Abstract
Growth differentiation factor 5 (GDF-5), a member of the transforming growth factor beta (TGF-β) superfamily expressed in periodontal tissues, promotes extracellular matrix (ECM) in periodontal ligament (PDL) cells. Matrix metalloproteinases (MMP) are proteolytic enzymes that degrade ECM and are expressed in PDL cells. To date, little is known about the regulation of MMP synthesis and secretion in PDL cells. The aim of this study is to examine the effects of GDF-5 on MMP production and activity in PDL cells. GDF-5 increased both collagen type I alpha 2 (Col Iα2) and MMP-2 gene expression in cells derived from mouse PDL tissues after 3 days of culture. Because PDL cells represent a heterogeneous population, we examined gelatinolytic activity and gene expression profiles in an osteoblast cell line. After 6, 12, 24, and 48 hours of culture, GDF-5 increased both Col Iα2 and MMP gene expression in osteoblasts. GDF-5 also promoted MMP-2 activity as revealed by gelatin zymography after 7 days of culture. In the presence of the p38 MAP kinase inhibitor SB202190, on the other hand, MMP-2 activity was blocked. Taken together, these results indicate that GDF-5 may increase simultaneously the gene expression of type I collagen and MMP-2 in the osteoblast-like cells among the PDL cells and p38 MAP kinase pathway in osteoblast could involve in the regulation of MMP-2 enzyme activity induced by GDF-5.
Keywords
Introduction
Matrix metalloproteinases (MMP) are a family of proteolytic enzymes involved in the remodeling of extracellular matrices (ECM), which are composed of collagens, laminins, fibronectins, and proteoglycans. These enzymes play a role in normal morphogenesis, including osteogenesis, and tissue remodeling, in which modification of the ECM and thereby of the microenvironments surrounding various types of cells is required for cell migration, proliferation, differentiation, and apoptosis. Among the MMP, many studies have focused on the gelatinases because of their various functions in pathological conditions as well as physiological events. The active gelatinases, namely MMP-2 and MMP-9, degrade denatured collagen (gelatin), native collagen (including type I) and proteoglycan core protein. The activation of these MMP occurs through multiple steps: they are initially synthesized as pre-proenzymes each containing a predomain, and are then secreted as inactive pro-MMP-2 and -9. Pro-MMP-2 and -9 are fully activated as MMP-2 and MMP-9 through the disruption of the bridge between the zinc ion and the cysteine contained in the predomain, which maintains the pro-MMP in a latent zymogenic form. 1 The expression of MMP-2 and MMP-9 is regulated by hormones and growth factors, including bone morphogenetic proteins (BMPs).2–7
Growth differentiation factor 5 (GDF-5), also known as cartilage-derived morphogenetic protein 1 (CDMP-1), is a member of the BMP family, which belongs to the transforming growth factor beta (TGF-P) superfamily Mutations of GDF-5 have been identified in association with skeletal growth and development in mice 8 and humans. 9 In addition to the chondro- and osteoinductive functions of GDF-5 and other BMPs both in vivo 10 and in vitro, 11 GDF-5 has been demonstrated to have a specific induction capacity for tendon- and ligament-like tissue when implanted ectopically in animals. 12
Periodontal tissue is a specialized supporting apparatus that consists of hard and soft tissues, including cementum, alveolar bone, and periodontal ligament (PDL). PDL, which plays a critical role in anchoring the tooth-covered cementum and the alveolar bone of the jaw, contains a heterogeneous mix of cell populations that include fibroblasts, cementoblasts, osteoblasts, osteoclasts, odontoclasts, and epithelial cell rests of Malassez;13–15 these cells are involved in PDL homeostasis. Several studies have demonstrated that PDL cells have an osteoblast-like or cementoblast-like cell character, expressing or secreting ECM including collagen type I alpha 2 (Col Iα2), osteopontin, and osteocalcin under regenerative conditions in vitro. 16 This osteoblastic or cemento-blastic cell character contributes to the regeneration of PDL as a functional attachment tissue that forms mineralized tissues on both the root surface and the alveolar bone. 17 To induce regeneration of the PDL through ordinary developmental processes including cell migration, proliferation, and differentiation, a number of studies have suggested the application of various growth factors such as platelet-derived growth factor (PDGF), insulin-like growth factor (IGF), basic fibroblast growth factor (bFGF), transforming growth factor beta (TGF-P), and BMP-2.18–21 Because GDF-5 is expressed in the development of PDL22,23 and ECM production is promoted by GDF-5 in cells derived from PDL, 24 recent studies have suggested that GDF-5 may be useful in wound healing or in the regeneration of periodontal tissues.25,49 MMP-2 and MMP-9 are also expressed in periodontal tissue wound healing,50 yet no studies have focused on the association of MMP expression with GDF-5. The purpose of this investigation is to determine whether GDF-5 promotes MMP and ECM production in PDL cells and osteoblasts.
