Abstract
Introduction:
The efficacy and safety of patent foramen ovale closure (PFOC) in cryptogenic stroke (CS) patients aged ⩾ 60 remain controversial. This study evaluates the efficacy and safety of PFOC in elderly (aged ⩾ 60) versus non-elderly (aged < 60) patients and examines potential age-related benefit.
Patients and methods:
A hospital-based cohort study (January 2013–June 2023) compared the efficacy and safety between PFOC and non-PFOC groups in patient with CS or cryptogenic TIA. The primary efficacy outcome was recurrent ischemic stroke, and safety outcomes included procedure-related adverse events and periprocedural atrial fibrillation (AF). Subgroup analyses, including various age ranges, were performed for the elderly group.
Results:
Among 239 patients (mean age 57.2 years), 120 were elderly. During a mean follow-up of 3.1 years, the PFOC group had significantly fewer recurrent ischemic stroke than the non-PFOC group (adjusted hazard ratio (AHR): 0.10, 95% CI: 0.03–0.29,
Discussion and conclusion:
PFO closure is safe and effective in elderly patients with CS. Advanced age should not be a contraindication for PFOC, as older patients may potentially derive more significant benefits from the procedure.

Introduction
Patent foramen ovale (PFO) closure effectively reduces recurrent ischemic stroke in younger cryptogenic stroke (CS) patients, but its efficacy and safety in elderly CS patients remain unclear.1–5
Previous clinical trials mainly focused on patients aged ⩽ 60, shaping international guidelines for this group.6,7 Nevertheless, recent studies suggest that the risk landscape for older patients may be changing.8–10 Notably, research shows that CS patients with PFO over 60 years old have a 1.64 times higher risk of recurrent stroke than younger patients. 8 Additionally, with an aging population—given that over 65% of stroke patients are older than 60 11 —there is a significant group that could potentially benefit from re-evaluation of PFO treatment. The current age-based restrictions on PFO closure recommendations do not account for the physiological and pathological complexities that may lead to a higher recurrence risk in older adults.
Our hypothesis posits that elderly patients (aged 60 years and above), presenting with CS and PFO, may derive similar or even more substantial benefits from PFO closure as observed in the non-elderly group (<60 years).
Patients and methods
Study design and participants
This hospital-based cohort study utilized data from a prospective stroke registry at a comprehensive stroke center in central Taiwan, including patients with cerebral ischemic events from January 2013 to June 2023. All cerebral ischemic events, including ischemic stroke and transient ischemic attack (TIA), were classified according to the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) study. 12 CS, or cryptogenic TIA, was diagnosed by neurologists based on a standardized comprehensive evaluation.
Patients were classified into two groups: (1) the PFOC group, which included patients who underwent transcatheter PFOC in addition to medical therapy and (2) the non-PFOC group, which included patients who received medical therapy alone. Demographic data, including risk of Paradoxical Embolism (RoPE) score, 13 the PFO-Associated Stroke Causal Likelihood (PASCAL) Classification 14 and ancillary tests at admission were recorded, with outpatient follow-ups and telephone surveys conducted throughout the study. The PASCAL classification was retrospectively applied to participants enrolled before 2022. Since patients aged over 60 can only be categorized as “possible” at most (the maximum RoPE score for patients aged ⩾ 60 is 6), and no revised version tailored for elderly currently exists, the PASCAL classification in this study was simplified into two categories: unlikely and likely (including both possible and probable). The follow-up period of each patient ended when (1) there was a recurrent cerebral ischemic event, (2) loss of follow-up, or (3) mortality of any cause.
The efficacy outcome was recurrent ischemic stroke during the follow-up period. The safety outcomes were procedure-related adverse events and new-onset periprocedural atrial fibrillation (AF) that occurred within seven days after PFOC. The efficacy and safety outcomes between the treatment groups were compared in the non-elderly (<60 years) and elderly (⩾60 years) groups. In addition, the safety outcomes were also compared between the younger elderly (age 60–70 years) and older elderly (age ⩾ 70 years) groups. Details of each case of procedure-related adverse events will be presented.
Additional subgroup analysis tailored to the elderly population, including various age ranges and modified PASCAL classification, were performed to investigate the efficacy of PFOC in elderly.
