaries. This infiltration leads to irreversible damage to muscles via production of humoral factors such as antibodies and cytokines. 4 In IP complicating PM/DM, CD8+ T cells play an important role in the pathogenesis and in unresponsiveness to corticosteroid.5–8
CD4+ cells mainly act as helper T cells and are classified into type 1 (Th1) and type 2 (Th2) according to the cytokines produced: interferon (IFN)-γ in Th1 and interleukin (IL)-4 in Th2. The Th1/Th2 balance shows a difference between PM and DM but reflects the disease activity in both disorders.9–11 Recently, Th type 17 (Th17) has appeared as a novel subpopulation of T cells characterized by production of IL-17. Th17 has been reported to contribute to the pathogenesis of autoimmune disorders such as rheumatoid arthritis and multiple sclerosis.12,13 Furthermore, pathological studies have demonstrated infiltration of IL-17-producing cells in muscles in PM/DM.14,15 The precise mechanisms by which naïve T cells differentiate into Th17 cells remain unclear, but some cytokines such as tumor growth factor-β (TGF-β), IL-1β, IL-6 and IL-21 are probably relevant to this process.16–18 TGF-β and IL-6 are important also as humoral factors affecting the pathogenesis of PM/DM.19,20
In the present study we performed flow cytometry with intracytoplasmic cytokine staining in order to investigate subpopulations of Th cells, particularly Th2 and Th17, in peripheral blood of PM/DM before and after treatment. Serum levels of IL-1β, IL-4, IL-6, IL-21, IL-23 and TGF-β were also determined by an enzyme-linked immunosorbent assay (ELISA). Significantly higher levels of Th2/Th1 and Th2/Th17 ratios than controls decreased after treatment in both PM and DM, and we postulate that balance of Th subpopulations may be important as a marker of the disease activity in these disorders.
Patients and Methods
Patients
We studied 10 patients with PM (2 men and 8 women; mean age 51.8 ± 20.2 years, range 17 to 81) and 15 with DM (6 men and 9 women; mean age 50.1 ± 14.5 years, range 24 to 75) who had been admitted to our hospital from January 2008 to August 2010. They were diagnosed as having PM/DM according to the classification criteria proposed by Bohan and Peter. 21 None of them presented with any abnormal findings suggestive of malignancy and none had received any immunomodulatory treatment prior to admission to our hospital. Based on clinical symptoms such as muscle weakness, skin eruptions, and increases in serum creatine kinase (CK), PM/DM was considered to be active in all of the patients. Eight of the DM patients met the clinical criteria of clinically amyopathic dermatomyositis (CADM) because of the presence of skin symptoms, including Gottron's sign and heliotrope rash, with no or little muscle involvement. 22 Serum rheumatoid factor was not detectable in any patients. All of the patients received prednisolone therapy with or without immunosuppressive agents (eg, cyclosporine A and tacrolimus) and reached clinical remission, which was defined as a state showing no or little musculocutaneous symptoms with normal CK in serum. As a control we used 11 age-matched healthy subjects (8 men and 3 women; mean age 51.0 ± 10.4 years, range 36 to 65), although the male-female ratio was different from that of the patients. The Local Ethical Committee approved this study and we obtained informed consent from all of the patients and controls.
Clinical Assessment of the patients
As clinical markers of disease severity we primarily used 3 indices. The first was muscle power. Manual muscle strength test (MMT) was performed in both sides of the deltoid, biceps, triceps, iliopsoas, quadriceps, and hamstrings before and after treatment. Each muscle power was semi-quantitatively graded using the rating scale from 0 to 5 (0: no contraction, 1: visible contraction but immovable, 2: movable without gravity but immovable against it, 3: movable against gravity, 4: slight decrease, 5: normal), and the sum was defined as the MMT score. The second clinical marker was laboratory data, particularly CK and C-reactive protein (CRP) in serum.
We obtained these data from the medical records of the patients.
