Abstract
Background
The role of organic anion transporting polypeptide 5A1 (OATP5A1) a member of a family of drug transporters that mediate cellular uptake of drugs has not been characterized so far.
Methods
Gene expression levels of OATP5A1 in small cell lung cancer (SCLC) cell lines were determined by real-time qPCR and chemosensitivity of HEK-293-SLCO5A1-transfected cells to satraplatin in MTT assays.
Results
Significant expression of this transporter was found at the mRNA level, primarily in drug-resistant SCLC cells, and SLCO5A1-transfected HEK-293 cells showed higher resistance to satraplatin. OATP5A1 is found preferentially in cytoplasmic membranes of tumor cells, including SCLC.
Conclusions
OATP5A1 seems to effect intracellular transport of drugs and may participate in chemoresistance of SCLC by sequestration, rather than mediating cellular uptake. Since satraplatin failed to improve survival in SCLC patients, the relation of OATP5A1 expression to clinical drug resistance and its use as marker of chemoresistance should be further investigated.
Introduction
Lung cancer is still one of the most causes of cancer deaths worldwide. 1 Tumors frequently reveal resistance to common chemotherapeutic drugs, such as cisplatin or etoposide, impairing the efforts to treat the patients and increase survival. 2 Several mechanisms implicated in chemoresistance of tumor cells including expression of drug efflux-mediating membrane proteins, eg, ATP binding cassette transporters like P-glycoprotein have been described besides increased DNA repair in response to cisplatin-induced DNA damage and others were described. 3 Newly discussed drug transporters include the group of organic anion transporting polypeptides (OATPs) that belong to the solute carrier organic anion (SLCO) transporting polypeptides family of the solute carrier (SLC) transporter superfamily, respectively.4–6 Eleven human OATPs classified in six distinct subfamilies (OATP1-6) have been identified so far. 7 They are expressed in a variety of tissues including intestine, liver, kidney and brain. 8 OATPs possess twelve transmembranespanning domains yielding six extracellular and five intracellular loops with both N- and C-termini facing the cytosol. OATP-mediated transport is independent from ATP and transmembrane Na+, Cl– or K+ gradients. OATP3A1, OATP4A1, OATP2B1 and OATP2A1 are widely distributed in various tissues and characterized by a broad spectrum of substrate specificity for amphipathic organic anions, neutral as well as few cationic organic compounds, while OATP1B1 and OATP1B3 are believed to be localized to the basolateral membrane of human hepatocytes and OATP6A1 expression is constricted to the testis. Among the substrates of the OATPs are endogenous substances like bile acids, bilirubin, eicosanoids, thyroid hormones, steroid conjugates and oligopeptides. Xenobiotics transported by OATPs include antibiotics, statins, fexofenadine and glibenclamide, as well as anticancer drugs like methotrexate, paclitaxel, docetaxel and imatinib, respectively. 9 Interestingly, a number of OATPs share substrate specificity with some cellular efflux pumps, such as multidrug resistance protein 1 (MDR1) and multidrug resistance-associated protein 2 (MRP2). 7 A common transport mechanism has been proposed for all OATPs, in which substrates are translocated through a central, positively charged pore in a rocker switch type mode. 10 However, it is still unclear whether this process involves the coupled movement of another solute across the membrane. Except for OATPs 1B1, 1B3, 1A2 and 2B1, actual tissue distribution, physiological functions and substrate specificities of the other OATPs have remained largely unknown. Due to the present lack of specific inhibitors assessment of a pharmacokinetic profile of substrates for each of those transporters will be a major challenge. 8
Several studies reported on expression of OATPs in different tumor entities. Marked overexpression of OATP1B3 was found in up to 80% of colorectal adenocarcinomas and immunostaining was absent in normal colonic tissue. 11 In breast cancer, six of the eleven OATPs were found, but there was no relation with either age, tumor size, hormone receptors or HER-2 status of patients. 12 Interestingly, mRNA expression of SLCOs 2B1, 3A1 and 4A1 was significantly higher in nonmalignant specimens in comparison to breast tumor tissue samples. In contrast to benign bone lesions mRNA levels of SLCOs were generally reduced in specimens derived from osteosarcomas and bone metastases. 13 Gene expression analysis revealed presence of all SLCOs except SLCOs 1C1 and 6A1 in the majority of liver cancer tissue samples [manuscript in press]. 14 Marked upregulation of SLCOs 4A1 and 5A1 at the mRNA and protein levels were observed in metastatic liver cancer.
