Abstract
Objective
Plasminogen system plays a crucial role in physiological and pathological events related to tissue regeneration, wound healing, immune response, angiogenesis, invasion and metastasis. It gets activated when plasminogen associates with its cell surface receptors. Latest information on some of the well-explored plasminogen receptors such as annexin II-S100A10, cytokeratin 8, α-enolase, plasminogen receptor (KT) (Plg-R(KT)) and histone H2B has been discussed in the present review. These receptors can pave the way for effective new therapeutic and diagnostic strategies to counteract malignant diseases.
Conclusion
The present review concludes the key role of plasminogen receptors in extracellular matrix degradation, infiltration into surrounding tissues, neovascularization, invasion, metastasis and drug resistance. This review also discusses the possible effect of blocking these plasminogen receptors with monoclonal antibodies and DNA-based vaccination or silencing plasminogen receptor gene using small interfering RNA or short hairpin RNA to counteract cancer invasion and metastasis.
Introduction
Plasminogen receptors are widely distributed and form a diverse group of cell surface proteins. These receptors are commonly expressed on cell surfaces, where they can interact with plasminogen and plasmin. Plasminogen is a 90-kDa glycoprotein secreted from the liver. It is a key zymogen protease which regulates extracellular proteolytic events such as fibrinolysis and activation of growth factor. It has N-terminal glutamic acid (Glu) residue and five Kringle regions containing lysine-binding sites (LBS). The LBS arbitrate their binding to cell surface receptor and activation along with a C-terminal protease domain. Plasminogen is activated by various tissue-degrading proteases, including high-affinity activators such as tissue plasminogen activator (tPA) and urokinase plasminogen activator (uPA), causing activation of plasmin. Plasmin is a serine protease involved in the degradation of the extracellular matrix (ECM) by activating matrix metalloproteinases (MMPs). 1 Cell-associated plasmin proteolysis contributes both to physiological and pathophysiological processes such as tissue remodeling and embryogenesis, invasion, metastasis and inflammation.2,3 An experiment conducted by Lund et al reported that knock-out mice for uPA and tPA were less impaired in wound healing when compared to plasminogen-deficient mice. This indicates that additional plasminogen activators such as factor XIa factor Xlla and kallikrein provide a sufficient plasmin to sustain the healing process even in the absence of uPA and tPA. 4
Plasminogen receptors exhibit differential number of binding sites ranging from 10 5 to 10 7 per cell, as well as divergence in expression on cell types such as αMβ2-integrin expression is confined to leukocytes and annexin A2/p11 to endothelial cells (ECs). The binding affinity of plasminogen on U937, a human lymphoma cell, was 6.87 × 10 4 at 37 ° C and decreased to 1/4 at 4° C due to a decrease in plasminogen receptor at this temperature, whereas in GM1380, a fibroblast cell, there was not much difference in plasminogen-binding affinity with a change in temperature. 5 However, histone H2B is abundant on the surface of human umbilical vein endothelial cells. This broad binding ability and expression allows plasminogen receptors to perform diverse biological functions, including fibrinolysis, inflammation, wound healing and angiogenesis. 6 Plasminogen receptors include integrins, urokinase-type plasminogen activator receptors (uPAR) and annexin II, which localizes to the leading edge of migrating cells and creates an advantageous microenvironment, in which one plasminogen receptor is particularly capable of plasminogen activation. However, a recent report has demonstrated that cell surface-bound plasminogen receptor can be efficiently activated by uPA bound to another cell. 7 In addition, new plasminogen receptors can be elicited by plasmin itself, which depends on the availability of uPA on the cell surface. As a consequence, a cell type may express a variety of plasminogen receptors, but only few can be differentially upregulated and exploited to mediate a specific cellular response. Cellular recruitment is a complex process requiring activation of different intracellular signaling pathways. Diverse plasminogen receptors activate distinct signaling events and cause efficient cell migration. Therefore, blocking of the signaling response induced by any one plasminogen receptor may lead to suppressed signaling and diminished cell migration.
The present review focuses on the importance of plasminogen receptors in activation of the plasminogen system, which plays a key role in cancer progression and metastasis. Thus, blocking plasminogen receptors may be a promising strategy to counteract invasion and metastasis.