The third clinical marker of disease severity was the CT score of associated IP. To assess pulmonary involvement in all of the patients before and after treatment, high-resolution computed tomography (HRCT) was serially performed using LightSpeed VCT VISION (GE Healthcare Japan; Hino, Japan), with 2-mm collimation at 15-mm intervals at end inspiration from the lung apices to the bases. All images were reconstructed with a high-spatial frequency algorithm and photographed at window widths of –1500 H.U. and window levels of –600 H.U. We selected and analyzed 6 transverse CT images taken at the levels of the upper and lower portions each of the aortic arch, the hilus and the lower lobe. Two raters (YS and MM) blinded to the clinical information evaluated the HRCT images, focusing on the following 6 findings: (1) nodular opacities, (2) bronchial dilatation and wall thickening, including consolidation around bronchovascular bundles, (3) pleural thickening, (4) reticular opacities, including honey-comb changes, (5) linear opacities and (6) increased lung opacities. To determine the distribution of IP changes, we used the visual estimation method described by Kinoshita et al. 23 Each CT slice level was divided into 4 areas according to a horizontal and a vertical line drawn by a rater, and scored in a semi-quantitative manner as follows: 0 = absent; 1 = less than 25% involved; 2 = 25%–50% involved; 3 = 50%–75% involved; and 4 = greater than 75% involved. The final judgment was reached by consensus of the 2 raters. Right and left lungs were evaluated separately, and the sum was defined as the total CT score.
Flow Cytometry
Heparinized whole blood taken both from patients before and after treatment and from the controls was separated into mononuclear cells by the Ficoll-Hypaque gradient method. After being washed twice with cold FACS flow buffer (Becton Dickinson, San Diego, CA, USA), cells were divided into those for detecting surface markers and those for intracytoplasmic staining. To detect surface markers of lymphocytes, cells were re-suspended at 5 × 10 6 /mL and an aliquot of 200 μL was put into a 10 mL tube; 20 μL of each appropriate monoclonal antibody (mAb) was then added to these tubes, incubated at 4 °C in the dark for 30 minutes and washed twice with cold FACS flow buffer.
For intracelluar staining of cytokines, cells were incubated at 37 °C for 4 hours in 5 mL of RPMI 1640 (Sigma, St. Louis, MO, USA) containing 5% fetal bovine serum (Gibco, Grand Island, NY, USA), 2 mM glutamine (Gibco), 2 μM monensin (GolgiStop, Becton Dickinson), 40 ng/mL of phorbol 12-myristate 13-acetate (PMA, Sigma) and 500 ng/mL of ionomycin (Sigma). After being washed twice with cold FACS flow buffer, the cells were incubated with 2% human serum at 4 °C for 30 minutes in order to block Fc receptors. After being washed twice with cold FACS flow buffer, the cells were re-suspended at 5 × 10 6 /mL and an aliquot of 200 μL was put into a 10 mL tube. Appropriate mAbs directed against a cell surface antigen were then added to these tubes and incubated at 4 °C in the dark for 30 minutes. The cells were washed twice with cold FACS flow buffer and treated with 250 μL of Cytofix/Cytoperm solution (Becton Dickinson) at 4 °C for 20 minutes. After being washed once with Perm/Wash solution (Becton Dickinson), 20 or 10μL of each appropriate anti-cytokine mAb that had been used for intracytoplasmic staining were then added to these tubes and were incubated at 4 °C in the dark for 30 minutes.
After being washed twice with cold FACS flow buffer, the labeled cells were analyzed by two- or three-color flow cytometry using FACSCalibur (Becton Dickinson). The gate was set on lymphocytes, and 1 × 10 4 cells were analyzed. The results were expressed as the percentage of cells positive for each mAb in whole lymphocytes. The following mAbs were employed in this study: fluorescein isothiocyanate-conjugated mAbs to CD4 (13B8.2), CD8 (B9.11), CD16 (3G8) and CD23 (9P25), phycoerythrin (PE)-conjugated mAbs to CD25 (B1.49.9), CD56 (N901), CD80 (MAB104), IL-4 (4D9), interferon (IFN)-γ (45.15) and IL-17 (41802), phycoerythrin-Texas red-conjugated mAbs to CD45RO (UCHL1) and CD45RA (2H4), and phycoerthrin-cyanin 5.1-conjugated mAbs to CD3 (UCHT1) and CD19 (J4.119). We purchased PE-conjugated mAb to IL-17 from R&D Systems (Minneapolis, MN, USA) and others from Immunotech (Marseille, France). Each above-mentioned fluorochrome-conjugated mouse IgG1 (Immunotech) was used as negative controls.