Up to now, far less is known in respect to tissue distribution and substrate specificity of OATP5A1. 9 The putative OATP5A1 protein with a molecular mass of 92 kDa consists of 848 amino acids. Realtime RT-PCR revealed SLCO5A1 mRNA expression in thymus, heart, skeletal muscle, prostate and fetal brain; however, these results have not been confirmed in immunohistochemistry assessing protein expression and functional studies are still pending. 15 The expression profile of OATP5A1 (available at: www.proteinatlas.org) revealed weak to moderate cytoplasmic protein expression in normal cells with highest levels in adrenal cortical and ovarian follicular cells. Moreover, aberrant expression of OATPs is frequently found in malignant tissues. In particular, elevated expression of OATP2A1 and OATP5A1 was detected in primary and secondary hepatic tumors at both the mRNA and protein levels. Strong OATP5A1 expression was also demonstrated for urothelial and renal tumors, besides minor appearance in colorectal, pancreatic and several other cancer tissues (www.proteinatlas.org). 16
In dependence of their lipophilicity, anticancer drugs may enter tumor cells generally by diffusion on the one hand or may be carried into the cells more specifically by a transport mechanisms on the other hand. Most SLCs are capable of moving various structurally unrelated molecules across the cell membranes by facilitated diffusion.17, 18 In addition to OATP1B3 mediating the cellular uptake of paclitaxel, OATP1B1 is now regarded as another paclitaxel-carrying membrane protein [Svoboda et al, submitted]. Increased OATP expression in tumors is likely to permit enhanced uptake of growth factors, hormones and nutrients; however, higher OATP expression was furthermore observed in tumors treated with cytotoxic drugs and, since the efficacy of the chemotherapeutics was frequently impaired, OATPs may be involved in mechanisms resulting in drug resistance. Since the processes leading to chemoresistance in SCLC are not fully characterized and etoposide as well as the novel lipophilic platinum complexes satraplatin and picoplatin may represent OATP substrates, the present study aimed at the investigation of the role of OATP5A1 that is known to be expressed in lung tumors in drug resistance of SCLC cell lines. 19
Materials and Methods
Chemicals
Unless indicated otherwise all chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA). Satraplatin (JM 216; bis-acetato-ammine-dichloridocyclohexylamine-platinum (IV)) was synthesized by Chiracon (Luckenwalde, Germany) according to standard procedures and kindly provided by IPSS (Berlin, Germany).
Cell lines and culture conditions
Cell lines were obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA), the European Collection of Cell Cultures (ECACC, Salisbury, UK) and the Department of Radiation Biology, Finsen Center, National University Hospital, Copenhagen, Denmark (all SCLC, except the NCI and DMS273 cell lines). Cells were grown in RPMI-1640 bicarbonate medium (Seromed, Berlin, Germany) supplemented with 10% fetal bovine serum (Seromed), 4 mM glutamine and antibiotics in a humidified incubator (5% CO2, 37 °C, 95% humidity). Cells were checked for mycoplasma contamination (Mycoplasma PCR ELISA, Roche Diagnostics, Vienna, Austria). Attached cells were subcultured by trypsinization (0.05% trypsin containing 0.02% EDTA) two times a week.
SLCO5A1 transfection of HEK-293 cells
The complete cDNA coding for SLCO5A1 was amplified by RT-PCR from normal ovarian RNA (Stratagene, Santa Clara, CA, USA) and cloned into a pcDNA3.1 vector containing a CMV promoter and an neomycin resistance marker. HEK-293 cells were transfected and selected in minimal essential medium (MEM) containing 10% fetal bovine serum, 100 U/ml penicillin, 100 μg/ml streptomycin, supplemented with 400 μg/ml geneticin (G418). Expression of SLCO5A1/OATP5A1 was confirmed by real-time qPCR and staining with antibody HPA025062 (Atlas Antibodies, Stockholm, Sweden).
