Abstract
Objectives
Many researches showed that Erythropoietin-producing hepatocyte kinase receptor A1 (EphA1) can promote the occurrence and development of malignant tumors and may be related to tumor microenvironment. But most of them are phenomenon studies, and there are few in-depth and complete mechanism studies. This study aims to understand how EphA1 promotes the progression of malignant tumors by regulating tumor microenvironment (focusing on Interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF)) from two experimental dimensions of in vitro and in vivo by using genetic engineering technology.
Material and Methods
We used genetic engineering technology to enhance and knock down EphA1 gene expression in SGC-7901 cells, respectively, and analyzed its influence on cell function and the expression levels of VEGF and IL-6 in cells. Subsequently, we constructed human EphA1 gene overexpression, EphA1 gene silencing, and normal expression of human EphA1 gene subcutaneous transplanted tumor models of SGC-7901 cells nude mice, and analyzed the differences in tumor development and the changes in the expression levels of VEGF and ILl-6 in tumor tissues.
Results
After EphA1 gene expression was enhanced, the proliferation, invasion and migration of SGC-7901 cells were enhanced, and apoptosis was weakened, and the expression levels of VEGF and IL-6 were increased. While the opposite results were found when EphA1 gene expression were knocked down. Meanwhile, tumor formation time and growth rate of subcutaneous transplantation in nude mice were advanced and the expression levels of VEGF and IL-6 in tumor tissues were increased when EphA1 gene expression were overexpressed by genetic engineering technology. Similarly, the opposite effect occurred in transplanted tumor model when EphA1 gene was silenced.
Conclusion
Our study showed that EphA1 can up-regulating VEGF and IL-6 expression, thereby enhancing the inflammatory environment and angiogenesis in the tumor microenvironment, and this helps to promote the progression of SGC-7901 cells and its transplanted tumor.
Introduction
Erythropoietin-producing hepatocyte kinase receptor A1 (EphA1) belongs to the family of Receptor tyrosine kinases, which have certified as key regulators in carcinogenesis among some solid cancers, and functions in bi-directional signal transduction via direct act on adjacent cells that expressing its specific ligand.1,2 More and more studies have shown that EphA1 can promote the emergence, growth, invasion and metastasis of malignant tumors, and is expected to become a new molecular therapeutic target for cancer.3,4 However, the studies on malignant tumor and EphA1 lack systematic in vitro and in vivo studies, and most of them are phenomenon studies, and there has not been a thorough and complete study on the possible mechanism.5,6
Lately, more and more studies focused on the influence of alteration of tumor microenvironment to cancer development, especially on inflammation and angiogenesis.7,8 VEGF (vascular endothelial growth factor) largely exists in tumor microenvironment as an important tumor angiogenesis promoter, which mainly functions on vascular endothelial cells, that stimulate endothelial cell survival, migration, and growth from vasculature by a network of signaling processes triggered for activated the VEGF-VEGFR signaling axis, and has been found to be associated with the invasion, metastasis and poor prognosis of various malignant tumors.9,10 Much researches suggests a connection between the chemokine IL-6 (interleukin-6) and tumor microenvironment, which is emerged as a key player in tumour cell growth, survival, and invasive migration through starting abundant signaling pathways and downstream mediators such as signal transducer and activator of transcription 3 (STAT3).11,12 Coincidentally, increasing data show that Ephs has been involed to the angiogenesis in tumor microenvironment and oxidative stress due to inflammation during occurrence of solid tumoures.13–15 In addition, studies have found positive relationship among the expression of EphA1 and tumor microenvironment marker proteins VEGF, IL-6 in clinical specimens of malignant tumors, but no further mechanism has been explored. 16
Taken together, We hypothesized that EphA1 may involved in the regulation of tumor microenvironment, and consequently facilitates tumorigenesis and progression through drives the expression of IL-6 and VEGF in tumor microenvironment. To testing and confirmation of this hypothesis, we elucidating the role of EphA1 in regulating IL-6, VEGF expression before and after EphA1 gene knockout by cell culture experiments and animal models.
