Abstract
A spectrophotometric method was developed for simultaneous determination of amlodipine (Aml) and valsartan (Val) without previous separation. In this method amlodipine in methanolic solution was determined using zero order UV spectrophotometry by measuring its absorbency at 360.5 nm without any interference from valsartan.
Valsartan spectrum in zero order is totally overlapped with that of amlodipine. First, second and third derivative could not resolve the overlapped peaks.
The first derivative of the ratio spectra technique was applied for the measurement of valsartan. The ratio spectrum was obtained by dividing the absorption spectrum of the mixture by that of amlodipine, so that the concentration of valsartan could be determined from the first derivative of the ratio spectrum at 290 nm. Quantification limits of amlodipine and valsartan were 10-80 μg/ml and 20-180 μg/ml respectively. The method was successfully applied for the quantitative determination of both drugs in bulk powder and pharmaceutical formulation.
Introduction
Amlodipine is a dihydropyridine calcium channel blocker. It is used for management of hypertension and angina pectoris. 1 Several methods were developed for amlodipine determination. The most recent reported methods include capillary electrophoresis (CE) methods which were used for amlodipine determination2–4 or for enantiomer separation and determination of amlodipine.5–7
HPLC methods were used for the determination of amoldipine alone either in human fluids or in tablet form.8–15 Other HPLC methods determine amlodipine in a mixture with another drug as metoprolol succinate,16,17 atrovastatin,18,19 benazepril20,21 and losartan. 22 Amlodipine was determined in tablets and biological fluids using voltametric method. 23 Spectrophotometric methods were used for the determination of amlodipine,24,25 amlodipine, enalapril mixture 26 or for monitoring amlodipine photodegradation. 27
Valsartan is an angiotensin II receptor antagonist used in the management of hypertension to reduce cardiovascular mortality in patients with left ventricular dysfunction following myocardial infarction and heart failure. 1
Several methods have been reported for valsartan assay including HPLC methods,28–32 spectrofluorimetric method 33 and capillary electrophoresis. 34
Valsartan and hydrochlorothiazide mixture were determined using HPLC methods,35–37 derivative spectrophotometric methods,38–40 TLC method 41 and CE method. 42
Two HPLC methods for the determination of valsartan and nebivolol were reported.43,44
Simultaneous determination of valsartan and amlodipine in post mortem whole blood was carried out using HPLC-MS technique. 45
The goal of antihypertensive therapy is to abolish the risks associated with blood pressure elevation without adversely affecting quality of life. Drug selection is based on efficacy in lowering blood pressure and in reducing cardiovascular end points including stroke, myocardial infarction and heart failure. The American Society of Hypertension presents a paper on combination therapy of hypertension. Specific combinations are designated as preferred or acceptable, less effective combinations are also identified. The study classifies the combination therapy of amlodipine and valsartan as acceptable. 46
Experimental
Chemicals and Reagents
Amlodipine and valsartan donated by Novartis, Egypt. Exforge tablets labeled to contain 10 mg of amlpdipine and 160 mg of valsartan. Batch number 0051, manufactured by Novartis, Egypt. Methanol analytical grade (Analar).
Apparatus
Ultraviolet/Visible spectrophotometer, Shimadzu, Japan 1601pc. Ultrasonic crest model 575T Cortland, New York 13045, USA.
Sample Preparation
Stock methanolic solution of amlodipine and valsartan of concentration 100 μg/ml and 200 μg/ml respectively were separately prepared by dissolving the appropriate amount of the respective substance in methanol. Working standard solutions used for method optimization were prepared freshly before application by mixing suitable aliquots of stock solutions of amlodipine and valsartan and diluted with methanol to obtain the required concentration ranges.
Stock methanolic solution of laboratory prepared mixture of amlodipine and valsartan containing 100 μg/ml and 1.6 mg/ml respectively was prepared by dissolving 25 mg amlodipine and 400 mg valsartan in 250 ml methanol.
Tablets working solution was prepared by dissolving an accurately weighed quantity of powdered exforge tablets sample containing an equivalent of 10 mg amlodipine and 160 mg valsartan was transferred into 100 ml volumetric flask, then 70 ml methanol was added. The mixture was sonicated for ten minutes and the volume was made up using methanol, then the sample was filtered.
Procedure
Aliquots (1-8 ml) or (1-9 ml) of amlodipine or valsartan stock solutions respectively were transferred into two separate series of 10 ml volumetric flasks and volumes were made up with methanol. The absorbance spectrum of each flask were recorded at zero order between 200-450 nm and stored in a personal computer.
Amlodipine (
The absorbance of amlodipine series were determined in zero order at 360.5 nm.
The UV absorption spectra of the series of valsartan solutions were divided wavelength by wavelength by a standard spectrum of amlodipine (40 μg/ml). The first derivative of the obtained ratio spectra with Δλ = 4 nm and scaling factor equal 10 were used for determination of valsartan concentration from the amplitude at 290 nm.
Application of the Method for the Determination of Laboratory Prepared Mixture
The method was applied for the determination of laboratory prepared mixture of amlodipine and valsartan containing 100 μg/ml and 1.6 mg/ml respectively. Aliquots (1-8 ml) and (1.5-10 ml) of the stock methanolic solution of laboratory prepared mixture were transferred into two separate series of 10 ml and 100 ml volumetric flasks, volumes were made up using methanol. The first series of (10 ml) volumetric flasks was used for the determination of amlodipine, while the other series (100 ml volumetric flasks) was used for the determination of valsartan applying the same mentioned procedure.
