Abstract
Background
MammaPrint® is a microarray-based gene expression test cleared by the US Food and Drug Administration to assess recurrence risk in early-stage breast cancer, aimed to guide physicians in making neoadjuvant and adjuvant treatment decisions. The increase in the incidence of invasive lobular carcinomas (ILCs) over the past decades and the modest representation of ILC in the MammaPrint development data set calls for a stratified survival analysis dedicated to this specific subgroup.
Study Aim
The current study aimed to validate the prognostic value of the MammaPrint test for breast cancer patients with early-stage ILCs.
Materials and Methods
Univariate and multivariate survival associations for overall survival (OS), distant metastasis-free interval (DMFI), and distant metastasis-free survival (DMFS) were studied in a study population of 217 early-stage ILC breast cancer patients from five different clinical studies.
Results and Discussion
A significant association between MammaPrint High Risk and poor clinical outcome was shown for OS, DMFI, and DMFS. A subanalysis was performed on the lymph node-negative study population. In the lymph node-negative study population, we report an up to 11 times higher change in the diagnosis of an event in the MammaPrint High Risk group. For DMFI, the reported hazard ratio is 11.1 (95% confidence interval = 2.3–53.0).
Conclusion
Study results validate MammaPrint as an independent factor for breast cancer patients with early-stage invasive lobular breast cancer. Hazard ratios up to 11 in multivariate analyses emphasize the independent value of MammaPrint, specifically in lymph node-negative ILC breast cancers.
Keywords
Introduction
Invasive lobular carcinoma (ILC) is the second most prevalent type of breast cancer based on histological criteria. Approximately 10%–15% of primary breast cancers fall into this category.1,2 These carcinomas are in general more estrogen receptor (ER)-positive, more human epidermal growth factor receptor 2 (HER2)-negative, and of lower histological grade compared to the more common invasive ductal carcinomas (IDCs).3,4 Pathological assessment of ILCs identified four different subtypes, illustrating heterogeneity in this group of tumors. There is a tendency to classify ILC as a type with low risk of relapse; however, available reports on survival data show heterogeneous outcome statistics. 2 As a result, in current clinical practice, it is unclear which ILC patients are at increased risk of tumor recurrence and whether there are patients who would benefit from specific treatment options. Treatment decisions for early-stage ILCs may benefit from clinical adoption of the MammaPrint® test, in addition to evaluation of clinicopathological parameters, as is already common in clinical practice for IDCs, especially because systemic treatment options for both types of tumors are currently almost identical. 4
The MammaPrint assay is a microarray-based test cleared by the US Food and Drug Administration, which uses the expression levels of the 70 MammaPrint genes to assess risk of recurrence in early-stage breast cancer.5–7 The test aims to guide physicians in making neoadjuvant and adjuvant treatment decisions. This assay was developed and validated in cohorts of breast cancer patients, which consisted of approximately 85% of the more prevalent type of IDC. This dominance of IDC in the MammaPrint training and validation sets could potentially introduce a bias in the prognostic performance of the MammaPrint assay in favor of IDC, with the gene signature being more correctly prognostic in IDC than in ILC. However, a pathway analysis of the 70 signature genes of the MammaPrint assay demonstrated that the test measures a number of universal aspects of (breast) cancer biology, including proliferation, angiogenesis, invasion, and ER signaling, and it is likely that these processes are very similar in breast cancers of different origins. 8 A recurrent clinical question about MammaPrint involves its prognostic value for specifically the ILC group of breast cancers. Some physicians feel that the smaller representation of invasive lobular cancers in the development data set calls for a survival analysis dedicated to this specific subgroup to determine the prognostic value of MammaPrint.
With the increase in the incidence of ILCs over the past decades,9,10 there is a clear clinical need to better evaluate the prognostic value of the MammaPrint test for specifically invasive lobular breast carcinomas. Hence, we have sought to validate the prognostic capacity of MammaPrint in primary invasive lobular breast cancers. The results of this evaluation are presented here.