Material and methods
Cell culture
The SGC-7901 cell line was bought from Shanghai fuheng biotechnology co., Ltd. (Shanghai, China). It originated from lymph node metastasis in a 56-year-old woman with gastric adenocarcinoma. It has the characteristics of metastatic invasion. And cultured in DMEM medium containing 10% fetal bovine serum at 37°C and 5% CO2. The medium was changed every 2–3 days, and 100 mg of streptomycin and 100 U penicillin were added to the medium.
Construction of EphA1 gene overexpression and interference lentivirus stable transfected cell lines
EphA1 gene fragment (bought from Shanghai quanyang biotechnology co., Ltd, Shanghai, China) was connected to vector PUC57 (bought from Shanghai quanyang biotechnology co., Ltd, Shanghai, China) to construct recombinant plasmid. The recombinant vector plasmid carrying the target gene was co-transfected with the packaged plasmid system (pSPAX2 vector plasmid and pMD2G vector plasmid) into 293T cells to obtain lentivirus particles. SGC-7901 cells were infected with an overexpressed EphA1 lentiviral vector or a negative control (NC) vector, and qRT-PCR was performed to detect gene delivery efficiency. Subsequently, cell lines with stable expression of the target gene were screened using puromycin resistance.
In accordance with shRNA design principle, the RNAi target sequence was designed according to the human EphA1 gene sequence (nm_005232.5) in GenBank database and double strand DNA oligo was synthesized (bought from Shanghai quanyang biotechnology co., Ltd, Shanghai, China). The shEphA1 target sequences (5′-3′) were as follows: Sense: CCGGGCCCTTGTTCCAGAAGGTAACCTCGAGG TTACCTTC TGGAACAAGGGCTTTTTG; Antisense: AATTCAAAAAGCCCTTG TTCCAGAAGGTAACCTCGAGGTTACCTTCTGGAACAAGGGC. The interferen -ce fragment was connected to the vector to construct the recombinant plasmid. Then the recombinant plasmid vector containing the interfering fragment was packaged with lentivirus and the virus particles were collected. The tested qualified venom was used to infect the target SGC-7901 cells. Finally, qRT-PCR was used to detect and verify the gene interference efficiency. Similarly, the SGC7901 cell line with stable transfection interference fragment screened by purinomycin resistance were used for further experiments.
MTT assay for cell proliferation
First, cells were incubated in 96-well plates, at a density of 1 × 104 cells per well for 24 h. After different time points, 10 μL of MTT dye (5 mg/mL; Sigma-Aldrich) was added and incubated for another 4 h at 37°C. And then, 150 mL of dimethyl sulfoxide was added to each well, and mixed for 10 min. Spectrometric absorbance was determined with a microplate reader (Bio-Rad, Hercules, CA, USA), at a wavelength of 490 nm. Each sample had three replicates.
Transwell assay for cell invasion
24-well, 8-μm pore sized transwell plates were used for transwell invasion assays according to the manufacturer’s instructions (Corning, New York, NY, USA). BD Matrigel Basement Membrane Matrix coarted in the bottom of transwell chamber. 1 × 105 cells in RPMI 1640 containing 5% FBS filled in the upper chamber. RPMI 1640 which containing 25% FBS as a chemo-attractant filled in the lower chamber. When the chambers were incubated for 24 or 48 h at 37°C later, noninvading cells on the upper side of the chamber were removed from the surface of the membrane by scrubbing, and invading cells on the lower surface of the membrane were fixed with methanol, mounted, and dried. A technician which blinded to the experimental settings counting the number of cells invading through the matrigel in four randomly selected microscopic fields of each filter. The test was conducted in three biological replicates.
Detection of apoptosis
The cultured adherent cells were digested with trypsin, and the cells were counted and collected. Pre-cooled PBS was used for washing cells twice time, centrifuged at 1500 r/min for 3 min, and 100 μL 1×Annexin V Binding Buffer and let it stand in the dark for 15 min. Add 10 μL of propidium iodide (PI), mix well, and stand for 5 min at rBuffer was added to resuscitate the cells. Add 5 μL Annexin V-APC oom temperature and avoid light. Cell cycle analysis was performed by flow cytometry (BD, USA). FlowJo7.6 software was used to analyze and map the images of flow cytometry apoptosis.
