Abstract
This paper deals with development and validation of a high performance liquid chromatographic method for the quantitative determination of disodium EDTA (Ethylenediaminetetraacetic acid) in Meropenem active pharmaceutical ingredient (API). EDTA was derivatized with Ferric chloride solution by heating at 70 °C in water bath for about 20 minutes and the chromatographic separation achieved by injecting 100 μL of the derivatized mixture into a Waters HPLC system with photodiode array detector using a Phenomenex Luna C18(2) column (250 × 4.6 mm), 5 μ, The mobile phase consisting of 5% methanol and 95% of 0.7 g/L solution of Tetra butyl ammonium bromide and 4.6 g/L solution of sodium acetate trihydrate in water (pH adjusted to 4.0 with the help of acetic acid glacial) and a flow rate of 1 milliliter/minute. EDTA eluted at approximately 6 minutes. The method was suitably validated with respect to specificity, linearity of response, precision, accuracy, ruggedness, stability in analytical solution, limit of quantitation and detection and robustness for its intended use.
Introduction
Ethylenediaminetetraacetic acid (EDTA) (Fig. 1) is a powerful chelating agent, forming stable complexes with most metal ions. Due to its ability to sequester metal ions, EDTA is widely used in medicine, chemical industry, food technology, agriculture and pharmaceutical technology. EDTA in its disodium salt or calcium disodium salt form is frequently used in pharmaceuticals because of its stability, compatibility and low toxicity. In the field of analytical chemistry, besides its use in complexometric titrations, EDTA has been reported to be very useful ligands for the complexation of metals, which enables their chromatographic separations.
1
Structure of EDTA.
Meropenem is an ultra-broad spectrum injectable antibiotic used to treat a wide variety of infections, including meningitis and pneumonia. It is a beta-lactam and belongs to the subgroup of carbapenem, similar to imipenem and ertapenem. EDTA is being used in the synthesis of Meropenem. Therefore the quantification of the residual EDTA is essential as per regulatory requirement.
Various analytical methods have been proposed for the determination of EDTA in a wide variety of sample matrices. 2 They include titrimetry, 3 spectrophotometry, 4 electrochemistry eg, polarography, 5 differential pulse polorography, 6 catalytic potential tirtrimetry, 7 differential pulse anodic stripping voltammetry, 8 amperometry, 9 capillary electrophoresis, 10 and chromatography. Among them, gas chromatography and HPLC (reverse phase ion-pair or ion exchange retention mechanism) appear to be the prevailing techniques, despite the fact that EDTA lacks volatility and exhibits low UV/visible absorptivity. The gas chromatographic methods always include a time consuming derivatization steps, in which EDTA is converted into methyl, ethyl, propyl and butyl esters to obtained volatility.11,12
This paper describes development and validation of derivatized method with direct UV-detection for the quantitative determination of disodium EDTA in Meropenem drug substance. This method also has advantages over some techniques as mentioned in above references,3–12 like here EDTA response is measured by direct UV detection with enhanced sensitivity and method is simpler, highly reproducible, specific and accurate, compare to using complex techniques like titrimetry, spectrophotometric, capillary electrophoresis or GC technique. As EDTA does not contain any chromophoric group, it is very difficult to determine EDTA by direct UV detection. Hence a method has been optimized and developed by derivatizing disodium EDTA with ferric chloride solution. The method has been optimized with respect to reaction time, derivatization temperature and derivatization reagent volume and suitably validate for its intended use.
Experimental
Reagents and Chemicals
Di-sodium salt of Ethylenediaminetetra-acetic acid dehydrate was purchased form Qualigens, Sodium acetate trihydrate was purchased from Qualigens (AR grade), Tetra butyl ammonium bromide was purchased from Spectrochem (AR grade), Methanol (HPLC grade), Acetic acid glacial was purchased from Rankem (AR grade), Ferric chloride anhydrous was purchased from Qualigens (AR grade) and Meropenem was obtained from Ranbaxy Labs. Ltd. (India). All the above materials were used without any further purification. Water (HPLC grade) was used for the preparation of solutions.
