Abstract
Intercellular communication is very important for cell development and allows a group of cells to survive as a population. Cancer cells have a similar behavior, presenting the same mechanisms and characteristics of tissue formation. In this article, we model and simulate the formation of different communication channels that allow an interaction between two cells. This is a first step in order to simulate in the future processes that occur in healthy tissue when normal cells surround a cancer cell and to interrupt the communication, thus preventing the spread of malignancy into these cells. The purpose of this study is to propose key molecules, which can be targeted to allow us to break the communication between cancer cells and surrounding normal cells. The simulation is carried out using a flexible bioinformatics platform that we developed, which is itself based on the metaphor chemistry-based model.
Introduction
Cell-to-cell communication is a necessary condition for the development and existence of multicellular organisms, and it is essential for maintaining vital physiological activities. 1 Through this intercellular communication, the cells work in coordination, facilitating their survival. It is known in the case of cancer cells that communication allows them to preserve their malignancy and through cell junctions to induce malignancy in neighboring cells. 2 Surrounding normal cells usually cannot control cancer cell growth. The formation of union between normal and malignant cells plays an important role in the control of a cellular focus that will result in a tumor. 3
The Wnt/β-catenin pathway plays an important role in the formation of intercellular communications. β-Catenin is the central part in this pathway, interacting with the E-cadherin and α-catenin protein, which is coupled to the cytoskeleton. If Wnt is absent, β-catenin is phosphorylated on the serine–threonine residue of the amino-terminal domain by glycogen synthase kinase 3 beta (GSK3β) and casein kinase (CKI), forming a complex with adenomatous polyposis coli (APC) and Axin. Phosphorylated residues are recognized by beta-transducing repeat containing protein and the E3 ubiquitin ligase, which induce proteasome degradation of β-catenin, and thus the formation of gap junctions is avoided (Fig. 1). 4

Wnt/β-catenin canonical pathway. When it is off, as most of the time occurs in normal cells, the β-catenin is linked to an enzyme complex that phosphorylates it? And this phosphorylation favors its ubiquitinylation and posterior emeute degradation in the proteasome. When the pathway is turned on, β-catenin promotes the transcription of genes involved in proliferation, differentiation, adhesion, and cell survival. Wnt cross talks with the MAPK pathway-related cytoskeletal rearrangement and cell survival way and G proteins. Red arrows indicate inhibition, green arrows indicate activation, previously modeled parts are shown in violet, and the new interactions added to the model in purple.
The Wnt/β-catenin pathway has been the subject of many studies. It is involved in the proliferation, differentiation, adhesion, cell survival, and death, and at the tissue level. It is also involved in tissue regeneration, inflammatory processes, and cell death. 5 It is also important for controlling the growth of tumor cells, as the concentrations of this pathway elements are modified in different cancers, including breast, 6 glioblastoma, 7 esophagus, 8 gastric, 9 prostate, 10 ovarian, 11 colorectal, 12 and liver cancers. 13
In the last few years, different computational tools that enable modeling and simulation of molecular and cellular biology phenomena have been developed. 14 However, the majority of these tools, such as E-Cell, 15 BetaWB,16,17 and Cell Illustrator, 18 own abstractions to model only intracellular behavior. Thus, they are not suitable to model cells in their social context, along with all those biological mechanisms that involve two or more cells, namely signal transmission that is important in the scenario discussed in this article. Therefore, in recent years, other major requirements in the simulation of intracellular and intercellular systems have emerged, guiding the development of new computational models and tools19,20: (1) topology and locality, respectively, used (a) to model complex topologies of signaling networks, which involve feed forward and feed backward relationships between signaling components placed into different intracellular compartments and structures and (b) to provide a global view of the signaling system as a composition of local interactions and (2) considering the amount of each component of the signaling pathways with respect to time, their location, and status (active, inactive, free, bound, etc.). Notable examples of platforms supporting multicompartment models are Bio-PEPA, 21 MCell, 22 COPASI, 23 Virtual Cell, 24 CompuCell 3D, 25 and BTSSOC-Cellulat. 26 They all account for movements and reactions of molecules within and between cells, implementing deterministic or stochastic models and providing tools to visualize simulation dynamics.
In our earlier study, we have successfully used a flexible bioinformatics platform, called BTSSOC-Cellulat, to simulate and explore proapoptosis and prosurvival signaling pathways and their integration. 27 In this article, we use an improved version and explore the complex role of cell junctions in intercellular communication.
In particular, in this article, we propose a model and an associated simulation of Wnt signaling pathway. Although a large amount of information on this pathway has been reported in the literature so far, part of this information is already stored in our platform, because this pathway has cross talk with pathways that we have modeled earlier, such as Epidermal growth factor (EGF)/Mitogen-activated protein kinase (MAPK)/Janus kinase-Signal transducer and activator of transcription protein pathway (JAK-STAT), 28 PI3K, 29 and caspases.30–32 We have integrated into the model the canonical and noncanonical pathways as well as Notch, 33 Hedgehog, 34 and Hypoxia 35 ones (Fig. 2). We believe that through modeling and simulation of cellular communication systems we will learn more about them and be able to find key interactions that allow us to modulate positively or negatively the communication between cancer cells and, thus, to prevent cancer cell proliferation. As a result of the different simulations of the Wnt signaling pathway, we have been able to find therapeutic targets for the control of cancer cells. We have included only two cells, one normal and another cancerous, to detect interactions easier, than in a tissue with a greater number of cells. This would introduce a greater number of receptors for the same signal, which would hide the possible targets. However, we are working on the engineering of the platform for experiments including a cancer cell in a normal tissue in which normal cells work together to isolate and eliminate the cancer cell.

