Abstract
Inhaled corticosteroids are the mainstay of asthma therapy, but there is now compelling evidence that addition of a long-acting inhaled β2-agonist, such as formoterol, gives better control in terms of reduced symptoms, improved lung function and reduced exacerbations in patients with mild to severe persistent asthma than increasing the dose of corticosteroids in patients not fully in control by low dose. This has led to development of fixed dose combination inhalers such as budesonide/formoterol.
Budesonide/formoterol combination in a single inhaler represents a safe, effective and convenient treatment option for management of patients with unstable asthma than inhaled steroid alone. This combination has shown effectiveness for both maintenance and rescue therapy. Clinical results show that the budesonide/formoterol by SMART approach prolongs the time to first severe asthma exacerbation, reduces frequency of exacerbation and maintains day to day asthma control at a reduced corticosteroid load and cost when compared with higher fixed maintenance dose of combination inhalers. With regular maintenance therapy by this approach it is more likely to improve patient compliance.
Budesonide/formoterol combination inhaler has shown to have a faster bronchodialatory effect compared with other combination inhalers, a quality highly in demand during exacerbation episodes. Due to this rapid onset of action, budsonide/formoterol in a single inhaler lends itself to be used as a rescue medication, as well.
Introduction
Asthma affects 7% of the population, or 20 million Americans 1 and 300 million worldwide. 2 Asthma is a chronic condition involving the respiratory system in which the airways occasionally constrict, become inflamed, and are lined with excessive amounts of mucus, often in response to one or more triggers. 3 These triggers may be exposure to stimulants as such allergen, environmental tobacco smoke, cold or warm air, perfume, pet dander, moist air, exertion and exercise or even emotional stress. In children, the most common triggers are viral illnesses such as those causing the common cold.4,5 The airway narrowing causes symptoms such as wheezing, shortness of breath, chest tightness, and coughing. Asthma attack essentially has the clinical symptoms of shortness of breath (dyspnea) and wheezing or strider. 6 International guidelines stipulate goals for optimizing asthma management, such as preventing chronic and acute symptoms, minimizing exacerbations and emergency care, minimizing the use of rescue β2-agonists and maintaining normal levels of physical activities. 3 Even though, there is still no cure for the asthma, based on large number of studies done in the past 2.5 decades many patients can reach a relatively good asthma control. 3 This can be achieved by combining inhaled corticosteroids (ICS) and long-acting β2-agonists (LABA) in a single inhaler. This article discusses specifically the place of combination of budesonide and formoterol in single inhaler in management of asthma.
Asthma and treatment drugs–-mechanism of actions
Asthma is clinically characterized by repeated episodes of wheezing, breathlessness, chest tightness, and coughing, usually in the presence of variable and reversible airflow obstruction. 6 At the heart of asthma pathophysiology is chronic airway inflammation, 7 with filtration of eosiniphils, mast cells, and CD4+ T lymphocytes that express T helper cell type-2 cytokines such as interleukins 4, 5 and 13. 8 Airway remodeling is another characteristic feature of chronic persistent asthma, which consist of smooth muscle hypertrophy, thickening of basement membrane, increased mucus production and denudation of airway epithelium. 7 The basic understanding of asthma and its pathogenesis has lead to the development of novel pharmacological therapies. These include inhaled corticosteroids (ICS) and long-acting β2-agonists (LABA).
There has been important advances in understanding the molecular mechanism for the anti-inflammatory effects of corticosteroids.9,10 ICS cross the cell membrane of target cells in the airways on which epithelial cells appear to be the most important. Corticosteroids bind to glucocorticoid receptors (GR) in the cytoplasm, which then rapidly translocate to the nucleus of the cell. Some genes have a recognition site(s) in their promoter region for the activated GR dimmer known as a glucocorticoid response element (GRE) and are activated by the interaction with the GR. An example of gene activation by corticosteroid is the β2-receptor, where a significant increase in β2-receptors in human lung in vitro can be detected as early as 2 h after exposure to a corticosteroid. 11 An important anti-inflammatory enzyme that is activated by corticosteroids is mitigen-activated protein kinase phosphatase (MKP)-1, which is the endogenous inhibitor of mitogen-activated protein (MAP) kinase pathways that are pro-inflammatory. Corticosteroids induce a rapid 10-fold increase in MKP-1 mRNA within 2 h of exposure and an increase in MKP-1 protein in 4 h, with simultaneous inhibition of MAP kinase cell signaling. 12
A rapid anti-inflammatory effect of ICs was demonstrated with a single high dose of inhaled budesonide (2400 μg) with significant sputum eosinophils reduction and increased protection against hypertonic saline-induced bronchoconstriction within 6 hour after treatment administration in patients with mild asthma. 13 The protective action of corticosteroid against adenosine monophosphate (AMP) bronchoconstriction effect in asthma appears to be due to its rapid inhibitory effect on mucosal mast cells. 14
Glucocorticoids inhibit virtually all steps in the inflammatory response including cytokine production on variety of cell types. For instance, budesonide and dexamethasone have been shown to inhibit vascular cell adhesion molecule (VCAM-1) and intercellular adhesion molecule (ICAM-1) expression on activated epithelial and endothelial cells15,16 and on fibroblasts. 17 Glucocorticoids also inhibit granulocyte-macrophage colony stimulating factor (GM-CSF), interleukins (IL-8 and IL-6) production by epithelial cells and fibroblasts.18,19
Long-acting β2-agonists (LABA) are generally considered to be smooth muscle relaxants. However, their anti-inflammatory effects are indicated by an inhibitory effect on granulocyte adhesion to epithelium 20 and on infiltration of inflammatory cells in the skin and lung of guinea pigs. 21 Formoterol has been shown to inhibit ICAM-1 and VCAM-1 upregulation on human lung fibroblasts as induced by different cytokines. 17
Human lung fibroblasts may be involved in the inflammatory of asthma. 22 Furthermore, lung fibroblasts produce large amounts of GM-CSF and IL-8 after stimulation by proinflammatory cytokines. These products are able to activate and/or attract eosinophils and neutrophils. The effects of the combination of inhaled budesonide and formoterol in vivo may therefore be partly achieved through modulation of lung fibroblast activation.
