Abstract
Background
Paragangliomas are relatively rare vascular tumors that develop from the neural crest cells of carotid bifurcation. They usually present as slow-growing, painless unilateral neck masses; bilateral presentation is rare and is mostly associated with familial forms. Bilateral total resection is not always possible for high-grade bilateral tumors, and radiotherapy is a good alternative, with cure rates similar to surgery.
Case Report
A 35-year-old female patient was admitted with a chief complaint of a bilateral, painless mass located on her neck. Subsequent magnetic resonance imaging (MRI) and angiographic imaging revealed bilateral hypervascular masses surrounding her carotid at 360°, and they were interpreted as stage 3 carotid paragangliomas according to the Shamblin classification protocol. Surgery was carried out on the left carotid paraganglioma and the mass was totally resected. It was thought that the patient could not tolerate bilateral surgery. Primary radiotherapy was planned on the right carotid paraganglioma: 59.8 gray (Gy) conformal, Linac-based multileaf collimator radiotherapy with a 180 cGy daily dosage, and five fractions per week were planned.
Results
Follow-up at 3 months following the conclusion of radiotherapy revealed no significant regression. A follow-up MRI 6 months and 24 months later revealed 59% regression. Grade 2 esophagitis and minimal neck edema were the only complications noted during the course of radiotherapy and during the 24-month follow-up period. No complications or relapse were observed except for edema following neck surgery.
Introduction
Carotid paragangliomas develop from the neural crest cells of the common carotid artery. They most commonly present as a slow-growing, painless mass in the neck, which is located at the carotid bifurcation. Carotid paragangliomas are the most common head and neck paragangliomas 1 that occur at any age, but they are typically diagnosed between the third and sixth decades of life.2–4 These tumors are sporadic 90% of the time, and of the sporadic form, 95% are unilateral; familial forms can be seen in 10% of cases, and the incidence of bilaterality is higher among familial forms. 5 Surgical classification of carotid body tumors was proposed by Shamblin et al. 6 in 1971:
Group 1: relatively small; surgical excision can be performed safely.
Group 2: larger; can be resected with precise surgical dissection.
Group 3: large neoplasms encasing the carotid arteries; can be resected only with arterial sacrifice.
Local invasion or aggressive behavior does not indicate malignancy of the tumor. Metastases to the lymph nodes or distant sites remain the hallmark of malignancy for paragangliomas, and distant metastases have a poor prognosis. 3 Bilateral resection is not possible in higher-grade (Shamblin grade 3) tumors, and contralateral radiotherapy is necessary.
Indium-111 octreotide scanning is useful in the evaluation of paragangliomas given that paragangliomas are neuroendocrine neoplasms and they may secrete somatostatin. Octreotide is a somatostatin analog, and this scanning is sensitive for detecting tumors greater than 1.5 cm. Octreotide scanning is also a useful test that can show multicentric or metastatic paragangliomas. It may also distinguish between scar tissue and residual tumor following surgery. 7
High-power hematoxylin and eosin stain shows a Zellballen growth pattern in paraganglioma specimens 8 S-100 protein immunohistochemical staining is a good indicator of a Zellballen growth pattern. These immunohistochemical studies revealed that malignancy rates are as high as 10%–50%.8,9 It is not always possible to predict malignant behavior based on histologic features alone. Some malignant histologic criteria include necrosis, extensive capsular or vascular invasion, increased mitotic activity, atypical mitotic figures, MIB-1 immunostaining, and loss of the S-100-positive sustentacular cell population.8–10
Recent literature has shown that some paragangliomas have been germline mutations. 11 Molecular studies revealed that six genes have been identified for the germline mutations of paragangliomas. These include RET, VHL, NF1, and succinate dehydrogenase (SDH). SDH subunits are SDHB, SDHC, and SDHD. SDHD and SDHB mutations are very important for head and neck paragangliomas. Paragangliomas may be hereditary and may be a part of genetic syndromes such as multiple endocrine neoplasia (MEN) 2A and MEN 2B, Von Hippel–Lindau syndrome, and neurofibromatosis type 1 (von Recklinghausen disease). Although many familial cases that are often associated with the abovementioned germline mutations go unrecognized, 11 germline mutations have been reported in 7.5%–24% of patients with pheochromocytomas or paragangliomas, and with their sporadic presentation. Plasma or urinary metanephrines and catecholamines should be evaluated for diagnosis and posttreatment biochemical follow-up. 12
Surgical resection is the treatment of choice, but surgery is very difficult because paragangliomas are very vascular tumors. Preoperative embolization and adrenergic blockade should be considered due to the high vascularity and chatecolamine levels of these tumors. Malignant paragangliomas have less than a 50% 10-year survival rate. 7
Clinical Case
A 35-year-old female patient was admitted to the Medical Faculty's Department of Otorhinolaryngology at Bezmialem Vakif University (Istanbul, Turkey) with the chief complaint of a bilateral, painless mass located on her neck. The mass was present and had been gradually enlarging for the last 4 years. A physical examination revealed that the mass was mobile along the horizontal plane, but firm along the vertical plane. Pulsation and murmur was present during auscultation.