Wnt cross talk. Cross activation of Wnt/β-catenin with Notch, EGF, Hh, TGFb, hypoxia, and other pathways that activate RTK interactions. All these pathways are incorporated in our model. For a better understanding of the figure, only some components of the signaling pathway are shown. Red arrows indicate inhibition, green arrows indicate activation, violet arrows indicate consequence, previously modeled parts are shown in light blue, and the new interactions added to the model in blue.
Biological Background
Control of cancer cells by the neighboring normal cells occurs due to intercellular communication, by means of direct contact, through physical connections, such as junctions, or through extracellular signals. An appropriate condition of intercellular junctions is essential to maintain a communication and cellular unity; this is seen in both normal cells and cancer cells. 2 In this article, we study the role of Wnt pathway in the formation of adherent and occluding junctions, since they are two of the types of connection more relevant in the formation of complex intercellular junctions. Adherent junctions are distributed along a wide fringe of the intercellular membrane and are mediated by a family of transmembrane glycoproteins, called cadherins. E-cadherin or cadherin-I forms intercellular bridges, similar to a zipper, between adjacent cells and, in its turn, is associated within the cell with the actin cytoskeleton and with other proteins that are related to the catenin family pathway. 4 Therefore, these intercellular junctions are important not only for the intercellular adhesion and cell architecture. Occluding junctions (also known as tight junctions) are intercellular junctions located in a narrow strip of the apical zone that are mediated by transmembrane proteins, among which are the claudins and ocludins, that interact with the actin cytoskeleton through the family of proteins ZO (Fig. 3).