Study inclusion and exclusion criteria
The inclusion criteria included (1) patients with CS or cryptogenic TIA, (2) patients with PFO, and (3) patients aged > 18 years old. The exclusion criteria included (1) patients who were diagnosed with pulmonary arteriovenous malformation (AVM) according to the transcatheter procedure, (2) a follow-up period of less than 6 months, and (3) patients with missing data.
Assessment of CS, cryptogenic TIA, and PFO
In our hospital, we arranged a routine survey of stroke etiologies based on the international guidelines in place at the time, specifically those from 2013, 15 including brain computed tomographic or magnetic resonance angiography, transcranial color-coded duplex (TCD) sonography, duplex sonography of cervical arteries, in-hospital heart-rhythm monitoring and electrocardiography (ECG). We performed further studies, including hypercoagulable state, autoimmune disease, hereditary diseases, malignancy, transthoracic echocardiography (TTE), and 24-h Holter ECG, when a routine survey did not reveal an attributable stroke etiology. While an etiology for the index ischemic event could not be identified, a microbubble test was performed to ascertain the presence of PFO. Patients with an abnormal microbubble test were assumed to have PFO. CS or cryptogenic TIA was diagnosed after excluding identifiable etiologies other than PFO.
The microbubble test used a gas-contrast TCD, focusing on the unilateral middle cerebral artery. Agitated saline-gas-blood contrast was injected intravenously, with tests performed at rest and during the Valsalva maneuver. 16 Definite high-intensity transient signals (HITS) were recorded within 30 s. The diagnosis of PFO was based on the microbubble test according to a validated study. 17 In addition, the current study also recorded whether there were more than 20 visible micro-embolic signals (MES) based on TCD, 18 and this methodology was used exclusively for the subgroup analysis based on the PASCAL classification.
Large PFO shunt size was defined using two methodologies in the current study: (1) HITS > 10: based on TCD, according to the original proposed method 17 ; and (2) Large shunt: MES > 20 based on TCD, according to the PASCAL classification developed in 2021. 14 To incorporate both methodologies, PFO size was classified only as large shunt or not, without further grading, since visible MES has been shown to have limited specificity. 18
High-risk PFO was defined as the presence of a large shunt PFO or atrial septal aneurysm (ASA). The diagnosis of ASA was based on transesophageal echocardiography (TEE). Patients who were suspected to have ASA on TTE underwent further evaluation with TEE for confirmation. Elderly patients with high-risk PFO were directly classified as “likely” according to the PASCAL classification.
Transcatheter PFO closure
CS patients with PFO were eligible for PFOC unless contraindicated by factors like active bleeding, contrast allergy, acute pulmonary edema, or systemic infection. A multidisciplinary team discussed risks and benefits with patients or families during shared decision-making conferences before obtaining consent. Details of PFOC are available in our prior study. 19
Patients who received PFO closure were treated with a standardized antiplatelet regimen, 1 consisting of clopidogrel 75 mg daily for 6 months, and aspirin 100 mg daily for long-term use. In the non-PFOC group, antiplatelet therapy was prescribed at the discretion of the treating neurologists, based on contemporaneous treatment guidelines during the study period.15,20 None of the included participants were treated with anticoagulants. While patients underwent PFOC, we conducted a 24-h Holter ECG on the day of the procedure and subsequently every 6 months to confirm the occurrence of any new-onset AF.
Statistical analyses
Descriptive statistics were presented as mean ± standard deviation (SD) or median and interquartile range (IQR). We used Fisher’s exact test, Chi-squared test for categorical variables, and Mann–Whitney
Cox regression analyses, adjusted by RoPE score and NIHSS at admission, were performed to investigate the effect of PFOC on the incidence of efficacy outcome. Adjusted hazard ratio (AHR) with a 95% confidence interval (CI) was calculated accordingly between the PFOC and non-PFOC groups. The efficacy and safety outcomes between groups were compared. Besides, the safety outcomes between the younger elderly (age 60–70 years) and the older elderly (age ⩾ 70) groups were also compared.
Additional subgroup analyses on the elderly group, including various age ranges, high-risk PFO features, and ROPE scores of 6, 5, and 4, were performed by adjusted Cox regressions and were visually presented in a Forest plot based on AHR.