Materials and Methods
Cell Culture
All animal studies conformed to the approved Animal Study Protocols of the Fukuoka Dental College (approval #08032). The mandibular first molars of C57BL/6J mice (Kyudo Co. Ltd., Tosu, Japan) were extracted by dissecting the molars with adherent PDL from the surrounding alveolar bone. The isolation of cementoblast (CMB)/PDL cells was carried out as described previously 13 with minor modifications. Briefly, molars were immersed in a 1.5-ml centrifuge tube containing phosphate buffered saline (PBS) with 1 mg/ml collagenase (Invitrogen, Tokyo, Japan) and 0.25% trypsin-EDTA (Invitrogen) and incubated for 2 h at 37 °C. After incubation, CMB/PDL cells were collected and seeded on a 6-well cell culture plate at 4.0 × 10 5 cells per well. After 4 days of culture, the cells in each well were subjected to experimentation. The cell culture medium for PDL cells was alpha modified Eagle's minimum essential medium (αMEM, Invitrogen) containing 10% fetal bovine serum (FBS, Equitech-Bio Inc., Tokyo, Japan) and antibiotics (100 U/ml of penicillin-G and 100 mg/ml of streptomycin) at 37 °C in a humidified atmosphere of 5% CO2 in air.
Cells from the mouse osteoblast cell line MC3T3-E1 were obtained from Riken Bioresource Center Cell Bank (Tsukuba, Japan). Osteoblast cells were cultured in the same medium as the CMB/PDL cells under identical conditions. The medium was changed twice per week. Before reaching confluence, the cells were passaged for use in experiments.
For each experiment, the medium was changed to high glucose Dulbecco's Modified Eagle Medium (D-MEM) (Invitrogen Life Technologies, Tokyo, Japan), containing 10% FBS and antibiotics (100 U/ml of penicillin-G and 100 mg/ml of streptomycin). Cells were treated with various concentrations of recombinant mouse GDF-5 (R&D Systems, Minneapolis, MN, USA) diluted in 0.1% bovine serum albumin (BSA)—Hank's balance salt solution (HBSS) and cultured for the indicated time.
Cell Proliferation
Proportional numbers of cells were determined using a Cell Counting Kit-8 (Dojindo Laboratories, Kuma-moto, Japan), which depends on the measurement of a highly water-soluble formazan dye produced from tetrazolium salts. Osteoblasts were seeded at 1.0 × 10 3 cells per well on a 96-well plate and the medium, which included 100 or 1000 ng/ml of GDF-5, was changed after one day of culture for cell attachment. We also used fibroblast growth factor basic mouse recombinant (bFGF) (R&D Systems, Minneapolis, MN) at 50 ng/ml diluted in 0.1% BSA-HBSS. After 1, 3, or 7 days of cell culture, cells were incubated with counting reagent for 1 h according to the manufacturer's instructions. The relative cell number was then determined by measuring light absorbance at a wavelength of 450 nm (PerkinElmer, 1420 Multilabel couter ARVO MX, MA, USA) using the formazan dye product in the cultures.
