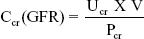Abstract
This study was undertaken to study the renal toxicity of mercuric chloride in rats at different periods of time. The following groups of rats were studied: i) control, ii) placebo, iii) rats injected with a single ip dose of 100 mg/kg body weight of 2, 3 dimercapto-1-propanesulfonic acid, iv) rats injected with a single ip dose of 100 mg/kg body weight of 2, 3 dimercapto-1-propanesulfonic acid (DMPS) followed by a single dose ip of 2.0 mg HgCl2/kg body weight one hour after DMPS injection v) rats injected with a single ip dose of 2.0 mg HgCl2/kg body weight. Results indicate that mercuric chloride was more toxic after 48 hours of its administration when compared to 24 hours. Mercuric chloride administration caused an impairment of renal function which was evident from a significant decrease in urine volume, urinary excretion of urea, creatinine and glomerular filteration rate (P < 0.001) when compared to other treated groups. There was an increased excretion of protein, albumin and γ–-glutamyltransferase in the urine of mercuric chloride treated rats. Administration of 2, 3 dimercapto-1-propanesulfonic acid before mercuric chloride treatment caused the altered indices to return to near normal levels.
Introduction
Within the earth's crust, cinnabar or mercuric sulfide is the principal ore of element mercury. The ubiquitous nature of mercury in the environment, its toxicity to humans at levels that are uncomfortably close to exposures experienced by a proportion of the population are some of the concerns associated with this pollutant. The disasters in Minimata, Japan in the 1950s and in Iraq in 1971-72 clearly demonstrated neurologic effects associated with the ingestion of methyl mercury. The studies of mercury exposure in the work place were mainly of elemental or inorganic mercury and effects that were observed at relatively low exposure levels were primarily neurologic and renal. 1 Humans may be exposed to various species of mercury, which includes charged inorganic mercurous (Hg1+), mercuric salts (Hg 2 +), neutral elemental metal (Hg0) and organic molecules. Exposure to mercury vapor and organic mercurials specifically affect the central nervous system, while kidney is the target organ for inorganic mercury compounds. 2 In general mercury toxicity derives from the fact that mercury binds to sulfide groups and disrupts the proper functioning of sulfhydryl enzymes. 3 Routes of exposure, toxicity, target organs and ultimately treatment strategies vary according to the species of mercury involved in the exposure.
The kidneys excrete waste products of metabolism and play an important role in maintaining the homeostasis by regulating the body water and solute balance. In addition to the excretory function, the kidneys also have an endocrine function producing hormones like renin, erythropoietin etc. Inorganic mercury (HgCl2) has been shown to accumulate in kidneys 4 along with in other organs. A specific concern associated with mercury exposure in humans is the need for effective therapy in dealing with intoxication. In this respect, chelation therapy is the most commonly used and seen as the least invasive. 5 Chelating agents compete with the in vivo binding site for the metal ion through the process of ligand exchange. 6 The toxic metal bound to the chelating agent is excreted from the body through the urine or feces. Among chelating agents currently available, the sodium salt of 2,3-dimercapto-1-propane-sulfonate (DMPS) has been found to be highly effective, particularly with respect to promoting mercury elimination following inorganic or elemental mercury exposure. 5 Among the species used for testing various toxins, the rat and mouse most successfully model the human reaction. 7 In our previous studies8,9 we have shown that HgCl2 treatment to rats damages the collagen which is reflected by increased levels of hydroxyproline in serum and an increased excretion of hydroxyproline in urine. The present study was carried out to study the effect of HgCl2 treatment on rat kidneys at different periods of time and the protective effect of DMPS on HgCl2 induced renal damage.
Materials and Methodology
Chemicals
The chemicals used were obtained from Sigma Chemical Company, St Louis, MO, USA. Glass distilled water was used throughout the study.
Animal care
Healthy adult male Wistar rats weighting 150-200 g were obtained from Breeding Laboratory, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia. The animals were labeled by identifying ear notches, housed in clean cages, and placed in the animal care room. The animals were housed singly in clean and properly suspended metabolic cages made of stainless steel wire-mesh floor and given water and standard rodent chow ad libitum. The animal room was maintained at 21 ± 1.5 °C. Food intake by the animals was monitored daily.
