Abstract
Acute renal infarction is still an underdiagnosed pathology. Most cases are secondary to arterial embolism in patients with atrial fibrillation or other cardiac illnesses; however, a less known etiology is the vascular affection of systemic lupus erythematosus (SLE). Renal infarction in lupus patients normally appears with positive antiphospholipid antibodies or lupus anticoagulant in the context of an antiphospholipid syndrome (APS). This is characterized by a state of hypercoagulability potentially affecting all segments of the vascular bed with thrombosis. A differential diagnosis with lupus nephritis, a very common pathology in SLE patients, must be carried out.
We have to suspect this pathology in patients with SLE and APS who come to the emergency department complaining of abdominal pains or a renal colic.
We present the case of a 69-year-old woman who was diagnosed of bilateral segmental renal infarction in the context of recently diagnosed SLE with no other vascular manifestations.
Keywords
Introduction
Many patients present daily to the emergency department complaining of abdominal or flank pain. The differential diagnosis in these cases is quite extended. Renal colic, gastrointestinal diseases, and appendicitis are the most suspected affections in this situation. However, other pathologies such as abdominal or retroperitoneal viscerae infarctions should be also taken into consideration. 1
Currently, acute renal infarction is still an underdiagnosed pathology. Most cases are secondary to arterial embolism in patients with atrial fibrillation or other cardiac illnesses. A less known aetiology is the vascular affection of systemic lupus erythematosus (SLE). 2
We present the case of a 69-year-old woman who was diagnosed with a bilateral segmental renal infarction in the context of recently diagnosed SLE with no other vascular manifestations. Written consent was obtained from the patient to reproduce information appearing in this work.
Case Report
A 69-year-old woman with a recent history of SLE presented to the emergency department complaining of pain in the left renal fossa irradiated to the ipsilateral iliac region and with sickness and vomiting. The symptoms had begun 5 days prior to the visit, but the intensity of the pain had increased acutely over the last 12 hours. The patient denied having suffered fever episodes, haematuria, or dysuria, or previous renal colics. Her medical history consisted of atrial fibrillation treated with acenocumarol. Six months before, Anti-Nuclear Antibodies were negative with slightly positive IgM anticardiolipin antibiodies and negative lupic anticoagulant.
Physical examination revealed a mild positive palpation at the right hypochondrium and a slightly positive ipsilateral renal percussion. The patient didn't complain of any discomfort on the left side during palpation. No edema or malar rash were identified.
A 12-lead electrocardiograph showed an irregular rhythm at 80 bpm in the context of her atrial fibrillation with regular QRS complex and no ST segment changes.
Complete urgent blood and urine tests were performed. The results were: urea, 43 mg/dL (normal range, 10–45); creatine, 0.84 mg/dL (normal range, 0.5–1.1); lactate dehydrogenase (LDH), 879 U/L (normal range, 135–250); C-reactive protein, 10.71 mg/dL (normal range, 0–5); hemoglobin, 15.5 g/dL (normal range,12–16); leukocytes, 10980/μL with 88.2% neutrophiles (normal range, 4500–10,800); platelets, 226,000/μL (normal range, 150,000–450,000); International Normalized Ratio, 2.01; activated partial thromboplastin time (APTT), 35.7 s (normal range, 27–40); urine pH, 6.5; proteins, 75 mg/dL; urine red cells 65/μL; bacteria, 19.1/μL.
An ultrasonographic examination using a 3 hertz convex transducer was performed. The patient was explored with B-mode and Doppler techniques showing symmetrical renal size and preserved parenchymal thickness. No parenchymal echogenicity abnormalities were seen in the right kidney; however, its lower pole showed mild parenchymal heterogeneities and a poor vascular pattern (Fig. 1).
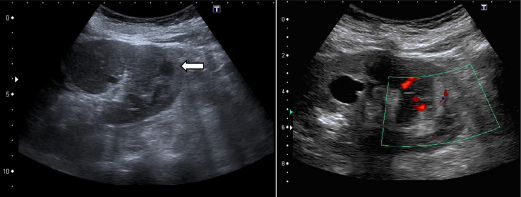
The lower pole of left kidney shows a non cystic, cortical heterogeneity (white arrow). The vascular pattern suggests a poor blood support. A cortical cyst is seen in the middle region.
The unspecific results of the previous examination led to the performance of a nonenhanced and postcontrast (120 cc of Ioversol) comuted tomography (CT) scan. Parenchymal phase showed an anterior, hypovascular area in the right kidney, with geographical limits, and the heterogeneous enhancement of the posterior region of the left kidney (Fig. 2).
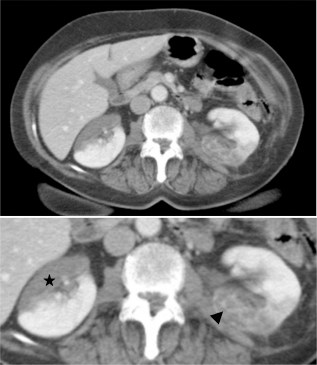
Axial contrast-enhanced image in the parenchymal phase shows an anterior, hypodense area in the right kidney, strongly suggestive of renal infarction (asterisk). The posterior region of the left kidney is heterogeneously hypodense, with fine enhancing lines (arrowhead), consistent with the Doppler-US findings showing persistence of poor vascular flow in this area.