Flow cytometric detection of OATP5A1
HEK-293 cells were harvested and washed with phosphate buffered saline (PBS). Pellets were fixed in 4% paraformaldehyde/PBS at 4 °C for 15 min and thereafter washed two times with PBS containing 0.5% bovine serum albumin (BSA) by centrifugation. After tapping, the pellet was treated with 0.1% Triton X-100/0.5% BSA at room temperature for 15 min to permeabilize the cell membranes. Cells were then incubated in blocking buffer (2% normal rabbit serum/0.1% Triton X-100/0.5% BSA) for 15 min and further incubated with 1:25 or 1:50 dilutions of anti-OATP5Al rabbit antibody (HPA025062, affinity purified) in the same buffer for 2 hrs. Following two wash steps with 0.1% Triton X-100/0.5% BSA in PBS cells were incubated with goat anti-rabbit IgG (whole molecule), F(ab′)2 fragment-FITC antibody (Sigma-Aldrich, F1262) in blocking buffer for 1 hr. Finally, washed cells were resuspended in PBS for flow cytometric analysis (Cell Lab Quanta SC, Beckman-Coulter, Brea, CA, USA).
Chemosensitivity assay
1 × 10 4 cells in 100 μL medium/well were distributed to 96 well microtiter plates (Greiner, Kremsmuenster, Austria), and substances to be tested were added in a volume of another 100 μL. All compounds were serially diluted in 6-10 twofold steps in triplicate. The microtiter plates were incubated under tissue culture conditions (RPMI-1640/10% fetal bovine serum, 4 mM glutamine; 37 °C, 5% CO2, 95% humidity) for four days and cell viability measured using a modified MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide) assay (EZ4U, Biomedica, Vienna, Austria). Optical density was measured at 450 nm in a microplate reader with an empty well as reference. Wells containing media alone served as control values that were set to 100% proliferation.
TaqMan® real-time qPCR
Total RNA from all cell lines was isolated using the RNeasy® Mini kit (Qiagen, Hilden, Germany) and treated with RNase free DNase (Qiagen) to remove genomic DNA, possibly present. Concentration, purity and integrity of RNA samples were determined using a Nanodrop ND-1000 (Kisker-Biotech, Steinfurt, Germany) and agarose gel electrophoresis. 1 μg of total RNA was reverse-transcribed in 20 μl reactions using the High Capacity cDNA RT kit (Applied Biosystems, Foster City, CA) with the random hexamer primers and RNAse inhibitor (Applied Biosystems) provided according to the manufacturer's instructions. TaqMan® Gene Expression Assay for SLCO5A1 (hs00229597_ml) and control assays for the ACTB (PN 4326315E) and HPRT (PN 4310890E) genes were purchased from Applied Biosystems. Prefabricated primers and probes for the endogenous control genes GAPDH and RPL13A were obtained from PrimerDesign (PrimerDesign Ltd., Southampton, UK). TaqMan® real-time qPCR was performed in an amplification mixture volume of 10 μl containing 5 μl 2x TaqMan® Gene Expression PCR Master Mix (Applied Biosystems), 0.5 μl of the respective Gene Expression Assay, 10 ng template cDNA diluted in 4 μl nuclease-free water and 0.5 μl nucle-ase-free water. Thermal cycling conditions were 2 min at 50 °C and 10 min at 95 °C, followed by 40 cycles of 15 s at 95 °C and 1 min at 60 °C on the ABI PRISM 7900HT Sequence Detection System (Applied Biosystems) equipped with a 96-well fast cycling block. Results were imported into DataAssist™ 2.0 software (Applied Biosystems) for automated data analysis using the comparative Ct (δδCt) method. Thus, a normalization factor was calculated by averaging the Ct values of the four endogenous control genes ACTB, GAPDH, HPRT and RPL13A via the geometric mean. Relative quantities for every sample were then determined according to the DataAssist™ 2.0 Software User Instructions (Applied Biosystems).