Activation of Plasminogen System by Receptors
The plasminogen system plays a key role in tissue regeneration, wound healing, tissue involution, immune response, angiogenesis, cancer invasion and metastasis.8–11 The plasminogen system is involved in the regulation of plasmin expression, which promotes activation of laminin, perlein, fibronectin, pro-uPA and pro-matrix metalloproteinases (pro-MMPs). All the plasminogen receptors have LBS, ie, C-terminal lysine or an internal amino acid residue that mimics C-terminal lysine for binding with plasminogen and plasmin. Plasminogen receptors increase plasminogen activation by either uPA or tPA, thus enhancing the catalytic efficiency of plasmin and also protecting bound plasmin from inactivation by plasmin inhibitors such as α2-antiplasmin and α2-macroglobulin. 12 In fact, some plasminogen receptors are reported to have one or more functional characteristics. Differences in the affinity of a specific subset of plasminogen receptors for plasminogen might be due to the influence of local conformation or amino acid residues adjacent to LBS-binding residue within receptor. The affinities for ligand appears to be similar in the case of many plasminogen receptors that exploit an internal residue to hold plasminogen with the potential exception of annexin A2/p11heterotetramer, where the proximity of multiple plasminogen-binding sites within a single molecular species could substantially enhance affinity. 13
Plasminogen System and Metastasis
The plasminogen system promotes tumor metastasis by several different mechanisms. One of the well-explored mechanisms is the uPA and uPAR system, which initiates the activation of MMPs as well as the conversion of plasminogen to plasmin followed by ECM degradation and reduced cellular interaction. Expression of uPA and uPAR is regulated by multiple factors such as mitogen, growth factors, oncogenes v-Src and v-Ras, cytokines, protein kinase C and also on ligation of integrin with extracellular matrix protein. Binding of uPA to uPAR can activate Ras-Raf-MEK-ERK pathway. The FAK has been implicated to mediate signal transduction events initiated by integrins through a recruitment of c-Src or other Src family tyrosine kinases. uPA-induced Ras-ERK signaling pathway is dependent on the downstream effectors Raf and MEK. uPA initiates cell migration via Rho-Rho kinase pathway, which helps to promote Ras-ERK–stimulated cell migration. 14
Plasminogen Receptors in Cancer Progression
Plasminogen receptors play a role in the proliferation, migration and metastasis of tumor cells in many cancer types and may serve as prognostic and diagnostic markers.
15
They are involved in mediating colocalization of plasminogen and its activators such as uPA and tPA on cell surfaces and markedly decrease the Km for plasminogen activation. Plasminogen receptors are expressed on the cell surface of most tumors and their expression frequently correlates with cancer diagnosis, survival and prognosis. Notably, they can trigger multiple specific immune responses in cancer patients, highlighting their role as tumor-associated antigens.
16
Except red blood cells (RBCs), plasminogen receptors are broadly distributed on the cell surface with a high binding capacity (3 × 10
7
molecules per cell).17,18 Cell surface receptors loaded with plasmin, which is protected from inhibitors, play a key role in cancer progression.18,19 Plow et al reported that plasmin enhances phagocytosis mediated by macrophages.
20
Therefore, macrophage-mediated phagocytosis of cancerous cells is an important strategy in cancer therapy.
21
In another study on MDA-MB-231 cells, it was analyzed that lipid raft (LR) region of cell membranes showed the predominance of ANG (angiogenin), annexin II and S100A10 were as nonlipid raft (NLR) regions was predominant with uPAR, suggesting that ANG interacts with uPAR at the junctions of LR and NLR regions. uPAR interacts with uPA causing plasmin formation and FAK phosphorytion, which are necessary for tumor migration and invasion.