Radiology
Magnetic resonance imaging (MRI) revealed a hypervascular 48 × 37 × 43 mm and 40 × 37 × 25 mm mass located on the left and right internal carotid artery (ICA) and external carotid artery bifurcation, respectively (Fig. 1). Magnetic resonance (MR) angiography showed hypervascular contours and flow signals at the same localizations (Fig. 2). The masses were surrounding the carotid arteries at 360°, and they were evaluated as stage 3 carotid paragangliomas according to the Shamblin classification. 6 Surgery was planned on the left carotid paraganglioma.
Octreotide scanning did not show any lymph node or distant metastasis, nor did it show synchronous pheochromocytomas.

Preoperative MR (Axial) (40 × 37 × 25 mm mass).

Preoperative MR Angiography (Coronal).
Pathology
There is founded some malignant histologic criteria that necrosis, perineural invasion (Figure 3), increased mitotic activity, atypical mitotic figures in pathologic specimen. Tumor elements round and oval core, granular large acidophilic cytoplasm and the cells were showing marked pleomorphism. Large areas of tumor hemorrhage, necrosis of small 3 mitosis in the fields. Perineural invasion, widespread non-fatty tissue capsule and capsule invasion was observed. immunohistochemical staining shows a S-100-positive sustentacular cell population. Immunohiystochemical studies did not reveal any SDHD or SDHB mutations.

Pathology, perineural invasion.
Laboratory analysis
No abnormal catecholamines were detected in urine or blood tests.
Surgical procedure
During the surgery, the tumor was firm, abundantly vascularized, and it surrounded the carotid artery completely. The common carotid artery and ICA were dissected after an embolectomy procedure. The tumor was fully resected, along with 5 cm of a carotid segment and saphenous vein graft repair was carried out.
Radiotherapy
Following the surgical procedure, 40 gray (Gy) external local radiotherapy was applied on the paraganglioma on the right side. Tumor dosage to the left side was calculated as 20 Gy at the time of the right-side radiotherapy. Subsequently, after 2 months, conformal radiation therapy (RT) was carried out: 19.8 Gy additional fractioned (180 cGy; eleven fractions) radiotherapy was applied to the right side of the neck with 6 MV photons. The left side of the neck received 15 fractions of 27 Gy total irradiation (Fig. 4). The total radiotherapy dosage was calculated; 59.8 Gy and 47 Gy three-dimensional conformal radiotherapy was applied to the right and left side of the neck, respectively.
