Abstract
The purpose of this study was to examine the function of metallothioneins (MT) in respect to the mobilization of heavy metals and superoxide anion (O2–) scavenging in aquatic organisms. Using an O2– generating system, liberation of free zinc from native and zinc MT (Zn-MT) was measured in vitro. Addition of the O2– generating system and H2O2 readily increased the di- and trimeric forms of MT as determined by gel electrophoresis analysis. To determine whether the proportion of oxidized MT could change in contaminated environments, metal-contaminated
Introduction
Since their discovery some 50 years ago, metallothioneins (MTs) have been identified as sulphur-rich and cadmium-binding proteins derived from equine renal cortex (Kagi and Vallee, 1960). MT is a small, heat-stable protein of 6–7 κDa containing about 25%–30% thiols as cysteine residues and devoid of aromatic amino-acids. MT is a transitional metal-binding protein capable of binding Ag(I), Au(I), Bi(III), Cd(II), Co(II), Cu(I), Fe(II), Hg(II), Pb(II), Pt(II), Tc(IV) and Zn(II) (Klaassen et al. 1999). Because of its ubiquity in many cellular compartments of prokaryotes and eucaryotes, the exact physiological role of MT is difficult to circumscribe. The general consensus, however, is that they are implicated in the detoxification of heavy metals by sequestering and preventing metals from binding to sensitive biochemical sites in cells. They also act as sinks for essential metals such as Cu(I), Fe(II) and Zn(II). More recently, it was demonstrated that MT has the ability to sequester reactive oxygen and nitrogen species such as superoxide anion and nitric oxide (Atif et al. 2006). During this process, the metal thiolate clusters in MT is readily oxidized, thus liberating metals from the protein (Kang, 2006). MT therefore co-exists in a reduced and oxidized form in the cytoplasm of cells, where oxidation processes involves the release of metals from MT and reduction involves the formation of metal-thiolate clusters in MT.
MT is a well recognized biomarker of heavy-metal contamination. It has even been viewed a “biomarker of exposure” against heavy metals in vertebrates and invertebrates (Amiard et al. 2006), in which it can be induced by various metals such as Ag, Cd, Cu, Hg and Zn. The induction potential of these metals varies and is subject to background variation that influences the mobilization of heavy metals. MT was successfully used as a biomarker of metal metabolism in freshwater mussels obtained from lakes contaminated by mine tailings and even proved predictive of effects at higher levels of biological organization (Couillard et al. 1995). Although MT can be an effective biomarker of the early biological effects of metal contamination, its role in the homeostasis of the redox state in cells limits its use as a universal biomarker of heavy-metal stress. This is especially true in situations where organisms are exposed to many other contaminants with the potential to cause oxidative stress (e.g. pesticides and polyaromatic hydrocarbons and inflammatory conditions) and in environmental conditions in which available oxygen levels vary with temperature, intertidal frequency and water characteristics (e.g. dissolved oxygen and organic carbon content). Legeay et al. (2005), for instance, found that cadmium-induced MT levels were higher in a low dissolved oxygen treatment group than in a high dissolved oxygen treatment group. An attempt was made to define the fundamental function of MT in a “simple” pro-karyote cell model. It showed that MT offered no protection from Cd or Zn contamination, although it was able to bind these metals in a wild-type strain of
The purpose of this study was to examine more closely the relationships between reduced and oxidized forms of MT in respect to heavy-metal mobilization and oxidative stress. First, the redox form of MT was examined in the laboratory in the presence of reducing and oxidizing conditions using free Zn measurements and gel electrophoresis analysis to track its oxidative-mediated polymerization. Second, we examined the effects of cofactors (NADH, NADPH, GSH) on the mobility of labile Zn in cell free extracts of digestive gland homogenates. Finally, the redox state of MT was examined in feral and heavy-metal-contaminated
Methods
MT preparation for in vitro experiments
Stock preparations of rabbit MT (containing, by weight, 3% Cd and 0.5% Zn) were purchased from Sigma Chemical Company and dissolved to 1 mg/mL in degassed (15 min under a negative pressure pump at 10 psi) 10 mM Tris-acetate, pH 7.4, containing 5 mM dithiothreitol (DTT) as a stabilizing agent. Zinc, copper-saturated MT and oxidized MT were prepared by adding each of 10 mg/L of zinc or copper sulphate and 2 mM H2O2 to 100 μg/mL stock MT for 30 min. MT was then precipitated by the addition of 80% acetone, stored at −20 °C for 30 min and centrifuged at 10 000 x
Oxidation of MT by superoxide anion generation system