22
Some well-explored and well-established plasminogen receptors in cancer metastasis and invasion are shown in Figure 1.23–27 uPAR is a 50- to 60-kDa extracellular glycoprotein, rich in cysteine and congregated in lipid rafts. uPAR is composed of three homologous domains D1, D2, and D3 belonging to the Ly-6/uPAR/alpha-neurotoxin protein domain family, which is attached to the cell membrane by a glycosyl phosphatidyl inositol (GPI) anchor and is responsible for high intramembrane mobility. uPAR binds with high affinity to uPA, pro-uPA and the ECM protein vitronectin (VN). The crystal structure of a soluble form of human uPAR reveals that the receptor-binding module of uPA engages the uPAR central cavity, thus leaving the external receptor surface accessible for VN and integrins. Somatic mutation of the X chromosome PIG-A gene in hematopoietic stem cells cannot synthesize the GPI moiety, which anchors many different types of proteins to the cell membrane. uPAR, which is a GPI-anchored protein expressed on activated monocytes and granulocytes, plays an important role in fibrinolysis by promoting the conversion of urokinase to its active form. Increased levels of soluble uPAR lacking the GPI anchor is associated with paroxysmal nocturnal hemoglobinuria (PNH) as well as in patients with certain types of malignancy. The absence of membrane uPAR on monocytes, granulocytes and platelets might contribute to thrombosis in PNH.
14
Plasminogen receptors and metastasis. Schematic representation of the plasminogen receptors involved in activation of plasmin in the presence of plasminogen and uPA. uPAR along with integrins activates FAK signaling, which activates PI3/AKT and on other side plasmin promotes ECM degradation, fibrin degradation, and activation of pro-MMPs, which finally leads to pathophysiological processes, such as cell proliferation, metastasis and invasion.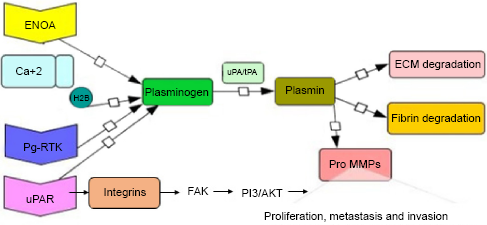
Annexin AII–S100A10 Heterotetrameric Complex
Annexin II (p36) is a pleiotropic, calcium-regulated, phospholipid-binding protein located in the cytoplasm, plasma membrane and the nucleus. It is grouped under annexin family with a molecular weight (MW) of 36 kDa. It is characterized by the canonical annexin folds, which consists of the amino acid sequence G-X-G-T-(38)-(D/E). 28 It exists as a soluble monomer or stable heterotetramer form without carboxyl-terminal lysine. It acts as a plasminogen receptor by binding with S100A10 (p11), a member of the S100 family protein. This stable heterotetrameric complex possesses two copies of a 36-kDa-heavy chain (p36) of annexin II and two copies of 11-kDa-light chain (p11) of S100A10. The C-terminal lysine residue of S100A10 acid protein forms a binding site for plasminogen activators, plasminogen and plasmin. The role of annexin II and S100A10 is interdependent, where annexin II anchor S100A10 on the cell surface and form ubiquitin-mediated degradation and in turn S100A10 imparts plasminogen-binding activity.
Majority of intracellular S100A10 is found in association with annexin II and the excess of S100A10 undergoes proteasomal degradation as shown in Figure 2. In a heterotetrameric complex, S100A10 dimer is positioned in the center with annexin II flanking on both sides. After phosphorylation at Tyr-23 through a Src-like tyrosine kinase–dependent mechanism with conformational changes, this heterotetrameric form of annexin II is translocated to the cell surface. However, expression of annexin II on the cell surface is modulated by a different mechanism.
29
At the endothelial cell surface, heterotetramer form AII-S100A10 (p11) accelerates tPA-dependent activation of the fibrinolytic protease plasmin. Previous report suggests that annexin II-S100A10 heterotetramer plays a crucial role in pathological conditions such as inflammation, thrombosis, autoimmune disease, and cancer progression.
30
It has also been reported that cancer cells overexpress annexin II–S100A10 complex, which causes an increase in the formation of plasmin, thus enhancing ECM degradation, infiltration into surrounding tissues, neovascularization, invasion, metastasis and drug resistance.31–34 Annexin II–S100A10 heterotetramer contributes to cancer invasion and metastasis by acting as a coreceptor for plasminogen, tPA, and pro–cathepsin B.
