Abstract
Changes in plasma IL-6, plasma VEGF and serum YKL-40 were determined in rheumatoid arthritis (RA) patients during treatment with etanercept alone or in combination with methotrexate. Twenty-five patients with active RA (DAS28 ≥ 3.2) were randomized to receive etanercept (25 mg sc. biweekly) plus methotrexate (n = 12) or etanercept alone (n = 13). Plasma IL-6, plasma VEGF and serum YKL-40 were determined by ELISA. The 3 biomarkers and DAS28 scores were evaluated at baseline and after 4, 8, 12 and 16 weeks of treatment. At inclusion all patients had significantly (p < 0.001) elevated plasma IL-6, plasma VEGF and serum YKL-40 compared to healthy subjects. Eighteen patients responded to treatment (pooled data from both treatment groups), and they had significant (p < 0.05 to p < 0.001) decreases in plasma IL-6, plasma VEGF, serum YKL-40, ESR and DAS28 after 4 weeks of treatment and throughout the study (except serum YKL-40 at week 16). Plasma IL-6 showed the largest reductions. Non-responders had unchanged biomarkers. At week 16 the patients with DAS28 < 3.2 had lower levels compared to baseline values in plasma IL-6 (p = 0.005), plasma VEGF (p = 0.014), and ESR (p = 0.024).
Plasma IL-6, plasma VEGF and serum YKL-40, which reflect different aspects of the inflammatory process, may provide useful information regarding early differentiation of responders from non-responders.
Introduction
Etanercept, a tumor necrosis factor-alpha (TNF-α) receptor antagonist, in combination with methotrexate has shown to be more effective than etanercept or methotrexate alone for control of disease activity in patients with rheumatoid arthritis (RA). 1 With the development of new targeted therapies for RA there is a need for more sensitive and specific assessment of treatment response. Molecular markers produced locally in the arthritic joint may reflect the inflammatory and destructive processes in RA, and quantification of these may be a useful adjunct to the clinical evaluation of these patients.
Interleukin-6, produced in the arthritic joint by T and B lymphocytes, macrophages, synoviocytes, osteoblasts and mast- and endothelial cells, plays important roles in the inflammatory processes seen in RA. 2 The sources of VEGF in RA patients are the synovial lining cells, macrophages, leukocytes, platelets, fibroblasts and endothelial cells. 3 VEGF stimulates angiogenesis (i.e. the formation of new vessels from pre-existing ones), 4 which constitute an early event in RA and may lead to cartilage and bone destruction in later stages of the disease. 4 YKL-40 (chitinase-3-like 1 protein) is produced locally in the arthritic joint by activated macrophages, chondrocytes, synoviocytes and neutrophils.2,5,6 YKL-40 is found to be a possible auto antigen in RA. 7
We studied the short time changes in plasma IL-6, plasma VEGF and serum YKL-40 in RA patients receiving either etanercept alone or in combination with MTX.
Materials and Methods
Patients and Therapy
Twenty-five Danish RA patients, who accepted participation in a prospective, randomized, international study (Protocol Number 0881A1–101136, The ADORE study), 8 were enrolled in this investigator-initiated amendment involving repeated measurements of plasma IL-6, plasma VEGF and serum YKL-40. All patients received methotrexate (for minimum 3 months prior to the study and a stable dose for 6 weeks prior to inclusion), but their disease activity was inadequately controlled (i.e. disease activity score (based on 28 joint counts and the erythrocyte sedimentation rate (ESR), DAS28 ≥ 3.2). 9 The patients were randomized to receive either etanercept plus methotrexate (n = 12, group A) or etanercept alone (n = 13, group B). Group B patient's tapered methotrexate during a period of 4 weeks. No patients received intraarticular or systemic steroid injections within 4 weeks before screening. Five patients received a stable dose of oral corticosteroid 4 weeks prior to and throughout the study. Clinical evaluation and blood samples were collected at baseline and after 4, 8, 12 and 16 weeks of treatment.
