Abstract
Dysregulation of the PI3K/AKT/ mammalian target of rapamycin (mTOR) pathway could contribute to the pathogenesis of autism spectrum disorders. In this study, phosphorylated Akt concentration was measured in 37 autistic children and 12, gender and age similar neurotypical, controls using an enzyme-linked immunosorbent assay. Akt levels were compared to biomarkers known to be associated with epidermal growth factor receptor (EGFR) and c-Met (hepatocyte growth factor (HGF) receptor) pathways and severity levels of 19 autism-related symptoms. We found phosphorylated Akt levels significantly lower in autistic children and low Akt levels correlated with high EGFR and HGF and low gamma-aminobutyric acid, but not other biomarkers. Low Akt levels also correlated significantly with increased severity of receptive language, conversational language, hypotonia, rocking and pacing, and stimming, These results suggest a relationship between decreased phosphorylated Akt and selected symptom severity in autistic children and support the suggestion that the AKT pathways may be associated with the etiology of autism.
Introduction
Autism spectrum disorders (ASDs) comprise a diverse group of conditions characterized by problems in social actions, communication deficits, and stereotypical repetitive behaviors. In all, 1 in 88 children in the US is thought to have ASD. 1
The PI3 K/AKT/ mammalian target of rapamycin (mTOR) signaling pathway plays an important role in the regulation of cell growth, proliferation, differentiation, motility, survival, metabolism, and protein synthesis. AKT, a serine/threonine-specific protein kinase, plays a fundamental role in cell survival and apoptosis. 2 Activated AKT can phosphorylate a series of downstream signaling molecules, including mTOR (which participates in various aspects of signaling, including nutrition (such as amino acids), growth factors (such as insulin), energy levels, and environmental pressures (such as hypoxia)). 3
Studies suggest that the dysregulation of the PI3K/AKT7 mTOR pathway possibly contributes to the pathogenesis of ASDs. For instance, mutations/deletions of PTEN, a negative regulator of the PI3K pathway, leads to autistic-like features. 4 Also, comorbidity of ASDs and tuberous sclerosis complex (TSC) neurogenetic disorder are very common. 5 In a mouse model of TSC disorder, researchers found that intraperitoneal injection of the mTORC1 inhibitor, rapamycin, can improve the behavior, 6 and the PI3K/AKT/mTOR pathway is likely involved in the pathogenesis of Rett syndrome, a rare form of autism that affects females. 7
In addition to its possible relationship to autism etiology, the PI3K/AKT pathway maybe important in the diagnosis and treatment of ASD. The mTOR-targeted inhibitor rapamycin has shown effects on intelligence recovery in autistic mice. 8 It will be of great significance to further study whether other elements in the PI3K/AKT/mTOR pathway could serve as therapeutic targets for ASDs. 9
Because of this, we studied pathway markers associated with the PI3K/AKT pathway. The epidermal growth factor receptor (EGFR, ErbB-1, HER1 in humans) is a member of the ErbB family of receptors, and when signaled by growth factors such as epidermal growth factor (EGF), its phosphorylation is a precursor for Akt pathway activation. 10–13 EGFR triggers quiescent astrocytes into reactive astrocytes 14 and has been associated with neurobehavioral disorders, such as amyotrophic lateral sclerosis (ALS) 15 and Alzheimer's. 16–18
Genetic variants of receptor tyrosine kinases (RTKs), such as MET, have been implicated in the etiology of autism.18,19 Growth factors, hepatocyte growth factor (HGF) and EGF, which are the signaling ligands for MET and EGFR, respectively, are decreased in autism.20,21 Attachment of growth factors to these receptors regulates many aspects of neuronal growth and differentiation,22–27 and they have been found to promote wound healing. 28 Since there is immune dysfunction in the nervous system29–33 in children with autism, it is plausible to think that growth factors are involved.
Gene mutations 34 and abnormal plasma levels35,36 of growth factors such as EGF have been linked to autism.
HGF has mitogenic, morphogenic, and motogenic effects on hepatocytes as well as endothelial, mesenchymal, and hematopoietic effects on cell types and demonstrates noticeable species cross-reactivity.37–48 Its ability to stimulate branching morphogenesis, cell migration, survival, and proliferation gives it a central role in angiogenesis, tissue regeneration, as well as tumorogenesis.37–43 It also plays significant roles in the development of the central nervous system. 46
Gamma-aminobutyric acid (GABA), the most abundant inhibitory neurotransmitter in the mammalian brain, where it is widely distributed, 47 has been found to be dysfunctional in autism and related disorders. 48 HGF has been shown to modulate GABAergic activity 49 and enhance 7V-methyl-D-aspartate currents in the hippocampus. 50
In this study, we measured phosphorylated Akt in WBCs of autistic children and compared these levels with putative biomarkers, such as EGFR, EGF, HGF, and GABA as well as the severity of 19 different behavioral symptoms.
