Abstract
Periampullary bleeding is an uncommon cause of upper gastrointestinal (GI) hemorrhage, which is typically iatrogenic in origin occurring as the result of endoscopic intervention of the papilla. Spontaneous, non-iatrogenic periampullary bleeding is extraordinarily rare with only a few cases reported in the literature to date. Vascular malformations, including angiodysplasia and Dieulafoy's lesions, have been implicated in several reports as the etiology but endoscopic intervention is often unsuccessful in achieving durable hemostasis with surgery being required for definitive management in many cases. Herein is reported the case of a 67-year-old male on anticoagulation for atrial fibrillation who presented with severe upper GI bleeding determined to be arising from underneath the hood of the major papilla. No distinct lesion was seen endoscopically but the presumed etiology was an unidentified vascular malformation. Successful treatment was achieved with argon plasma coagulation (APC) applied circumferentially around the papilla. No subsequent endoscopic or surgical intervention was required for durable hemostasis and the patient was able to resume anticoagulation shortly after the procedure. This is the first reported case of spontaneous periampullary bleeding successfully treated with APC.
Case Report
A 67-year-old man on anticoagulation with warfarin at a dose of 5 mg daily for atrial fibrillation presented to hospital with a 3-week history of fatigue, weakness, and presyncope. On admission to hospital, his hemoglobin was found to be 3.8 g/dL (normal range: 12.6-16.5 g/dL) but his international normalized ratio was in the therapeutic range at 2.8 (therapeutic range: 2.0-3.0). He denied all manifestations of overt gastrointestinal (GI) blood loss including hematemesis, melena, hematochezia, and blood per rectum. He denied nausea, vomiting, abdominal pain, change in bowel habit, jaundice, icterus, and change in stool or urine color. There had been no change to his normal diet or fluid intake. After medical stabilization, reversal of anticoagulation, and blood transfusion, he was referred for gastroenterology assessment. The patient had a colonoscopy 2 years prior which was unremarkable. He never had an esophagogastroduodenoscopy (EGD). There was no family history of colon, gastric, or esophageal cancer.
Inpatient EGD demonstrated blood and clot in the second part of the duodenum overlying the expected position of the major papilla. The straight-viewing gastroscope was exchanged for a side-viewing duodenoscope for better visualization. Copious washing revealed the bleeding to be emanating from directly underneath the hood of the major papilla (Fig. 1). Despite copious washing and a clear view of the papilla, no definitive vascular malformation could be identified (Fig. 2). As no distinct lesion was seen, it was initially felt that the bleeding source may have been intraductal and so the procedure was terminated. Colonoscopy complete to the terminal ileum was negative for a source of bleeding. Imaging of the abdomen by contrast-enhanced computed tomography scan, however, revealed no abnormalities of the pancreaticobiliary ductal system, solid organs, or vasculature.

Periampullary bleeding which appears to arise from underneath the hood of the major papilla.

No distinct lesion of the papilla or periampullary mucosa is apparent even after blood is cleared away with copious washing.
The patient continued to bleed despite treatment with a continuous infusion of pantoprazole at a dose of 8 mg/hr and endoscopy was repeated 6 days later. This again revealed bleeding emanating from underneath the hood of the papilla but no distinct lesion was identified. It was then presumed that bleeding was occurring from an unidentified vascular malformation, either an angiodysplastic or Dieulafoy's lesion. On the basis of this presumed diagnosis, argon plasma coagulation (APC) treatment of the periampullary mucosa underneath the hood was applied circumferentially at a current of 40 W and flow rate of 1.4 L/min to achieve hemostasis. Special care was taken to avoid thermal injury to the papilla itself. Thermal ablation of the periampullary mucosa with APC resulted in progressive reduction of bleeding until complete hemostasis was achieved (Fig. 3A-C). The procedure was then terminated. His post-procedure hemoglobin after transfusion was 7.9 g/dL and no further decrease in hemoglobin was detected in hospital. The patient was discharged home and 1 week later, his hemoglobin was 8.6 g/dL. He was then deemed to be fit to resume anticoagulation which was restarted shortly thereafter with no further evidence of blood loss.