The study was performed in accordance with the Helsinki Declaration II and approved by the local ethical committee.
Biochemical Analysis
Blood samples were centrifuged within 3 hours after venipuncture and stored at –80 °C until analysis. ESR was determined by routine method. The biomarkers were determined by ELISA: plasma IL-6 and plasma VEGF (R&D Systems, Oxon, U.K), and serum YKL-40 (Quidel, CA, U.S.A.). Samples from each patient were analyzed on the same ELISA plate with internal control samples on each plate to confirm assay precision.
Healthy Subject Population
The plasma IL-6 concentration in 318 healthy subjects (median age 48 years, range 18–64, 122 women and 196 men) was median 1.3 ng/l (90th percentile = 3.3 ng/l). The plasma VEGF level in 306 healthy subjects (median age 48 years, range 18–64, 116 women and 190 men) was median 45 ng/l (90th percentile = 109 ng/l). The serum YKL-40 in 245 healthy subjects (median age 49 years, range 18–79, 134 women and 111 men) was median 43 μg/l (90th percentile = 95 μg/l).
Statistical Analysis
Results are given as median, range and percentage changes from baseline. Comparison between and within groups was done by Mann-Whitney and Wilcoxon-Pratt tests, respectively. Correlation was calculated by Spearman's test. P < 0.05 were considered statistically significant. The statistical tests used are all non-parametric since the interleukin-6, vascular endothelial growth factor and YKL-40 are not normally distributed according to a normal probability plot. Areas under the curve (AUC) values, i.e. weighted means, were calculated for each parameter. The patients were divided according to the EULAR response criteria, into clinical responders (EULAR good or moderate responders) and clinical nonresponders (EULAR non-responders) (www.das-score.nl). For statistical calculations, Sigma Stat (SPSS Inc., Chicago, IL, USA, version 3.1) was used.
Results
Clinical and biochemical parameters before treatment with etanercept.

ESR at baseline is missing in 2 patients.
The proportion of patients with elevated baseline level of the parameter.
Figure 1 illustrates the individual changes in clinical and biochemical markers in the RA patients according to the EULAR DAS28 response criteria. Table 2 shows the percentage decreases compared to baseline values. Significant decreases from baseline values were found in plasma IL-6, plasma VEGF, ESR, and DAS28 at 4, 8, 12 and 16 weeks and in serum YKL-40 at 4, 8 and 12 weeks (p = 0.03 to p < 0.001). Plasma IL-6 and DAS28 showed the most pronounced reductions. All biomarkers, HAQ and DAS28 decreased in both treatment arms, and there was no significantly differences between group A and B. No significant changes were found in the biomarkers of the non-responders. The only AUC value that correlated was plasma IL6AUC versus ESRAUC (rho = 0.79, p = 0.004).