Elisa
Serum samples for cytokine analysis were also obtained from the patients and controls. Whole blood was allowed to clot at room temperature for 60 minutes followed by separation by centrifugation. To prevent the degradation of protein, 1000 kallikrein inhibitory units of protease inhibitor (aprotinin, Sigma) was added to each milliliter of serum at the time of the sample collection. Serum samples were subsequently frozen and stored at –80 °C until assay for cytokines. All assays were performed in a blinded fashion on coded sterile samples.
We used commercially available ELISA kits in order to determine the serum concentrations of IL-1β, IL-4, IL-6, IL-21, IL-23 and TGF-β. All of the kits were purchased from R&D Systems (Minneapolis, MN, USA) except that for IL-21 (Abnova, Atlanta, GA, USA). The cytokine present in the sample or the standard was bound to the mAb adsorbed on polystyrene microwells. Horseradish peroxidase (HRP)-conjugated mAb was subsequently added and bound to the cytokine captured by the first antibody. Unbound enzyme-conjugated mAb was removed by washing, and a substrate solution reactive to HRP was added to each well. A colored product was formed in proportion to the amount of the cytokine present in the sample. The reaction was terminated by adding acid, and the absorbance of each well was measured at appropriate wave length using a Multiskan Spectrum spectrophotometer manufactured by Thermo Labsystems (Franklin, MA, USA). A standard curve was prepared from standard dilutions of cytokines in duplicate. The cytokine concentration in each sample was determined from the standard curve. To detect heterophilic antibodies in the samples we used microtiter plates (Flow Laboratories, McLean, VA, USA), pre-coated overnight with 1 μg/mL of mouse IgG1 (eBioscience, San Diego, CA, USA) in 20 mM Tris pH 9.0 and then blocked with NET/NP-40 buffer containing 0.5% bovine serum albumin and 0.05% NaN3 instead of those in the ELISA kits.24,25
Statistical Analysis
To detect significant differences in phenotypes of lymphocytes and serum cytokines among PM, DM, and controls, we used the Mann–Whitney U test. Wilcoxon's signed rank test was employed for comparison between results before treatment and after clinical remission. Relationships between phenotypes of lymphocytes and serum cytokines or clinical indices, such as CK and the total CT score, were investigated using Spearman's correlation coefficient test. The results were expressed as mean ± standard deviation, and a P-value less than 0.05 was regarded as statistically significant. Commercially available statistics software was used for data analysis (StatView for Macintosh, Abacus Concepts, Berkeley, CA, USA).
Results
Phenotypes of Peripheral Blood Lymphocytes(Pbl)
Results of flow cytometry and comparison among PM, DM, and controls are demonstrated in Table 1. Both PM and DM showed significant increases in the percentages of CD4+CD25+ (activated Th, P < 0.05) and CD4+IL-4+ (Th2, P < 0.01) cells, and significant decreases in those of CD3″CD16/CD56+ (natural killer cells, P < 0.01 and P < 0.05, respectively) and CD19+CD80+ (activated B cells, P < 0.005 and P < 0.001, respectively) cells when compared with controls. Both PM and DM showed a decrease in the percentage of CD4+IL-17+ cells (Th17) and statistically significant differences were seen between controls and either PM (P < 0.0005) or DM (P < 0.005). This subpopulation was lower in PM than in DM, although there was no statistical difference. The percentage of CD3+CD4–CD8–IL-17+ cells were quite low and no significant difference was present between controls and either PM or DM. Both PM and DM showed significant increases in Th2/Th1 (P < 0.05 and P < 0.005, respectively), Th1/Th17 (P < 0.005 and P < 0.01, respectively) and Th2/Th17 ratios (P < 0.005) when compared with controls. There was no significant difference in phenotypes of lymphocytes examined in the present study between PM and DM. Figure 1 demonstrates actual values of phenotypes and ratios which showed significant differences between controls and PM or DM. IP was seen in 5 patients with PM and 9 with DM. DM with IP showed significantly higher values in the percentage of Th2 cells (P < 0.01) and the ratios of Th2/Th1 (P < 0.01) and Th2/Th17 (P < 0.05) when compared with DM without IP (Fig. 2). The Th2/Th1 ratio was significantly higher in DM with IP also in comparison with PM with IP (P < 0.05). There was no significant difference in these indices of PM between with and without IP.