The protein expressions of EphA1, IL-6, VEGF in the three cell lines were detected by Westernblot
After adding RIPA cell lysate to the collected cells, the cells were lysed on ice for 30 min, centrifugated at 12,000 r/min for 10 min, and supernatant containing total cell protein was taken. SDS-PAGE gel was prepared and the sample protein was electrophoretic, and the protein tape was transferred to PVDF membrane. After the protein membrane was fully rinsed, Western blocking solution was added and placed on a shaking table for sealing at room temperature for 2 h. Primary and secondary antibodies were added to incubate according to the instructions. Follow the instructions and use the ECL luminescence kit to detect the protein.
Gene expression of EphA1, IL-6, VEGF in three cell lines was detected by qRT-PCR
Comparative analysis of EphA1, VEGF and IL-6 gene expression levels in three groups of cells (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, normal: normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group).

Tumorigenesis experiment in vivo
To examining tumorigenicity, we using five-week-old male nude athymic BALB/c nu/nu mice for the subjects. Three groups of SGC-7901 cells were prepared (Normal: Normal SGC7901 cell line group; OV-EphA1: EphA1 overexpressed SGC7901 cell line group; Si-EphA1: EphA1 gene silencing SGC7901 cell line group), and then inoculating subcutaneously into the flanks of nude mice (2 × 106 cells in 0.2 mL volume). Record the time of tumor formation and tumor size was measured every 2 days. After 16 days, the mice were sacrificed, and tumor masses were measured. Immunohistochmesitry staining of IL-6, VEGF and EphA1 was used on the mice tumor tissue slides. The experiments were performed using five mice per group, and all animal experiments were performed in strict accordance with the principles and procedures approved by the Ethics Committee of Anhui Provincial Cancer hospital.
Statistical analysis
All the experimental data are expressed as mean ± SEM form, and each experiment was repeated at least three times. Student’s t-test was used for evaluating continuous variables. Bivariate correlations between study variables were calculated by Spearman’s rank correlation coefficients. Statistical analyses were performed with SPSS25.0 software (SPSS Inc., Chicago, IL, USA).
Results
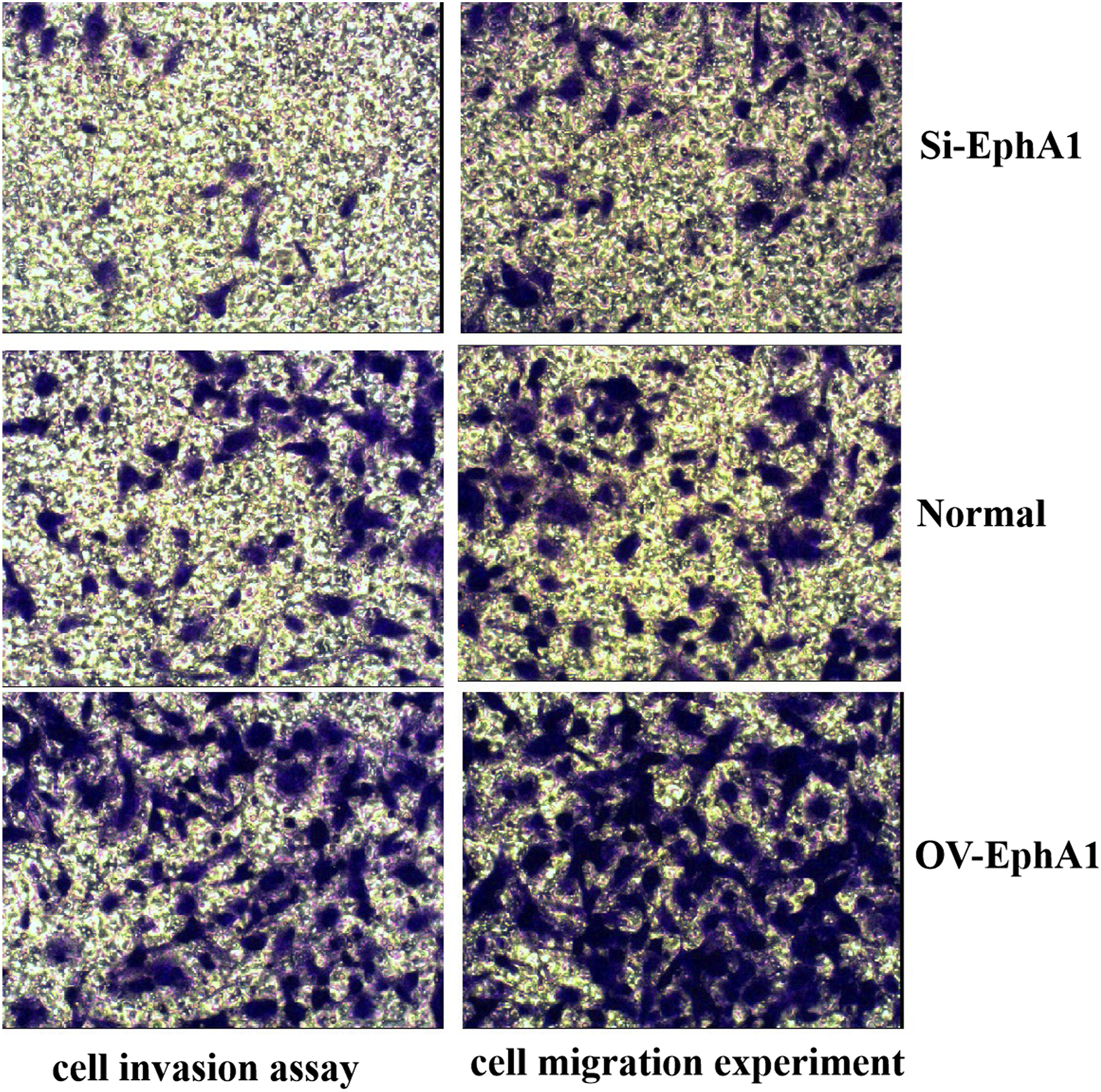