Modified PASCAL classification for elderly
Building on the theoretical framework of the PASCAL classification, we evaluated whether integrating the RoPE score and high-risk PFO features is applicable for risk stratification in elderly patients. The modified PASCAL classification system was constructed for elderly group by incorporating the presence of high-risk PFO features and extending the eligible RoPE score threshold from 7 to 6 or 5. Patients were categorized into a “likely” group (with either determinant) or an “unlikely” group (with neither determinant). Subgroup analyses based on the modified PASCAL classification were conducted using adjusted Cox regression and presented in Forest plot.
Concordance analysis of large shunt PFO, sample size calculation and power analysis
We used the McNemar test to evaluate the consistency between the two methodologies for defining large shunt PFO.
To calculate the required sample size for comparing two proportions, we used the standard formula for two independent proportions. According to previous studies, the DEFENSE-PFO trial 21 and Gore REDUCE trial, 4 we assumed that the recurrent stroke rate in the PFOC group would be 1.4% and 12.9% in the non-PFOC group. With an estimated dropout rate of 10%, a minimum of 86 patients per group (total 172) is required to detect a statistically significant difference with 80% power and a 5% significance level.
Since the DEFENSE-PFO trial, 21 a rare controlled study that included patients aged over 60 years, reported no ischemic stroke among those who received PFO closure, reference data for calculating the sample size in the current study were limited. Therefore, we also performed post hoc power analyses based on the primary efficacy outcome, recurrent ischemic stroke, to assess the adequacy of our sample size.
Results
Participants
Based on the inclusion and exclusion criteria, 239 patients were considered eligible to participate in our study (Figure 1). The cohort included 119 patients in the non-elderly group and 120 patients in the elderly group. In the total cohort, the participants’ mean age was 57.2 ± 15.2 years; the mean follow-up period was 3.1 ± 1.8 years, and most patients had CS as an index event (92.5%) with a median NIHSS score (IQR) of 2 (1–6). The mean RoPE score was 5.5 ± 1.9, and 100 (41.8%) had high-risk PFO features.