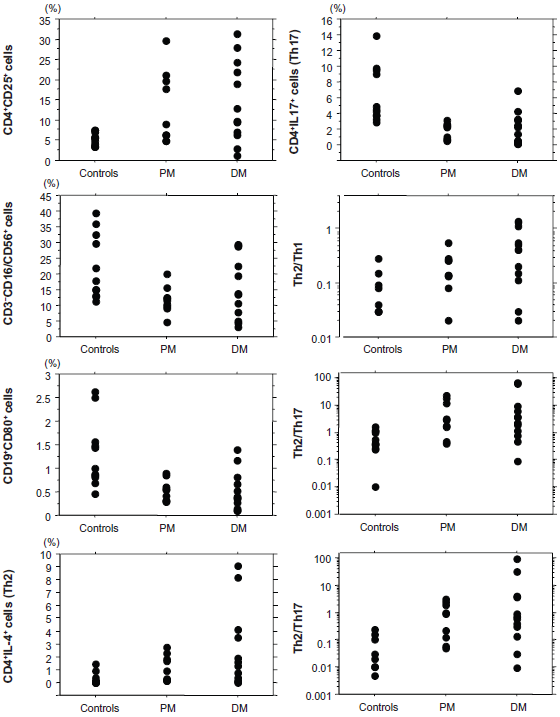
Actual values of phenotypes and ratios which showed significant differences between controls and PM or DM.

Th2/Th1 and Th2/Th17 ratios and the percentage of Th2 cells in PM/DM before treatment.
Results of flow cytometry and comparison among PM, DM and controls.

Comparison of PBL phenotypes in PM and DM between results before treatment and after clinical remission is shown in Figure 3. The percentages of Th17 cells in both PM and DM were significantly higher after clinical remission than before treatment (P < 0.05 and P < 0.01, respectively). In contrast, the percentages of Th2 cells in PM and DM significantly decreased after clinical remission compared with before treatment (P < 0.05 and P < 0.01, respectively). The Th2/Th1 and Th2/Th17 ratios in DM and the latter in PM significantly decreased after clinical remission compared with before treatment (P < 0.01 and P < 0.05, respectively). Other lymphocyte phenotypes showed no significant change in percentage after clinical remission. There was no significant relationship between PBL phenotypes and the clinical markers of disease severity, such as the MMT score, serum CK, and the total CT score (data not shown).

Comparison of PBL phenotypes in PM and DM between before treatment and after clinical remission.