Spoelstra and colleagues 23 investigated the effect of formoterol on the anti-inflammatory action of budesonide reflected by inhibition of adhesion molecule (ICAM-1, VCAM-1). These authros showed that formoterol exerts an additive effect on anti-inflammatory properties of budesonide, partially by the additive inhibition of cellular infiltration resulting from diminished adhesion molecule expression and GM-CSF production of lung fibroblast. 23
Inhaled corticosteroids (ICS) are considered the most effective drugs to suppress asthma-associated airway inflammation, largely through inhibitory interactions with pro-inflammatory DNA-binding transcription factors, such as nuclear factor-kappa B (NF-κB) and activator protein-1 (AP-1). 24 Inhaled long-acting β2-agonists, acting as bronchodilators, provide long-lasting relaxation of airway smooth muscles. 25 As evident in the rest of this article, asthmatic subjects treated with combination of inhaled corticosteroids and LABA tend to have better lung function and symptom control, and lower incidence of exacerbation compared with those given either drug alone. 26 In addition to their major anti-inflammatory effects, corticosteroids recently have been suggested to improve β2-adrenergic bronchodilation. Through delayed transcriptional action on airway cells, corticosteroids could increase β2-receptor numbers 27 and restore β2-receptor coupling to G proteins that mediate adenylyl cyclase simulations. 28 The proposed intracellular mechanisms for interaction between β-agonists and corticosteroids is depicted in Figure 1. 29
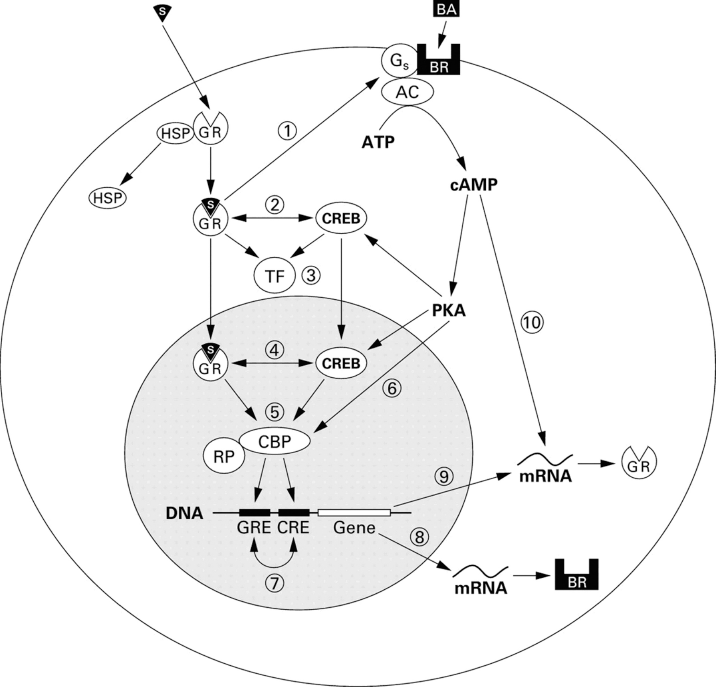
Proposed intracellular mechanisms for interaction between β-agonists and corticosteroids. (
A number of polyspecific cell membrane transporters, such as the potential-dependent organic cation transporters (OCTs) 1 to 3 and the pH-dependent organic cation/carnitine transporters (OCTNs) 1 and 2, have been shown to play a central role in determining the pharmacokinetics of structurally diverse cationic drugs at the tissue and cellular level. 30 The corticosteroid-sensitive uptake of norepinephrine by OCT3 in vascular muscle, an action that is likely influencing sympathetic neurogenic tone of air way blood vessel has been describe by Horvath et al.31,32 Most inhaled β2-agonists are cations with positive net charge at physiologic pH and thus likely substrate for OCTs expressed in the airway cell. Another study suggested an active absorption mechanism for cationic drugs in human epithelia, which is accomplished by means of polarized apical expression of the pH-dependent OCTN1 and OCTN2. 33 Since the carrier-mediated disposal of inhaled drugs absorbed into airway tissue is still incompletely understood, Horvath et al, 34 investigated OCT expression and uptake of two long-acting β2-agonists, the cationic formoterol and the lipophilic salmeterol, by bronchial and vascular smooth muscle cells. Furthermore, since corticosteroids can potentiate β2-agonists’ actions by inhibiting drug disposal in the airway, the corticosteroid sensitivity of β2-agonists uptake was also assessed. The authors concluded that addition of ICS, in deed, enhances the β2-agonist effects, when combined. In other words, the combination of LABAs and ICS is more effective in the treatment of obstructive lung disease than either drug alone. The proposed mechanism is expected to increase the tissue concentration of a cationic β2-agonist by inhibiting its local disposal on the one hand and by delaying the vascular clearance by causing vasoconstriction through diminished local disposal of norepinephrine on the other hand. This was the first time the immediate interplay of corticosteroids and long-acting β2-agonists was demonstrated in vitro. Furthermore, it was shown that this beneficial interaction could only apply to cationic β2-agonists (e.g. formoterol) but not to lipophilic ones (e.g. salmeterol). 34
Mast cells play a key role in exacerbations of asthma through the release of bronchoconstrictor mediators, such as cysteinyl-leukotrienes, prostaglandins and histamine. Human lung mast cells express β2-adrenoceptors, although in rather low density, and β2-agonists inhibit the release of bronchoconstrictor mediators, such as cysteinyl-leukotreines histamine. 35 This can be demonstrated in asthmatic patients in-vivo by the greater protective effect of β2-agonists against adenosine-induced brochoconstriction, which is mast cell dependent, compared to direct bronchoconstrictors, such as histamine or methacholine. 36
A corticosteroid has been shown to actually reverse the tolerance to β2-agonists in mast cells in vitro, by Chong et al. 37 This suggests that regular treatment with inhaled corticosteroid can reverse and prevent the tolerance to the mast cells stabilizing effects of β2-agonists. This may be an important mechanism in reducing asthma exacerbation, as indicated in the Formoterol and Corticosteroid Establishing Therapy (FACET) studies. 38
Pharmacokinetics
When pharmaceutical agents with known efficacy and tolerability are combined in a new formulation, it is necessary to investigate possible interactions of the components. The first study to explore the pharmacokinetics of budesonide (BUD) and formoterol (FF) administered together in one pressurized metered-dose inhaler (pMDI) in healthy adults was conducted by Eklund and colleagues. 39 They investigated the pharmacokinetics of BUD and FF using a series of single-drug and combination inhalers in healthy subjects, in four open-label, randomized and cross-over studies.
