Abstract
Background and purpose
Previous studies have suggested embolic mechanisms for bihemispheric subcortical infarcts involving the anterior and posterior circulation. However, the mechanism of bihemispheric subcortical infarcts in middle cerebral artery (MCA) territories remains uncertain. We describe a patient with acute bihemispheric subcortical infarcts in restricted MCA territories suggesting an embolic mechanism.
Case description
A 62-year-old woman with a history of hypertension and hyperlipidemia suddenly presented with left hemiplegia. Diffusion-weighted and T2-weighted magnetic resolution imaging of the brain showed multiple subcortical high intensity in the MCA territories. There were no acute infarctions in the cerebrum, brain stem, or cerebellum, including cortical lesions. The patient had no carotid, internal carotid artery, or MCA disease.
Conclusion
Bihemispheric subcortical infarcts in the MCA territory are likely to have a proximal embolic source and such infarcts could be associated with multiple subcortical infarcts due to small vessel disease.
Keywords
Introduction
Various mechanisms of small subcortical infarcts, such as intracranial arteriosclerosis, microatheroma, or emboli from the carotid or aortic arch or the heart, have been suggested. Multiple subcortical infarcts often include bihemispheric infarcts. Previous studies have suggested embolic mechanisms for bihemispheric subcortical infarcts involving the anterior and posterior circulation.1,2 However, confirmation of the mechanism of bihemispheric subcortical infarcts in middle cerebral artery (MCA) territories is difficult because bihemispheric subcortical lesions in the MCA or perforating arteries arising from M1 are distant from the site of infarction. Findings providing clues to the mechanism involved can contribute to a better understanding of etiology of multiple subcortical infarcts. We describe a patient with acute bihemispheric subcortical infarcts in restricted MCA territories suggesting an embolic mechanism.
Case Presentation
A 62-year-old woman with a history of hypertension and hyperlipidemia received a calcium-channel blocker before the onset of stroke and suddenly presented with left hemiplegia. The blood pressure was 139/60 mmHg. Diffusion-weighted and T2-weighted magnetic resolution imaging (MRI) of the brain showed multiple subcortical high intensity in the MCA territories (Fig. 1). There were no acute infarctions in the cerebrum, brain stem, or cerebellum, including cortical lesions. There were old subcortical infarcts in the anterior and middle cerebral artery territories on T1- and T2-weighted images. The results of ultrasonography of the carotid and vertebral arteries and echocardiography were normal. Electrocardiography showed no abnormal arrhythmias. MR angiography showed no stenosis or occlusion in intracranial, extracranial or cervical major arteries. Routine serum laboratory values were normal expect for hyperlipidemia. The serum levels of total cholesterol and triglycerides were 283 mg/dL and 99 mg/dL, respectively. The serum levels of thrombin-antithrombin complex and D-dimer were not elevated, and that of antithrombin-III was not decreased. Antinuclear and anti-β2-glycoprotein I antibodies were negative. The patient received intravenous ozagrel and edaravone and was subsequently given warfarin. After 1 month, he was discharged, with mild residual signs and symptoms in the left limbs. The high-intensity areas on diffusion-weighted MRI had disappeared later (Fig. 1).
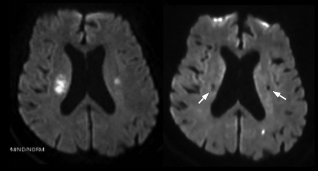
Diffusion-weighted magnetic resolution images of the brain, bilaterally showing high intensity in the centrum ovale (left panel). Both high-intensity regions disappeared later (right panel, white arrows). An asymptomatic high spot was also evident in the left occipital lobe.