Cytokines in Sera
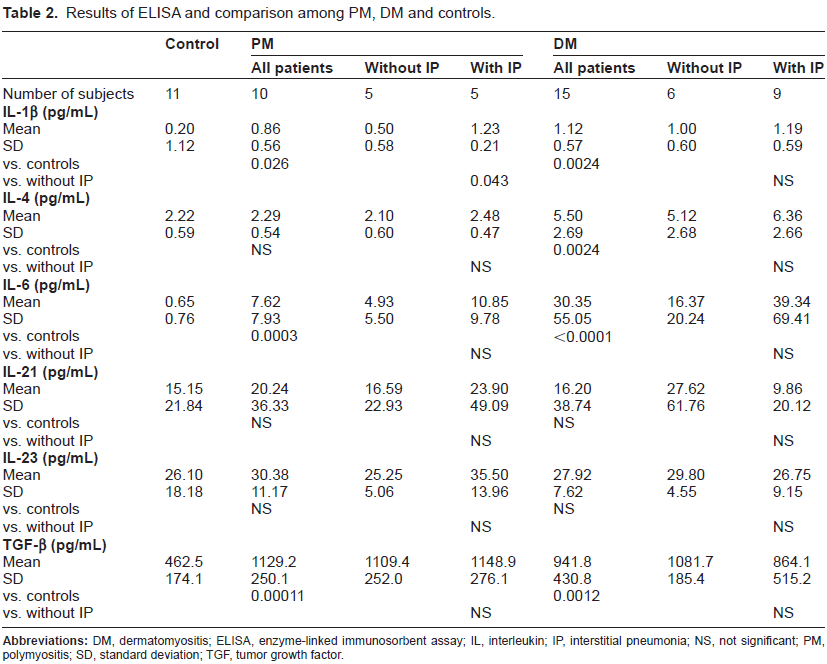
Results of ELISA and comparison among PM, DM, and controls are shown in Table 2. No positive results were detectable in any microtiter plates pre-coated with mouse IgG1. Both PM and DM showed significantly higher values in IL-1β (P < 0.05 and P < 0.005, respectively), IL-6 (P < 0.0005 and P < 0.0001, respectively) and TGF-β (P < 0.0005 and P < 0.005, respectively) than controls. IL-4 was also significantly increased in DM (P < 0.005) when compared with controls but not in PM. IL-21 and IL-23 were slightly higher in both PM and DM than in controls despite the lack of significant differences.
Results of ELISA and comparison among PM, DM and controls.
Comparison of serum cytokines in PM and DM between before treatment and after clinical remission is shown in Figure 4. Serum TGF-β in PM significantly decreased after clinical remission compared with before treatment (P < 0.05). DM showed a tendency to decrease in TGF-β after clinical remission, but no significant difference was seen in comparison with before treatment. IL-6 also decreased after clinical remission in both PM and DM. PM showed no significant difference between results before treatment and after clinical remission, while in DM, IL-6 decreased significantly (P < 0.005). IL-1β, IL-4, IL-21, and IL-23 showed a tendency to decrease after clinical remission in both PM and DM compared with before treatment, but no significant differences were present. No significant correlations were seen between any cytokines and either the clinical markers of disease severity, such as the MMT score, serum CK, and the total CT score, or the phenotypes of PBL, including Th17 cells (data not shown).

Comparison of TGF-β in PM (A) and IL-6 in DM (B) between before treatment and after clinical remission.
Discussion
Recent Reports From Other Institutes Have so Far Demonstrated Alterations of Pbl Phenotypes in the Active Phase of pm and Dm, Such as Decreased numbers of CD4+, CD8+ and CD8+CD28+ cells, decreased percentages of CD3+ and CD8+ cells, and increased percentages of CD4+CD28null cells.26–29 In the present study on surface antigens of PBL the proportion of CD4+CD25+ cells indicating activated Th cells were significantly increased in both PM and DM, when compared with controls, as shown in the previous report. 11 CD4+CD25+ cells may include regulatory T cells, but we do not have data enough to mention Foxp3, which is a marker specific for this subpopulation. PM and DM showed significant decreases in the proportion of CD3–CD16/CD56+ cells indicating natural killer (NK) cells irrespective of associated IP, in contrast to idiopathic pulmonary fibrosis, in which this subpopulation is increased.30,31 As acute IP sometimes precedes muscle and skin symptoms in both PM and DM, an obvious decrease in the proportion of NK cells in PBL of IP patients may suggest the existence of these disorders in background. The present study on intracytoplasmic cytokines demonstrated significant increases in the proportion of Th2 cells and the Th2/Th1 ratio in both PM and DM when compared with controls. The previous reports have also shown an increase in these indices in active phase of DM as well as PM.10,11 Considering that both the proportion of Th2 cells and the Th2/Th1 ratio were higher in DM than in PM despite the absence of a significant difference, Th2-predominancy is probably more marked in the former than in the latter. Associated IP further enhanced Th2-predminancy in DM. These results suggest that the increased Th2/Th1 balance may contribute to the pathogenesis of PM and DM, particularly the latter with IP often resulting in poor life prognosis. The significantly elevated serum level of IL-4 in DM patients also supports this hypothesis.