Chromatography
The analytical separations were carried out on a Waters HPLC system, equipped with a 2695 separation module and 2996 photodiode array detector. The analytical column was a Phenomenex Luna C18 (2) column (250 × 4.6 mm), 5 μ. The mobile phase consisted of premixed and degassed solution of buffer and methanol in the ratio of [95:5] [v/v]. Mobile phase was prepared by dissolving 4.1 gm of sodium acetate trihydrate and 0.64 gm Tertrabutyl ammonium bromide in 950 mL of water and than added 50 mL of methanol, mixed and pH adjusted to 4.0 with acetic acid glacial. The mobile phase was filtered through a 0.45 μm membrane filter. Water used as a diluent for the preparation of solutions. The flow rate was 1 mL/min and runtime was 10 minute for EDTA standard solution and 60 minutes for Meropenem sample solutions. Column temperature was maintained at 30 °C. UV detection was measured at 254 nm and the volume of sample injected was 100 μL. The control of the HPLC system and data collection was by Empower software.
Derivatization Process of Standard and Sample Solution
Standard Solution Preparation
A standard stock solution at 1 milligram per milliliter (mg/mL) was prepared by dissolving disodium salt of EDTA in diluent (Accurately weighed and disolved disodium salt of EDTA dihydrate 63.5 mg equivalent to 50 mg of EDTA in 50 mL of diluent). The derivatization process was carried out by transferring 10 mL of standard stock solution into 100 mL volumetric flask. Add 5 mL of Ferric chloride solution (About 0.64 gm of Ferric chloride anhydrous transfer to 200 mL volumetric flask and dissolve with 5 Ml 1 N solution of hydrochloric acid and make up the volume to 200 mL with water). Shake well and keep in water bath at 70 °C for 20 minutes, finally make-up the volume with diluent after it attains room temperature. Dilute the solution with diluent to achieve the final concentration 0.0002 mg/mL.
Sample Solution Preparation
A sample solution was prepared by dissolving 500 mg of Meropenem sample in diluent, in 25 mL volumetric flask. The derivatization process was carried out by adding 2 mL of Ferric chloride solution. Shake well and keep in water bath at 70 °C for 20 minutes, finally make-up the volume with diluent after it attains room temperature. The final concentration is 20 mg/mL.
Results and Discussion
Method Validation
Specificity
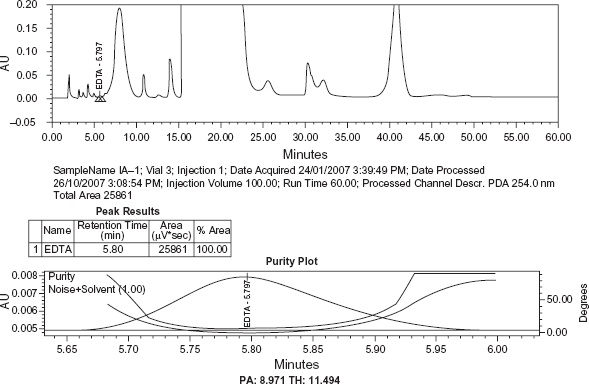
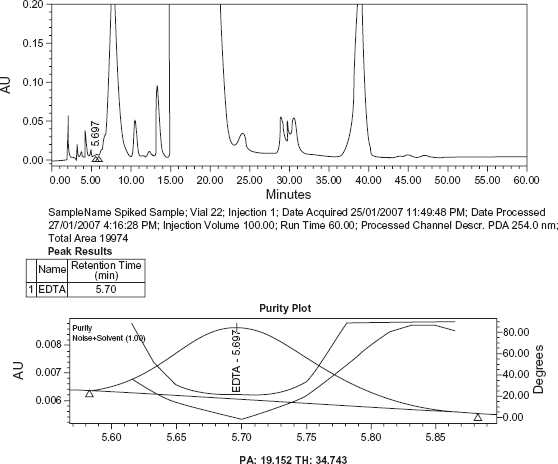
Blank, sample solution and sample solution spiked with EDTA (at 0.001% of sample concentration) along with other known related substances of Meropenem were chromatographed individually as per the method to examine interference, if any, with EDTA peak.
No peak from the blank was observed at the retention time of EDTA peak. The peak purity plot of EDTA shows that the peak is pure and has no co eluting peaks, indicating specificity of the method (Figs. 2–4).