The first three studies were single-dose:
Study-I, included eight inhalations of budesonide pMDI (160 μg), formoterol DPI (4.5 μg, Oxis® Turbuhaler®), budesonide pMDI (160 μg) + formoterol DPI (4.5 μg).
Study-II, included eight inhalations of budesonide/ formoterol pMDI (160/4.5μg) (Symbicort®), budesonide pMDI (160 μg) + formoterol DPI (4.5 μg).
Study-III, included twelve inhalations of budesonide/ formoterol pMDI (40/4.5, 80/4.5 and 160/4.5 μg, Symbicort®).
Study-IV, assessed the steady-state pharmacokinetics of budesonide/formoterol pMDI (160/4.5 μg, Symbicort®) with two and four inhalations twice daily for 5 days and 4-inhalations, single-dose.
Study-I was designed to investigate the pharmacokinetics of interaction of the two drugs when administered simultaneously compared with each product administered alone.
Study-II was designed to investigate the relative systemic bioavailability of the two drugs when inhaled from the budesonide/formoterol pMDI compared with budesonide pMDI + formoterol DPI (concomitant administration) and to compare the relative plasma and urine sampling when estimating the relative systemic bioavailability of FF.
Study-III investigated the dose proportionality of BUD, measured as systemic bioavailability, and the relative systemic bioavailability of FF when administered via three different formulation strengths of budesonide/formoterol pMDI.
Study-IV was designed to investigate; (i) the dose proportionality of BUD and FF, measured as systemic bioavailability, when inhaled from budesonide/ formoterol pMDI at two different doses for 5 days and (ii) the relationship between single- and repeated-dose PK of BUD and FF when inhaled from budesonide/ formoterol pMDI.
In study-I, BUD and FF did not show any difference in any PK parameters when administered concomitantly (budesonide pMDI + formoterol DPI) or individually as mono-components (BUD or FF). For AUC0-∞ and Cmax, the 90% CIs for the mean treatment ratios were within the established bioequivalence limits of 80% to 125%.
In study-II, the mean plasma concentration versus time curves for BUD and FF for each treatment showed 39 that for BUD, the AUC0-∞ and Cmax ratios comparing BUD/FF with BUD + FF (concomitant) were close to 100%, with 90% CIs for the mean treatment ratios. The budesonide T1/2 was similar for both treatments.
Plasma concentration of FF were slightly lower after treatment with BUD/FF than after BUD + FF (concomitant) (AUC: 82.2%, Cmax: 92.9%; combo/ mono). Formoterol T1/2 was similar for both treatments. 39 The estimated relative systemic bioavailability for FF, comparing budesonide/formoterol pMDI to budesonide pMDI + formoterol DPI (concomitant), was 81.5% based on Ae (in urine), that was similar to the estimated systemic bioavailability from plasma sampling (AUC0-∞, 82.2%).
In study-III (PK dose proportionality), the mean plasma concentration versus time curves for BUD and FF for each treatment showed 39 that the dose-adjusted budesonide AUC0-∞ and Cmax values decreased with increased nominal BUD dose. The 12 inhalations of BUD/FF 80/4.5 μg resulted in approximately 10% lower values than the12 inhalations of BUD/FF 40/4.5 μg, and the 12 inhalations of BUD/FF 160/4.5 μg resulted in 10 to 15% lower values than the 12 inhalations of BUD/FF 80/4.5 μg. The budesonide T1/2 was similar for the three treatments.
The relative systemic bioavailability for formoterol was comparable 39 when administered via the three different formulation strengths of budesonide/formoterol pMDI. The T1/2 for formoterol was similar among the three treatments.
In study-IV, the mean plasma concentration versus time curves for BUD and FF for each treatment showed 39 that for BUD, the dose-adjusted AUC0–12 h and Cmax ratios comparing 4 vs. 2 inhalations twice daily for 5 days were close to the expected 100%, with the 90% CIs, indicating dose proportionality. Repeated dosing resulted in higher plasma concentration of BUD than the corresponding single dose as measured by AUC (AUC0–12 h for repeated dosing and AUC0-∞ for single dosing) and Cmax. The Rac for BUD, computed as the ratios of AUC0–12 h for repeated dosing over the AUC0–12 h for single dosing was 132.3%. There was no meaningful difference in T1/2 between the single and repeated dosing.