EphA1 modulates cell proliferation, migration, invasion and apoptosis in gastric cancer cells
We successfully constructed EphA1 overexpressed SGC7901 cell line (OV-EphA1 group) and EphA1 gene silencing SGC7901 cell line (Si-EphA1 group) by genetic engineering. We analyzed three groups’ (OV-EphA1 group, Si-EphA1 group, Normal SGC7901 cell line group) cellular function and found that: After 24 h observation, the cell metabolic activity of the OV-EphA1 group was significantly higher than that of the normal control group ( Cell proliferation activity of the three groups (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, Normal: Normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group). Effect of EphA1 gene enhancement and silencing on migration and invasion of SGC7901 cells (×100). (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, Normal: Normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group). Cell apoptosis detection and analysis in three groups (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, Normal: Normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group).

EphA1 regulates the expression of IL-6 and VEGF in SGC7901 cells
We used qRT-PCR and Western blot to detect and analyze the gene and protein expression levels of EphA1, VEGF and IL-6 in the OV-EphA1 group, the Normal control group and the Si-EphA1 group. The results show that: After overexpression of EphA1 gene, the expression levels of VEGF and IL-6 in cells were also significantly higher than those in the normal control group ( The expression levels of VEGF and IL-6 genes in the three groups were compared (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, Normal: Normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group). The protein expression levels of EphA1, VEGF and IL-6 in the three groups were compared and analyzed (OV-EphA1: EphA1 overexpressed SGC7901 cell line group, Normal: Normal SGC7901 cell line group, Si-EphA1: EphA1 gene silencing SGC7901 cell line group). Western blot analysis of EphA1, VEGF and IL-6 protein expression in three groups of cells (1: Normal group, 2: NC group, 3: OV-EphA1 group, 4: Normal group, 5: NC group, 6: Si-EphA1 group). (OV-EphA1: EphA1 overexpressed SGC7901 cell line, Normal: Normal SGC7901 cell line, Si-EphA1: EphA1 gene silencing SGC7901 cell line, NC: No-treatment Control).