Immunolocalization of OATP5A1 in SCLC
For immunofluorescence staining, 4 μm sections were generated with a Cryostat-Microtome HM 500 OM (Microm, Heidelberg, Germany) from frozen tumor samples (stored at –80 °C). Tissue sections were fixed with acetone and blocked with 5% BSA/PBS. Dilutions for primary antibodies were 1:50 for OATP5A1 (HPA025062, Atlas Antibodies, Stockholm, Sweden) and CD34 (Acris, Herford, Germany). Incubation with the primary antibodies was done overnight. After washing, sections were incubated with Alexa Fluor® 488 anti-rabbit IgG (1:2000) or Alexa Fluor® 568 anti-mouse IgG (1:1000), respectively. Cell nuclei were counterstained with 0.5 μg/ml bisbenzimide/PBS (Hoechst 33342). Sections were mounted in Mowiol 4–88 (Carl Roth, Karlsruhe, Germany) and fluorescent staining was visualized with an Axioplan 2 microscope (Carl Zeiss, Jena, Germany). Images were captured using an AxioCam HRc2 Color CCD digital camera and Axiovision 4.6 software (Carl Zeiss Vision GmbH, Aalen, Germany).
Statistical analysis
Values are demonstrated as mean ± SD. Statistical analysis was performed using Student's t-test. Differences with *P < 0.05 were regarded as statistically significant.
Results
Expression of SLCO5A1 in normal tissues and SCLC cell lines
Expression of SLCO5A1 mRNA was quantified by realtime qPCR using RNA from selected normal tissues, the human embryonic kidney cell line HEK-293, a panel of SCLC cell lines as well as the non-small cell lung cancer (NSCLC) cell line A549 for comparison (Fig. 1). Significant mRNA expression of this transporter was found for the thyroid gland and HEK-293 cells, representing normal tissues, as well as for most of the SCLC lines in varying quantities. Normal liver and lung tissues exhibited low expression and comparatively minor expression in A549 indicated an insignificant role of OATP5A1 in NSCLC. Highest SLCO5A1 levels were detected in the GLC-19, GLC-14, and GLC-16 cell lines derived from the same SCLC patient during a longitudinal follow-up (Fig. 1). 20
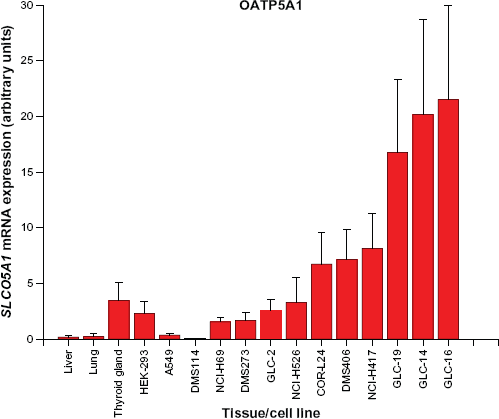
Transcript levels of SLCO5A1 in normal tissues and in cell lines. Relative expression of SLCO5A1 mRNA (arbitrary units) is presented as mean ± SD (n = 5). SLC05A1 mRNA levels of normal HEK-293 human embryonic kidney and A549 NSCLC cells were compared to a panel of SCLC cell lines.
Chemosensitivity of SLCO5A1-transfected HEK-293 cells to satraplatin
HEK-293 cells were either transfected with empty vector pcDNA3.1 (EV) or vector containing the coding sequence of SCL05A1 and both resulting cell lines tested for their chemosensitivity to dilutions of satraplatin in MTT cell viability assays. Three permanently transfected clones were cultivated with G418 and revealed a 15–20-fold overexpression of SCLO5A1 mRNA in real-time qPCR. Immunofluorescence staining of fixed and permeabilized cells with HPA025062 antibody revealed a weakly positive signal in untransformed and significantly elevated expression in SLCO5A7-transformed HEK-293 cells in flow cytometry (Fig. 2). HEK-293 control cells transfected with empty vector (EV) exhibited a 2.8 ± 0.64 fluorescence ratio (antibody signal/nonrelevant antibody control signal) in good agreement with the result obtained in real-time qPCR. HEK-293 cells transfected with a vector containing SCLO5A1 showed a marked increase in reactivity with the HPA025062 antibody: fluorescence ratio of 13.8 ± 0.25, translating to a 4.9-fold increased expression of OATP5A1 (values calculated for three independent clones; Figure 2: clone B3-right column, 1:25). G418 supplementation was omitted in the cytotoxicity tests, since former experiments had revealed no differences in the results obtained in comparison to standard tissue culture medium. HEK-293-SLCO5A1-transfected cells proved to be significantly more resistant to satraplatin at concentrations of 5, 2.5 and 1.25 μM than control HEK-293-EV cells (Fig. 3). The increased survival of the transfected HEK-293 cells corresponded to an upward shift of 0.8 μM in IC50 for satraplatin. Sensitivity of HEK-293-EV-and HEK-293-SLCO5A1-transfected cells to cisplatin was not different (data not shown).