Materials and Methods
Patient samples
MammaPrint results, 6 clinicopathological data, and survival data were collected for all early breast cancers of the invasive lobular type from Agendia's clinical series database. The study population consisted of 217 unique cases that were derived from five clinical series, including the RAtional THerapy for breast cancER (RATHER) ILC series, 11 and the microarRAy-prognoSTics-in-breast-cancER (RASTER) series 12 (refer Supplementary File 1 for detailed information).11–14 Clinicopathological data included age at surgery, differentiation grade, lymph node (LN) involvement, surgery type, and administration of adjuvant chemotherapy and hormone therapy. Additionally, information on ER status and HER2 status, as assessed by the TargetPrint assay, 15 was available for analyses. TargetPrint readout was as described previously by Roepman et al. 15 The mean follow-up time for this study cohort was 85 months (range: four months–22 years).
Research was performed according to the principles of the Declaration of Helsinki. All patient samples and data were anonymously coded in accordance with national ethical guidelines (“Code for Proper Secondary Use of Human Tissues”, Dutch Federation of Medical Scientific Societies), and the study samples had institutional review board approvals for the anonymized use of archival tissues.11–14 This study was performed based on the guidance of the REporting recommendations for tumor MARKer prognostic studies (REMARK) (National Cancer Institute-European Organisation for Research and Treatment of Cancer [NCI-EORTC]). 16
Data analysis
Statistical analyses, survival analyses, and visualization of data were performed using the statistical package SPSS 22.0 for Windows (SPSS Inc, Chicago, IL, USA).
The relationship between MammaPrint results (MammaPrint index values dichotomized to Low Risk and High Risk) and known clinicopathological parameters was investigated using the Pearson chi-squared test or Fisher's exact test.
MammaPrint results were used in survival analyses. The Cox proportional-hazards model was used to analyze the association between MammaPrint results for survival at 10 years after surgery. Overall survival (OS) was defined as the time from surgery until death by any cause. 17 Distant metastasis-free interval (DMFI) was defined as the time from surgery until the diagnosis of a distant recurrence. Distant metastasis-free survival (DMFS) was defined as the time from surgery until the diagnosis of a distant metastasis or death by any cause. 17 Differences in survival between patient groups are presented as hazard ratios (HRs). The MammaPrint Low Risk group was used as the reference group for all survival analyses. Kaplan-Meier curves were used to visualize the univariate survival associations. Multivariate survival analyses were performed to account for the effects of other variables or confounders on survival and to account for potential differences in distribution of clinicopathological factors between Mamma Print Low Risk and High Risk groups. Multivariate models included the following predetermined clinically important covariates: age at surgery, LN involvement, differentiation grade, adjuvant chemotherapy, ER status, and HER2 status, irrespective of statistical significance. Age at surgery and differentiation grade were entered as continuous variables into the multivariate models. All tests were two-tailed types, and P-values < 0.05 were considered statistically significant.
Results
Patient characteristics
Clinicopathological and survival data were available for n = 217 invasive lobular cases. These cases originate from multiple clinical study series, as described in the “Materials and Methods” section. The association of MammaPrint verdict results (Low Risk and High Risk for MammaPrint) with clinicopathological parameters is described in Table 1. Analyses were performed for the entire study cohort (n = 217), as well as for the group of LN-negative cases (n = 144). MammaPrint verdict results of the entire study cohort correlated with the established prognostic parameter of LN involvement, with the MammaPrint Low Risk group containing slightly more LN-negative tumors. Most patients had ER-positive and HER2-negative disease. The mean patient age at surgery was 58 years (range: 29–93 years). As expected, a higher percentage of breast-conserving therapy was the choice of surgery for MammaPrint Low Risk patients, and less patients in the MammaPrint Low Risk group received adjuvant chemotherapy. Interestingly, we observed more patients older than 55 years in the MammaPrint High Risk group compared to those in the Low Risk group.
Associations of the study cohort with clinicopathological parameters.

ER status and HER2 status were assessed by TargetPrint 15 .
A Fisher's exact test was used in the analyses for the LN-negative cases. cTwo missing values for specification surgery.
Prognostic value of MammaPrint in the ILC subgroup
Results of univariate survival analyses are shown in Table 2, and visualized in Kaplan–Meier curves (Fig. 1). A significant association between MammaPrint High Risk and poor clinical outcome was shown in univariate analyses for OS, DMFI, and DMFS. Based on the univariate analyses, patients with tumors classified as High Risk showed a 3.6 times higher chance to develop a distant metastasis within 10 years after surgery (DMFI HR: 3.6; 95% confidence interval [CI]: 1.6–7.8) or to die within 10 years after surgery (OS HR: 3.6; 95% CI: 1.8–7.0), and a 3.3 times higher chance to present with either event (DMFS HR: 3.3; 95% CI: 1.8–6.1).