Study flow diagram.
Baseline characteristics between PFOC and non-PFOC in each age group
We compared the sociodemographic and PFO-related factors for patients who underwent PFOC or not (non-PFOC) in the total cohort and in each age group (Table 1). Compared to the non-PFOC group in the total cohort, the PFOC group was younger (53.9 ± 15.2 vs 60.9 ± 14.3,
Baseline clinical characteristics between the non-elderly and elderly groups.
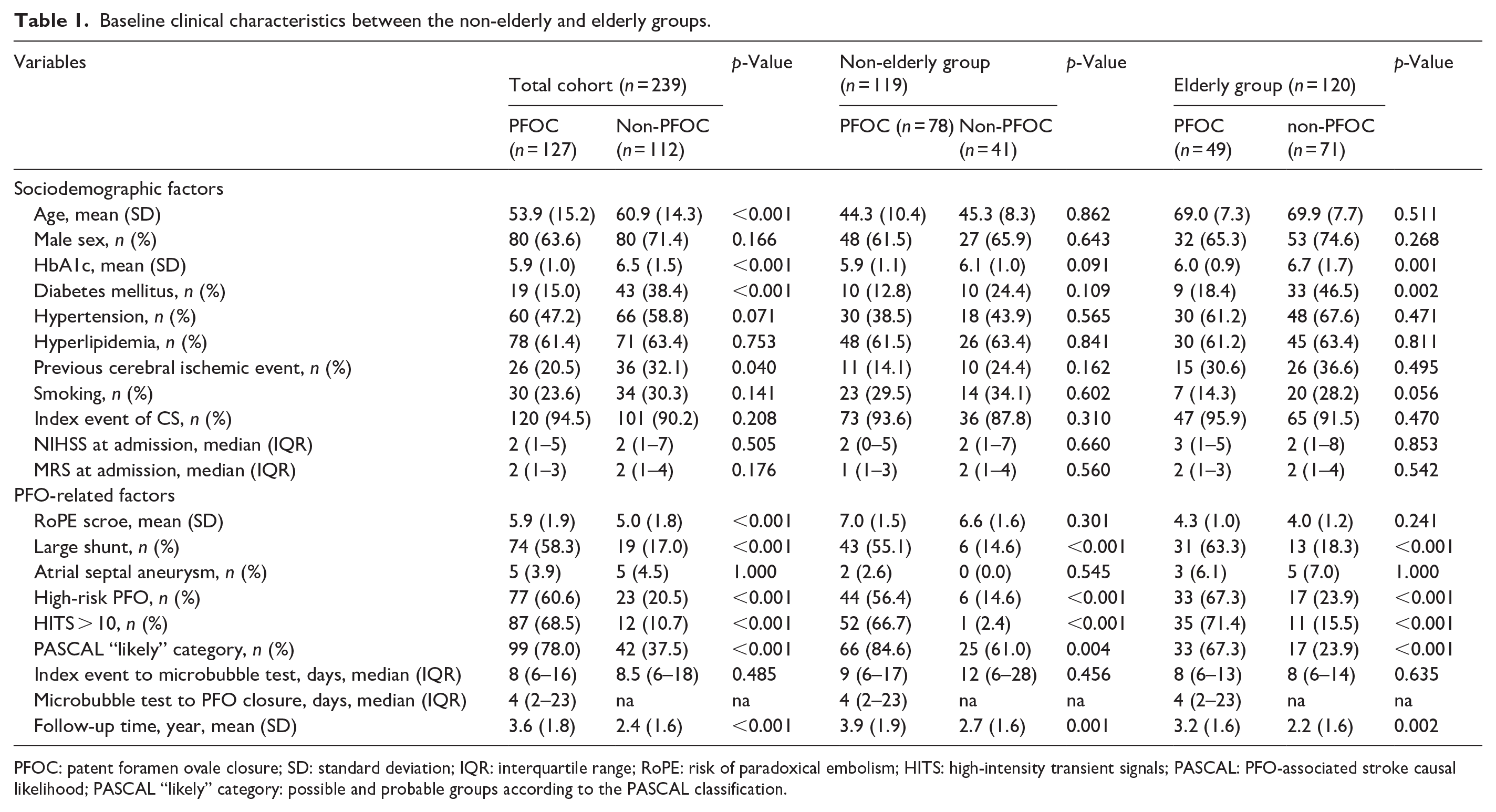
PFOC: patent foramen ovale closure; SD: standard deviation; IQR: interquartile range; RoPE: risk of paradoxical embolism; HITS: high-intensity transient signals; PASCAL: PFO-associated stroke causal likelihood; PASCAL “likely” category: possible and probable groups according to the PASCAL classification.
Among the non-elderly group, the baseline sociodemographic factors were comparable between the PFOC and non-PFOC groups. Compared to the non-PFOC group, the PFOC group had a higher frequency of HITS > 10 (66.7% vs 2.4%,
Among the elderly group, the PFOC group had a lower mean HbA1c (6.0 ± 0.9 vs 6.7 ± 1.7 %,
Baseline comparison between elderly and non-elderly groups
A direct comparison of baseline characteristics between elderly and non-elderly patients is presented in Supplemental Table 1. Compared to the non-elderly group, elderly patients had higher rates of hypertension (40.3% vs 65.0%,
Outcomes
Thirty-one patients (13.0%) developed recurrent ischemic stroke during the follow-up period. Among these, recurrent ischemic stroke occurred in 5 of 127 patients (3.9%) in the PFOC group and 26 of 112 patients (23.2%) in the non-PFOC group during a mean follow-up of 3.1 years, respectively. The adjusted Cox regression revealed a significantly lower risk of recurrent ischemic stroke in the PFOC group compared to the non-PFOC group (AHR: 0.10, 95% CI: 0.03–0.29,
Recurrent ischemic stroke between PFOC and non-PFOC in each age group a .