Time–-course response of HgCl2 administration
Following one-week acclimatization period, rats were randomly divided into different groups (5 rats/group) and individually housed in stainless-steel metabolic cages (Mini Mitter Co., Inc., Bend, Oregon, USA). Rats were allowed free access to food (Purina rodent chow) and tap water for one day. Baseline values for body weight, food and water intake, urine output and urine contents were obtained during a 24-hour period running from 09:00 AM to 09:00 AM the following morning. All data collected during this 24-hour period were designated day 0. Food was withheld between 0:00 to 13:00 hours in order to get urine free of food contamination. Urine samples were collected on dry ice, centrifuged and stored at −10 °C till assayed. After one day control rats were divided into five different groups each group consisting of 10 rats. The following groups were studied: i) rats which received no treatment (Control group); ii) rats injected intraperitonally (ip) normal saline solution (Placebo group); iii) rats injected with a single ip dose of 100 mg of 2, 3 dimercapto-1-propanesulfonic acid (DMPS)/kg body weight; iv) rats injected with a single ip dose of 100 mg/kg body weight of 2, 3 dimercapto-1-propanesulfonic acid (DMPS) followed by a single dose ip of 2.0 mg HgCl2/kg body weight one hour after DMPS injection v) rats injected with a single ip dose of 2.0 mg HgCl2/kg body weight.
Half the rats from each group were sacrificed after 24 hours of the HgCl2 treatment and the other half of rats were sacrificed after 48 hours of the HgCl2 treatment.
Sample collection
Urine samples were collected on dry ice, centrifuged and stored at −10 °C till assayed. At appropriate time intervals, the rats were asphyxiated by diethylether inhalation. The kidneys were removed, weighed and placed in separate containers and used for histological studies.
Evaluation of kidney function
Renal clearances of creatinine was used to determine the glomerular filtration rate (GFR), urinary excretion of γ–-glutamyltransferase and total protein were used as indicators of renal cellular injury.
Estimation of glomerular filtration rate
The renal clearance of creatine (Ccr) was used as an indicator of glomerular filtration rate (GFR) and overall renal function using the following equation
where Ucr and Pcr are the concentration of creatinine (mg/mL) in the urine and serum, respectively and V is the rate of excretion, or flow of urine in mL/min.
Biochemical analysis
Biochemical determinations in urine sample were used to evaluate the extent of renal injury and renal function. These included the concentration of γ-glutamyltransferase in the urine as indicator of mercuric chloride toxicity, 10 urea, creatinine, total protein, albumin, sodium and potassium in the urine. These parameters were determined using Roche/Hitachi 917 Biochemistry Analyzer, Roche Diagnostic Corporation, USA.
Histopathological examination
The right kidney was dissected out, weighed cut and fixed in 10% neutral buffered formalin solution. Fixed tissues were embedded, sectioned at maximal thickness of 2–4 μm and stained with hematoxylin and eosin (H and E) before microscopic examination. Tissues from treated animals were compared to tissues from control animals.
Statistical Analysis of the Results
Data from each sample were run in duplicate and expressed as the mean ± SD. One–-way ANOVA was used for comparison between the groups. For multiple comparison, P values were adjusted using Dunnett's multiple comparision test. Bartlett's test was used for homogeneity of variance. Spearman correlation analysis was used to examine the association between variables. All statistical tests were run at a 95% confidence interval and P < 0.05 was taken as the level of statistical significance. Statistical analysis were made using InStat personal computer program (GraphPad Software, Inc., San Diego, CA, USA). Grafit software for Windows (Erithancus Software Ltd, UK) was used for creating graphs.
Results
Table 1 shows the effect of HgCl2/DMPS treatment on various biochemical indices in the urine. HgCl2 treatment caused a significant increase in the urinary excretion of total protein, albumin and sodium but a decrease in the excretion of urinary potassium (P < 0.001) when compared to the control and other treated group of rats. DMPS treatment before HgCl2 injection caused these parameters to return to near normal levels.
Effect of 2 mg mercuric chloride (HgCl2/kg body) on urine biochemical parameters in rats at 24 or 48 hour post treatment.