34
Studies conducted by Connell et al
35
and Swisher et al
36
suggest that in S100A10-deficient mice, macrophage invasion across an inflammatory peritoneal membrane was decreased with a decrease in the generation of plasmin and MMP-9. This indicates that annexin II-S100A10 heterotetramer dramatically affects tumor cell-mediated pericellular proteolysis, tumor invasiveness and metastasis. It has been reported that overexpression of cell surface annexin II is associated with an increase in both mRNA and total cellular expression, suggesting translational regulation.
37
Zhang et al
38
reported that small interfering RNA (siRNA) targeting annexin II not only decreased annexin II messenger RNA and protein levels but also downregulated the levels of S100A10 and c-Myc in invasive breast cancer. The silencing of annexin II inhibited tPA-dependent plasmin generation and reduced cell motility.
Schematic representation of overexpressed annexin II–S100A10 heterotetramer in cancer cells. Plasminogens bind to the S100A10 domain of heterotetramer and uPA binds to uPAR and causes overexpression of plasmin, leading to ECM degradation, invasion, and metastasis.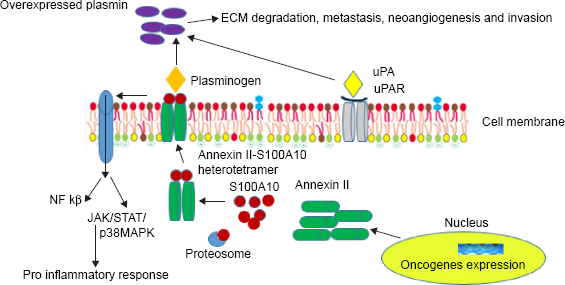
Anti–annexin II antibodies reduce cancer progression by affecting metastasis. Annexin II–specific targets have promising therapeutic benefits in animal models, but are yet to be elucidated in human trials. 39
Cytokeratin 8
Cytokeratin 8 (CK8) is a 52-kDa basic and one of the major structural proteins forming cytoplasmic network of intermediate filaments with variable expression on epithelial cells, differentiation stage and epithelial cell-derived neoplasms. 40 Cytokeratin 18 (CK18) is a 45-kDa acidic intermediate filament protein that codes for a type I intermediate filament protein. It is normally coexpressed with CK8 and is found in most simple ductal and glandular epithelia. Constantly, coexpression of CK8 and CK18 is important for the formation of intermediate keratin filament on epithelial and cancer cells. 41 CK8/18 are associated with flexible intracellular scaffolding in order to stabilize the structure of cytoplasm and mitochondria. It is also required to maintain tissue integrity and is involved in apoptosis and cell cycle progression. CK8/18 are typically coexpressed as the primary cytokeratin pairs in simple epithelial cells and their expression is maintained during malignant transformation. 42 Studies on different cancer cells have proved that expression of CK8/18 varies based on posttranslational modifications, including site-specific phosphorylation, O-GlcN-arylation and acetylation. These modifications regulate the solubility, organization and stability of the filament, emphasizing the role of CK8/18 in cancer progression. 43 CK8 exhibits a unique feature among cytokeratins by having a carboxyl-terminal lysine. Recently, it has been demonstrated that CK8 is a primary plasminogen-binding protein on the plasma membrane. 44 A most acceptable mechanism for the role of CK8 in cancer progression is activation of plasminogen. In breast cancer cells, it has been reported that it acts as a receptor for uPA, which suggests a model in which CK8 in complex with uPA, plasminogen, and fibronectin constitutes a signaling platform capable of modulating invasion, metastasis, cell adhesion, and growth. 45