Gel Zymography
Osteoblasts were cultured in the absence or presence of various concentrations of GDF-5. For the inhibitory experiment, the p38 MAP kinase inhibitor SB202190 (Merck Bioscience, San Diego, CA, USA) was used, and the cell culture medium was added to a final volume of 20 μM with or without GDF-5. After 7 days of culture, the culture medium was changed to a serum-free medium containing only antibiotics and incubated for 8 h at 37 °C in a humidified atmosphere of 5% CO
Each serum-free conditioned medium was collected. Aliquots of 10 μl were treated with or without 10 mM
Reverse transcription polymerase chain reaction (RT-PCR)
CMB/PDL cells or osteoblasts were cultured in the absence or presence of 500 ng/ml of GDF-5 in D-MEM containing 10% FBS and antibiotics. After 6, 12, 24, or 48 hours of culture for osteoblast cells and 3 days of culture for CMB/PDL cells, total RNA was isolated from the cultured cells using an RNeasy Mini Kit (Qiagen, Inc., Valencia, CA, USA) according to the manufacturer's instructions. One-step reverse transcription polymerase chain reaction (RT-PCR) was performed using a SuperScript One-Step RT-PCR with a Platinum Taq kit (Invitrogen) according to the manufacturer's instructions, and an MJ Mini Thermal Cycler PTC-1148 (Bio-Rad, Tokyo, Japan). The gene expression levels of collagen type I alpha 2 (Col Iα2), MMP-2 and MMP-13 were assessed relative to that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Amplimers were designed as follows: for mouse MMP-2 antisense primer, 5′- ACC ATC GCC CAT CAT CAA-3′, base pairs 953–970; for sense primer, 5′- ACT TTA CGC GGA CCA CTT-3′, base pairs 387–404 (Genebank Accession number NM-008610); for mouse MMP-13 antisense primer, 5′-TGC GGG AAT CCT GAA GAA-3′, base pairs 169–186; and for sense primer, 5′- TCA GCA GTG CCA TCA TAG-3′, base pairs 481–498 (Gene Accession number NM-008607). The amplimers designed for Col Iα2 and GAPDH have been previously reported (Hatakeyama et al, 2004). The PCR products were subjected to electrophoresis. Digital images were obtained and analyzed using ImageJ software (NIH, Bethesda, MD, USA). Three cell populations of each cell type were studied.
Statistical Analyses
All data are expressed as the mean ± standard deviation (SD). Statistical significance was determined using one-way analysis of variance (ANOVA) followed by a post-hoc
Results
In order to examine whether GDF-5 regulates MMP-2 gene expression in PDL cells, we performed RT-PCR on PDL cells after one, 2 and 3 days of culture. After 3 days of culture, the gene expression level of MMP-2 was increased significantly in CMB/PDL cells cultured in the presence of GDF-5 (Fig. 1A). GDF-5 also increased the gene expression level of Col Iα2 under the same culture conditions (Fig. 1B). After one and 2 days of culture, the gene expression of neither MMP-2 nor Col Iα2 was increased significantly (data not shown).
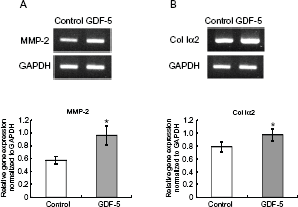
GDF-5 promotes gene expression in both MMP-2 and collagen type I alpha-2 (Col Iα2) in PDL cells. PDL cells were isolated from first molars extracted from mice and cultured for 3 days in the absence (Control) or presence (GDF-5) of 500 ng/ml of GDF-5. Relative gene expression was assayed by RT-PCR and normalized to that of GADPH. Levels of both MMP-2 (
The CMB/PDL cells isolated from the PDL tissues utilized in this study were a heterogeneous cell population. Based on previous reports that GDF-5 causes no obvious promotion of fibroblast cell proliferation in vitro, 26 we tested whether GDF-5 promotes osteoblast cell proliferation in an assay of general cell activity. At any time point, 1, 3 and 7 days of culture, GDF-5 at dose of 100 and 1000 ng/ml did not increase osteoblast cell numbers significantly compared with the control (Fig. 2, GDF-5 and Control). In the presence of 50 ng/ml bFGF, on the other hand, osteoblast cell numbers were significantly increased after 1 day of culture compared with the control (Fig. 2, bFGF 50 ng/ml and Control) as well as at subsequent time points (bFGF 50 ng/ml, 1, 3, and 7 days).
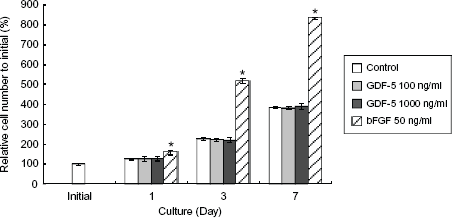
GDF-5 had no effect on osteoblast cell proliferation. Osteoblast cells were cultured in the absence (Control) or presence of GDF-5 at doses of 100 and 1000 ng/ml. Relative cell numbers were assayed by absorbance of formazan dye; the number present at initial cell culture was taken as 100%. Neither 100 nor 1000 ng/ml of GDF-5 increased cell numbers after culture for 1, 3, or 7 days. bFGF as a positive control resulted in increased cell numbers in all cell culture periods.