Materials and Methods
Subjects
Cellular phosphorylated Akt concentration was measured in 37 autistic children and 12, age and gender similar neurotypical, controls.
The diagnostic criteria used in this study were defined by Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) criteria. In 2012, the separate diagnostic labels of autistic disorder, Asperger's disorder, and pervasive developmental disorder not otherwise specified were replaced by one umbrella termed autism spectrum disorder.
Plasma and white blood cells from consecutive individuals with diagnosed autism (n = 37, 29 males, mean age 10.1 years) and controls (n = 12, 8 males, mean age 9.4 years) were obtained from patients presenting at the Health Research Institute (HRI)
*
over a two-year period. All autistic individuals who presented to
The HRI is a comprehensive treatment and research center, specializing in the care of with neurological disorders, including autism.
The AGRE is a repository of biomaterials and phenotypic and genotypic data to aid research on ASDs.
Patient consent was obtained from all patients involved in this study, and this study was approved by the institutional review board of the
Enzyme-linked immunosorbent assays (ELISAs) were used to measure cellular Akt and other biomarkers (eBioscience).
50 μL/well of 1 × Cell Lysis Mix (negative control) and 50 μL/well of positive control cell lysate (positive control) were used to separate assay wells for controls.
40 μL of lysis buffer (containing a combination of detergents, phosphatase inhibitors, salts, and buffers) was added to each of the control and experimental wells.
10 μL of buffy coat cells (experimental and control) were added to appropriate wells and mixed gently.
50 μL/well of antibody cocktail mix (detection antibody and horse radish peroxidase (HRP)-conjugated antibody) was added to all the assay test wells. The plate was incubated for one hour at room temperature on a microplate shaker (~300 rpm).
Wells were washed with 300 μL/well 1 × Wash Buffer four times.
100 μL of detection reagent (3,3’,5,5‘-tetramethylbenzidine) was added to each well, and the wells were incubated for 10–30 minutes.
After color development, 100 μL of Stop Solution was added to each well.
Absorbance was measured using a colorimetric (spectrophotometric) plate reader (BioRad) set at 450 nm.
To ensure reproducibility of results, samples were run in duplicate, and reported concentrations were the result of the average of at least two separate assays.
Serums
Buffy coat cells, obtained from the patients at the HRI, were treated in an identical fashion - frozen at –70 °C immediately after collection and cell/serum separation and then stored at –70°C until thawed for use in ELISAs.
Severity of disease
The Pfeiffer questionnaire, severity criteria, and statistical methodology have been previously reported. 21
An autism symptom severity questionnaire was used to evaluate symptoms. The questionnaire (Pfeiffer questionnaire) asked parents or caregivers to assess the severity of the following symptoms: awareness, expressive language, receptive language, (conversational) pragmatic language, focus, attention, hyperactivity, impulsivity, perseveration, fine motor skills, gross motor skills, hypotonia (low muscle tone), tip toeing, rocking/pacing, stimming, obsessions/fixations, eye contact, sound sensitivity, light sensitivity, and tactile sensitivity. The symptoms were rated by parents/guardians on a scale of 0–5 (5 being the highest severity) for each of these behaviors.
Statistics
Inferential statistics were derived from unpaired t-test and odds ratios with 95% confidence intervals. Pearson moment correlation test was used to establish degree of correlation between groups using 95% confidence intervals.
Results
We found that cell phosphorylated Akt concentration is significantly lower (P = 0.04; 95% confidence interval) in individuals with autism (Fig. 1). We also found a correlation between these levels and that high EGFR (r = –0.5; P = 0.05) (Fig. 2) has a negative correlation with Akt and HGF (r= –0.82; P = 0.0005) (Fig. 3) has a superior negative correlation with Akt. We also found that low phosphorylated Akt correlates well with low GABA (r= 0.5; P = 0.02) (Fig. 4) in the individuals with autism.

Cell Phosphorylated Akt is significantly lower in individuals with autism (P= 0.04).

Cell Phosphorylated Akt correlates significantly with EGFR in individuals with autism (r = – 0.5; P = 0.05).
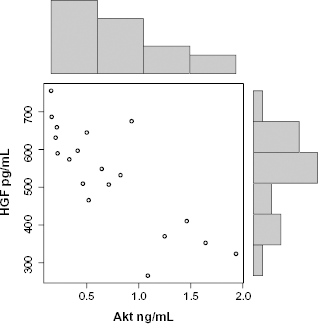
Cell Phosphorylated Akt correlates significantly with HGF in individuals with autism (r= –0.82; P = 0.0005).

Cell Phosphorylated Akt correlates significantly with GABA in individuals with autism (r=0.5; P= 0.02).
We found that low Akt levels correlated with high severity of rocking and pacing (r= –0.62; P = 0.001), high severity of hand and finger stimming (r = –0.42; P = 0.02), high severity of obsessions and fixations (r = –0.4; P = 0.03), and severity of hypotonia (r = –0.5; P = 0.009).