Chromatogram of blank. Chromatogram and Peak purity plot of EDTA in sample. Chromatogram and Peak purity plot of EDTA in Sample spiked with known related substances of Meropenem.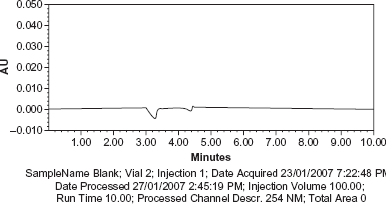
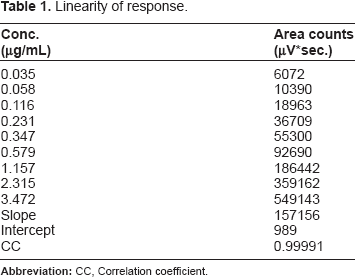
Linearity of Response
Linearity of response.
Precision
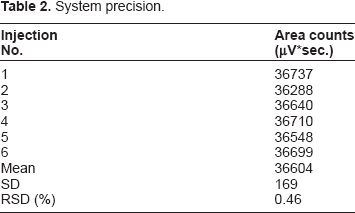
System Precision
System precision.
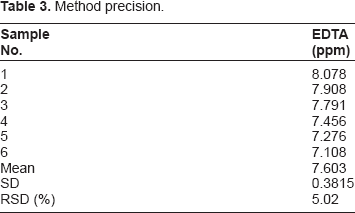
Method Precision
Method precision.
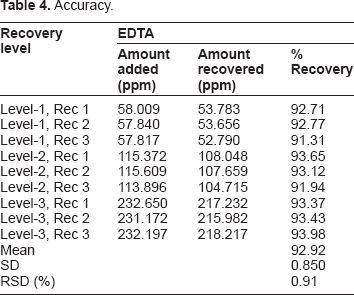
Accuracy
Accuracy.
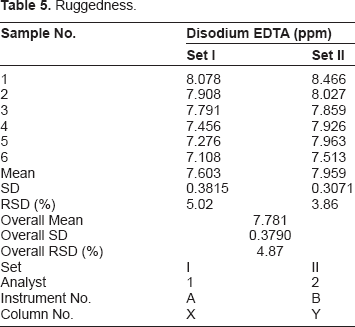
Ruggedness
Ruggedness.
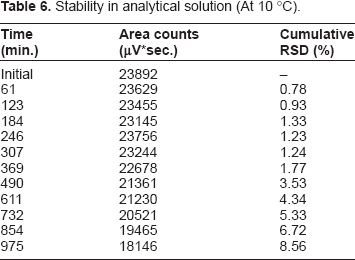
Stability in Analytical Solution
Stability in analytical solution (At 10 °C).
Limit of Quantitation and Detection
Limit of quantitation and detection.
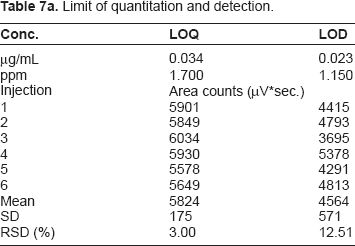
Summary of LOQ and LOD values.

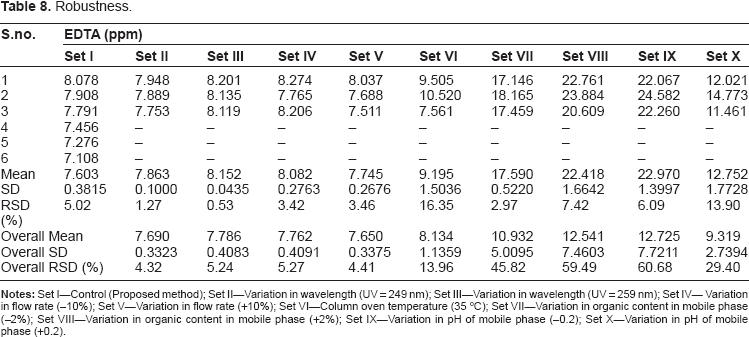
Robustness
Robustness.
Conclusion
A simple isocratic reverse phase method for the determination of EDTA was developed and validated using direct UV detection. The method is selective, precise and accurate and was successfully applied to the analysis of commercially available Meropenem drug substances. EDTA is not easily detected by HPLC using UV detection because of absence of a chromophoric group. Derivatization with Ferric chloride solution is a simple and very effective means of enhancing the chromatographic detection of the compound.
Footnotes
Acknowledgment
Authors are thankful to Ranbaxy Research Laboratories for providing necessary facilities and for permission to publish this article.
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