Univariate survival associations for invasive lobular breast cancer.
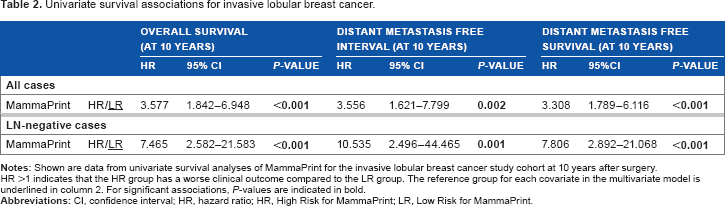
HR >1 indicates that the HR group has a worse clinical outcome compared to the LR group. The reference group for each covariate in the multivariate model is underlined in column 2. For significant associations, P-values are indicated in bold.

Univariate survival curves for invasive lobular breast cancer stratified by MammaPrint.
Results of multivariate survival analyses are shown in Table 3. In multivariate analyses, MammaPrint was validated as an independent factor for DMFI and DMFS. MammaPrint High Risk status was associated with worse clinical outcome in invasive lobular breast cancer. In these analyses, accounting for the effect of confounders or differences in distribution of clinicopathological factors between analyses groups, patients with a tumor classified as MammaPrint High Risk showed a 2.4 times higher chance to develop a distant metastasis within 10 years after surgery (DMFI HR: 2.4; 95% CI: 1.0–5.6). The chance to develop a distant metastasis or die within 10 years after surgery was 2.1 times higher in the MammaPrint High Risk group (DMFS HR: 2.1; 95% CI: 1.0–4.1). Multivariate survival analyses were further performed by including only those clinicopathological parameters that showed a significant association with MammaPrint outcome (Supplementary File 2). These analyses confirm MammaPrint as independent factor for OS, DMFI, and DMFS.
Multivariate survival associations for invasive lobular breast cancer.

For age at surgery, the HR is given per unit increase, with one unit representing 10-year increase in age.
ER status and HER2 status were assessed by TargetPrint. 15
Prognostic value of MammaPrint in ILC without LN involvement
In the patient group without LN involvement (n = 144), MammaPrint was validated for OS, DMFI, and DMFS in univariate (Table 2) and multivariate (Table 3) survival analyses. Additionally, Kaplan-Meier curves were plotted to visualize the univariate survival associations (Fig. 2). The lower number of cases in the LN-negative subgroup as compared to the whole study cohort inherently resulted in wider CIs. Based on univariate analyses, patients with tumors classified as High Risk showed a 7.5–10.5 times higher chance for an OS-related event (OS HR: 7.5; 95% CI: 2.6–21.6), DMFI (DMFI HR: 10.5; 95% CI: 2.5–44.5) or DMFS-related event (DMFS HR: 7.8; 95% CI: 2.9–21.1). Based on multivariate analyses, the MammaPrint High Risk group showed, for DMFS, a 6.4 times higher chance for an event within 10 years after surgery (DMFS HR: 6.4; 95% CI: 2.1–19.2). Patients with tumor classified as MammaPrint High Risk showed an 11.1 times higher chance to develop a distant metastasis within 10 years after surgery (DMFI HR: 11.1; 95% CI: 2.3–53.0), or a 5.1 times higher chance to die within 10 years after surgery (OS HR: 5.1; 95% CI: 1.5–17.2). Additionally, multivariate survival analyses were performed (Supplementary File 2) by including only those clinicopathological parameters with a significant association with MammaPrint outcome as shown in Table 1. These analyses confirm MammaPrint as an independent factor for OS, DMFI, and DMFS in the patient group with LN-negative cases. Patient numbers were too low to report any results for the LN-positive subanalysis.
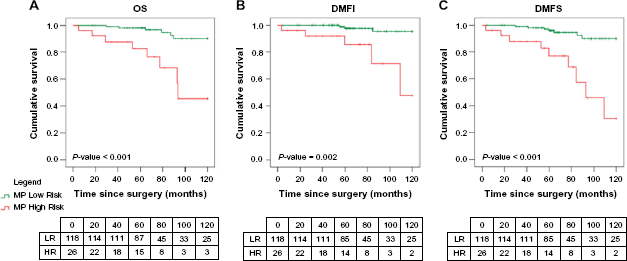
Univariate survival curves for invasive lobular breast cancer without lymph node involvement stratified by MammaPrint.