For formoterol, the dose-adjusted AUC0–12 h and Cmax ratios, expressed as percentages, comparing 4 vs. 2 inhalations twice daily for 5 days were higher (117.6% and 109.7%, respectively). 39 No meaningful differences were found between the two repeated-dosing treatments for T1/2. The repeated dosing resulted in higher plasma concentrations of FF than the corresponding single dose as measured by AUC (AUC0–12 h for repeated dosing and AUC0-∞ for single dosing) and Cmax. The Rac for FF, calculated as the ratios of AUC0–12 h for repeated dosing over the AUC0–12 h for single dosing was 117.4%. The T1/2 for the repeated dosing was relatively larger compared to the single dosing (131.2%).
Similarity of pharmacokinetic parameters for BUD and FF administered simultaneously with separate inhalers and as individual monocomponents in study-I, indicates that there is no pharmacokinetic interaction between BUD and FF. These results also suggest consistent dose delivery from the investigated drug formulations on the different experimental days. The lack of pharmacokinetic interactions between the two drugs is consistent with a previous study on budesonide/formoterol DPI. 40 These findings by Eklund complement those reported by Cazzola, 41 in which improvements in lung function were observed with formoterol/budesonide DPI compared with formoterol DPI alone without a concomitant modification of heart rate, indicating a lack of interaction between BUD and FF, translating to a systemic effects.
Study-II demonstrated that systemic exposure to BUD from the combination pMDI was nearly identical to that from the budesonide pMDI, which had the same formulation except for the FF presence. The systemic exposure (AUC) to FF from combination pMDI was about 20% lower than from the formoterol DPI.
Pharmacokinetic dose proportionality after treatment with budesonide/formoterol pMDI was examined in studies-III and IV. In study-III, the formulation strength of BUD was varied, while the FF dose was kept constant. For BUD, the 90% CI for the treatment comparison ratios of the dose-adjusted AUC0-∞ were within the established bioequivalence limits for adjacent doses (12 inhalations of 40/4.5 μg vs. 80/4.5 μg or 12 inhalations of 80/4.5 μg vs. 160/4.5 μg), indicating that the plasma concentration of BUD changed in proportion with the nominal delivered dose for two adjacent formulation strengths. The AUC0-∞ of FF was similar across the three BUD formulation strengths, with 90% CIs of the mean treatment comparison ratios for AUC0-∞ and Cmax within established bioequivalence limits. These data indicate that switching between formulation strengths with varying doses of BUD has no effect on the plasma concentration of FF.
In study-IV, the AUC0–12 h and Cmax ratios data for BUD demonstrate that an increase in dose of budesonide/formoterol pMDI from two to four inhalations would increase the systemic exposure to BUD proportional to the dose. For FF, the 90% CI for the dose-adjusted AUC0–12 h ratios (four vs. two inhalations) fell just outside the bioequivalence limits (25.1%), but Cmax fell within the bioequivalence limits. Study-IV also compared single and repeated dosing with budesonide/formoterol pMDI. Increased systemic exposure with repeated dosing was reflected in Rac (ratio of AUCs0–12 h), which was 132% for budesonide and 177% for formoterol.
In summary, the four studies demonstrated that there were no pharmacokinetic interactions between BUD and FF when delivered via one inhaler or individually as monocomponents. Additionally, systemic exposure to BUD from the combination pMDI was similar to that from budesonide pMDI, and the systemic exposure after an increase in BUD dose was well tolerated. The systemic exposure to FF from combination pMDI was about 20% lower than that from formoterol pMDI, as mono. The studies also indicate comparable bioavailability of FF when administered via three different formulation strengths of budesonide/formoterol pMDI. Lastly, the studies suggest that an increase in dose of BUD/FF from two to four would result in an increase in systemic exposure proportional to dose.
Safety and Efficacy
Concurrent ICS and LABA therapies vs. individual drug therapies
It has been shown that adding a long-acting β-agonist (LABA) to a low dose inhaled corticosteroids (ICS) results in better asthma control than increasing the dose of ICS. 38 To investigate if this was due to an additional reduction of airway inflammation, Overbeek et al 42 studied the effect of 8-weeks treatment with 100 μg budesonide bid (BUD200) compared with 100 μg BUD bid (BUD100) plus 12 μg formoterol bid (BUD200 + FF), followed by another 8-weeks of treatment with 400 μg BUD bid with and without FF on the markers of inflammation in bronchial biopsy specimens and exhaled air (eA). Prior to the run-in period, patients used a mean dose of 520 μg ICSs daily. Increasing BUD200 to BUD800 led to a further significant increase in FEV1, whereas increasing BUD200 + FF to BUD800 + FF did not produce a further significant change in FEV1. Exhaled nitric oxide (eNO) decreased significantly from baseline in both (BUD200) and (BUD200 + FF) groups after 8 weeks of treatment, with no detectable significant difference between the two. Also, increasing the ICS to 800 μg resulted in a significant decrease in eNO compared with the BUD200 group, while there was no such effect detectable in the (BUD200 + FF) group. In the biopsy evaluation, the epithelium and the major basic protein (MBP) was significantly reduced in the (BUD200) group after 8-weeks of treatment, while this was not the case in the (BUD200 + FF) group. Increasing the ICS dose to 800 μg did not significantly change MBP in either treatment groups. With respect to mast cells, (BUD200) resulted in a significant reduction of tryptase in the epithelium compared to baseline, but hardly affected tryptase in the subepithelium. (BUD200 + FF) did not show any significant reduction in tryptase in both the epithelium and subepithelium compared with baseline. Additionally, no significant differences between treatments were detected for any of the variables, or after increasing the ICS dose to 800 μg. It was concluded the treatment with BUD at low dose already had significant anti-inflammatory effects in the bronchial mucosa of patients with mild asthma. As a result, no significant additional anti-inflammatory effects on the inflammatory markers analyzed could be demonstrated whether by adding formoterol or by increasing the dose of BUD.
