Abstract
Introduction
There is support for the role of gamma aminobutyric acid (GABA) in the etiology of mood disorders. Recent research has shown that hepatocyte growth factor (HGF) modulates GABAergic inhibition and seizure susceptibility. This study was designed to determine and correlate plasma levels of HGF and GABA as well as symptom severity in individuals with obsessive-compulsive disorder (OCD).
Subjects and Methods
Plasma from 15 individuals with OCD (9 males, 6 females;, mean age 38.7 years) and 17 neurotypical controls (10 males, 7 females; mean age 35.2 years) was assessed for HGF, GABA, urokinase plasminogen activator (uPA), and urokinase plasminogen activator receptor (uPAR) concentration using enzyme-linked immunosorbest assays ELISAs. Symptom severity was assessed in these OCD individuals and compared with HGF and GABA concentrations.
Results
In this preliminary study, individuals with OCD had significantly decreased HGF levels, decreased plasma levels of GABA and decreased uPA. We found that both uPA and uPAR levels correlate with HGF. Both low uPA and low uPAR levels correlate with high symptom severity in individuals with OCD. Low GABA levels in OCD individuals also correlate with high symptom severity.
Discussion
These results demonstrate a preliminary association between HGF, GABA, uPA levels, and OCD and suggest that plasma GABA and uPA levels are related to symptom severity in individuals with OCD.
Introduction
Obsessive-compulsive disorder (OCD) is an anxiety disorder in which people have unwanted and repeated thoughts, feelings, ideas, sensations (obsessions), or behaviors that make them feel driven to do something (compulsions). 1
Obsessions are often characterized as persistent and unwanted intrusive thoughts that cannot easily be dismissed. These obsessions are generally perceived as foreign and recognized as being irrational or excessive. The anxiety associated with obsessions is often described as a feeling that something is incomplete or wrong or that terrible consequences will ensue if specific actions are not taken. Many patients engage in repetitive, compulsive behaviors that aim to discharge the anxiety associated with obsessional thoughts.2,3
The lifetime prevalence of OCD is estimated to be 1% to 3%, based on population-based surveys conducted in many communities nationally and internationally. 4 Although the disorder affects individuals of all ages, the period of greatest risk is from childhood to early adulthood.5,6 Patients experience a chronic or episodic course with exacerbations that can substantially impair social, occupational, and academic functioning; according to the World Health Organization, OCD is among the 10 most disabling medical conditions worldwide.7,8
There is evidence for the role of gamma aminobutyric acid (GABA) and of excessive glutamatergic and reduced GABAergic activity in OCD. 9 Cerebrospinal fluid (CSF) from patients with OCD found CSF glutamate levels to be significantly elevated in subjects with OCD. 10 Magnetic resonance spectroscopy (MRS) findings also implicate dysregulation of glutamate neurotransmission in cortico-striato-thalamo-cortical (CSTC) circuits in OCD.11–16
Candidate genes also support the possible relationship between glutamate, GABA, and the etiology of OCD. Recently, there have been reported associations between OCD and the BDNF locus, 17 glutamate N-methyl-D-aspartate (NMDA) subunit receptor gene, 18 and GABA type B receptor 1 (GABBR1) gene has been observed to be overtransmitted at the A-7265G polymorphism, 19 OLIG2, 22 and myelin oligodendrocyte glycoprotein (MOG) gene. 20
The relationship between glutamate and GABA is an intricate one. Most GABA synthesis in the brain is catalyzed in inhibitory neurons from L-glutamic acid by the enzyme glutamic acid decarboxylase 67 (GAD67). In vivo, knockdown of GAD67 in the amygdala disrupts fear extinction and the anxiolytic like effect of diazepam in mice. 21
GABA is the most abundant inhibitory neurotransmitter in the mammalian brain, where it is widely distributed. 22 While in the mature brain, GABA acts as an inhibitory transmitter, and during the embryonic and the perinatal period, it depolarizes targeted cells and triggers calcium influx, regulating different developmental processes from cell proliferation migration, differentiation, synapse maturation, and cell death. 23
Hepatocyte growth factor (HGF), an 82 kDa, 674 amino acid residue heterodimeric glycoprotein, was originally isolated from rat platelets.24,25 HGF has mitogenic, morphogenic, and motogenic effects on hepatocytes, as well as endothelial, mesenchymal and hematopoietic cell types.26–28 HGF regulates cell growth, cell motility, and morphogenesis by activating a tyrosine kinase-signaling cascade after binding to the proto-oncogenic c-Met receptor (translated by the MET gene). HGF has been shown to modulate GABAergic activity 29 and enhance NMDA currents in the hippocampus. 30
uPAR is a membrane bound receptor for uPA, otherwise known as urokinase. When uPA is bound to the receptor, there is cleavage between the glycosylphosphatidylinisotol (GPI)-anchor and the uPAR, releasing suPAR (soluble uPAR). 31 Specific evidence to support HGF modulation of GABA activity is data associated with a mouse model of autism, the uPAR–/– mouse, which displays a spatially selective defect in interneuron migration such that the frontoparietal cortices of these mice show 50% less calbindin-positive interneurons (with a near absence of PV cells), whereas more caudal cortices are spared.32,33 These mice display autistic-like behaviors with increased anxiety and altered socialization, as well as interictal epileptiform EEG activity and an increased susceptibility to seizures.32,33 uPAR encodes an urokinase plasminogen activator that is required for the proper processing of the hepatocyte growth factor (HGF). In turn, HGF, through its receptor MET, has been shown to be a critical motogen for interneuron migration and is able to rescue the interneuron migration defect and seizure susceptibility of uPAR–/– mice.33,34
The aim of this preliminary study was to determine and correlate plasma levels of HGF, GABA, GAD, uPA, uPAR, as well as symptom severity in individuals with OCD and neurotypical controls.