We also tested whether or not GDF-5 promotes gelatinolytic activity in osteoblasts using gelatin zymography. In all samples obtained from a serum-free medium after 7 days in the absence or presence of GDF-5, no pro-MMP-9 was detected in conditioned-medium-cultured osteoblasts through gelatin zymography (Fig. 3, pro-MMP-9). MMP-9 activated with APMA, which was evident at a mass of 92 kD, was nevertheless not detected under these conditions (Fig. 3, pro-MMP-9, APMA-activated). Both pro-MMP-2 and MMP-2 were detected in the conditioned medium after 7 days of culture, but not in the initial culture period (Fig. 3, Int). After 7 days of culture, MMP-2 activity was increased in the presence of a high concentration of GDF-5, while MMP-2 activated with APMA in the conditioned medium was increased in the presence of GDF-5 at all doses (Fig. 3, APMA-activated), compared to their levels after culture in the absence of GDF-5 (Fig. 3, APMA-activated, 0 ng/ml).
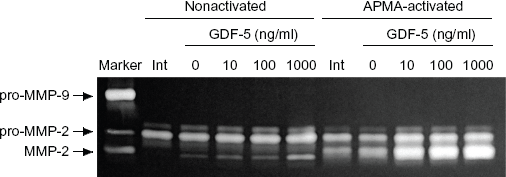
GDF-5 promoted gelatinolytic enzyme activity. Osteoblasts were cultured in the absence (0) or presence of GDF-5 at 10, 100, and 1000 ng/ml for 7 days. Gelatinolytic activity was assayed by zymography containing gelatin with a loaded conditioned medium treated without (nonactivated) or with APMA (APMA activated). Cell culture medium at the initial (Int) time point had no MMP-2 activity. During culture, however, GDF-5 promoted MMP-2 activity at each concentration (left panel, nonactivated, 10, 100, and 1000). Activation of pro-MMP-2 by APMA resulted in an increase in latent MMP-2 in the conditioned medium (right panel, APMA activated, 10, 100, and 1000).
In order to investigate how GDF-5 increases MMP-2 production and activity, and whether GDF-5 increases the levels of other MMP and ECM in osteoblasts, we performed RT-PCR for MMP-2, MMP-13, and collagen type 1α2. In the presence of 500 ng/ml GDF-5 in the medium, the gene expression level of MMP-2 was significantly increased at all time points, ie, 6, 12, 24, and 48 h (Fig. 4A). MMP-13 was expressed in a pattern similar to that of MMP-2 (Fig. 4C). The gene expression level of Col 1α2 was also significantly elevated at all time points, similar to that of MMP (Fig. 4B).
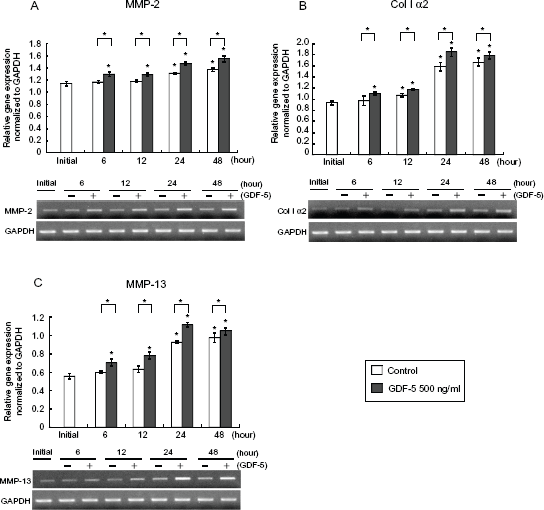
GDF-5 increased gene expression levels of MMP and Col Iα2. Osteoblasts were cultured in the absence (0 or Control) or presence of GDF-5 at 500 ng/ml (500 or +). Gene expression levels were assayed by semiquantitative RT-PCR in total RNA isolated from cultured osteoblasts at each time point. MMP-2 (
To study the mechanism by which GDF-5 regulates MMP-2 production and activity, we performed gelatin zymography in the conditioned medium in the presence of p38 MAP kinase inhibitor. Osteoblasts were cultured for 7 days in the absence or presence of 1000 ng/ml GDF-5 in the medium and/or p38 MAP kinase inhibitor SB202190 diluted in dimethyl sulfoxide (DMSO). After 7 days of culture, MMP-2 was observed only in the APMA-activated medium sample (Fig. 5, 9th line from left); it was not observed in the conditioned medium of osteoblasts cultured with only SB202190 (Fig. 5, 4th line from left). Also, the level of pro-MMP-2 was dramatically lower in the presence of SB202190 than in control cultures (Fig. 5, 1st line from left). This decrease was not reversed by the presence of 1000 ng/ml of GDF-5 (Fig. 5, 5th line from left). DMSO, the chemical carrier of SB202190, resulted in slight decreases in pro-MMP2 and MMP2 activity (Fig. 5, 4th line from left). In all of the samples obtained in each experiment group, pro-MMP-2 was activated to MMP-2 by means of APAM treatment (Fig. 5, APAM-activated).