In a similar study, the effect of adding a LABA, formoterol, to low doses of an inhaled corticosteroid, BUD, was assessed for a year in subjects with mild persistent asthma. 43 Group A, with 698 corticosteroid free patients was assigned to twice daily treatment with 100 μg BUD, 100 μg BUD + 4.5 μg FF, or placebo, while Group B, with 1,272 corticosteroidtreated patients was assigned to twice daily treatment with 100 μg BUD, 100 μg BUD + 4.5 μg FF, 200 μg BUD, or 200 μg BUD + 4.5 μg FF. The main outcome variables were time to the first severe asthma exacerbation and poorly controlled asthma days. In group A, BUD alone reduced the risk for severe exacerbation by 60% and the poorly controlled days by 48%. In this group, adding FF increased the lung function with no change in other end points. By contrast, in group B, adding FF reduced the risk for first severe exacerbation by 43% and the poorly controlled days by 30%. The results of this study indicated that in corticosteroid-free patients when adding low dose inhaled budesonide alone reduced the exacerbations and improved asthma control. However, in patients already receiving ICS, doubling the BUD dose produced little clinical benefit. Addition of FF showed greater efficacy with regards to the time of the first severe asthma exacerbation, the rate of asthma exacerbations, and the number of poorly controlled asthma days. The doses of inhaled BUD used in this study were small and the long-term adverse effects were minimal. The two main adverse effects with the long-term use of ICS, growth in children and bone mineral density in adults, do, however, appear to be relevant when low doses of ICS are used for asthma management.44,45 These results are consistent with those reported by Overbeek 42 based on the inflammation marker.
There has been emerging concerns that the regular use of LABAs may potentially mask an increase in the underlying inflammatory process in patients who have been stepped down to a lower maintenance dose of ICS. 46 To address this concern, the effect of once-daily combination therapy on surrogate inflammatory markers was evaluated by Aziz et al. 47 Patients with atopic persistent asthma were evaluated in a randomized study with 1-week placebo washout period, comparing the following once-daily nighttime treatments: (1) FF, 12 μg and 24 μg; or (2) BUD, 400 μg and 800 μg; or (3) FF 12 μg + BUD 400 μg and FF 24 μg + BUD 800 μg, each for 2 weeks. The inflammatory markers, adenosine monophosphate (AMP) bronchial challenge, exhaled nitric oxide (eNO), and serum eosinophilic cationic protein (ECP) were evaluated at 12 h post-dosing after administration of each placebo and after 4 weeks of each treatment. The results of AMP challenge (provocative concentration causing a 20% drop in FEV1) at 4 weeks showed significant (p < 0.05) improvement after patients had received all active treatments compared to placebo (20 mg/ml) with FF + BUD at 261 mg/ml being superior (p < 0.05) to FF alone at 82 mg/ml, but not to BUD at 201 mg/ml. eNO and ECP showed significant reductions (p < 0.05) compared to placebo with FF + BUD or BUD alone, but not with FF alone. The combination therapy was associated with optimal patient preference and compliance with the rank order of FF + BUD > FF > BUD; p < 0.0005. The combination therapy was also associated with the highest peak expiratory flow (PEF), and the lowest rescue inhaler usage. It was concluded that patients preferred once-daily combination therapy, while this had no greater effect on inflammatory markers than therapy with budesonide alone. Further, FF had no anti-inflammatory activity but exhibited brochoprotection. This emphasized the importance of optimizing anti-inflammatory control with ICS before considering adding a regular LABA.
These studies show that combination therapy with ICS and LABA, or treatment with high doses of ICS, can improve asthma control when low-dose ICS is not sufficient. However, it is not clear which of these treatment options is more effective in sustaining asthma control. In 2008, O'Byrne et al 48 evaluated the effect of increasing ICS versus adding LABA on time spent with well-controlled asthma or poorly controlled asthma, by a post hoc analysis of Formoterol and Corticosteroid Establishing Therapy (FACET) study, which compared a four-fold increase in BUD dose with and without FF. Their results showed that the time with well-controlled asthma was improved by 19% (p = 0.017) by adding FF 24 μg/d to BUD 200 μg/d compared to 2% (p = 0.76) with BUD 800 μg/d alone. The time with well-controlled asthma was further improved by 29% (p < 0.001) by adding FF to BUD 800 μg/d. The time with poorly controlled asthma was significantly reduced by the same interventions by 43%, 22% and 50%, respectively. Adding FF to BUD was significantly more effective in increasing time with well-controlled asthma when compared to increasing BUD dose by four-folds. O'Byrne and his co-workers concluded that the addition of FF to low dose BUD increases the probability of well-controlled asthma compared to a substantial increase in the dose of ICS.
Even after all the benefits achieved by the common practices in treating asthma patients with inhaled corticosteroids or ICS plus LABAs, these approaches still have encountered inadequacies. As an example, in a large study the attitude and actions of over 3400 asthma patients aged 16 and over were examined in eleven countries. 49 Patients were prescribed regular maintenance therapy with ICS or ICS plus LABA. They observed that patients receiving regular maintenance therapy still had high levels of inadequately controlled asthma. The study also found that patients recognized deteriorating asthma control and adjusted their medication during episodes of worsening. However, they often adjusted treatment in an inappropriate manner, which represented a window of missed opportunity.