The patient was admitted to the urology department, and treatment was begun with continuous infusion of unfractionated heparin with daily determinations of APTT. Control blood tests showed an increase in LDH levels up to 922 U/L and normal renal function. IgM anticardiolipine antibodies were also detected at 14.1 MPL (normal range, 0–10). A complete thrombosis study was performed. The results were: fibrinogen, 696 mg/dL (normal range, 130–400); VIII factor, 155% (normal range, 60–120); von Willebrand factor, 160% (normal range, 60–120); and XI factor, 95% (normal range 60–120); positive unspecific antiphospholipid antibody. The patient's sickness and pain then disappeared.
A control CT scan was performed 13 days after admission, with no significant changes in the size of the hypoecogenic area or any new other lesions. The patient was discharged at day 14 with daily 5000 U of bemiparin sodium.
Discussion
Lupus is a chronic inflammatory disease affecting 4 to 250 per 100 000 inhabitants, where 70% of patients correspond to SLE. It is 9 times more frequent in women than in men. The first symptoms appear when the patient is between 15 and 44 years old. Approximately 30% to 50% of cases show clinical manifestations of renal damage, but up to 85% of biopsies or necropsies already show changes in patients with none-declared kidney affections. 3
It is well-know the frequent renal involvement in SLE, lupus nephritis. Glomerular pathological changes are emphasized and vascular lesions are sometimes overlooked at the World Health Organization (WHO) classification of this pathology, although their presence can affect the prognosis of the renal disease. 4
The lupus vasculopathy has been reported to be associated with the WHO class IV lupus nephritis. 5 Renal infarction in lupus patients normally appears with positive lupus anticoagulant in the context of an antiphospholipid syndrome, which is characterized by a state of hypercoagulability potentially affecting all segments of the vascular bed with thrombosis.6,7
Renal infarction in lupus patients normally appears with positive antiphospholipid antibodies or lupus anticoagulant8,9 in the context of an antiphospholipid syndrome (APS). This syndrome is characterized by a state of hypercoagulability potentially affecting all segments of the vascular bed with thrombosis. 10 Renal thrombotic events were among the first reported manifestations. 6 In a cohort of 1000 patients with APS, renal thrombotic complications were reported in only 2.7% of cases. 11 According to the revised classification criteria for the antiphospholipid syndrome, our patient would meet one clinical and one laboratory criteria. 7
Imaging findings of lupus-related kidney disease are unspecific and usually depend on the stage of the illness. The differential diagnosis of a renal infarction in a patient with SLE should include lupus-related nephritis. 12
Ultrasound is unable to specifically diagnose acute glomerulous damage, although a loss of corticomedullary differentiation with an hypoechoic cortical rim due to edema has been reported in lupus nephritis. The Doppler technique provides a good overview of the abnormalities of the vascular pattern and information of the parenchymal consequences of vascular involvement (renal enlargement, triangular hypoechogenic areas). 13
We suggest that the CT standard protocol for the diagnosis of a nonsuspected kidney disease should also include precontrast and postcontrast images. In a previously known SLE, renal failure is particularly something to take into account. Nonenhanced CT will allow the radiologist to rule out the presence or renal haemorrage, calcifications, or masses. Administration of intravenous contrast is needed to depict vascular abnormalities and irregularities of parenchymal enhancement. 12
An abnormal enhancement pattern with wedge-shaped hypodensities can be seen either in renal lupus nephritis or focal infarction. Although the diagnosis of lupus nephritis is mainly clinical and anatomopathological, its appearance on image tests can lead to misinterpretation. The cortical rim sign is a late feature of infarction, which reflects the blood support from collateral circulation. 12 All these findings must be correlated with clinical and laboratory data.
Conclusions
Renal infarction is an unusual pathology that must be taken into account in the context of abdominal pain. This suspicion is bound to be greater in patients with LES and positive antiphospholipid antibodies because of the state of hypercoagulability. Other diagnoses that must be considered are intra-abdominal haemorrhage (as the patient is on anticoagulation) and peritoneal serositis.
A differential diagnosis between renal infarction and lupus nephritis is sometimes difficult and can be clarified by anatomopathological studies. Imaging findings can help in showing occluded vessels, but they mostly show unspecific signs.
Renal infarction does not always lead to renal functional impairment, but patients must be closely followed up.
Author Contributions
Wrote the first draft of the manuscript: BPF, DGC. Contributed to the writing of the manuscript: MFLG, MMI. Agree with manuscript results and conclusions: CMR, JGG. Jointly developed the structure and arguments for the paper: BPF, DGC, CMR. Made critical revisions and approved final version: MFLG, MMI. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