The changes in plasma IL-6, plasma VEGF, serum YKL-40, ESR and DAS28(ESR) in 25 RA patients during the 16-week study period, divided by being EULAR good/moderate responders or EULAR non-responders. Values are given as box and whiskers plots. Boxes indicate 25th, 50th, 75th centiles. Whiskers indicate 5th-95th percentiles. The y-axis is logarithmic for the biomarkers. *, ** and *** indicate statistically significant changes from baseline (Wilcoxon-Pratt): *p < 0.05, **p < 0.01, ***p < 0.001. Relative decrease in clinical and biochemical parameters compared to baseline values. The relative decreases, expressed in%, compared to baseline values are given as mean values (standard error of mean). Negative values indicate an increase compared to baseline level. p < 0.05 p < 0.01 p < 0.001.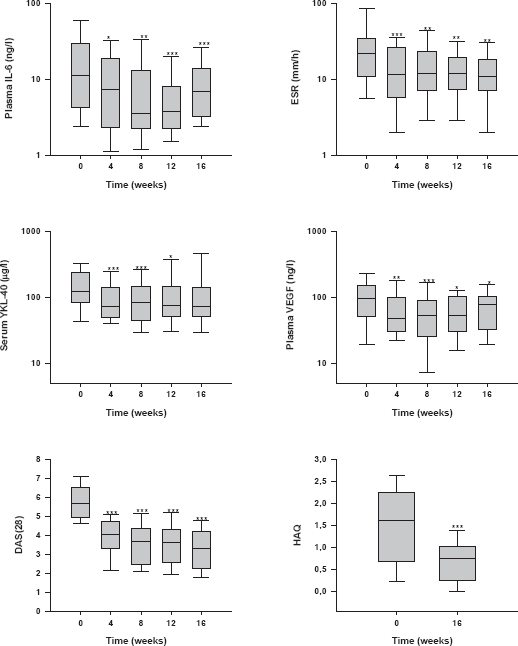

At week 16, eleven patients had low disease activity (DAS28 < 3.2), and 7 of whose were in remission (DAS28 < 2.6). Patients with DAS28 < 3.2 at week 16 had lower plasma IL-6 levels at this time point compared to baseline level (4.1 vs. 9.6 ng/l, p = 0.005), ESR (8 vs. 16 mm/h, p = 0.024) and plasma VEGF (74 vs. 126 ng/l, p = 0.014). No significant change was found in serum YKL-40. At week 16 plasma IL-6 was significantly higher in RA patients who were in remission compared to healthy subjects (4.1 vs. 1.3 ng/l, p < 0.005) whereas plasma VEGF (81 vs. 45 ng/l, p = 0.318) and serum YKL40 were not (66 vs. 43 μg/l, p = 0.063).
Discussion
In this study we focused on changes in circulating levels of IL-6, VEGF and YKL-40 in RA patients during anti-TNF-α therapy. TNF-α, IL-1, and IL-17 augment the production of IL-6 by synovial cells and IL-6 is found to be responsible for many of the clinical and biochemical changes in RA. 2 TNF-α and several pro-inflammatory cytokines (i.e. IL-1, IL-6, IL-8) induce VEGF production and these cytokines are responsible for overexpression of VEGF seen in RA. 3 IL-1 and TNF-α induces YKL-40 expression and synthesis by chondrocytes and requires activation of nuclear factor kappa B. 10 In accordance with others11–13 we found that plasma IL-6, plasma VEGF and serum YKL-40 were significantly increased in RA patients compared to healthy subjects, and that these markers decreased during anti-TNF-α therapy.5,6,14 Etanercept, a construct comprising two human p75 TNF-α receptors coupled to the Fc portion of a monoclonal human antibody (IgG1), binds both TNF-α and TNF-β and thereby reducing cytokine production in patients with RA. 15 Methotrexate is known to reduce cytokine production through apoptosis of peripherally active T-cells and reduce the number of macrophages in the synovial tissue. 16 Even though etanercept was able to decrease plasma IL-6, plasma VEGF and serum YKL-40, the level of plasma IL-6 was significantly higher in RA patients in remission than in healthy subject, whereas VEGF and YKL-40 were not. The disease process in the joints not being fully suppressed, despite the low clinical disease activity could explain this. As suggested by others non-responsiveness to TNF-α blockade and residual disease activity, indicate that TNF is not the sole responsible biological target in RA. 17 Prospective studies including a larger number of RA patients are needed to evaluate if these biomarkers, can provide useful information regarding early differentiation of responders from non-responders.
Dsiclosure
The authors report no conflicts of interest.
Footnotes
Acknowledgements
Wyeth Pharmaceuticals is acknowledged for financial support. The expert technical assistance of Tonni Løve Hansen and Debbie Nadelmann, Herlev Hospital is gratefully acknowledged. The staff of Department of Clinical Immunology, Nykøbing Falster County Hospital is thanked for collection of blood samples from healthy volunteer blood donors.