Different types of intercellular junctions, particularly Wnt/β-catenin regulates the formation of adherents junctions. As shown, both cells should produce the same protein junctions. If this does not occur, junctions do not form.
The Wnt family of secreted proteins consists of 19 glycoproteins, characterized by six conserved cysteine residues. Furthermore, they are lipid modified, and this lipid mark is important for secretion and efficient signaling. 36 The Wnt pathway leads to the activation of two pathways: the canonical and the noncanonical. 37 The canonical pathway was first identified and delineated from genetic screens in Drosophila, worm, frog, fish, and mouse, and in all cases its function is the same: the accumulation and translocation of the adherens junction-associated protein β-catenin into the nucleus. The noncanonical Wnt pathway increments the endoplasmic reticulum Ca2+ output and also increases the Ca2+ content in the cytosol. This Wnt pathway stimulates Rho/Rac, and in a previous study we model the MAPK pathway where Rho participates, so that we already have studied the reactions that arise from it. 38 The canonical Wnt pathway (Wingless and INT-1) regulates the expression of numerous genes involved in cellular differentiation, proliferation, and survival. 39 Wnt is a soluble protein ligand that initiates the signaling Wnt pathway by coupling with a transmembrane Frizzled complex associated with the receiver of low-density lipoprotein (LRP). The following steps of this pathway depend critically on the β-catenin, which acts as a control center for signal transmission from the plasma membrane to the nucleus. In the absence of Wnt, β-catenin is a part of a destruction complex, formed among others by the proteins APC, Axin, CKI 1α (CK1α), and GSK3β. In this complex of destruction, the β-catenin is phosphorylated by GSK3β and CK1α, which is followed by the ubiquitination and proteasomal degradation of β-catenin. 40 A degradation of β-catenin in the absence of a Wnt prevents the formation of intracellular connections. The Wnt coupling with Frizzled/LRP gives rise to the accumulation of β-catenin in the cell, and the excess of it is transferred to the nucleus, where it acts as coactivator of proteins, 41 such as CBP, BCL9, TCF, and LEF (Fig. 2). The β-catenin is a structural component of the cell–cell interactions through the formation of adherent junctions, with the participation of cadherins and phosphorylated catenins.42,43 The participation of the catenins is essential for the establishment of the adhesion between cells, and they represent an important point for the regulation of complex of accession, as defects in the expression or function of catenins are associated with loss of adhesion and metastasis. 44 For example, the expression of α-catenin has antiproliferative effects, basically inhibiting transcription mediated by β-catenin. For the correct establishment of adherent connections, essential requirement is the association between cadherin and α-catenin mediated by the β-catenin.
Materials and Methods
In this section, we first present the platform upon which we intend to build the model and execute simulations, giving also a sketch of the methodology used at the purpose. We then describe the model, specifying the part of the entire Wnt signaling pathway we consider and how its interacts with other important pathways of the cell.
The Bioinformatics Framework
Modeling cell signaling requires a robust approach accounting for the important features that govern the activity, interaction, selectivity, inhibition, and evolution of such a sort of complex system, such as situatedness, adaptation, diversity, eternity, topology, and locality. Such features have found a suitable support in our bioinformatics framework, which is itself a chemical metaphor-based model as introduced in Ref. 45. In fact, the cell detects the spatial context (the cellular compartments) of signaling elements, enabling or disabling actions and interactions accordingly (situations), dynamically adapts to external changes (adaptively), can change its function over time and then the activities being executed (presumption and diversity), and is able to tolerate runtime changes in terms of structure, elements, and function (eternity). Moreover, the chemical metaphor-based model supports topology and locality, respectively, to model the cellular structures involved in cell signaling and to provide a view of the signaling system as a composition of local interactions. Finally, and more generally, its biochemical inspiration makes BTSSOC-Cellulat, an infrastructure particularly suitable for simulating systems characterized by two types of components, chemical reactions and concentrations, as is the case of cellular signaling systems. A detailed explanation of BTSSOC-Cellulat, including all technical details, can be found in Refs. 27-32. Here, we will just highlight the benefits that the bioinformatics platform provides the user for simulating cellular signaling systems, such as multiple compartmentalization, representation of different types of chemical reactions, representation of signaling molecules and activation and deactivation signals, management of kinetic parameter of chemical reactions, and a wide range of visual and interactive tools.
In BTSSOC-Cellulat, different biological structures, such as cellular compartments, cells, and tissues, can be represented as part of the simulated cell system. For example, the user can create a tissue comprising several cells within each cell to create the required intracellular compartments. That is, the bioinformatics platform supports the simulation of biological processes that require communication and interaction among two or more cells, which is useful for the user. With respect to chemical reactions, the bioinformatics platform allows the user the specification of the following types: collaborative activation (aA* + bB* + cC → cC*), alternative activation (aA* + bB → bB*), formation of protein complexes (aA + bB + cC* → mABC*), decomposition reactions (AB → A + B), standard equation for enzymatic reaction (E + S ↔ ES → E + P), and interaction from and to extracellular space (→bB, bB→). Moreover, BTSSOC-Cellulat allows both specifying the measurement units (millimole, micromole, nanomole, and picomole) and amounts of reactants, and the concentration values can be entered as floating point values. Regarding the kinetic parameters, in BTSSOC-Cellulat, chemical reactions are executed with the proper rate. Every molecule is explicitly modeled and every reaction, in which they can participate, is explicitly simulated on the basis of a stochastic algorithm, Gillespie's stochastic simulation algorithm. 46 Once the system has been initialized, ie, molecules, reactions, and reaction rates are defined, the simulation proceeds choosing the next reaction to occur on the basis of a random number and its propensity function that is calculated based on the reaction rate and on the number of reactants. The time interval to update the simulation time is also computed step by step depending on a random number and on the sum of propensity functions of all reactions. The iteration of these steps constitutes the simulation. As a final point, the bioinformatics platform offers the user a wide range of tools that show in every moment the status of each of its elements and the incremental structure of the resulting signaling network, among which are concentration–time charts, the activity map, and the dynamic graph.
Figure 4 illustrates the main graphical user interfaces of BTSSOC-Cellulat with quite a lot of highlights previously described. The bioinformatics framework BTSSOC-Cellulat (in its Executable Jar File version) and the simulation file can be downloaded from: https://app.box.com/s/epojx6oqcazzbpij2zn5ix2b4udpnrp4. Note that BTSSOC-Cellulat was developed as a standalone application and not as a Web application. Therefore, for proper installation, follow the instructions included in the README file.