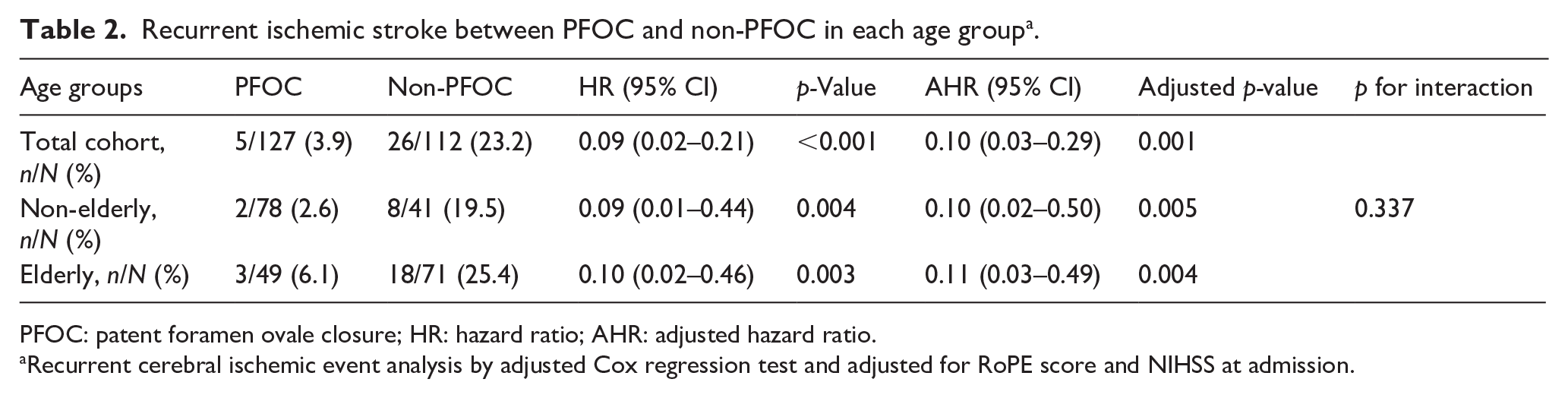
PFOC: patent foramen ovale closure; HR: hazard ratio; AHR: adjusted hazard ratio.
Recurrent cerebral ischemic event analysis by adjusted Cox regression test and adjusted for RoPE score and NIHSS at admission.
The subgroup analyses of total cohort (Supplemental Figure 1) demonstrated significant risk reduction of recurrent ischemic stroke with PFOC across patients stratified by gender, presence of large shunt, HITS > 10, and “likely” category in the PASCAL classification. The risk reduction was significant (AHR: 0.08, 95% CI: 0.02–0.26,
Regarding the safety outcomes, seven procedure-related adverse events (5.5%) and six new-onset periprocedural AF (4.7%) were observed in the total cohort. The occurrence rates of procedure-related adverse events were comparable between the non-elderly and elderly groups (5.1% vs 6.1%,
Safety outcome between age groups.

Subgroup analyses on the elderly group
In the subgroup analyses for the efficacy outcome of elderly group (Figure 2), patients were categorized into the following age ranges: 60–65 years versus ⩾65 years, and 60–70 years versus ⩾70 years. In terms of the efficacy of PFOC in risk reduction of recurrent ischemic stroke, the risk reduction was significant (AHR: 0.01, 95% CI: 0.00–0.66,
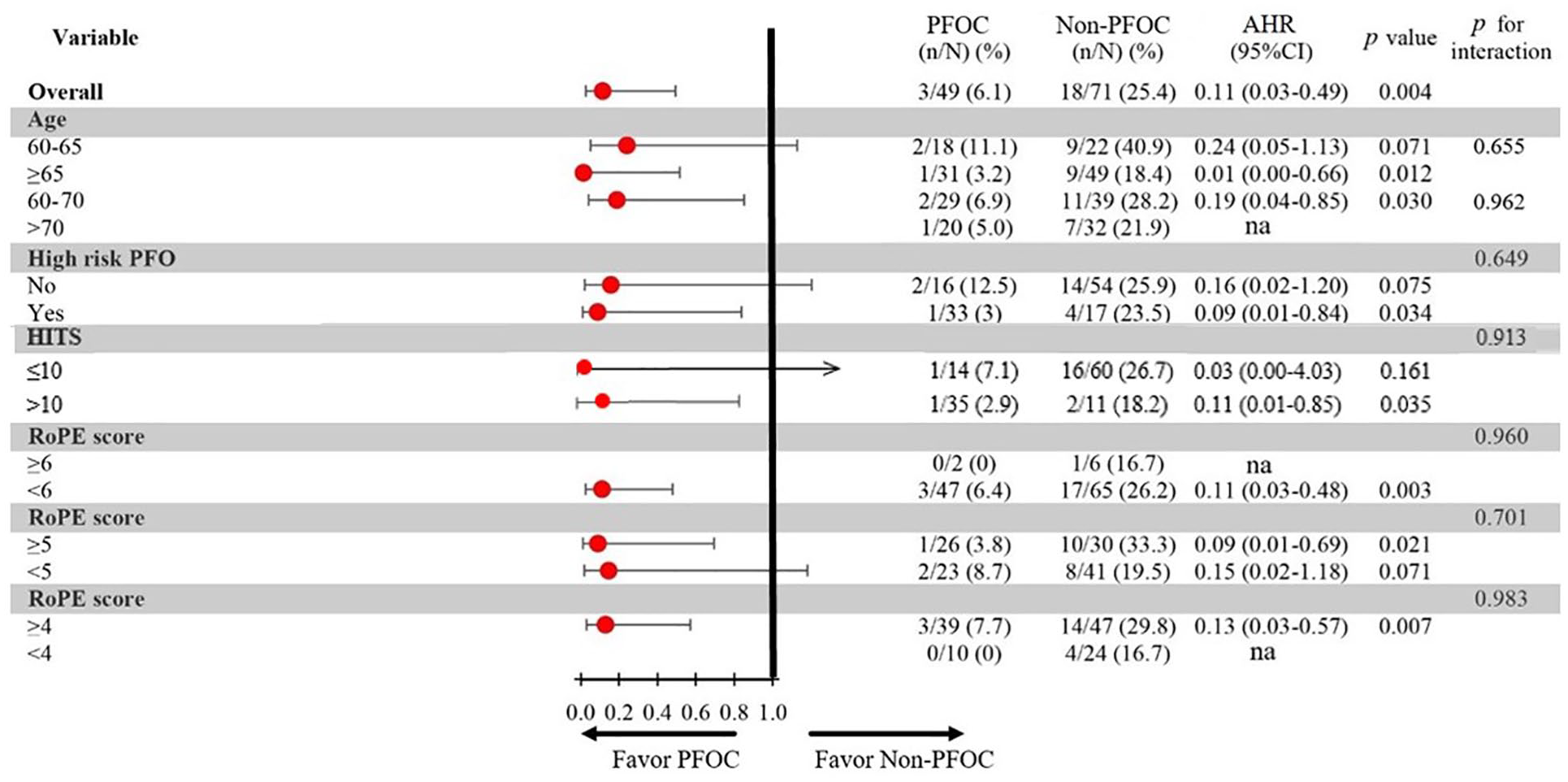
Subgroup analyses on the elderly group.
Regarding the subgroup analyses for the safety outcomes of elderly group, patients were categorized into age ranges: younger elderly (aged: 60–70) versus older elderly (aged ⩾ 70). The safety outcomes were comparable between the younger elderly and older elderly groups (procedure-related adverse events: 6.9% vs 5.0%,
Safety outcome between younger elderly (aged 60–70) and older elderly (aged ⩾ 70) groups.

AF: atrial fibrillation.
Modified PASCAL classification for elderly
According to the original PASCAL classification, the risk reduction was significant (AHR: 0.09, 95% CI: 0.01–0.84,
Concordance analysis of large shunt PFO, and post hoc power analysis