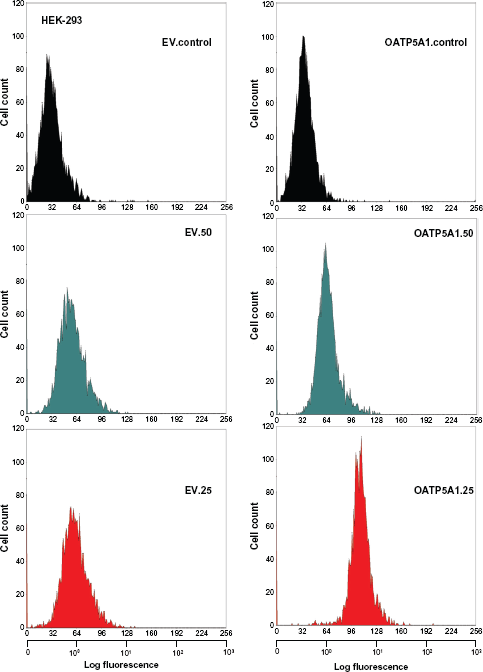
Protein expression of OATP5A1 in transfected HEK-293 cells. OATP5A1 was detected in the cells using the HPA025062 antibody in indirect immunofluorescence. Flow cytometry histograms are shown for empty vector controls (EV, left column) and SLCO5A1-transfected cells (right column) for normal serum controls (top row) and antibody dilutions 1:50 (middle row) and 1:25 (bottom row), respectively.
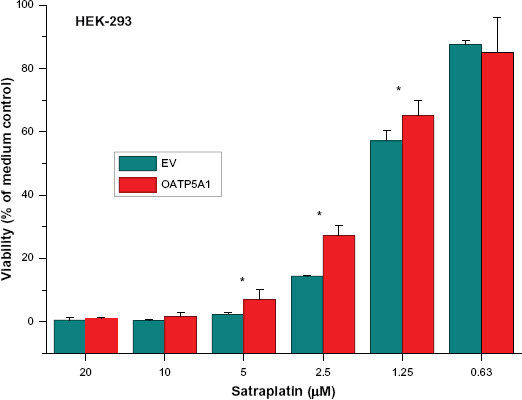
Cytotoxic effect of satraplatin on SLCO5A1-transfected HEK-293 cells. SLCO5A1-transfected and control HEK-293 cells were exposed to the indicated concentrations of satraplatin for four days and cell viability assessed by MTT assays. SLCO5A1-transfected cells showed higher viability than control cells at concentrations of 5–1.25 μM satraplatin (mean ± SD; n = 3; *P < 0.05).
SLCO5A1 expression and effect on satraplatin chemosensitivity of SCLC cell lines
SCLC cell lines were grouped according to their sensitivity to satraplatin, which had been previously determined in MTT assays (data not shown). A satraplatin concentration of 1.7 μM was used as cutoff for the IC50 values and the mean SLCO5A1 expression for the two groups is shown as box plot (Fig. 4).
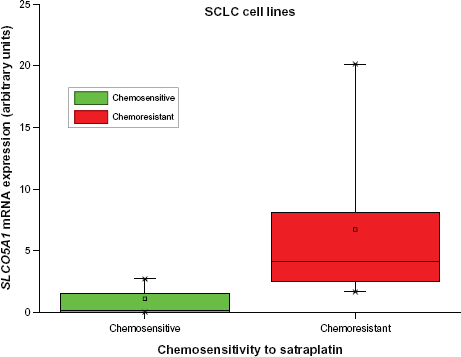
Mean values of SLCO5A1 mRNA expression levels in satraplatin-resistant and -sensitive SCLC cell lines. Cell lines were grouped according to their satraplatin chemosensitivity (IC50 < 1.7 μM satraplatin: DMS114, NCI-H69, DMS273, GLC-2; IC50 > 1.7 μM satraplatin: remaining SCLC cell lines); mean expression of SLCO5A1 mRNA was calculated and is presented as box plot. The difference between the two groups did not reach statistical significance (P = 0.072).