Single-inhaler Combination Therapies
Fixed-dose BUD/FF combination
Effective control of asthma and maintenance of optimal lung function are critical for the long-term management of patients with persistent asthma. Use of inhaled glucocorticosteroids as first-line of treatment for the management of patients with all but the intermittent asthma is already established.3,50 Increasing the dose of ICS or addition of LABA to low-dose ICS have been the therapeutic options. The first efficacy and safety study of a new single inhaler containing budesonide and formoterol (Symbicort® Turbohaler®, AstraZeneca, Lund Sweden) was conducted in 2001, 51 where, BUD/FF and the concurrent administration of BUD and FF in two separate inhaler were compared with BUD alone in patients with symptomatic asthma despite previous regular treatment with ICS. This study was the first to demonstrate that BUD/FF via single inhaler was more effective than BUD alone in patients whose asthma was not previously fully controlled by ICS treatment alone. The effect was evident in lung function, symptom scores and use of rescue medication. Additionally, it was well tolerated as the concurrent treatment with the mono products, BUD plus FF via separate inhalers. Furthermore, during the first 30 days of treatment, therapy with BUD/FF showed more rapid improvement in lung function, reduction in symptom scores and reduction in rescue-medication usage compared with the separate inhaler therapy.
The improvements attained in the 12-weeks study by BUD/FF via single inhaler, were similar to those achieved over 1 year period in the study conducted by Pauwels et al, 38 where inhaled FF was added to high or low dose BUD, individually. In later studies, the efficacy and safety of low-dose BUD/FF 80/4.5 μg, bid in a single inhaler (Symbicort®) was compared with an increased dose of BUD 200 μg bid, in adult patients with mild to moderate asthma that were not fully controlled on low dose of ICS alone. 52 In this randomized study, 432 patients aged 18 to 78 years completed a 12-week study. The increase in mean morning peak expiratory flow (PEF), the primary efficacy measure, was significantly higher for BUD/FF compared with the BUD alone (16.5 L/min vs. 7.3 L/min, p = 0.002), so was the evening PEF (p < 0.001). Additionally, the percent of symptom-free days and asthma-control days were significantly improved in the BUD/FF group. Relative risk of an asthma exacerbation in the BUD/FF group decreased by 26% (p = 0.02), compared to BUD alone and the adverse effects were comparable between the two treatment groups.
In another study, the long term safety, efficacy and patient preference of BUD/FF (Symbicort®) in a single inhaler in treatment of asthma were compared with concurrent use of individual BUD and FF inhaler. 53 After a 12-month treatment period, there was no clinically important differences between the single inhaler product and BUD plus FF administration via separate inhalers in adverse events, vital signs, ECG or laboratory measurements. BUD/FF was also at least as effective as BUD plus FF in improving the lung function, asthma control and health-related quality of life throughout the length of the study. However, there were significantly fewer patients who withdrew in the BUD/FF group compared with the BUD plus FF group (5.5% vs. 14.6%, p = 0.005). This was partly attributed to the greater simplicity and convenience of single inhaler therapy compared with two-inhaler therapy resulting in patient adherence to treatment. Furthermore, health-economic analysis of the data in this study demonstrated that long-term treatment with BUD/FF was indeed associated with a significantly lower average annual cost of treatment compared with BUD plus FF (8,914 vs. 10,509, Sweden Kronor).
In evaluation of long-term safety and asthma control with regular use of LABA with ICS, a study was conducted by comparing the use of high dose BUD/FF using pressurized metered-dosed inhaler (pMDI) {BUD/FF hydrofluoroalkane (HFA) pMDI; Symbicort Inhalation Aerosol, AstroZeneca LP, Wilmington, DE} vs. HFA pMDI budesonide in asthma patients. 54 In this 52-week study, 702 patients ≥12 years of age with moderate to severe persistent asthma previously receiving ICS, were randomized into three groups to receive budesonide/formoterol pMDI (640/18 μg) bid, budesonide/formoterol pMDI (320/9 μg) bid or budesonide pMDI (640 μg) bid. Across the range of assessments, including chemistry, hematology, ECG, Holter monitor, and hypothalamic-pituitary-adrenal axis assessment, budesonide/formoterol pMDI 640/18 μg bid and 320/9 μg bid showed acceptable safety and adverse effect (AE) profiles relative to budesonide pMDI 640 μg bid alone, with no significant or unexpected patterns of abnormalities observed for up to 1 year of treatment. In this regard, all the three treatments were concluded to be well tolerated. Treatment with BUD/FF in a single pMDI was more effective than treatment with budesonide pMDI alone in improving the lung function and measures of symptom control. In addition to improvements in diary and spirometry-based measures of asthma control, budesonide/formoterol pMDI was associated with a lower incidence of asthma exacerbations, which resulted in a significant reductions in systemic corticosteroid use compared to budesonide pMDI alone. Overall, the findings from this study was reassuring and supported the long-term use of budesonide/formoterol pMDI dose (320/9 μg bid) as maintenance therapy in adolescents and adults with moderate to severe persistent asthma and suggest minimal increased safety risks when used at double the labeled dose (640/18 μg bid) in this patient population.