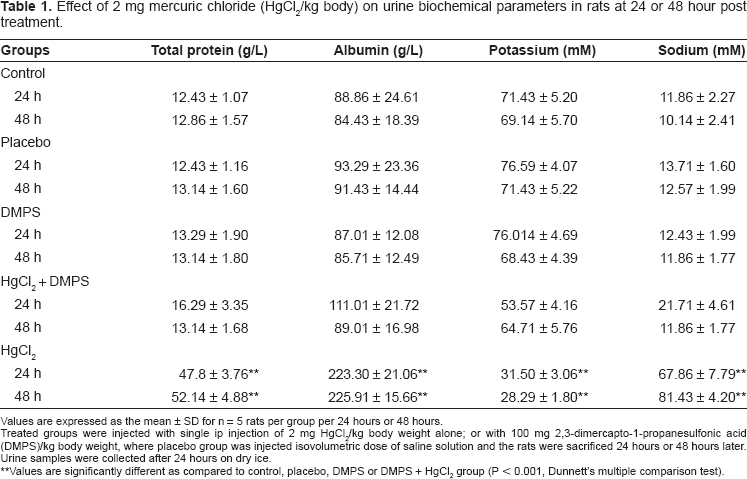
Values are expressed as the mean ± SD for n = 5 rats per group per 24 hours or 48 hours.
Treated groups were injected with single ip injection of 2 mg HgCl2/kg body weight alone; or with 100 mg 2,3-dimercapto-1-propanesulfonic acid (DMPS)/kg body weight, where placebo group was injected isovolumetric dose of saline solution and the rats were sacrificed 24 hours or 48 hours later. Urine samples were collected after 24 hours on dry ice.
Values are significantly different as compared to control, placebo, DMPS or DMPS + HgCl2 group (P < 0.001, Dunnett's multiple comparison test).
Figure 1 shows the effect of HgCl2/DMPS treatment on urine volume collected 24 and 48 hours after the HgCl2 treatment. Results demonstrated that in HgCl2 treated rats there was a significant increase in urine volume for 24 hours post treatment (P < 0.001) when compared to other groups. However there was a significant decrease in urine volume collected for 48 hours after the HgCl2 treatment when compared to other groups (P < 0.001). DMPS treatment before HgCl2 injection restored the urine volume to near normal levels.
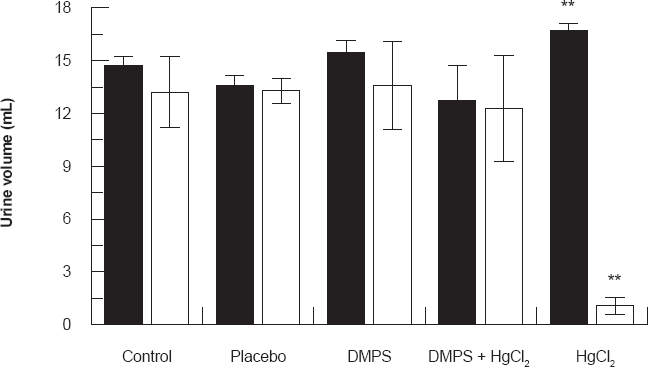
Effect of HgCl2/DMPS treatment on urine volume collected 24 and 48 hours after the HgCl2 treatment. Values are expressed as the mean ± SD for n = 5 rats per group per 24 [■] hours or 48 [□] hours.
Figures 2 and 3 show the effect of HgCl2/DMPS treatment on urinary excretion of urea and creatinine 24 and 48 hours after the HgCl2 treatment respectively. Results demonstrated that in HgCl2 treated rats there was a significant decrease in urinary excretion of urea and creatinine 24 and 48 hours after the HgCl2 treatment when compared to control and other treated groups (P < 0.001). DMPS treatment before HgCl2 injection caused these indices to return to near normal levels.
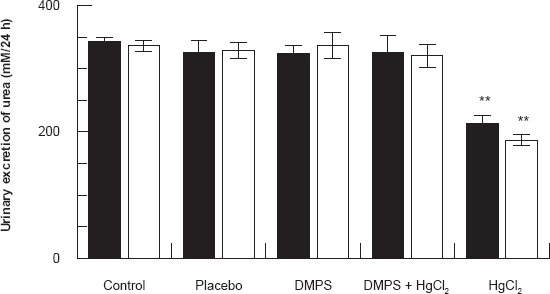
Effect of HgCl2/DMPS treatment on urinary excretion of urea 24 and 48 hours after the HgCl2 treatment. Values are expressed as the mean ± SD for n = 5 rats per group per 24 [■] hours or 48 [□] hours.