The McNemar test yielded a
Based on post hoc power analysis using the primary efficacy outcome, the current study achieved a statistical power of approximately 99.7%, confirming that the sample size for this trial was deemed sufficient.
Discussion
Our study demonstrated that PFOC in elderly cryptogenic TIA and CS patients is reasonable in significantly reducing the incidence of recurrent ischemic stroke with a favorable safety profile compared to non-PFOC.
For the multivariable regression analyses, we adjusted for the RoPE score and baseline NIHSS at admission. The RoPE score is a composite index that incorporates age, vascular risk factors (e.g., hypertension, diabetes), previous cerebral ischemic event, smoking history, and lesion location, while baseline NIHSS is linked to ischemic stroke recurrence and mortality. 19 This adjustment was primarily intended to account for the baseline differences between the PFOC and non-PFOC groups, particularly regarding age, diabetes, and previous cerebral ischemic events. Additionally, because the RoPE score includes age and vascular comorbidities, the major differentiating factors between elderly and non-elderly patients in our cohort (Supplemental Table 1), this adjustment also helped minimize potential selection bias when comparing treatment efficacy across age groups. Due to the real-world design and extended enrollment period (2013–2023), treatment decisions regarding PFO closure were individualized, based on clinical assessment and shared decision-making between the multidisciplinary team and patients or their families. As shown in Table 1, patients who received PFO closure more frequently had high-risk PFO features, larger PFO shunt size (HITS > 10 or >20 microbubble signal), and higher RoPE scores, which likely guided therapeutic choices. In terms of the subgroup analysis of the total cohort, the effect on reducing recurrent ischemic stroke may be more pronounced when the index event is CS rather than cryptogenic TIA.