Superoxide anion was produced in vitro by a nonenzymatic generating system (Ewing and Janero, 1995). Stock solutions of MT were diluted to 1 μg/mL in 10 mM Tris-acetate, pH 7.2, and mixed with one volume of freshly prepared NADH and phenazine methanosulfate (PMS) at 100 μM each. The reaction mixture was allowed to stand at room temperature for 0, 5, 10, 15, 20 and 30 min. At each exposure time an aliquot was collected for free zinc analysis and gel electrophoresis. Free zinc was determined using the TSQ probe methodology (Gagné and Blaise, 1996). The probe was dissolved to obtain a 50 μM concentration in 10% dimethylsulfoxide (DMSO) with phosphate-buffered saline (140 mM NaCl, 5 mM KH2PO4, pH 7.4, buffered with 10 mM Hepes), as previously described. The DMSO served two purposes: first, it acted as the solvent vehicle to dissolve the fluorescent probe; second, it protected the probe against attack by reactive oxygen radicals (DMSO traps oxygen radicals). For gel electrophoresis analysis, after each incubation times, the reaction mixture was diluted 1/10 in loading buffer (0.001% bromophenol, 2% SDS, 10 mM Tris-HCl, pH 7.4, containing 1 mM EDTA) with no reducing agent. The samples were loaded on high-resolution gradient (4%–2%) polyacrylamide gels (NuPAGE precast gels, Invitrogen). The buffer system consisted of 50 mM MOPS, pH 7.2, containing 2% SDS and 1 mM EDTA. Gels were stained using the commercial Coomassie blue G250 staining kit (Biorad, Canada). Molecular weight protein standards from 6 κDa to 200 κDa were used for gel calibration (Biorad, Canada).
Modulation of MT redox status in cell-free extracts
The influence of digestive gland cell-free extracts in the presence/absence of various reductive cofactors (NADPH, NADH and GSH) on the time-dependent redox status of MT was examined in clams. Oxidized (MT treated with H2O2) and Zn (reduced)-MT (10 μg/mL, 10 μ L) were mixed with either 100 μM GSH or NAD(P)H (100 μL) in the presence of 100 μL of the S15 fraction (90 μg/mL) from the homogenate fraction of digestive gland of
In situ assessment of metallothionein-like protein redox in Mya arenaria clams
Clams were collected during low tide in the St. Lawrence Estuary at both pristine and polluted sites by heavy boat traffic with a confirmed history of metal contamination (Gagné et al. 2006). Intertidal clams (
Data analysis
The experiments were repeated N = 3 times and the means with standard errors were reported. The data were verified for homogeneity of variances and normality using Levene's test. In cases where the data deviated from normality, they were log10-transformed. The data were then subject to an analysis of variance followed by the least square difference test to highlight significant changes within the various treatments. A multiple regression analysis was performed to highlight relationships between the redox state of MT with Zn/Cd availability, superoxide dismutase activity and LPO. Significance was set at p < 0.05.
Results
The levels of labile Zn in prepared native MT and Zn-saturated MT were below the operational detection limit of the assay methodology, indicating that MT precipitation in 80% acetone was effective in removing unbound or weakly bound Zn. The liberation of Zn from MT was examined using a superoxide generating system (Fig. 1). In native MT, the levels were somewhat increased during the incubation time, significant liberation of Zn being observed after 10 minutes. For zinc-saturated MT, the liberation of Zn was much more evident, suggesting that Zn is released more effectively than Cd in the presence of superoxides. During the exposure period (30 min), MT samples were analyzed by gel electrophoresis under denaturing conditions but without reducing agents such as β-mercaptoethanol and DTT (Fig. 2). It is noteworthy that the Zn-saturated MT band was in the monomeric form at 6 κDa but a large fraction was found in the dimeric form (12 κDa). The relative proportion of the dimeric form of MT was enhanced in the presence of the superoxide generating system and upon incubation of Zn-MT in the presence of H2O2. In the latter treatment, the trimeric form was easily observed with an increase in proportion. We quantified the proportion of oxidized MT (≥ 12 κDa) and reduced the metallic form of MT (6 κDa) by densitometric analysis of the protein band density, as shown in Figure 3. About 46% of the Zn-MT was oxidized in the original preparation. The oxidized proportion rose to 55% in the presence of the superoxide generating system for 10 minutes, thus confirming the release of labile Zn in the aforementioned experiments. The same was observed for copper-saturated MT (Cu-MT) and the oxidized proportion reached 75% after 30 min in the presence of the superoxide generating system. The addition of the reducing agent DTT during the incubation period reduced the proportion of oxidized MT to 55%, suggesting that thiol reagents prevented the oxidation of MT. The addition of hydrogen peroxide to the Zn-MT preparation increased the proportion of oxidized MT from 45 to 60% after a 10-minute incubation period. These results demonstrate that oxidation of MT implicates the release of zinc in the media.