Discussion
The results of this study validate MammaPrint as an independent factor for early-stage invasive lobular breast cancer. The significantly high HRs (up to 11 for DMFI) in multivariate analyses emphasize the independent value of MammaPrint, specifically in LN-negative invasive lobular breast cancers.
The study data set is comparable to ILC cohorts described previously. 2 The current study showed a distribution of differentiation grade, ER status, and HER2 status that is comparable to the overall characteristics reported in a review on ILC by Guiu et al. 2 Additionally, the percentage of MammaPrint Low Risk versus High Risk tumors, as well as the percentage of ILC patients, is comparable to that in the studies reviewed by Guiu et al. 2 The authors of the review supported the need for personalized treatment by using gene expression assays, such as MammaPrint, for patients with lobular tumors. Because the patient numbers were low in the reported studies, we combined the results of multiple studies. In the current study, follow-up data of 217 ILC patients have been combined, and the results show a significant difference between Low Risk and High Risk of recurrence.
The study cohort comprises patients from five different clinical studies, and therefore systemic therapy decisions and adherence to treatment might not be fully comparable. This could have effect on the long-term outcome.
Demonstrating the usefulness of a test in clinical practice – or clinical utility – may be the most significant hurdle for clinical adoption of diagnostic tests. Recently, the results of the large Microarray In Node-negative and 1 to 3 positive lymph node Disease may Avoid ChemoTherapy (MIND-ACT) trial have been reported, 18 demonstrating the clinical utility of MammaPrint in early-stage breast cancer with Level 1a evidence. 18 This prospectively randomized study enrolled 6693 patients, of whom 500 were classified as having ILC. A separate analysis of the five-year outcome data of this ILC group will be part of further subanalyses of the MINDACT clinical trial data and will provide a larger number of patients for whom comprehensive data are available at the level of both clinical risk and genomic risk. The current study was planned to validate the prognostic value of the MammaPrint test for ILC breast cancer patients, thereby supporting the clinical adoption of MammaPrint prior to the comprehensive MINDACT analysis. The clear difference in 10-year outcome between MammaPrint Low Risk and High Risk patients indicates the utility of MammaPrint as an aid in systemic treatment decision for patients with early-stage ILCs, especially in the LN-negative group. Although IDC and ILC are recognized as different subgroups of the same disease with distinct clinical features, 3 MammaPrint has demonstrated prognostic values in the combined subgroups. The current study emphasizes the prognostic power of MammaPrint, specifically in primary invasive lobular breast cancers.
The ongoing research on breast cancer is currently focusing on further stratification and substratification of IDC and ILC. For both breast cancer subgroups, the future lies in creating focused treatment options based on insights in patient-specific variation 19 and the combination of clinicopathological parameters and multiple genetic classifiers that reflect tumor biology for individual tumors. Clinicopathological parameters and established genetic classifiers, such as MammaPrint and BluePrint, 20 can be combined with new genetic markers to even further identify patient subgroups with distinct relapse risks or benefit from specific treatment options. A specific example for the ILC subgroup is the identification of an immune-responsive subpopulation 11 that can be distinguished using gene expression classification.
The independent value of MammaPrint in LN-negative early-stage invasive lobular breast cancer indicates the beneficial effect of risk of recurrence determination by MammaPrint and subsequent adjuvant chemotherapy decision.
Author Contributions
Conceived and designed the experiments: IJB, LJvtV, RB, AMG. Data acquisition: IJB, AW, S-FC, S-JS, MS, CC, SL. Analysis: IJB, AW, CD. Interpretation of the data for the work: IJB, MP, CD, S-FC, S-JS. Wrote the first draft of the manuscript: IJB, MP. Contributed to the writing of the manuscript: IJB, MP, AW, CD, S-FC, S-JS, MS. Agree with manuscript results and conclusions: IJB, MP, AW, CD, S-FC, S-JS, MS, CC, SL, LJvtV, RB, AMG. Jointly developed the structure and arguments for the paper: IJB, RB, LJvtV, AMG. Made critical revisions and approved final version: CC, SL. All authors reviewed and approved of the final manuscript.