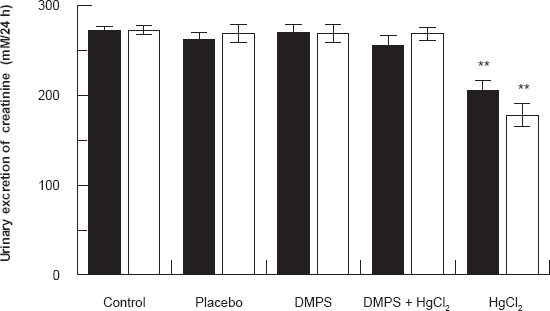
Effect of HgCl2/DMPS treatment on urinary excretion of creatinine 24 and 48 hours after the HgCl2 treatment. Values are expressed as the mean ± SD for n = 5 rats per group per 24 [■] hours or 48 [□] hours.
Figure 4 shows the glomerular filtration rate in differential experimental groups of rats 24 and 48 hours after HgCl2/DMPS the treatment. Results show that HgCl2 treatment caused a significant decrease (P < 0.001) in glomerular filtration when compared to control rats. DMPS treatment before HgCl2 injection increased the glomerular filtration rate to almost normal levels.
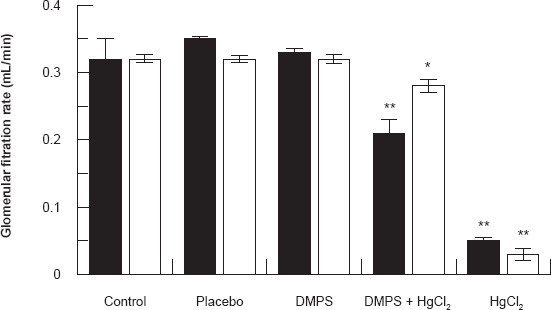
Effect of HgCl2/DMPS treatment on the glomerular filtration rate 24 and 48 hours after the HgCl2 treatment. Values are expressed as the mean ± SD for n = 5 rats per group per 24 [■] hours or 48 [□] hours.
Figure 5 shows the effect of HgCl2/DMPS treatment on urinary excretion of γ-glutamyl transferase (γ-GGT) 24 and 48 hours after the treatment. HgCl2 treatment caused a significant increase (P < 0.001) in γ-GGT when compared to other experimental groups of rats. DMPS treatment before HgCl2 injection caused these parameters to near normal levels.
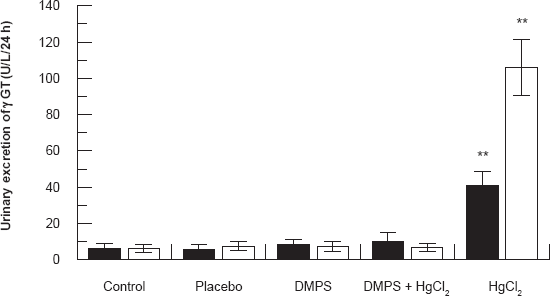
Effect of HgCl2/DMPS treatment on urinary excretion of γ-glutamyl transferase (γ-GGT) 24 and 48 hours after the HgCl2 treatment. Values are expressed as the mean ± SD for n = 5 rats per group per 24 [■] hours or 48 [□] hours.
Histological examination of renal sections from control, HgCl2 treated and DMPS plus HgCl2 treated groups sacrificed at 24 and 48 hours after the treatment are shown in Figures 6, 7 and 8. Kidneys of rats treated with HgCl2 showed glomerular mesangial hypercellularity and proximal tubular necrosis. DMPS treatment before HgCl2 injection however reversed the damage caused by HgCl2 to a large extent.
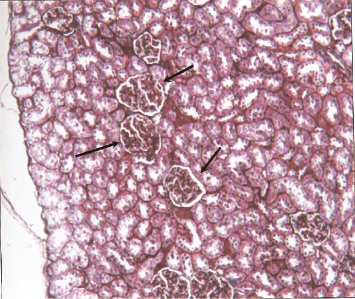
Photomicrograph of cross section of rat kidney (control group) at 24 hours showing normal renal structure.
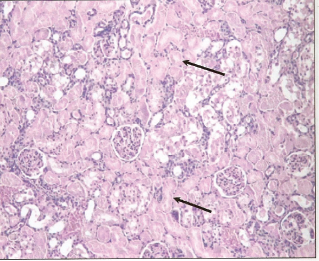
Photomicrograph of cross section of rat kidney treated with 2.0 mg HgCl2/kg body weight at 24 hour post-treatment showing almost total necrosis of the tubules with less severe changes within the glomeruli.
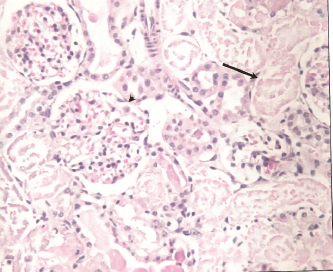
Photomicrograph of cross section of rat kidney treated with 2.0 mg HgCl2/kg body weight at 48 hour post-treatment showing tubular necrosis (arrow).