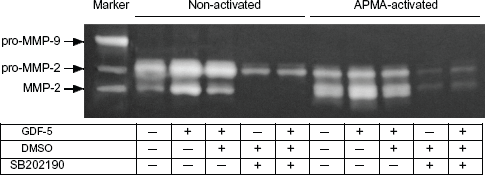
MMP-2 enzyme activity induced by GDF-5 was inhibited by p38 MAP kinase inhibitor. Osteoblast cells were cultured for 7 days, after which each conditioned medium was subjected to zymography containing gelatin. MMP-2 enzyme activity was observed in both the absence (indicated by—in table, 1st line from left) and the presence of 1000 ng/ml of GDF-5 (indicated by
Discussion
Various growth factors that affect MMP and ECM production in PDL cells have recently been examined as a means of evaluating cellular regulation in tissue regeneration in PDL. GDF-5 is one of the growth factors that promotes the production of ECM in cells derived from PDL tissues in vitro; 24 MMP are proteolytic enzymes involved in the remodeling of ECM. Previous studies have reported that GDF-5 22 and MMP 27 are both expressed in the development and repair of Our study demonstrated that GDF-5 increased the gene expression of Col Iα2 mouse primary CMB/PDL cells after 3 days of culture (Fig. 1). This result is similar to those of previous studies utilizing TGF-β.28,29 TGF-P down-regulated the expression of type I collagen mRNA after 3 days of culture, but increased it after 1 day of culture, in osteoblast-like PDL cells isolated from human PDL containing a heterogeneous cell population. 28 Thus, we assayed an osteoblast cell line to clarify the effect of GDF-5 on type I collagen gene expression. The gene expression level was significantly increased after 24 h and 48 h (Fig. 4). TGF-β1 transduces its signal through a combination of heteromeric complexes of two different types of serine/threonin kinase receptors, the TGF-β type I and type II receptors; the receptors of GDF-5, in contrast, are BMP receptor type IB (BMPR-IB) and BMPR-II or Activin type I receptor (ActR-II). 30 This difference may affect the profile of type I collagen gene expression in CM/PDL cells and osteoblasts, and may result in the increase in type I collagen gene expression that occurs after 3 days of culture. In addition, the fact that the ligands use different receptors may affect the expression of MMP-2 in CM/PDL cells. In human PDL fibroblasts, TGF-P may suppress MMP-2 production and secretion. 31 Therefore our results suggests that the increase in Col Iα2 gene expression may be dependent on the effect of GDF-5 on osteoblast-like PDL cells in a heterogeneous mix cell population. The effect on type I collagen may persist through 3 days of culture because of the different combinations of receptors required by GDF-5 and TGF-β1 as well as MMP-2 gene expression.
It has been reported that GDF-5 increases PDL cell numbers, 24 but it remains unclear whether GDF-5 acts on osteoblast-like PDL cells or on other cell components of PDLs such as fibroblasts. TGF-β1 as well as GDF-5 increases PDL cell numbers, 32 but the effect of TGF-β1 on osteoblast cell proliferation is dependent on the condition of the cell culture-like serum that is present in the medium, the stage of cell differentiation, 33 and the expression pattern of receptors for TGF-β1, 34 even for an osteoblast cell line that includes MC3T3-E1. Our results have revealed that GDF-5 has no effect on osteoblast cell numbers at a dose of either 100 or 1000 ng/ml in any period of osteoblast cell culture. These results suggest that GDF-5 could have a distinct cell proliferation effect, not on osteoblast-like cells, but on other cell components of PDL. It has been reported, however, that GDF-5 induces a very weak antiproliferative effect in fibroblasts rather than promoting cell proliferation. 35 Further studies are thus required to clarify the mechanism of cell proliferation induced by GDF-5 in fibroblasts isolated from PDL tissues.