EphA1 promotes the occurrence and development of SGC-7901 cell line xenograft tumor in vivo
The tumor formation rate and the time of the first subcutaneous tumor transplantation were observed in three groups of nude mice (OV-EphA1: nude mice subcutaneously inoculated with EphA1 overexpressed SGC7901 cell line group, Normal: nude mice subcutaneously inoculated with Normal SGC7901 cell line group, Si-EphA1: nude mice subcutaneously inoculated with EphA1 gene silencing SGC7901 cell line group).


a and b are the general drawings of subcutaneous tumorigenesis in nude mice. c are the comparison of subcutaneous tumor weight in three groups of nude mice (OV-EphA1: nude mice subcutaneously inoculated with EphA1 overexpressed SGC7901 cell line group, Normal: nude mice subcutaneously inoculated with Normal SGC7901 cell line group, Si-EphA1: nude mice subcutaneously inoculated with EphA1 gene silencing SGC7901 cell line group).

The growth trend of subcutaneous transplanted tumor in three groups of nude mice (OV-EphA1: nude mice subcutaneously inoculated with EphA1 overexpressed SGC7901 cell line group, Normal: nude mice subcutaneously inoculated with Normal SGC7901 cell line group, Si-EphA1: nude mice subcutaneously inoculated with EphA1 gene silencing SGC7901 cell line group).
EphA1 reduced the levels of IL-6 and VEGF in xenograft nude model tumour tissues
The relative expression levels of EphA1, VEGF and IL-6 genes in tumor tissues of three groups of nude mice transplanted subcutaneous (OV-EphA1: nude mice subcutaneously inoculated with EphA1 overexpressed SGC7901 cell line group, Normal: nude mice subcutaneously inoculated with Normal SGC7901 cell line group, Si-EphA1: nude mice subcutaneously inoculated with EphA1 gene silencing SGC7901 cell line group).

The relative expression levels of EphA1, VEGF and IL-6 proteins in tumor tissues of the three groups of nude mice transplanted subcutaneous (OV-EphA1: nude mice subcutaneously inoculated with EphA1 overexpressed SGC7901 cell line group, Normal: nude mice subcutaneously inoculated with Normal SGC7901 cell line group, Si-EphA1: nude mice subcutaneously inoculated with EphA1 gene silencing SGC7901 cell line group).

Discussion
EphA1 is a transmembrane protein consisting of an extracellular region that binds ligands, a transmembrane region and an intracellular region that acts as both receptor and enzyme. Studies have shown that most Eph receptors function through Eph/Ephrin signal transduction pathways.17,18 So how does EphA1 work when activated? We don’t know yet, and further research is needed. For a long time, most of the research on cancer has focused on the level of tumor cells, but little attention has been paid to the tumor microenvironment. More and more studies suggest that the analysis of tumor microenvironment is of great significance for exploring the occurrence and development of tumor and the treatment of metastasis.19,20 The environment around tumor cells is the tumor microenvironment, among which the vascular endothelial growth factor promoting tumor angiogenesis and the important inflammatory cytokines constituting the inflammatory environment have the greatest influence on tumor cells. Since some previous resarches have indicated that Ephs is related to angiogenesis in tumor microenvironment and oxidative stress induced by inflammation during solid tissue tumorigenesis,13–15 the design concept of this study was derived. That is, we speculated that EphA1 may enhance the expression of VEGF and IL-6 in tumor microenvironment, thus promoting the tumorigenesis and progression of malignant tumors.