Progressive reduction in bleeding with circumferentially applied APC (current: 40 W, flow rate: 1.4 L/min) beginning at the 12 o'clock position (
Discussion
Periampullary bleeding—bleeding arising from the immediate vicinity of either the major or minor papilla—is an uncommon cause of upper GI hemorrhage. Most cases of periampullary bleeding are iatrogenic and occur as the result of endoscopic intervention of the papilla including sphincterotomy, 1 balloon dilatation, 2 and stent manipulation. 3 As bleeding is often apparent during endoscopy, hemostasis can occasionally be achieved by endoscopic intervention during the procedure with a variety of techniques. 1
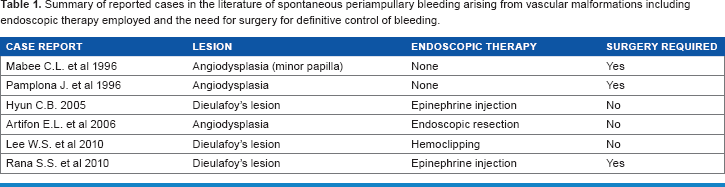
Vascular malformations of the upper GI tract are estimated to account for 30-40% of all causes of obscure GI bleeding. 4 These lesions are typically found in the stomach and duodenum. Periampullary vascular malformations are a rare cause of upper GI bleeding with only a few cases reported in the literature (Table 1). Mabee and colleagues reported the first case of periampullary bleeding, the origin of which was a large vascular malformation of the minor papilla. 4 Of the small number of reported cases, vascular malformations including angiodysplasia4–6 and Dieulafoy's lesions7–9 have been repeatedly implicated as the source of bleeding. A number of endoscopic techniques have been utilized to control hemorrhage originating from these lesions including epinephrine injection, 7 hemoclipping, 8 and endoscopic resection. 5 Despite the well-established efficacy of treating vascular malformations endoscopically, half of the reported cases of periampullary bleeding ultimately required surgery for definitive management (Table 1). Potential explanations for the failure of endoscopic therapy in controlling periampullary bleeding include challenging anatomic positioning, difficulty in identifying culprit lesions, and inadequate available endoscopic technology.
summary of reported cases in the literature of spontaneous periampullary bleeding arising from vascular malformations including endoscopic therapy employed and the need for surgery for definitive control of bleeding.
Herein is reported a case of severe spontaneous periampullary bleeding, felt to be arising from an unidentified vascular malformation, successfully treated by APC. To my knowledge, this is the first such case to be reported in the literature. No definitive vascular malformation was identified at endoscopy (Fig. 2), perhaps because the lesion was deeply buried in the fold of the hood of the major papilla. However, once an intraductal lesion had been ruled out by imaging, an unidentified vascular malformation was the logical choice as the bleeding origin. Bleeding was ongoing despite medical therapy and so empiric endoscopic treatment became medically necessary. If endoscopic therapy had failed, then referral to an advanced endoscopy center may have been considered but, with the severity of bleeding, surgery would ultimately have been the only available alternative.
Argon plasma coagulation was chosen as the treatment modality for several reasons. As no distinct lesion was identified that could be specifically targeted, APC allowed for the ablation of a wide area of mucosa. The application of thermal energy could be easily controlled and was applied circumferentially to avoid damage to the papilla. Thermal ablation of the mucosa also provides durable hemostasis as the vascular malformation is destroyed, whereas other modalities such as epinephrine injection may provide only temporary hemostasis. 9 As a side-viewing duodenoscope was employed for better visualization, maneuvering and deployment of endoclips may have proved challenging. In contrast, the APC probe was easily maneuvered by use of the elevator and scope movement. In this case, effective hemostasis was achieved after initial treatment with APC. However, if further bleeding occurred, subsequent sessions of APC could have been easily attempted before proceeding to surgery.
Argon plasma coagulation has been reported, by Kua and colleagues, 10 to treat bleeding arising from a non-vascular malformation, specifically a periampullary diverticulum. While this initially achieved hemostasis, the patient experienced recurrent hemorrhage 2 months later and required surgery for definitive bleeding control. This highlights the importance of the proper use of APC by the endoscopist to treat superficial vascular malformations only, as was done in the case presented herein. The use of APC alone to treat bleeding from non-vascular lesions is not likely to result in durable hemostasis.
In conclusion, this is the first reported case of successful treatment of severe spontaneous periampullary bleeding treated by APC. Although a distinct lesion was not identified, the origin was presumed to be an unidentified vascular malformation. Argon plasma coagulation was the ideal modality choice to effect hemostasis in this scenario and may certainly prove useful to other endoscopists who encounter this rare cause of upper GI bleeding in the future.
Author Contributions
Conceived the concepts: AA. Analyzed the data: AA. Wrote the first draft of the manuscript: AA. Made critical revisions: AA. The author reviewed and approved of the final manuscript.
Informed Consent
The patient described in the article has provided informed written consent to the use of all clinical information presented herein including demographic data, clinical history, physical examination findings, laboratory results, imaging studies, endoscopy images and discussion of treatments employed.