Main GUI of bioinformatics platform BTSSOC-Cellulat.
Modeling and Simulation Methodology
The methodological workflow of the major activities to be executed during the modeling and simulation of the Wnt signaling pathways is shown in Figure 5. As can be seen in Figure 5, steps 1 and 2 belong to modeling phase, and these are referred to the proper construction of the signaling pathway model, including the definition of cellular structures, signaling components, interactions among signaling components, chemical reactions, reactants, and initial concentration values, whereas steps 3-8 correspond to simulation phase on bioinformatics platform BTSSOC-Cellulat.

Modeling and simulation workflow.
The Model
As discussed in the “Introduction” section, in order to induce cell death of cancer cells, in previous study, we have modeled different scenarios of cancer cell death, so that our platform already has the necessary information on those scenarios. We model the Wnt pathway that regulates the formation of intracellular communication channels, as we believe that by interfering the cancer cell communication with its surroundings, it is possible to cut for the cellular oxygen and nutrient supply and prevent its growth and multiplication. The BTSSOC-Cellulat platform since its inception has been modified and enriched so that it is possible to model different cell compartments of two neighboring cells. The objective of our modeling and simulation is to propose key molecules that allow us to avoid communication between cancer cells and surrounding normal cells and prevent cancer from spreading.
The Canonical Pathway of the Wnt Signaling Cascade
As mentioned in the “Biological background” section, the Wnt pathway can be activated in two ways: the canonical pathway that is involved in cell differentiation and the non-canonical pathway that is involved in controlling the cell polarity and mobility. We are most interested in the canonical pathway, because this pathway is related to other signaling pathways in cancer: it is dependent on the β-catenin involved in adhesion and cell polarity (Fig. 1). The Wnt/β-catenin initiates the construction of intercellular junctions between neighboring cells and is associated with changes in the regulation of different types of cancer.6–13 In normal cells, the Wnt/β-catenin pathway is usually inactive. 47 The causes of this deregulation are associated with genetic mutations that affect multiple pathway components 48 and epigenetic mechanisms that alter the expression of regulatory genes through Wnt. 49
In order to model the canonical pathway, we will consider all the reactions that take place there and we will use their kinetic values obtained from the literature. In Table 1 and Figure 6, some reactions are exemplified.
Chemical reactions.