In the present study, we constructed human EphA1 gene overexpression SGC7901 lentivirus-stable transfected cell lines and human EphA1 gene interference SGC7901 lentivirus-stable transfected cell lines, and then implanted them under the skin of nude mice to construct subcutaneous xenograft tumor animal model of SGC7901 cells. To verify our hypothesis, we analyzed and compared the changes in cell function of the three cell lines (OV-EphA1 group, Normal control group, Si-EphA1 group) and the changes in the expression levels of VEGF, IL-6 genes and proteins in the cells, and further analyzed the changes in the tumorigenesis of the three groups (OV-EphA1 group, Normal control group, Si-EphA1 group) of nude mice and the changes in the expression levels of VEGF, IL-6 genes and proteins in the tumor tissues. Results show that the enhanced EphA1 gene expression, the increased number of SGC7901 cell proliferation and reduced apoptosis, cell migration and invasion occurred more, transplanted tumor significantly speed up, characterized by growth curve becomes steeper than normal control group; whereas inhibition of gene expression after EphA1 appeared opposite results. These results validate the EphA1 may promote the occurrence and development of malignant tumor cell SGC7901 hypothesis. We also found that after the up-regulation of EphA1 gene expression, the expression levels of VEGF and IL-6 in SGC7901 and transplanted tumor tissues also increased. While the expression levels of VEGF and IL-6 genes and proteins in SGC7901 cells and nude mice decreased significantly compared with the normal control group when the EphA1 gene was knocked down.
In-depth analysis of our experimental results
The activation of a receptor tyrosine protein kinase caused trepidation of angiogenesis factor and inflammatory cytokines, whether through what kind of signaling pathways, one thing is certain, the biggest impact on this change is tumor microenvironment, because both vascular excessive proliferation and continue uncontrolled chronic inflammation can cause interstitial high pressure formation, hypoxia and acidosis, etc., and these are the unique characteristics of the tumor microenvironment, this feature in the progression and metastasis of tumor plays an important role. 21 So how does EphA1 regulate the expression of VEGF and IL-6? Through reading the analytical literature and thinking in the experiment, we preliminarily speculated that it was related to activation of other signal systems on the cell surface. For example, the RAS-MAPK pathway and jak-STAT pathway, as well as the VEGF-VEGFR signaling axis of the angiogenesis signaling pathway. We can reasonably speculate that: Abnormal expression of EphA1 gene variants leads to abnormal activation of inflammatory signaling pathways and angiogenic signaling pathways, which leads to abnormal expression of cytokines in these signaling pathways (such as IL-6 and VEGF), and the high secretion of these cytokines in turn enhances the activation of these signaling pathways. Cycling to form the uncontrolled inflammatory response in cells and angiogenesis, could eventually lead to cancerous cells, and tumor formation, microenvironment of tumor markers protein VEGF in tumor microenvironment, abnormal secretion of IL-6 and strengthens the special micro environment and promoting tumor angiogenesis, inflammation promoting tumor cell proliferation and malignant invasion. This study for the first time found that EphA1 is linked to tumor microenvironment through the link of VEGF and IL-6 from the perspective of mechanism. Of course, there are still some limitations and shortcomings in our study: for example, only one cell line is used for the study, the experimental sample size is too small because the sample size calculation in this study was not done, and the effect of subcutaneous transplanted tumor model is not as good as that of in-situ transplanted tumor model. We will perfect and improve these deficiencies in future research.
Conclusions
In summary, this study reports that EphA1 functions as an tumour microenviroment promoter by enhancing the IL-6 and VEGF expression and hence has an important role in cancer occurrence and development. EphA1 is therefore an excellent candidate for the development of a targeted cancer therapy that will opens up a new way for tumor gene therapy.
Supplemental Material
Supplemental Material - Erythropoietin-producing hepatocyte kinase receptor A1 facilitating the prgression of SGC-7901 cells and its transplanted tumor by increasing the expression of interleukin-6 and vascular endothelial growth factor in tumor microenvironment
Supplemental Material for Erythropoietin-producing hepatocyte kinase receptor A1 facilitating the prgression of SGC-7901 cells and its transplanted tumor by increasing the expression of interleukin-6 and vascular endothelial growth factor in tumor microenvironment by Yong-Cang Wang, Wen-Lin Zheng, Wei Yu, Rui-Liang Quan and Ya-Jun Zhao in European Journal of Inflammation
Footnotes
Author contributions
Yong-Cang Wang, Wen-Lin Zheng and Wei Yu had the idea for the article, performed literature search and data analysis. Yong-Cang Wang drafted the article and Rui-Liang Quan, Ya-Jun Zhao critically revised the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the ethical committee of Anhui Provincial Cancer Hospital.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