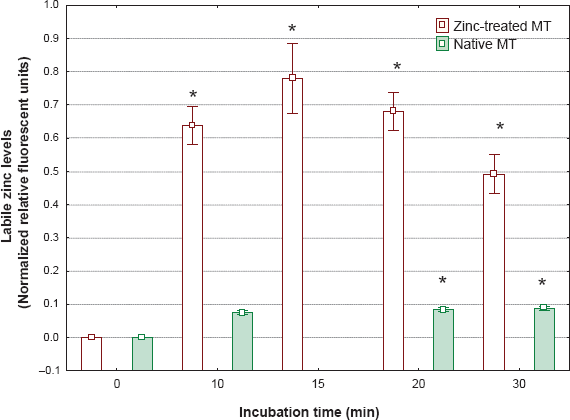
Zinc mobilization from MT exposed to superoxide anion.
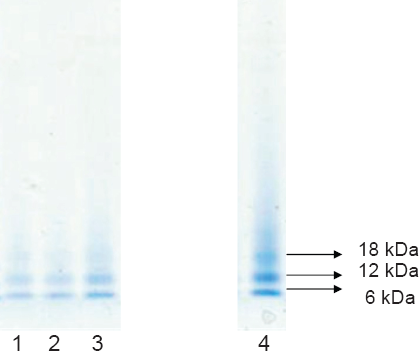
Gel electrophoresis of zinc-saturated MT (Zn-MT) under various oxidizing conditions.
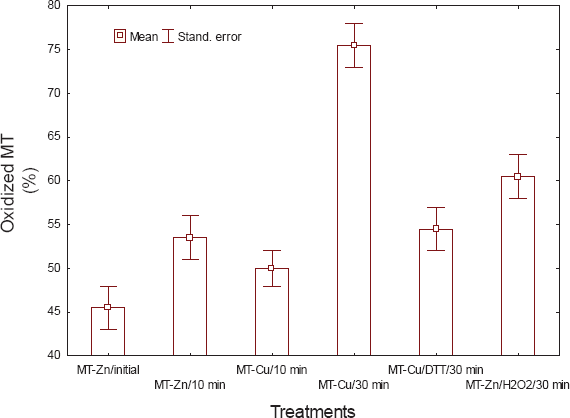
Changes in the proportion of oxidized MT under various oxidizing conditions.
We tracked the time-release of zinc in the presence of clam digestive gland cell-free extract (S15) and various cofactors such as reduced and oxidized MT, NADH, NADPH and GSH (Table 1). In the presence of Zn-MT alone (without the S15), labile Zn levels decreased over time, suggesting the gradual binding of Zn by MT. When the cell-free extract (S15) was added to Zn-MT, a moderate increase in Zn was observed, suggesting oxidation. When the reducing agents GSH and NADH were added to the S15 fraction, the significant reduction in free Zn indicated to the gradual binding of Zn to MT in reducing conditions. However, the addition of NAPDH had the reverse effect; that is, it increased free Zn levels. The same pattern was observed when oxidized MT (by H2O2) was added to the samples, suggesting that oxidized MT could bring about the release and capture of free Zn in the post-mitochondrial fraction of cells. The reducing effects of GSH on Zn levels were much more evident with oxidized MT than with native MT. Taken together, a conceptual framework between the redox state of MT and metal mobilization is proposed (Fig. 4). The redox state of the intracellular environment (e.g. NADH, GSH) could influence the redox status of MT in cells where oxidizing conditions favor the mobilization of metals (release of Zn) and reducing conditions decrease the mobilization of metals by maintaining the reduced (metallic) pool of MT. In this physiological model, NADH and GSH are more directly involved in the reduction of MT to favor metal sequestration than NADPH. NADPH seems involved in the reduction of reduced glutathione (glutathione reductase) or other enzymatic systems (e.g. NADPH cytochrome P450 reductase), which leads to the oxidation of NADPH into NADP+, where an excess of NADP+ could favor the oxidized form of MT and liberate metals.