OATP5A1 immunofluorescence staining of a tissue section from non-cancerous and adjacent cancerous regions of a SCLC
Immunolocalization of OATP5A1 was performed using paraffin-embedded sections of an SCLC specimen obtained from a patient and the anti-OATP5Al antibody HPA025062 (Fig. 5). Blood vessels were stained with an anti-CD34 antibody and the cell nuclei were visualized by counterstaining with bisbenzimide. An overlay of the different fluorescences revealed OATP5A1-staining preferentially in the cytoplasm and near the cell membrane in the majority of the cells.
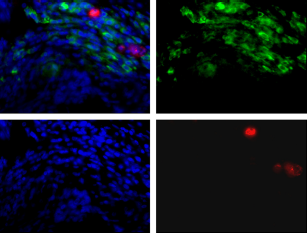
Immunohistochemical staining of an SCLC with anti-OATP5A1 antibody. The paraffin-embedded SCLC specimen was stained with anti-OATP5A1 and anti-CD34 antibodies, which recognizes blood vessels (top left). Cell nuclei are shown in blue due to counterstaining of the section with bisbenzimide. Controls for anti-CD34 (top, right), anti-OATP5A1 (bottom, right) and the counterstained section (bottom, left) were acquired using the respective bandpass filters (top region-tumor cells, bottom region-normal tissue). Controls using nonrelevant primary antibodies exhibited no specific fluorescence signals.
Discussion
Gene expression analyses of human tissue samples indicated absence of SLCO1A2 and the two liver-specific SLCOs 1B1 and 1B3, as well as 1C1, 5A1 and 6A1 in the lungs. 21 However, SLCOs 2B1, 3A1 and 4A1 were detected in the same tissue, while SLC04C1 was moderately present. Normal bronchial cells expressed high levels of SLCOs 3A1 and 4A1. 21 Furthermore, respiratory epithelial cells exhibited a weak cytoplasmic/membranous staining with anti-OATP5A1 antibody HPA025062.
OATP1B1 and 1B3 are known to mediate the transmembrane transport of paclitaxel, but tumor cells are expected to downregulate their expression to avoid cell death in response to cytotoxic compounds. Other OATPs that are upregulated in tumor cells are likely to participate in mechanisms leading to chemoresistance. Detection of OATP5A1 in U-251 MG glioblastoma and A431 epidermoid carcinoma cells with antibody HPA025062 revealed granular/fibrous staining in the cytoplasm. The majority of malignant cells were either OATP5A1-negative or displayed weak cytoplasmic staining, with exception of a fraction of ovarian and lung and a large number of renal cancer specimens, which showed moderate to strong cytoplasmic and nuclear immunoreactivity Several OATPs, such as human 1A2, 1C1, 2B1, 4A1 and 6A1, possess a C-terminal postsynaptic density-95(PSD-95)/Discs-large/ZO-1 (PDZ) consensus sequence.8,9 Recently it was shown that binding to PSD-95/Discs-large/ZO-1 domain-containing 1 (PDZK1) is essential for plasma membrane incorporation of rat Oatp1a1.23–25 Thus, a similar mechanism could be responsible for the plasma membrane localization of all OATPs with a C-terminal PDZ consensus sequence. It is therefore suggested that some OATPs may be involved in the intracellular transport of compounds across vesicle membranes instead of facilitating transmembrane cellular drug uptake. According to our own results obtained with HEK-293-SLCO5A1 cells, cisplatin does not serve as a substrate of OATP5A1, since its cytotoxicity is not altered; in contrast, introduction of lipophilic moieties to the basic structure of cisplatin, i.e. a cyclohexyl residue and axial ester ligands in the case of satraplatin, seem to render this compound suitable for transport by this carrier. 19 Hence, OATP5A1 may have an impact on cellular drug resistance by sequestration of satraplatin by intracellular binding and/or transport into cytoplasmic organelles, respectively. At concentrations exceeding 5 μM satraplatin this mechanism may become saturated, since the capacity to store drugs intracellularly is expected to be limited, in contrast to the effects of the multidrug-resistance efflux transporters. At lower concentrations of satraplatin handling of this compound seems to be shifted preferentially to degradation to its metabolites by hydrolysis and reduction. 