Application of the Method for the Determination of Exforge Tablets
Aliquots of 1.5, 4 and 8 ml of tablets working solution were transferred into two separate series of 10 ml and 100 ml volumetric flasks for determination of amlodipine and valsartan respectively.
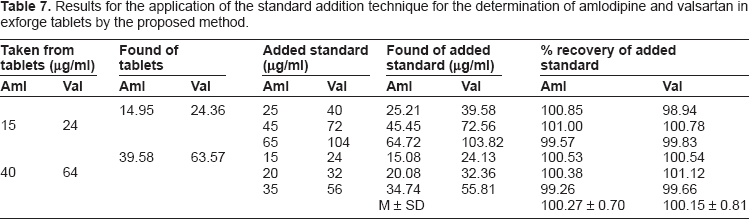
To study the accuracy of the proposed method and check the interference from excipients present in the dosage form, recovery experiment were carried out by standard addition method. This study was performed by addition of different amounts of amlodipine and valsartan to a known concentration of the commercial tablets.
Result and Discussion
The UV spectra of amlodipine and valsartan in zero order show that amlodipine has a significant peak at 360.5 nm at which the spectrum of valsartan does not interfere (Fig. 2). So methanolic solution of amlodipine was quantitatively determined at 360.5 nm, Beer's law was obeyed over concentration range 10-80 μg/ml. The amlodipine concentration could be calculated using the following equation.
UV spectrophotometric spectrum of methanolic solution of (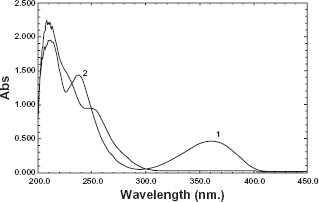
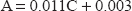
Valsartan spectrum in zero order shows a complete overlapping with that of amlodipine (Fig. 2). Applying first, second and third order spectra did not resolve valsartan peaks. So first derivative of the ratio spectra was applied to determine valsartan in presence of amlodipine.
The influence of Δλ and the effect of the divisor concentration on the calibration graph for the proposed mixture was studied in order to select the best factor for the determination. Results indicate that Δλ = 4 was the most suitable one, while divisor concentration range 10-60 μg/ml gave good results, 40 μg/ml was used overall.
For the determination of valsartan the absorption spectra of valsartan was divided by that of standard amlodipine solution (40 μg/ml). The first derivative of the developed ratio spectra were calculated with Δλ = 4 and scaling factor equal 10. (Fig. 3) shows that valsartan could be determined by measuring the amplitude at 290 nm.
First derivative of ratio spectra of different concentrations of valsartan using spectrum of amlodipine (40 μg/ml) as a divisor spectrum.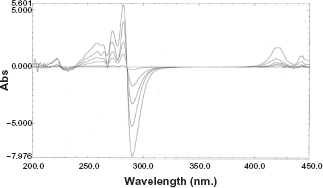
The proposed method is applicable over the concentration range 20-180 μg/ml. Valsartan concentrations could be determined applying the following equation.
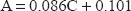
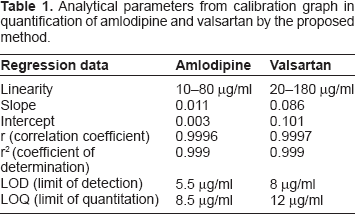
Analytical parameters from calibration graph in quantification of amlodipine and valsartan by the proposed method.
Results of quantitative analysis of amlodipine and valsartan in bulk powder by the proposed method.

Aml = amlodipine
Val = valsartan.
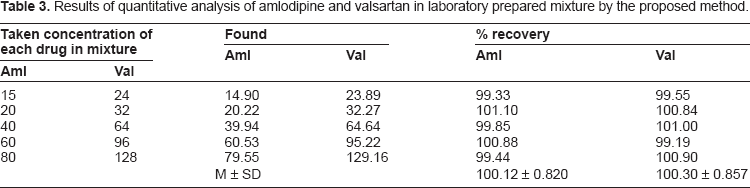
Results of quantitative analysis of amlodipine and valsartan in laboratory prepared mixture by the proposed method.
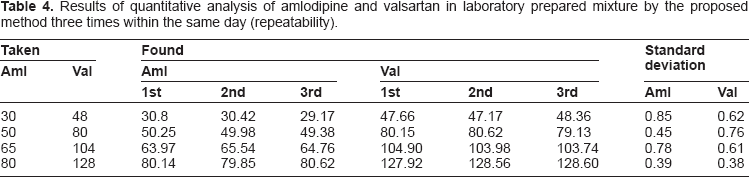
Results of quantitative analysis of amlodipine and valsartan in laboratory prepared mixture by the proposed method three times within the same day (repeatability).
Results of quantitative analysis of amlodipine and valsartan in laboratory prepared mixture by the proposed method over three different days (reproducibility).
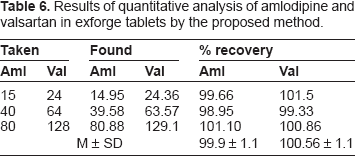
Results of quantitative analysis of amlodipine and valsartan in exforge tablets by the proposed method.
Results for the application of the standard addition technique for the determination of amlodipine and valsartan in exforge tablets by the proposed method.
Conclusion
In this study a new spectrophotometric method was developed to identify amlodipine and valsartan in mixture without previous separation. The method was optimized and validated. The method was successfully applied for the determination of amlodipine and valsartan in exforge tablets.
This method can yield results in a short time and does not require relatively expensive apparatus like HPLC.
Footnotes
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