Simplification of the interactions modeled in this study.
Signaling Pathways Required and Previously Simulated
We are interested in regulating the growth of tumor cells, and in our virtual laboratory, we already have ~325 reactions, including EGF/MAPK/JAK-STAT, 20 PI3K, 29 and caspases30–32 pathways. We integrate the canonical Wnt pathway and other pathways, such as Notch, 33 Hh, 34 and hypoxia stress pathways (Fig. 2).
Results
Our simulation scenario consists of two cells: a normal cell and a cancer cell. The cancer cell junctions are used to share information with the normal cell and to send signals of malignancy. On the other hand, the normal cell sends signals of cell death to defend itself. In our model, each cell is programed, on top of the bioinformatics platform, to contain all the necessary information: cellular compartments, chemical reactions, and reactants corresponding to the signaling pathways described in the previous section. Once the platform is loaded with this information, we can conduct experiments to find proteins that prevent the formation of gap junctions and leave the cancer cell uncommunicated. After a series of possible combinations, we selected two proteins, APC and NDK2. APC is a part of the complex that favors β-catenin ubiquitination and, thus, its destruction, 4 and NKD2 inhibits the disruption, promoting the complex of β-catenin. Usually, when the Wnt/β-catenin pathway is inactive, ie, Wnt has not joined its Frizzled signalling WNT receptor (FZD) receptor, β-catenin is linked to the degradation complex (Axin, APC, CKI, and GSK-β), and in the β complex, catenin is phosphorylated at residues 33, 35, 41, and 47. This marks the protein for ubiquitination and subsequent degradation in the proteasome. NKD2 inhibits DVL, and DVL in turn prevents β-catenin phosphorylation and degradation (Fig. 7). For our experiments, we use increasing concentrations (0.001, 0.01, 0.1, 1.0, 10.0, 100.0, and 1,000.00 μM) of NKD2 or APC. We consecutively increase the initial concentrations of these regulators, while keeping all other factors constant.
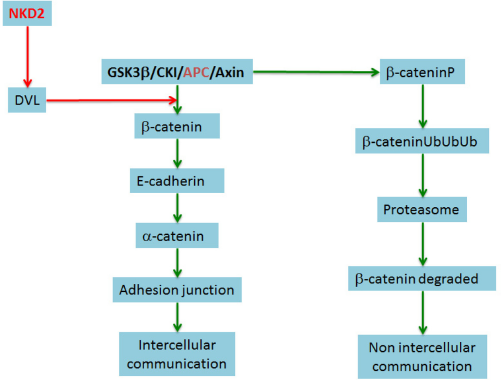
The general role of the proteins used for in silico experiments. APC protein is a part of the complex that favors ubiquitination of β-catenin and thus its destruction and NKD2 inhibits the disruption, promoting the complex of β-catenin. Usually, when the Wnt/β-catenin pathway is inactive, ie, Wnt has not joined its FZD receptor, β-catenin linked to the degradation complex (Axin, APC, CKI, and GSK-β), and in the β complex, catenin is phosphorylated at residues 33, 35, 41 and 47. Then the protein is ubiquitinated and subsequently degraded by the proteasome. NKD2 inhibits the DVL, and DVL prevents β-catenin phosphorylation and degradation.
APC
By using increasing concentrations of APC, with the lowest concentration of 0.001 μM, we should expect -2,500 ms to observe the disappearance of β-CateninCyt and DVL. At the concentration of 0.01 μM, it was observed that β-CateninCyt disappears at 1,000 ms and DVL at 3,000 ms, something similar happens with the concentration of 1 μm. For concentrations of 10, 100, and 1000 μM, the disappearance occurs at similar times, but excessive APC appears. Note that in all cases, the concentration of the FZD receptor is maintained or increases apparently because Wnt concentration, which is bound to FZD, diminishes, and new receptors appear, while Wnt still exists in the cytoplasm. The observed responses occur between 1,000 and 3,000 ms, which is 10 times less than the range observed, for example, in response to the activation of intracellular by different Wnt ligands and subsequent translocation of β-catenin into the nucleus (Figs. 8–10). 55

APC = 0.001 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. With the lowest level, we should expect about 2,500 ms to observe the disappearance of β-CateninCyt and DVL. Only the receptor and Wnt remain present.

APC = 0.01 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. At this level, it was observed that β-CateninCyt and DVL disappear within 1,000-3,000 ms, something similar happens with concentrations of 0.1 and 1 μM. Only the receptor and Wnt remain present.

APC = 10 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. For concentrations of 10, 100, and 1,000 μM, DVL is not observed, and β-CateninCyt disappears after 2,500 ms. Note that in all cases the concentration of the FZD receptor is maintained or increases apparently due to having no response to the binding of Wnt; new receptors appear, since Wnt exists in the cytoplasm.
NKD2
When using increasing concentrations of NKD2 observed for the first concentration of 0.001 μM, the disappearance of β-catenin and DVL is observed at 1000 ms. The disappearance is even faster (500 ms) when using a concentration of 0.01 μM. Interestingly, with 0.1 μM, the disappearance of DVL occurs at 3,000 ms, and that of β-CateninCyt occurs at 100 ms, something similar happens when we use a concentration of 1 μM, but the disappearance of β-CateninCyt occurs at 50 ms (data not shown). By using a concentration of 10 μM, DVL disappears at 10 ms, and β-CateninCytUb appears at 1,500 ms and disappears at ~2,200 ms. For a concentration of 100 and 1000 μM, something similar happens. In all cases, despite the absence of Wnt, junctions are formed (Figs. 11–15).