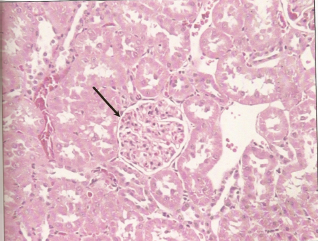
Photomicrograph of cross section of rat kidney treated with DMPS + 2.0 mg HgCl2/kg body weight at 48 hour post-treatment showing normal glomeruli (arrow) and normal tubules.
Discussion
There is evidence that chronic exposure to low concentration of metals like mercury causes tissue or organ damage. 11 This study demonstrates that administration of DMPS prior to HgCl2 treatment appears to moderate the renal damage caused by HgCl2. The administration of HgCl2 brings about an impairment of renal function which is evident by an decrease in urinary volume. Other indicators of renal damage caused by HgCl2 are decrease in the urinary excretion of creatinine and glomerular filtration rate and an increased excretion of γ-GGT. The urinary excretion of γ-glutamyltransferase (γ-GGT) has been used as a general indicator of acute nephrotoxicity. 12 In our earlier studies we have demonstrated that HgCl2 treatment causes a significant increase in serum creatinine and serum urea nitrogen indicating an impaired renal function. 8 Other manifestations of mercury toxicity include impairment of electrolyte, water and nonelectrolyte transport in variety of cells and tissues, the principal target organ being the kidneys. 13 In the present study decrease in urine volume 48 hours after HgCl2 could be due to decreased in fluid intake. 8 A decreased glomerular filtration rate further supports the fact that HgCl2 causes renal damage and that a reduced glomerular filtration rate is due to renal insuffiency. Inorganic mercury (HgCl2) has been shown to accumulate in the renal cortex and affect the morphology and function of the proximal tubules. 14 We have also reported earlier that in rats treated with HgCl2 there was a decrease in the serum albumin concentration which was accompanied with albuminuria. 9 Proteinuria indicates renal disease which may be the first reaction of the kidney to HgCl2 assault which is followed by decline in glomerular filtration rate. 15
The kidneys are paired bean-shaped organs located on either side of the spinal column. The kidneys perform a variety of functions for the body, the most important being removal of unwanted substances (waste and surplus) from the plasma, homeostasis of the body's water, electrolyte and acid/base status and participation in endocrine regulation. Human acute renal failure is often caused by ischemic and nephrotoxic insults commonly acting in combination. 15 There are very few agents that inhibit the acute renal failure in mercuric chloride induced model of nehrotoxicity. One of them is DMPS, which has been shown to afford protection to HgCl2 induced nephrotoxicity even if it is administered after HgCl2. In this study the renal damage was assessed by biochemical parameters measured in the urine along with histologic damage. The relative magnitude and degree of necrosis in proximal tubules based on quantitative histopathological analysis showed that the degree of renal damage observed was higher after 48 hours of HgCl2 treatment. Our studies have shown that at low doses of mercuric chloride, a very mild level of cellular necrosis was detected while severe cellular necrosis involving primarily the pars recta of proximal tubules was observed in rats treated with 2.0 and 3.0 mg of HgCl2/kg body weight (unpublished data). It has been reported that at small doses, HgCl2 affects the S3 segments in the cortico-medullary area. As the dose of HgCl2 is increased the injury spreads to involve the S1 and S2 segments of the proximal tubules. The lesions observed in the group of rats given HgCl2 alone were localized in the S3 segment of the proximal tubules. 16 The renal damage caused by HgCl2 was reversed by administration of DMPS before HgCl2. The administration of DMPS before HgCl2 restored the altered indices to near normal levels. Our previous studies 17 have shown that HgCl2 treatment of rat damaged the normal collagen content of the kidneys which was not restored by DMPS treatment.
Conclusion
HgCl2 treatment to rats impairs renal function which is reversed if DMPS is administered before HgCl2.
Disclosure
The authors report no conflicts of interest.
Footnotes
Acknowledgements
The authors would like to thank the Research Center, College of Science, King Saud University, Riyadh, Saudi Arabia (Grant number ST/Bio/1421/01) and King Abdulaziz City For Science and Technology, Riyadh, Saudi Arabia (Grant number SPR-9-41) for the financial support.