Materials and Methods
Enzyme-linked immunosorbest assay (ELISA) was used to measure serum HGF, GABA, uPA, uPAR (ELISA kits, R&D Systems, Minneapolis, MN and USCN Life Sciences, Wuhan, China).
All reagents and specimens were equilibrated to room temperature before the assay was performed. A 1:51 dilution of the patient samples was prepared by mixing 10 μL of the patient's sera with 0.5 mL of serum diluent. One hundred microliters of calibrators (20–200 Eu/mL antibodies), positive and negative control serums, serum diluent alone, and diluted patient samples were added to the appropriate microwells of a microculture plate (each well contained affinity purified polyclonal IgG to HGF or GABA). Wells were incubated for 60 minutes (±5 minutes) at room temperature, then washed 4 times with wash buffer. One hundred microliters of prediluter antihuman IgG conjugated with HRP was added to all microwells, incubated for 30 minutes (±5 minutes) at room temperature, and then washed 4 times with wash buffer. One hundred microliters of enzyme substrate was added to each microwell. After approximately 30 minutes at room temperature, the reaction was stopped by adding 50 μL of 1M sulfuric acid, and then the wells were read at 405 nm with an ELISA reader (BioRad Laboratories, Inc., Hercules, CA, USA).
Plasma
All plasma samples, experimental and control, were treated in an identical fashion: frozen at −70 °C immediately after collection and cell/serum separation (centrifugation at 1500 rpm for 10 minutes), then stored at −70 °C until thawed for use in ELISAs.
Subjects
Experimental and control
Plasma from 15 individuals with OCD (mean age 38.7 years; 8 males and 7 females) and 17 neurotypical controls (mean age 36.2 years; 9 males and 8 females) were obtained from patients presenting at the Health Research Institute/Pfeiffer Treatment Center* and the Autism Genetic Resource Exchange**. The autistic individuals met the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) criteria and many were diagnosed using the Autism Diagnostic Interview-Revised (ADI-R).
The Pfeiffer Treatment Center, Warrenville, Il, is a comprehensive treatment and research center, specializing in the care of with neurological disorders, including autism.
The Autism Genetic Resource Exchange is a program of Autism Speaks.
Patient consent was obtained from all patients involved in this study, and this study was approved by the institutional review board (IRB) of the Health Research Institute/Pfeiffer Treatment Center.
Severity of disease
A modified questionnaire asking patients to rank the severity of the signs and symptoms of OCD, found in the Mood Disorder Questionnaire, 35 was used to determine the overall severity. Patients were asked to rate on a scale of 0 to 5 (5 being the highest) behaviors such as irritability and anger, lack of ability to focus/concentrate, obsessive behavior, trouble sleeping, paranoia, migraines, intrusive thoughts, anxiety, depression, and panic. We evaluated the overall severity of OCD by establishing the mean of all of the scores for each patient. The overall OCD behavior assessment includes mean assessment of all the behaviors (above).
Statistics
Inferential statistics were derived from unpaired t test and odds ratios with 95% confidence intervals. ANOVA was used to assess variance between groups. The Pearson product moment correlation test was used to establish the degree of correlation between groups.
Results
In this study, individuals with OCD had significantly decreased HGF levels (P = 0.0001) (Fig. 1) decreased plasma levels of GABA (P = 0.01) (Fig. 2), and decreased uPA (P = 0.002) (Fig. 3). We found that in 14 patients where uPA and uPAR were both measured, both uPA (Fig. 4) and uPAR (Fig. 5) levels, measured in 14 patients, correlated with HGF (r = 0.8; P = 0.0005 and r = 0.5; P = 0.03, respectively). In 13 of these patients (1 patient did not provide symptom severity data), both low uPA (r = −0.5) (Fig. 6) and low uPAR (r = −0.3) levels correlate with high symptom severity in individuals with OCD, and these same individuals low GABA levels correlate with high symptom severity (r = 0.3).
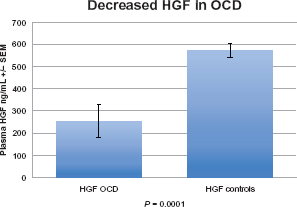
Plasma HGF in individuals with OCD. Individuals with OCD had significantly decreased HGF levels (P = 0.0001).