Mobilization of zinc in trout liver extract: effect of MT oxidation and cofactors.

Rate of change of Zn was expressed as 1000 *(zinc levels at 30 min-zinc levels at t = 0). Each treatment was corrected against appropriate controls. For example, the rate of change in Zn for GSH/S15 was corrected against the rate of change of GSH with MT alone. For the S15, the rate of change of Zn was corrected against the change of Zn of S15 alone (without addition of MT).
2) The cofactors were all added at 100 μM concentration.
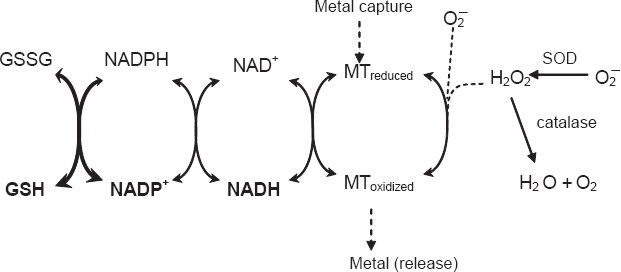
Proposed conceptual framework between the redox cycling of MT and metal mobilization.
Feral
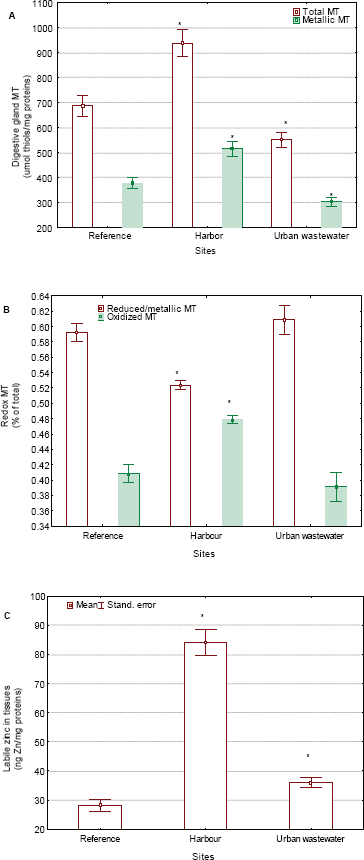
Change in the levels of total, reduced and oxidized MT in clam populations under pollution exposure.
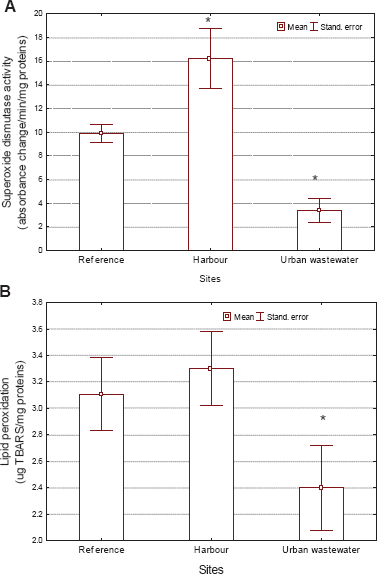
Evaluation of oxidative stress in
Multiple regression analysis of total MT and its redox state with Zn mobilization and oxidative stress endpoints in feral

Significant (p < 0.05) correlations are highlighted in bold and a marginal significance (0.05<p<0.1) is highlighted in italics and bold.
Discussion
Both metallic and oxidized MT was detected in vitro and in vivo, suggesting that MT co-exists in different states in cells. Indeed, the reported stoichiometrics for Zn-saturated MT (i.e. 7 Zn ions with 20 fully reduced thiols) does not necessarily represent its biologically active form (Maret, 2008). MT appears to have a dual physiological role: 1) the sequestration of divalent heavy metals and 2) oxygen radical scavenging activity of superoxide anion and other oxidants such as nitric oxide (Kroncke et al. 1994). The Zn-thiolate cluster in MT is thermodynamically stable while maintaining the kinetic reactivity of Zn and it confers antioxidant activity. The oxidization of the metallic (reduced) form of MT releases Zn in the intracellular environment under oxidizing conditions (Formigari et al. 2007). Conversely, the reduction of MT brings about the sequestration of zinc in cells. The generation of superoxide anion increased free Zn in vitro both with native and Zn-saturated MT. MT might function as a target for oxidants because the cysteines involved in the metal-thiolate clusters remain labile and freely exchange native metals with electrophiles and oxidants such as H2O2 (Quesada et al. 1996). When oxidized by nitric oxide or H2O2, the cysteines appeared structurally very close, which seemingly indicates the formation of intramolecular disulfide bond (Kroncke et al. 1994). MT is capable of forming not only intramolecular disulfide bridges, but intermolecular bridges as well, leading to dimeric and trimeric assemblies, as revealed by gel electrophoresis analysis. The labile Zn assay might provide clues on the redox state of the Zn-thiolate clusters in MT which, in turn, depends on the redox state of cells. This apparently holds true for bivalve MT, where about 40% of MT was present in oxidized form in this study, the value increasing to 50% at the harbor site known to be contaminated by various toxic metals such as Ag, Cd, Cu, Fe, Hg, Zn and organotins (Gagné et al. 2005). The study revealed a lack of metallic MT induction and a concomitant increase in oxidative stress in clams collected at the heavy metal-contaminated harbor site.