Early research, a meta-analysis by Overell et al., 22 linked PFOs to ischemic stroke in patients under 55 but not in older adults. This influenced major clinical trials (2010–2020) that excluded those over 60, shaping guidelines that recommend treatment only for younger patients.1–5 However, recent meta-analyses 8 and real-world study 10 highlight the need to reconsider PFO closure in those aged 60 and above, acknowledging their unique characteristics and the rising prevalence of cryptogenic stroke in this group. 23
Our results align with recent U.S. real-world data supporting broader PFOC use in older patients. 24 Notably, nearly half underwent PFOC for non-approved indications, 24 highlighting that strict age-based limits may overlook its real-world clinical benefits.
Moreover, the PFO Japan Post-Marketing Surveillance study provides evidence supporting the safety and efficacy of PFO closure in older patients, 9 specifically within the first 30 days following the procedure. In this study, nearly 30% of the participants were over 60 years of age, and the adverse event rate was low (3.4%). While short-term outcomes are promising, our cohort, with a mean follow-up of 3.6 years (3.2 years in the elderly group), provides stronger evidence that advanced age should not be a barrier to PFO closure. Our findings further confirm its long-term safety and efficacy, emphasizing its benefits given the increased risk of recurrent stroke in this population. 25
Elderly CS patients exhibit two distinct PFO characteristics. First, PFO prevalence is notably higher, with over 40% having a concomitant PFO—three times more common than in the general population. 26 Secondly, increasing age is associated with a higher risk of recurrent ischemic stroke in patients with CS and PFO, 8 due to greater susceptibility to right-to-left shunting, 27 increased PFO diameter 27 and higher incidence of venous thrombosis. 28 Growing evidence supports the importance of PFO closure in the elderly. A sub-study from the DEFENSE-PFO trial suggests that its benefits are even greater in patients aged 70 and older compared to younger individuals. 29 In line with emerging trends, our subgroup analysis of elderly patients showed a significant reduction in recurrent ischemic stroke following PFOC in those aged ⩾ 65 years. In contrast, the 60–65 age group did not reach statistical significance. This finding may reflect the smaller sample size and lower event rate in this subgroup rather than indicating a true absence of benefit. Nevertheless, the relatively low event rate observed in patients aged ⩾ 65 years who received PFOC (3.2% within 3.2 years follow-up) may further support the potential role of PFOC in reducing recurrent ischemic stroke in advanced age. Further studies with larger sample sizes are needed to confirm this finding.
Safety is another key concern for PFO closure in elderly. Complications include a fourfold increased AF risk and ⩾5% procedure-related events. 16 In our study, 7 patients (5.5%) experienced procedure-related adverse events, including 5 with femoral hematoma, 1 with vasovagal bradycardia, and 1 with pericardial effusion. Over a span of more than 10 years since we performed PFOC across all age groups, no patient encountered life-threatening complications. There was one case who experienced pericardial effusion, requiring pericardiocentesis by an interventional cardiologist and resulting in an additional two days of hospitalization. Even in the older elderly (aged ⩾ 70), there was only one patient (5%) suffering from a procedure-related adverse event, which was femoral hematoma (Supplemental Table 1). Additionally, the incidence of periprocedural AF was comparable between younger elderly and older elderly (Table 4). Namely, the complication rate of PFOC does not increase as age increases. The literature generally highlights an increased risk of periprocedural AF in the elderly, with age being a significant factor contributing to AF incidence. 30 However, our subgroup analysis found no age-related increase in periprocedural AF, suggesting that advanced age alone may not predict a higher AF risk. Furthermore, all PFOC patients underwent 24-h Holter every six months, with no new AF detected. Thus, advanced age may not be a safety concern for PFOC in CS patients with PFO.
From a current perspective, risk stratification is crucial for CS patients with PFO. However, no standardized risk stratification method exists for elderly patients, and the widely used PASCAL classification is not applicable to those over 60.