Plasma GABA in individuals with OCD. Individuals with OCD had significantly decreased plasma levels of GABA (P = 0.01).
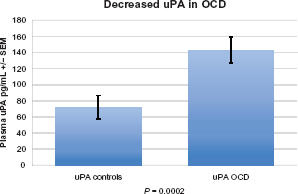
Plasma uPA in individuals with OCD. Individuals with OCD had significantly decreased uPA (P= 0.002).
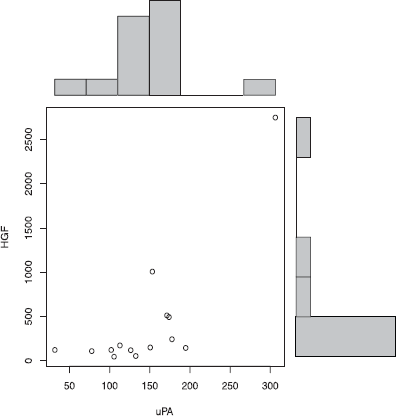
Plasma uPA correlates with HGF in individuals with OCD. uPA levels correlate with HGF (r= 0.8; P = 0.0005) in individuals with OCD.
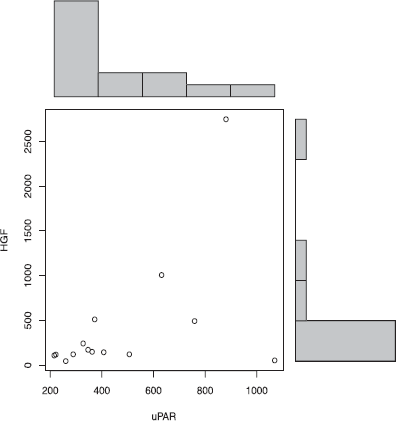
Plasma uPAR correlates with HGF in individuals with OCD. uPAR levels correlate with HGF (r= 0.5; P = 0.03) in individuals with OCD.

Low plasma uPA levels correlate with high symptom severity in individuals with OCD. Low uPA (r= −0.5) levels correlate with high symptom severity in individuals with OCD.
Discussion
OCD is a complex genetic disorder with unknown genetic and environmental bases. There are likely common genetic influences of modest effect (possibly in addition to other less common genetic factors). Moreover, the biological basis of OCD is largely unknown, preventing a more focused genetic search in particular metabolic pathways.
Other studies implicate glutamatergic excess in the pathophysiology of OCD. CSF glutamate levels are significantly raised in OCD patients. 36 Several single photon emission computed tomography, positron emission tomography, and functional magnetic resonance imaging studies have demonstrated increased cerebral blood flow, metabolism, and activation in the CSTC circuitry of individuals with OCD.37–40 These well-established neuroimaging findings in OCD have led to the hypothesis that glutamatergic dysfunction contributes to the regional metabolic hyperactivity seen in OCD.41,42
Our data, demonstrating significantly lower levels of GABA in the plasma of individuals with OCD compared with controls, support the work of others who have found altered GABA levels in individuals with mood disorders 43 and is logical when considering that glutamate is increased in patients with OCD. Histological, biochemical, and molecular approaches have demonstrated altered levels and distribution of GABA and GABA receptors in peripheral blood and plasma, as well as in the brain, including decreased GABA-A receptors and benzodiazepine binding sites in the hippocampal formation in other behavioral disorders such as autism.44–46 A recent study has reported that individuals with OCD may have decreased medial prefrontal cortex GABA levels. 47 There is also evidence for the gamma-aminobutyric acid type B receptor 1 (GABBR1) gene as a susceptibility factor in obsessive-compulsive disorder. 48 Although our study measures putative biomarkers in plasma of individuals with OCD, correlation between CSF and plasma GABA levels has been found in animals49,50 and humans. 51
To our knowledge, this is the first study to show significantly decreased GABA and HGF levels and the correlation between these putative markers and controlling molecules such as uPA in individuals with OCD. It is reasonable to suggest that decreased GABA, mediated by HGF dysregulation, is related to the glutaminergic dysfunction in OCD. Glutamate is a precursor to GABA, so if GABA is decreased in these patients, this could result in excess (unconverted) glutamate. We found decreased uPA and correlations between uPA, its receptor uPAR, and HGF. This suggests an involvement between decreased HGF and GABA modulation in OCD. In this study, we also show correlation between these markers and symptom severity, which supports the role of these putative biomarkers in the etiology of abnormal behavior found in individuals with OCD.
Abbreviations
Hepatocyte growth factor (HGF), Gamma-Aminobutyric acid (GABA), glutamic acid decarboxylase (GAD), urokinase plasminogen activator (μPA), urokinase plasminogen activator receptor (μPAR, PAUR), enzyme linked immunosorbest assay (ELISA), N-methyl-D-aspartate (NMDA), MET or MNNG HOS Transforming gene (c-Met).
Author Contributions
AR and SP carried out the immunoassays, participated in the design of the study and performed the statistical analysis. AR and SP conceived of the study, and participated in its design and coordination. AR and SP drafted and approved the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