In human PDL fibroblasts, TGF-P suppressed MMP-2 production and secretion 31 while TGF-β decreased MMP-2 gene expression. 29 In osteoblasts, TGF-β did not alter MMP-2 secretion, although gelatin zymography revealed MMP-2 activity but not MMP-9 activity. 36 Various studies have examined the expression and secretion of MMP-9 in osteoblasts. MMP-9 mRNA was not detected in osteoblastic cells by means of in situ hybridization, 37 but secreted MMP-9 expression was identified in tibial-derived rat osteoblasts by means of gelatin zymography and Western blotting of culture medium. 38 Our results have revealed that GDF-5 causes potent promotion of MMP-2 activity, though neither promotion nor induction of MMP-9 in osteoblasts was revealed by gelatin zymography; GDF-5 was also demonstrated to promote MMP-2 gene expression. These results suggest that GDF-5 could increase the gene expression and activity of MMP-2 in osteoblast-like cells among the PDL cells without affecting the gene expression of MMP-2 in other components of PDL such as fibroblasts.
The expression of MMP-2 is regulated not only by TGF-P but also by BMP-2,2,5,6 BMP-4, 7 and BMP-7 3 in various cells. Among these growth factors and hormones, BMP-2 and -7 possess functions similar to that of GDF-5 in chondrogenesis and osteogenesis in that they suppress MMP-2 mRNA in osteoblasts on a collagen scaffold. 6 Also, BMP-2 does not increase MMP-2 levels, as revealed by zymograms and immunoblots in a conditioned medium of chondrocyte cell culture. 2 Our results have demonstrated that GDF-5 promotes MMP-2 activity in osteoblasts on a normal plastic coat plate. This result suggests that MMP-2 production and increasing activity may depend on the ECM around the osteoblasts: specifically, GDF-5 could promote MMP-2 activity in the absence of ECM and could also simultaneously promote the production of type I collagen. Another possible explanation of the difference between the effects of BMP-2 and GDF-5 on the production and activity of MMP-2 in osteoblasts may be their different signaling pathways. The receptors for BMP-2, BMP-4, and GDF-5, as well as for TGF-β, are BMPR-IA, BMPR-IB, and ActR-I and -II; BMPR-II and Smads are known to be the downstream targets of these receptors. 30 It has been reported that BMP-4 binds to BMPR-IA while BMP-2 binds to both BMPR-IA and BMP-IB, but GDF-5 binds preferentially to BMPR-IB and not at all to BMPR-IA in osteoblasts. 30 It has been suggested that different BMP type I receptors regulate different Smads in the induction of alkaline phosphatase activity, which is one of the markers of osteoblast differentiation. 39 Other signaling pathways may be involved in MMP activity promoted by GDF-5, given that various studies have demonstrated that the induction of MMP (including MMP-2, MMP-9, and MMP-13) involves the p38 MAP kinase signaling pathway in various types of cells under chemical or mechanical stress or responding to signaling molecules.40–42 It has been reported that chondrogenesis induced by GDF-5 may involve the p38 MAP kinase pathway and that chondrogenesis is inhibited by the p38 MAP kinase inhibitors SB202190 and SB203580.43,44 It has also been suggested that p38 MAP kinase could be involved in the transcription of osterix, which is essential for osteoblast differentiation induced by BMP-2. 45 Our data have revealed that MMP-2 activity was blocked by SB202190 despite the simultaneous presence of GDF-5. Therefore these results indicate that GDF-5 could regulate the gene expression of gelatinolytic MMP and the p38 MAP kinase signaling pathway, which may involve MMP-2 activity induced by GDF-5 in osteoblasts.
It has been suggested that MMP-2 stimulated by TGF-P may remove degraded and denatured collagen at new matrix formation sites. 46 Another study has suggested that MMP-2 functions in PDL cell motility given its specific degradation of type I collagen. 47 Indeed, it has been reported that MMP-2 cleaves native type I collagen under appropriate conditions. 48 In addition to its effect on PDL cells, MMP-2 is also involved in osteogenesis and fracture healing, both in vivo and in vitro. 46 Recent studies have also examined the possibility of applying GDF-5 to promote wound healing and the regeneration of periodontal tissues.25,26 These reports suggest that the function of MMP-2 stimulated by GDF-5 in osteoblast-like cells in PDL is the regulation of matrix formation in PDL and that GDF-5 therefore has a potential application in PDL regeneration. The effect of GDF-5 on other component cells of PDL remains unclear, however. Further studies are required to clarify the role of GDF-5 in MMP expression and activity in PDL before its application in regeneration can be considered.
Footnotes
Acknowledgments
This work has been supported by the Advanced Science Research Center and by Grants-in-Aid for Scientific Research (No. 23592726) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