The most notable finding in the present study is that the proportion of Th17 cells in PBL was significantly decreased in the active phase of both PM and DM, when compared with controls. Together with an increase in the proportion of Th2 cells, the ratios of Th1/Th17 and Th2/Th17 were increased in both disorders before treatment. No report has so far described such findings in either PM or DM, although a recent study demonstrated a significantly higher proportion of Th17 in CXCR5+CD4+ T cells in peripheral blood of juvenile DM. 32 The proportion of Th17 cells significantly increased after clinical remission in both PM and DM, but there were no correlations between PBL phenotypes examined, including this parameter, and the severity indices, such as the MMT score, CK, and the total CT score. In contrast to our results, however, recent pathological studies demonstrated infiltrations of IL-17-producing cells in skeletal muscles of PM/DM.14,15 The precise reason for the discrepancy in IL-17-producing cells between peripheral blood and muscle tissue is unclear, but 2 possible mechanisms may have been relevant to the significantly decreased proportion of Th17 in the former. One is migration of Th17 cells from peripheral blood into muscles in the active phase of PM/DM. Increased production of inflammatory cytokines, including IL-6, may enhance the permeability of endomysial small vessels, 33 and make the Th17 cells easily infiltrate into the muscle tissue. The other possible mechanism is suppressed differentiation into Th17 cells in peripheral blood. Th2-predominancy in DM may have produced negative effects on differentiation into Th17 cells in peripheral blood via cytokines, such as IL-4, although elevated levels of TGF-β and IL-6 in serum (as shown in the present study) can increase these cells. 34 CD3+CD4–CD8– cells designated as double negative T cells have autoimmune reactivity, 35 and among them the IL-17-producing subpopulation (CD3+CD4–CD8–IL-17+ cells) has recently been reported to show an increase in collagen vascular disorders such as systemic lupus erythematosus. 36 In the present study, however, no significant increase in the proportion of CD3+CD4–CD8– IL-17+ cells was seen in the active phase of either PM or DM.
We determined serum levels of IL-1β, IL-6, TGF-β, IL-21, and IL-23, which are all involved in differentiation into Th17 cells.16–18 According to several recent reports, TGF-β is increased in both the skeletal muscle tissue and sera in PM/Dm19,39 and serum IL-6 is correlated with the disease activity of DM. 20 Also in the present study, IL-6 and TGF-β demonstrated significantly higher levels before treatment in both PM and DM when compared to controls. After clinical remission significant decreases were seen in IL-6 in DM and in TGF-β in PM. IL-1β was also higher in both PM and DM before treatment than in controls, but no significant decreases were seen after clinical remission. These results suggest that IL-6 and TGF-β may be usable as a clinical marker indicating the disease activity of DM and PM, respectively, although the general status of the patients at sampling and some factors other than these disorders, such as infection, often affect serum cytokine levels. IL-21 has been reported to contribute to the pathogenesis of some autoimmune disorders with regard to promoting proliferation of T cells, including Th17.18,40 In the present study, however, serum IL-21 showed no significant difference between controls and PM or DM before treatment, and there was no evidence suggesting the involvement of this cytokine in the pathogenesis.
In conclusion, Th2-predominancy with an increase in Th2 and a decrease in Th17 forms a basic immunological background in the active phase of PM and DM. Increased Th2/Th1 and Th2/Th17 ratios reflect the disease activity of PM and DM but not the severity, and may be useful as a therapeutic marker in the treatment of both disorders. These hypotheses should be confirmed in a larger set of patients.
Author Contributions
Conceived and designed the experiments: YS, WI, MM. Analysed the data: YS, MM. Wrote the first draft of the manuscript: YS. Contributed to the writing of the manuscript: MM, SI. Agree with manuscript results and conclusions: All authors. Jointly developed the structure and arguments for the paper: All authors. Made critical revisions and approved final version: YS, MM. All authors reviewed and approved of the final manuscript.
Funding
This work was supported by a grant from the Chugai Pharmaceutical Company, Ltd and the Health and Labor Sciences Research Grant on Intractable Diseases (Neuroimmunological Diseases) from Japan's Ministry of Health, Labor, and Welfare.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgment
We are grateful to S. Itoh and E. Nomura for their help in flow cytometry.