Studies using specific antibodies have revealed that both plasminogen and CK8 are located on the surfaces of certain epithelial cells, including hepatocytes, hepatocellular carcinoma cells and various breast cancer cells. This novel study has demonstrated the mechanism of protein penetration through the plasma membrane or binding of secreted CK8 to other cell surface molecules. In vitro and in vivo studies have also demonstrated that cancer cells are known to secrete plasminogen-binding CK8-containing protein complexes. These complexes bind with plasminogens as well. The plasminogen-binding activity of CK8 is unique among intermediate filament proteins, probably because its sequence includes a carboxyl-terminal Lys residue. However, a CK8 mutant that lacks the C-terminal Lys still binds with plasminogen, albeit with decreased affinity. CK18 does not bind with plasminogen; however, CK8 and CK18 equally bind to tissue-type plasminogen activators (tPA), which may be important in the mechanism, whereby CK8/18 complex promotes plasminogen activation by tPA. High levels of CK8 expression are associated with an increased migration and invasion of certain cancer cells. These correlations are most easily explained by the function of intermediate filament proteins in determining the rigidity of the cytoskeleton; however, the function of cell surface CK8 as a plasminogen receptor merits consideration. 46
Enolase
Enolase (ENOA; 2-phospho-D-glycerate hydrolase) is a highly conserved cytosolic ubiquitous metalloenzyme with a MW of 48 kDa. It catalyzes the dehydration of 2-phospho-D-glycerate to phosphoenolpyruvate in the glycolytic pathway. It exists in three different isoforms, namely, alpha, beta, and gamma based on three independent loci ENO1, ENO2, and ENO3, respectively. These isoforms are expressed in a tissue-specific manner. α-Enolase (ENOA) is present in almost all adult tissues, β-enolase is expressed in muscle tissues, and γ-enolase is found in neurons and neuroendocrine. 47 ENOA exists as a homodimer or heterodimer, consisting of alpha-alpha, alpha-beta, and alpha-gamma subunits. All these forms act as plasminogen receptors.48,49 ENOA as a plasminogen receptor is predominantly responsible for plasminogen activation via C-terminal lysine. It is involved in the activation of plasmin and in ECM degradation and also supports anaerobic proliferation (Warburg effect). ENO1 induces hypoxic stress through hypoxia-responsive elements in various cancer cells.50–54 Cell surface ENOA promotes cancer invasion when subjected to specific posttranslational modifications, such as acetylation, methylation and phosphorylation. 55
Kuan-Chung Hsiao et al 56 in their study suggested that a-enolase has a significant role in cancer metastasis as α-enolase colocalized with uPA and tPA proteins and was present at the site of pericellular degradation of ECM components. On the other hand, treatment with antibody against α-enolase in vitro suppressed cell-associated plasminogen and MMP activation, collagen and gelatin degradation and cell invasion. Thus, it was demonstrated that surface α-enolase promotes ECM degradation and invasion of cancer cells and that targeting surface α-enolase is a promising approach to suppress tumor metastasis.
Plasminogen Receptor (KT) (Plg-R(KT))
Plg-RKT is a single 147–amino acid protein with a molecular mass of 17.3 kDa. 57 It is an only integral membrane plasminogen receptor, which contains two transmembrane helices with a C-terminal lysine residue as the integral membrane protein. Studies demonstrated that the expression of Plg-RKT is induced during differentiation of monocytes. It is broadly expressed in small aggregates in human tissues, blood monocytes and lymphocytes but is absent in RBCs and colocalized with the urokinase receptors uPA and tPA. Hence, on the cell surface, Plg-RKT allows plasminogen and uPA to be in close proximity. This mechanism of colocalization of plasminogen, its receptor and two major plasminogen activators illustrates that Plg-RKT regulates plasminogen activation on the cell surface. 37 It is also involved in chemotaxis and chemokinesis of human monocytes. Plg-RKT also plays an important role in macrophage migration–dependent activation of MMPs. 58 Plg-RKT may be a novel target to plasminogen biology as it is widely expressed on the cancer cells and serves to modulate plasminogen activation.