The redox state of cells and metal-thiolate clusters in MT could modulate the mobility of essential metals such as zinc. It was proposed by Kang (2006) that the more oxidative the cells, the more efficiently Zn is released from MT, a finding that is corroborated by the present study. They have found that reduced GSH inhibits Zn release while GSSG stimulates the release of Zn from MT, which is in agreement with the proposed physiological model (Fig. 4). Indeed, when MT was oxidized by H2O2, the capture of Zn was increased more by GSH than with Zn-MT. Thus, the metal binding potential of MT is modulated by both the availability of metals and the redox state of the intracellular environment, including MT. Under oxidizing conditions (i.e. excess NAD(P)+ and GSSG), the proportion of oxidized MT increases with the release of metals from MT; under reducing conditions (i.e. excess NADH and GSH), the proportion of oxidized MT decreases and sequesters metals. NADPH does not appear to be directly implicated in the reduction of MT; rather, it is implicated in other pathways such as glutathione reductase or NADPH cytochrome P450 reductase activity, which explains the releasing effects of free Zn associated with the oxidation of MT. When NADPH is added to the S15 fraction, it is readily oxidized by other enzyme systems such as glutathione reductase and NADPH cytochrome P450 reductase, leading to increased NADP+ that oxidizes MT and releases Zn. The equilibrium between NADPH and NADH in the intracellular environment might also influence the redox state of MT in cells. This hypothesis is consistent with the finding that both MT and SOD mRNA levels were induced in flounder exposed to benzo[a]pyrene (An et al. 2008). The biotransformation of benzo[a]pyrene by cytochrome P4501A1 requires NADPH for activation of cytochrome P450 reductase, and the formation of NADP+ that would favour the oxidation of MT (liberates metals) and the increase in total MT expression (mRNA). Furthermore, this hypothesis was supported by the observation that total MT levels were highly correlated with the oxidized form of MT, but not the reduced form of MT in
Based on these experiments, it appears that a redox cycle of MT exists where NAD(P)H and GSH are coupled and the co-factor NADH is a major driver. The antioxidant potential of MT was greater than that of reduced glutathione (Miura et al. 1997): the peroxide radical scavenging activity of MT was estimated to be 100 times greater than that of reduced GSH. The study also revealed that the inhibitory activity of MT against LPO was explained by its ability to scavenge lipid peroxyl radicals on the membrane surface. However, this protection will depend on the type of metals in the thiolate clusters since their release (the oxidant cupric ions) could, in turn, produce LPO and induce MT (e.g. Zn or Cu). This suggests that MT represents the first line of defense against reactive oxygen species. A multiple regression analysis revealed that the metallic form of MT (reduced form) was negatively related with free Zn in cells and positively so with lipid peroxidation, a biomarker of oxidative damage. Conversely, the oxidized form of MT was positively correlated with free Zn in cells and marginally so with SOD activity. It appears, therefore, that the oxidized form and the total levels of MT are related to Zn mobility and the increase in superoxide anion elimination (SOD activity), with no evidence of oxidative damage as measured by LPO (i.e. a detoxification mechanism), although the reduced form of MT was related to oxidative stress (enhanced toxicity). The use of the MT biomarker in field investigations should consider the redox properties of the protein, since the reduced and oxidized forms of MT have different physiological consequences.
Footnotes
Acknowledgments
The authors thank Patricia Potvin for editing the English manuscript. This research was funded by Environment Canada and the St. Lawrence Action Plan. The technical assistance of Sophie Trepanier on the clam biomarker analyses is recognized.
Disclosure
The authors report no conflicts of interest.