SMART-treatment
In a novel asthma management strategy, BUD/FF (Symbicort®) in a single inhaler for both maintenance therapy and symptom relief, known as SMART, was compared with higher dose of BUD plus as-needed terbutaline. 55 It was a 6 month study with 697 patients with mild-to-moderate asthma, 11 to 79 years of age. Patients were randomized to a regiment of either BUD/FF (80/4.5 μg), two inhalations q.d in the evening plus BUD/FF as needed (for both maintenance and relief) or double the dose of BUD (160 μg, 2 inhalations q.d in the evening) plus terbutaline (0.4 mg as needed). All the drugs were administered via dry powder inhalers (DPI) that dispensed the drugs as delivered dose rather than metered dose. Patients were allowed to receive a maximum of 10 as-needed inhalation of either BUD/FF or terbutaline, each day. As a result, they had a total daily dose of BUD/FF up to 960/54 μg for both maintenance and relief. In the study, the rate of a severe exacerbation requiring medical intervention was reduced by 76% among the patients receiving the BUD/FF for both maintenance and relief compared with the higher-dose budesonide group (0.08 event/patient-year vs. 0.35, respectively). More notably, patients in the BUD/FF group had a significant reduction (about 90%) in hospitalization treatments (1 event vs. 10 events in the higher-dose BUD group). Furthermore, patients receiving BUD/FF for both maintenance and relief used less as-needed medication compared to those receiving the traditional therapy with BUD plus separate as-needed reliever medication. Overall, 22 patients in the BUD/FF group used more than 8 inhalations of reliever medication on any 1 day during the treatment period, compared with 53 patients in the BUD group. In this subgroup, a total of 41 severe exacerbations were recorded, of which only 4 occurred in the BUD/FF group and the rest in the BUD group (1 to 10 ratio). These results strongly suggest that additional as-needed inhalation of BUD/FF may prevent worsening asthma control from developing into an exacerbation compared with the use of traditional reliever medications, such as salbutamol or terbutaline. Furthermore, as patients with higher as-needed medication use are generally considered to be more at risk for severe exacerbations, the findings of this study showed that the tolerance to the LABA did not develop in the SMART group.
In terms of symptom-free days and asthma-control days, patients on SMART regimen had significantly lower asthma symptoms scores (p < 0.001) as well as significantly more symptom-free days (p < 0.01) and asthma-control days (p < 0.01). Figuratively, the mean difference in asthma-controlled days corresponded to an extra month per year with total freedom from asthma symptoms and reliever use for patients with the SMART treatment. Safety wise, in the incidence, frequency, and profile of adverse events were similar between treatment groups, both being mild to moderate in intensity and well tolerated. This study demonstrated that low-dose BUD combined with FF in a single inhaler, used for both maintenance and relief symptoms, reduced the incidence of severe exacerbations more effectively compared with the higher maintenance dose of BUD plus-short acting β2-agonist (SABA) for relief. Thus, for some patients who need a maintenance treatment with BUD/FF, the inhaler can also be used as rescue when needed, allowing asthma patients managing their asthma with one inhaler.
To evaluate the importance of the ICS component given as needed medication in the Symbicort as Maintenance and Relieve Therapy (SMART), a 12-month study was conducted 56 with 3394 patients, aged ≥12 years. All patients, with moderate to severe asthma not well controlled on ICS alone, were put on BUD/FF 160/4.5 μg, one inhalation twice daily for maintenance therapy and were randomly assigned to treatment with one of the three reliever medications (1:1:1 ratio) for 12 months: additional inhalation of BUD/FF 160/4.5 μg; FF 4.5 μg (Oxi® Turbohaler®, AsteraZeneca); or terbutaline 0.4 mg (MDI, AsteraZeneca). The primary outcome was to measure the time to first severe exacerbation. Secondary outcomes included total number of exacerbations, number of emergency treatments or hospitalizations, asthma symptom scores; mild exacerbations; FEV1; morning and evening peak expiratory flow (PEF); and reliever medication use. Results of the study showed that the time to first exacerbation was prolonged with BUD/FF for maintenance and relief (SMART) versus BUD/FF plus FF (p = 0.005) or terbutaline (p < 0.0001). The as-needed BUD/FF prolonged the risk of severe exacerbation by 27% vs. FF and by 45% vs. terbutaline. The risk reduction with the as-needed FF vs. terbutaline was 24%. This shows that the BUD component of the BUD/FF combination when used as-needed plays an important role in patients who remain symptomatic despite treatment with combination therapy. The as-needed FF was also seen to provide better exacerbation control compared with terbutaline. Therefore, the reduction in severe exacerbations can be attributed partly to the as-needed formoterol component of the combination, as well.
In a similar study in Canada, Sears et al 57 showed that BUD/FF as maintenance and reliever therapy (SMART) achieved similar or improved clinical control compared with the conventional best practices, with significantly lower total ICS dose and lower cost, while maintaining similar control of eosinophilic inflammation.
SMART compared to other combination therapies
In a 12-months dose-titration study, the effectiveness of BUD/FF for maintenance plus relief with a control group using salmeterol/fluticasone for maintenance plus salbutemol for relief was assessed. 58 In this study 2143 patients with persistent asthma, age ≥12 were randomized for treatment with either BUD/FF (Symbicort®) 160/4.5 μg b.i.d. plus additional inhalation as needed BUD/FF or salmetreol/fluticasone (Seretide® Diskus®; GSK, UK) 50/250 μg b.i.d. plus salbutamul for rescue medication (DPI or pMDI; Ventolin®, GSK). The maintenance plus as-needed BUD/FF prolonged the time to first severe exacerbation compared to salmeterol/fluticasone (25% risk reduction). The total number of severe exacerbations was significantly reduced in the BUD/FF group compared with the salmeterol/fluticasone group (255 vs. 329). Both treatments provided improvement in the symptoms, as needed-use, quality of life and FEV1, with the difference in favor of BUD/FF group for as-needed use (0.58 vs. 0.93 inhalations day-1) and FEV1. The mean ICS dose was similar for both groups (653 μg budesonide.day-1 vs. 583 μg fluticasone. day-1). Both treatments were well tolerated with no notable difference between the groups in the number of severe adverse events. However, a greater number of salmetrol/fluticasone patients withdrew from the study due to asthma vs. BUD/FF patients (11 vs. 3 patients, respectively).