Discussion
The data reported here suggest a relationship between Akt levels and autism and also suggest that the EGFR and c-Met pathways are associated with lower activated Akt levels. These data also show a strong relationship between low activated Akt and low GABA.
If ASDs are linked to the AKT signaling pathway, there are implications for the diagnosis and treatment of these disorders. The diagnosis of ASDs currently relies on behavioral symptoms, which usually lead to late or missing diagnosis. Molecular diagnosis may be the answer to this deficiency. So far, in addition to AKT signaling molecules, many other candidate genes, and their resulting proteins, have been selected for ASDs, such as reelin (RELN), human serotonin transporter (SLC6A4), gamma-aminobutyric acid receptor (GABR), neuroligin (NLGN), human oxytocin receptor (OXTR), and c-Met. 51
Linking ASDs to the dysregulation of AKT signaling may also lead to therapeutic modalities. Recombinant human insulin-like growth factor-1 (RhIGF-I), known to signal the AKT pathway, has been shown to play important roles in brain damage, diabetes, growth retardation, osteoporosis, and chronic renal failure. RhIGF-I was approved by the US Food and Drug Administration (FDA) in 2005 for treatment of short stature patients with severe primary insulin-like growth factor-1 (IGF-I) deficiency. Several studies have reported that there are some adverse effects of RhIGF-I, such as headache, vomiting, and hypoglycemia. However, these adverse events are mostly transient, easily handled, and infrequently lead to noncompliance. 52 Given the ease of operability and the safety of IGF-I, it may be a potent therapy for ASDs.
The mTOR-targeted inhibitor rapamycin is a macrolide compound that is purified from Streptomyces hygroscopicus. It was originally designed as an antifungal agent before it was found to have strong immunosuppressive and antiproliferative effects. Rapamycin was approved by the FDA as an immunosuppressant for renal transplantation in 1999. Recently, this drug has been used as a coronary stent coating and may be valuable as an anticancer treatment because of its antiproliferative effects.53–55 Interestingly, rapamycin also showed effects on intelligence recovery in autistic mice, 8 and may, therefore, provide another potent therapy for ASDs. It is possible that other elements of the AKT pathway serve as therapeutic targets for ASDs.
Our data suggest that in autistic children, decreased Akt levels strongly correlate with high RTK (EGFR), high growth factor (HGF), and low GABA. As you can see in Figure 5, summarizing the potential events associated with the Akt pathway (Fig. 5), HGF is a major growth factor that attaches to RTKs, such as c-Met. Subsequent phosphorylation (activation) of the receptor causes a chain of marker activation, which eventually activates Akt. Akt, in turn, affects the plasticity of GABAA receptors. It is plausible that the etiology of at least a subpopulation of autistic individuals, perhaps those with low GABA, is associated with an aberrant HGF/c-Met/Akt pathway. Particularly, high plasma HGF (the result of abnormal, but high levels of RTKs (ie, EGFR, c-Met)) and the inability to attach to these RTKs lower signaling, which, in turn, lowers Akt and GABA.
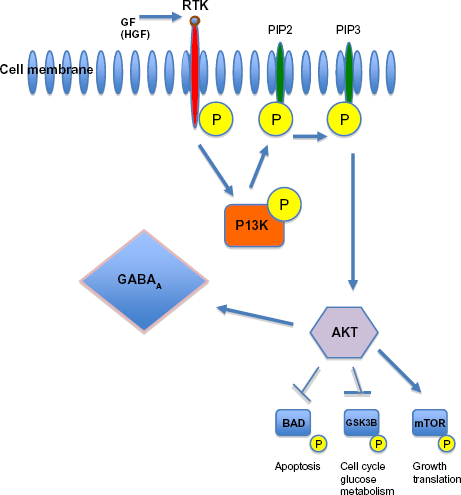
AKT is activated by PI3K, which itself is activated by several upstream signaling pathways such as receptor tyrosine kinases (RTK). After activation, it targets several downstream molecules and changes their activity by phosphorylation (
Some research supports our finding of downregulation of the Akt pathway associated with autism,56,57 while other data suggest that, in PTEN knockout mice, upregulation of the pathway results in autism-like behavior.4,58 It is possible that high Akt levels exist in a distinct subpopulation of individuals with Phosphatase and tensin homolog (PTEN) gene mutation.
Author Contributions
AJR carried out the immunoassays, participated in the design of the study, and performed the statistical analysis. AJR conceived the study and participated in its design and coordination. AJR drafted and approved the final manuscript.
Footnotes
Acknowledgements
The author would like to acknowledge the financial support from the Autism Research Institute and the resources provided by the Autism Genetic Resource Exchange (AGRE) Consortium and the participating AGRE families. The Autism Genetic Resource Exchange is a program of Autism Speaks and is supported, in part, by grant 1U24MH081810 from the National Institute of Mental Health to Clara M. Lajonc.