NKD2 = 0.001 μM. On the chart, the x-axis represents the time in milliseconds and the y-axis represents the concentration of reactants in micromolar. For the first concentration of 0.001 μM, β-CateninCyt disappears after 1,000 ms, and DVL is not observed.

NKD2 = 0.01 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. The disappearance of β-CateninCyt is even faster, 500 ms, when using a concentration of 0.01 μM.

NKD2 = 0.1 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. Interestingly, for this concentration, DVL disappearance occurs after 3,000 ms and β-CateninCyt after 100 ms, something similar happens with a concentration of 1 μM.

NKD2 = 10 μM. On the chart, the x-axis represents the time in milliseconds, and the y-axis represents the concentration of reactants in micromolar. For this concentration, β-CateninCyt is not observed and DVL disappears after 2,000 ms.

NKD2 = 100 μM. On the chart, the x-axis represents the time in milliseconds and the y-axis represents the concentration of reactants in micromolar. For 100 and 1,000 μM concentrations, similar behavior is observed. β-CateninCyt and DVL are not seen.
Discussion
As was observed in the simulation, increasing the concentration of NKD2 and APC inhibits the Wnt signaling pathway by preventing the formation of intercellular junctions, since the β-CateninCyt is destroyed and disappears. Thanks to our platform, there is no need for the in vitro experiment that allows us to determine the appropriate concentration of these key molecules. To conduct the experiment in cancer cells, we use 0.1 μM concentration in both cases; using this concentration, β-CateninCyt quickly disappears, and thus the formation of gap junctions is avoided. The platform can incorporate new information on the different interacting signaling pathways, in which the component values are changing over time. With our virtual laboratory, we were able to determine that the regulation of the Wnt pathway is necessary for cell communication, facilitating the spread of cancer cells, but its complete removal prevents the normal cell transformation for the cells surrounding the cancer cell. The cancer cell can be sent to death. If the cancer cell is not communicating with its environment, it cannot get nutrients and oxygen, cannot send signals of death or malignancy to neighboring cells, and eventually dies.
Problems Found during Simulation
During the simulation, the following problems related to the stability of the simulation solutions subjected to parameter adjustment were found: (1) the concentration value of some reactants did not match the required value, which prevented the expected solution to be reached, (2) the estimated rate of some chemical reactions did not meet the required value, avoiding that such reactions were executed at the appropriate time by Gillespie's algorithm, (3) the relationship between the rates of the various chemical reactions of the simulation was not properly adjusted, having as a result that some slower reactions were executed before the faster reactions.
The mentioned problems were solved one at a time through the feedback loop provided by the modeling and simulation workflow (steps 8, 6 and 7 in Fig. 5). The parameter adjustment was carried out suggesting the new values of concentration and rate from the available theoretical and experimental knowledge of Wnt-dependent signaling pathways, allowing us to estimate some unreported or missing values. At this point, it is worth noting that after parameter adjustment it was possible to observe the effects of perturbations, such as adding elements (ie, reactants and chemical reactions) and taking them out as knockouts on the simulated intercellular system.
Future Study
During the development of this study, we model the Wnt pathway, which is activated to form junctions between two cells. The next step is to increase the size of the cell population and to include other Wnt cross talk pathways, such as HIPPO, which regulate cell size and inhibition contact and then the cell growth. If we can control this pathway, we can avoid tissue invasion and metastasis.
Author Contributions
Conceived the cell death signaling model, designed the intercellular communication model, coordinated the modeling and simulation process, and wrote the first draft of the article: MC-G. Coordinated the software reengineering study, participated in the modeling and simulation of intercellular communication model, and wrote the first draft of the article: PPG-P. Contributed to the writing of the article, jointly developed the structure and arguments for the article, made the critical revisions, and approved the final version: SM. Carried out the software reengineering study and the simulation study: OSC. Collected the intercellular communication data and participated in the design of the model: EHC. All authors reviewed and approved the final article.