16
Therefore, we attempted to identify suitable risk stratification factors for elderly patients. Our subgroup analysis identified high-risk PFO features and a RoPE score of 5 as potential factors, but the non-significant interaction
Our study’s strengths include a relatively large sample of elderly CS patients who underwent a microbubble test, yielding meaningful insights into PFOC efficacy and safety. However, the sample size remains insufficient for statistical significance in elderly subgroup analyses. Nonetheless, the long-term follow-up provided robust outcome data. Several limitations exist. First, AF exclusion was based on a 24-h Holter ECG rather than prolonged monitoring, raising the possibility of undetected AF. This reflects guideline-based practice at the study’s inception in 2013. However, in-hospital heart-rhythm monitoring, pre-diagnosis Holter ECG, and regular follow-ups may have mitigated this limitation. Second, as a non-randomized study, selection bias remains a potential limitation. Although we adjusted for the RoPE score, further adjustment for large shunt PFO was constrained by the limited number of non-PFOC patients with large shunt, rendering propensity score matching infeasible. Third, the diagnosis of large shunt PFO and ASA was not systematically confirmed by TEE in all patients, potentially leading to under-detection of high risk PFO in the current study, which may have limited the discriminatory power of subgroup analyses based on the PASCAL classification for effective risk stratification. Fourth, outcome assessment relied on clinically diagnosed recurrent ischemic stroke with imaging evidence rather than new ischemic lesion detected by brain imaging, potentially reducing event detection and limiting statistical power in elderly subgroup analyses. Despite this limitation, the 23.2% stroke recurrence rate in the non-PFOC group over a 2.4-year follow-up is consistent with previous studies in Asian populations, 31 supporting the generalizability of our results.
Conclusion
PFO closure is effective and safe in elderly CS patients, significantly reducing recurrent cerebral ischemic events, comparable to younger populations. Our findings suggest a potential age-related benefit, with enhanced efficacy and no increased complications. Thus, advanced age should not be a contraindication for PFOC. Further research is needed to refine patient selection and optimize outcomes in elderly patients.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251341764 – Supplemental material for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit
Supplemental material, sj-docx-1-eso-10.1177_23969873251341764 for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit by Chi-Sheng Wang, Yu-Hsuan Wu, Yun-Ching Fu, Sheng-Ling Jan, Ming-Chih Lin, Chiann-Yi Hsu and Po-Lin Chen in European Stroke Journal
Supplemental Material
sj-jpg-2-eso-10.1177_23969873251341764 – Supplemental material for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit
Supplemental material, sj-jpg-2-eso-10.1177_23969873251341764 for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit by Chi-Sheng Wang, Yu-Hsuan Wu, Yun-Ching Fu, Sheng-Ling Jan, Ming-Chih Lin, Chiann-Yi Hsu and Po-Lin Chen in European Stroke Journal
Supplemental Material
sj-jpg-3-eso-10.1177_23969873251341764 – Supplemental material for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit
Supplemental material, sj-jpg-3-eso-10.1177_23969873251341764 for Assessment of patent foramen ovale closure in elderly patients with cryptogenic transient ischemic attack or stroke: Efficacy, safety, and potential age-related benefit by Chi-Sheng Wang, Yu-Hsuan Wu, Yun-Ching Fu, Sheng-Ling Jan, Ming-Chih Lin, Chiann-Yi Hsu and Po-Lin Chen in European Stroke Journal
Footnotes
Acknowledgements
The authors appreciate all the participants in this study. In addition, authors sincerely thank Professor Shih-Ann Chen, superintendent of Taichung Veterans General Hospital during the conduct of this research, for establishing the Heart-Brain Team and for organizing a collaboration between neurologists, cardiologists, and pediatric cardiologists, whose advanced insights and data contributed to this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Institutional Review Board (IRB) of Taichung Veterans General Hospital (CG19339B). The Institutional Review Board of Taichung Veterans General Hospital granted a waiver for consent.
Consent to participate
The IRB granted a waiver for consent.
Consent for publication
All authors reviewed and approved the manuscript.
Guarantor
Po-Lin Chen, MD, serves as the guarantor for this manuscript. In this role, he assumes full responsibility for the integrity of the work, including the data and its analysis, and guarantees that all aspects of the manuscript meet the highest standards of scientific rigor and ethical conduct.
Contributionship
Chi-Sheng Wang: Conceptualization, data curation, formal analysis, methodology, writing – original draft preparation. Yu-Hsuan Wu: Conceptualization, data curation, methodology, validation. Yun-Ching Fu: Conceptualization, data curation, methodology. Sheng-Ling Jan: Data curation, methodology. Ming-Chih Lin: Data curation, methodology. Chiann-Yi Hsu: Data curation, formal analysis, software. Po-Lin Chen: Conceptualization, funding acquisition, investigation, project administration, resources, supervision, writing – review and editing.
Data availability
The datasets of the current study are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