26 This ultimately results in formation of the highly cytotoxic metabolite JM118 being not a substrate of OATP5A1 (data not shown). However, the narrow dose window of 1.25–5 μM satraplatin for the action of OATP5A1 matches closely the peak plasma concentrations of this drug, and relatively small changes in cellular resistance in this range may result in profound clinical effects. 27 This mechanism of chemoresistance in highly refractory SCLC cell lines may be induced by the potential OATP substrate etoposide that is commonly used in conjunction with cisplatin for standard chemotherapy. 2 However, further investigations are necessary to prove the functional significance of OATP5A1 in lung cancers, with special respect to gene sequencing and an assessment of the specific drug transport characteristics. OATP-mediated chemoresistance is suggested to play a role in a new mechanism of drug inactivation that affects novel cisplatin analogs exhibiting lipophilic moieties.
Conclusions
OATPs were characterized as cellular uptake transporters for a number of important physiological substrates and can in some cases act in conjunction with efflux pumps effecting transcellular flux. Most of the eleven members of this gene family were found in tumors and suggested to be involved in the cellular uptake of hormones and growth factors. Expression of OATPs mediating uptake of chemotherapeutic drugs such as taxanes, imatinib and methotrexate28–30 would be expected to become downregulated in order to evade cytotoxic anticancer therapy. In the present study we confirmed significant expression of SLCO5A1 mRNA in SCLC cell lines. Expression of OATP5A1 had been previously reported for some lung cancer specimens by immunohistochemistry. Since OATPs are known to transport lipophilic compounds, novel platinum anticancer agents like satraplatin and picoplatin may be suitable substrates. Transfection of HEK-293 cells with SLCO5A1 resulted in increased chemoresistance to satraplatin for a clinically relevant range of concentrations, and SCLC cell lines, which showed an approximately fourfold higher mean expression of this transporter, tended to exhibit increased resistance to the same drug. However, the relationship of OATP5A1 and resistance of SCLC cells to satraplatin is complicated by the fact that in a part of the cell lines the transporter may be nonfunctional either due to mutations or expression of splice variants of the SLC05A1 gene. However, the granular cytoplasmic localization of OATP5A1 indicates an intracellular sequestration of satraplatin in organelles or inactivation by transporter protein binding, rather than to a role of OATP5A1 in increased drug uptake. There seems to be no general increase in cellular resistance to apoptosis, since the cytotoxic effect of cisplatin was not impaired in HEK-293-SLC05A1 cells. In breast cancer, both OATP3A1 and OATP5A1 were reported to become additionally expressed in the cytoplasm of cancer cells in contrast to their localization restricted to the plasma membrane of epithelial cells in normal breast tissue. 31 In conclusion, intracellularly expressed OATPs are expected to play a role in mechanisms leading to drug resistance of tumor cells, which may explain in part the low efficacy of novel cisplatin analogs with lipophilic moieties. 14 In addition to the established role of human organic cation transporters, at least the anion transporter OATP5A1 seems to be involved in the disposition of selected platinum complexes. 32
Author Contributions
Ulrike Olszewski-Hamilton did the chemosensitivity assays, calculation of data, as well as assistance in the preparation of the manuscript.
Martin Svoboda did the real-time qPCR measurements and transfection.
Therese Thalhammer guided real-time qPCR measurements and transfection.
Veronika Buxhofer-Ausch provided the clinical specimen.
Klaus Geissler guided design and interpretation of the study.
Gerhard Hamilton guided in the evaluation of the assays, statistical analysis and writing of the manuscript.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgments
This work was supported by a grant from the research fund of the National Bank of Austria, project number 13345.