Histone H2B
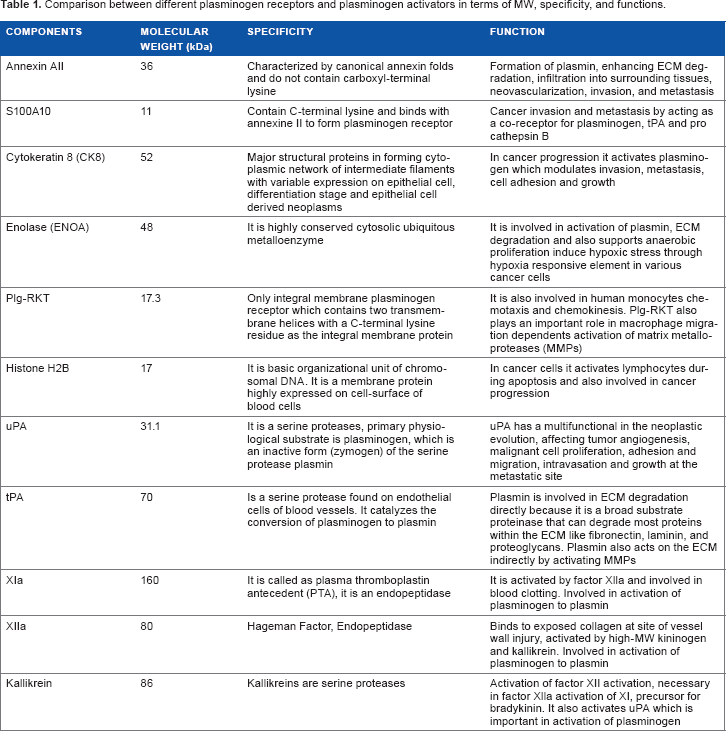
Comparison between different plasminogen receptors and plasminogen activators in terms of MW, specificity, and functions.
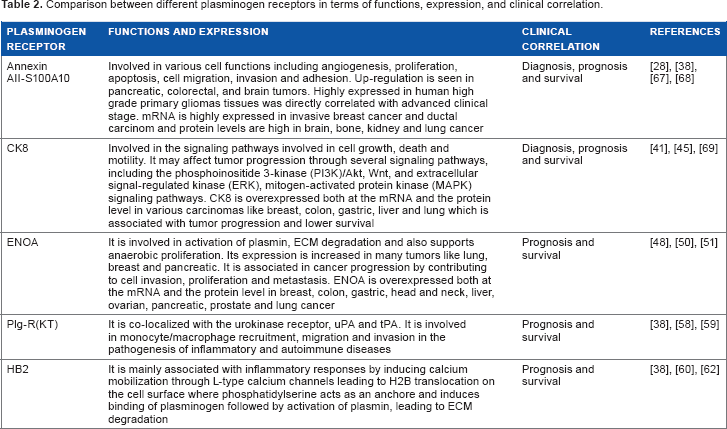
Comparison between different plasminogen receptors in terms of functions, expression, and clinical correlation.
Plasminogen Receptors as Therapeutic Targets in Cancer
Despite heterogeneous numbers of plasminogen receptors existing in eukaryotic cells, only a few receptors such as annexin II-S100A10 heterotetramer, ENOA and CK8 have a renowned role in human cancer progression by promoting plasmin-dependent tumor invasion. These receptors form a multiprotein complex with tPA, uPAR and integrins without interacting with plasminogen activators. 62 They also interact with the actin cytoskeleton and promote the migration of tumor cells independent of plasminogen binding, emphasizing their importance as potential targets for therapeutic purposes. 63 Thus, blocking plasminogen receptors with monoclonal antibodies and DNA-based vaccination or silencing plasminogen receptor genes using siRNA or short hairpin RNA may be a promising strategy to counteract invasion and metastasis. 64 Stereospecific radiation treatment offers a distinct opportunity for temporal and spatial regulation of gene expression at tumor sites by means of inducible promoters. Rao et al 65 confirmed this by the cell death and decrease in migration and invasion by downregulation of uPA and uPAR in meningioma cell lines. Utilization of specific plasminogen receptors to mediate tissue-specific or cell-specific responses is envisioned like annexin II-S100A10 heterotetramer is the major Plg-R on endothelial cells (ECs), H2B, is abundant on monocytoid cells, integrins and uPAR localizes to the migrating cells. The tissue-specific expression of plasminogen receptors can be explored to target specific plasminogen receptors. 66
Author Contributions
Analyzed the data: RM. Wrote the first draft of the manuscript: SK. Both the authors contributed to the writing of the manuscript and agree with manuscript results and conclusion, jointly developed the structure and arguments for the paper, made critical revisions and approved final version.
Footnotes
Acknowledgment
The present review was supported by GITAM University.