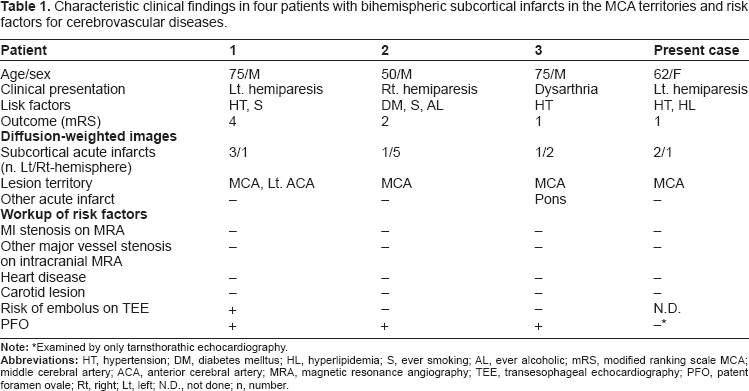
We identified three patients similar to the present case among 396 patients with acute cerebral infarcts from January 2000 through March 2010 and analyzed the risk factors for cerebrovascular diseases of three patients (Table 1). Patients 1 and 3 had a history of cerebral infarction and transient ischemic attacks, respectively. The intracranial and extracranial major vessels showed no stenosis in any patient. No patient had carotid lesions or heart disease. PFO was evident in three patients. “Heart disease” in the Table was defined as having a history of heart attack or heart disease, or as having abnormal findings such as rhythm abnormalities associated with atrial fibrillation, valvular dysfunction, or myocardial hypofunction on chest radiography, 12-lead electrocardiography, 24-Holter electrocardiography, monitoring for at least 48 hours after admission, or transthoracic Doppler echocardiography. “Carotid lesion” was defined as a causative lesion for cerebral infarction, such as at least 50% luminal stenosis 3 or mobile or ulcerative plaque on carotid Doppler ultrasonography or cervical MR angiography. The risks of embolus on transesophageal echocardiography (TEE) were positive if the thoracic aorta had mobile or ulcerative plaque or complicated plaque (defined as a lesion thicker than 4.0 mm or a 3.0-mm lesion with wall irregularity) 4 or if the heart showed left atrial or ventricular thrombus, atrial dysrhythmias, valvular strands, valvular calcification, mitral valve prolapse, spontaneous echo contrast within the left atrium, vegetation, tumor, or atrial septal aneurysm. 5 Patent foramen ovale (PFO) was identified by microbubble testing on TEE.
Characteristic clinical findings in four patients with bihemispheric subcortical infarcts in the MCA territories and risk factors for cerebrovascular diseases.
Discussion
Infarcts in this patient were located in the centrum ovale, the central white substance of the cerebral hemispheres. The centrum ovale is supplied by long medullary arteries that perforate it and course towards the upper part of the lateral ventricle. Centrum ovale infarcts have been attributed to a hemodynamic mechanism associated with ipsilateral carotid disease or artery-to-artery embolism in some patients with severe carotid disease.6,7 A previous study of 12 patients with subcortical centrum ovale infarcts showed extracranial internal carotid artery (ICA) steno-occlusive lesions in 7 patients, carotid artery stenosis in 1, intracranial ICA lesions in 6, and focal or diffuse MCA main trunk stenosis in 3. 7 However, our patient had no carotid, ICA, or MCA disease. In our patient, an embolic mechanism occurring more proximal than the carotid arteries is suggested.
Given that we have not identified definite embolic sources, how likely are emboli to enter the perforating arteries? One study tested embolic materials injected into the internal carotid artery of monkeys over a 10-minute period. Histological brain sections revealed that 6% of the injected spheres entered small penetrating vessels. 8 Autopsy studies have confirmed cholesterol atheromatous emboli in subcortical infarcts.9,10 Multiple lesions occurring simultaneously in different deep perforating artery territories, found in our patient, also suggest that embolic mechanisms are likely. Bogousslavsky et al found that acute multiple bihemispheric infarcts of the anterior circulation were associated with embolisms, usually arising from the heart. 2 Although we did not investigate leakage between the right and left circulation in lung vessels, which can potentially cause positive results of microbubble testing on TEE, we found potential PFO in three patients. A previous study reported on a patient with bihemispheric infarcts in the anterior circulation who had PFO, 4 and the potential PFO in our patients might have been associated with bihemispheric subcortical infarcts in the MCA territories. Other known risk factors for ischemic stroke, such as hypertension, hyperlipidemia, and smoking, might also be involved.
The present case might be classified as only cryptogenic stroke, but bihemispheric subcortical infarcts in the MCA territory are likely to have a proximal embolic source; however, confirmation must await larger studies.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