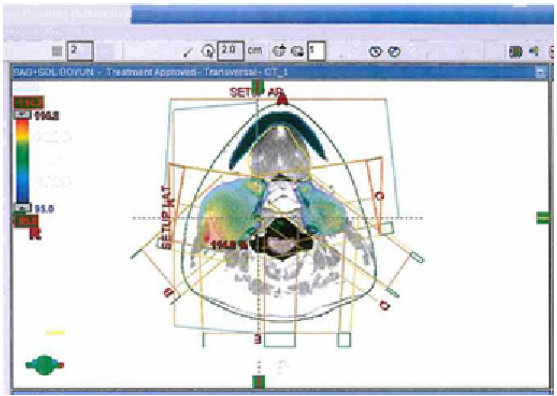
Radiotherapy 3D planning Isodose Curve, horizontal section.
Results
A neck MRI scan was obtained following the conclusion of radiotherapy at the 6th postoperative month: the carotid paraganglioma located on the right carotid bifurcation was 31 × 21 × 23 mm (Fig. 5). The size of the right carotid paraganglioma regressed by 59%. This response continued throughout the 24-month follow-up period. No tumor relapse occurred on the left-side tumor during this period. The patient was content with her general health status and quality of life, and the swelling resolved eventually. Grade 2 esophagitis and local edema were the only complications observed during the course of radiotherapy and during the 24-month follow-up period. No complications were observed except for edema following the neck surgery.

Right carotid bifurcation following adjuvant radiotherapy 6th month after conclusion of radiotherapy (31 × 21 × 23 mm mass).
Discussion
The extirpation of carotid paragangliomas requires careful preoperative evaluation given that paragangliomas are very rich vascular tumors and are surrounded by significant neurovascular structures. 13 Bilateral surgery can lead to disastrous postoperative complications such as stroke and myocardial infarction due to baroreflex failure. 14 In Shamblin group 2 or 3 patients, the risk of carotid vessels and cranial nerve injury was approximately 40%–50% due to the surgery. Large tumors are more difficult to resect, and they were associated with a significantly higher rate of neurovascular injury. 15
Bilateral total resection was not possible in this case since they were bilateral and high-grade tumors (grade 3 according to the Shamblin classification). 6 Therefore, radiotherapy was applied to the paraganglioma on the right side. Radiotherapy was also applied to the paraganglioma on the left side post-operatively because the tumor met some malignancy criteria. Radiotherapy has been shown to be effective in the long term: tumor control rates were reported as being similar to those achieved with surgery.16–19
Initially, paragangliomas may not respond to radiotherapy but, eventually, these tumors become smaller over a period of 2 years. Long-term cure rates are similar for surgery and radiotherapy.16–19 Kyrch et al. 17 reported the tumor control results associated with radiotherapy (external beam RT and stereotactic radiosurgery [SRS] methods) over a follow-up period of 10 years; the tumor control rate was 92%.
Mendenhall et al. 19 reported the results of a comparison between radiotherapy and surgery: radiotherapy was applied in a wide isodose 30–70 Gy range. Although the radiotherapy group was comprised of inoperable and large-sized tumors, similar results were obtained when compared to surgery (73% versus 78%, respectively). Kim et al. 20 reported the correlation between response rates: the recurrence rate was 1% above 40 Gy and 25% below 40 Gy. The consensus regarding the optimal dosage was 45 Gy.
SRS will probably be used as the primary treatment for new and recurrent paragangliomas, as its median local control was 100%. The complication rate was 3%; most complications included an increase in vertigo and transient cranial neuropathies. A surgical series described a lower local control rate and the onset of severe complications. A RT series showed a slightly lower local control rate than SRS. 18
Response rates in our clinical case were concurrent with those in the literature.17,19 Persistence of the tumor should not always be evaluated as a tumor; fibrosis of the tumor can result with a persevering mass. The stationary state of the mass can be interpreted as a successful outcome. The current case was positive for some closely malignancy criteria; therefore, 59.8 Gy, which is a relatively high dose, was preferred. Further multicentered studies are essential for understanding malign paragangliomas.
Author Contributions
Designed the study: HSK. Suggested the main idea: HSK. Acquired the data: AHE, BV. Wrote the manuscript: HSK, BO. Edited the manuscript: HSK, BO, AHE, BV. All authors reviewed and approved of the final manuscript.