An earlier study had shown that BUD/FF as maintenance and reliever (SMART) is more effective at reducing exacerbations and improving daily asthma control compared with the same maintenance dose of BUD/FF plus as needed SABA. 59 The concern about the SMART approach was that some patients might end up using the combination inhaler too frequently and receive unacceptable high dose of ICS. However, this turned out not be the case as the mean number of additional dose of BUD/FF was only one dose per day and very few patients required high doses. A comparison of the efficacy of SMART with that of a higher maintenance dose of ICS/LABA was needed to answer two important questions: could SMART approach be as effective at reducing exacerbations at higher fixed maintenance dose of combination therapy and would SMART result in a greater day-to-day fluctuations in asthma control? To that effect, Kuna et al 60 conducted a large study, randomizing patients to one of the three treatments groups for 24 weeks: BUD/FF 160/4.5 μg one inhalation bid for maintenance plus additional inhalations as needed (SMART), fixed-dose salmeterol/fluticasone (Seretide/Advair® Evohaler; pMDI, GSK, UK) 25/125 μg two inhalations bid plus terbutaline as reliever medication or BUD/FF 320/9 μg one inhalation bid plus terbutaline. They concluded that compared with a two fold higher fixed maintenance dose of BUD/FF or a corresponding dose of salmeterol/fluticasone plus SABA for relief, the SMART approach reduces the incidence of severe exacerbations and maintains similar daily asthma control at lower overall drug load. With this combination of increased efficacy and simplicity, SMART approach represents a significant improvement over the fixed, twice-daily combinations of higher-dose ICS/LABA, which have until recently been regarded as the most effective way to manage moderate and severe persistent asthma.
Children with asthma, have been treated with fixed-combination therapy (ICS/LABA), without any protection seen against asthma exacerbations.61–63 This might partly be because pediatric asthma is often an episodic disease with periods of few symptoms and near-normal lung function interrupted by periods of symptom increases and exacerbation. 64 Traditional dosing strategy emphasizes on the fixed-dose that is decided based on the history and that is eventually going to lag behind variation in disease activity and carry the risk of over or under treatment. This fixed-dosing treatment paradigm in pediatrics with moderate to severe persistent asthma was challenged by Bisgaard. 65 In his new approach, he emphasizes on the intermittent ICS/LABA use in addition to the regular low-dose use by dynamically titrating the steroid dose to the disease activity. This treatment approach where patients receive ICS/LABA for maintenance and reliever therapy in response to symptoms differs from the traditional treatment regimens, since it involves patients using one inhaler to control and treat their asthma symptoms without the requirement for a separate short-acting rescue inhaler. 65 In this study, 388 children aged 4-11 years with asthma were enrolled in a 12-month trial. Patients were randomized to one of the three treatments: BUD/FF (Symbicort®)) 80/4.5 μg qd plus additional doses as needed (SMART), BUD/FF 80/4.5 μg qd plus terbutaline 0.4 mg for rescue medication (fixed combination) or a 4-fold-higher maintenance dose of BUD 320 μg qd plus terbutaline 0.4 mg for rescue medication (fixed-dose BUD). During the study, the reliever medication was allowed to be used whenever patients judged that it was needed, but not more than eight inhalations in a single day. Overall, 14% of patients receiving the SMART regimen had an exacerbation vs. 38% and 28% of patients in the fixed dose combination and fixed-dose BUD groups, respectively. The SMART regimen significantly prolonged the time to first exacerbation vs. the fixed-dose combination (p < 0.001) and the fixed-dose BUD (p = 0.02). The sudden risk of having an exacerbation was 66% lower with SMART vs. the fixed-dose combination and 51% lower vs. the fixed-dose BUD. The number of adverse events per patient was significantly lower with SMART vs. the fixed-dose combination (p = 0.017); and lower with fixed-dose BUD vs. the fixed-dose combination (p = 0.073). Exacerbations requiring medical intervention occurred in 8% in the SMART, 31% in the fixed combination and 20% in the fixed-dose BUD. This is a significant reduction in instantaneous risk by 75% in the SMART vs. the fixed-dose combination (p < 0.001), and by 60% vs. the fixed-dose BUD (p = 0.016). Similarly, the rate of exacerbations requiring intervention was reduced by 70% to 79% with SMART vs. the two fixed-dose groups.
Safety wise, the SMART regimen was associated with an increased yearly growth rate vs. the fixed-dose BUD. This suggests that the regimen improved asthma control while minimizing systemic effects compared with a higher dose BUD. The SMART regimen was well tolerated compared with both the fixed-dose regimens and also had a potentially more favorable safety profile compared with the 4-fold-higher maintenance dose of budesonide.
In a study that examined whether asthma control improved if patients adjusted the maintenance dose (AMD) of BUD/FF according to the asthma severity compared with the traditional fixed dose (FD) regimens, the patients on budesonide/formoterol AMD had a lower rate of exacerbation over the study: 40% lower vs. salmeterol/fluticasone FD and 32% lower vs. budesonide/formoterol FD. Additionally, budesonide/formoterol AMD patients used less reliever medication in the open extension (0.58 vs. 0.92 occasions/day) for budesonide/formoterol FD, (p = 0.001) and 0.80 occasions/day for salmeterol/fluticasone FD (p = 0.011). Concluding that adjustable maintenance dosing with BUD/FF provides more effective asthma control by reducing exacerbations and reliever medication usage compared with the fixed dose salmeterol/fluticasone. 66 In a different study, comparing the adjustable budesonide/formoterol pMDI and fixed-dose budesonide/formoterol pMDI and fluticasone/salmeterol DPI in asthma patients, Busse et al 67 concluded that the adjustable-dose and fixed-dose budesonide/formoterol pMDI indicated no differences in asthma control or tolerability versus fixed-dose fluticasone/salmetrol DPI.
Cost factor
To evaluate the cost-effectiveness in addition to efficacy, in a recent study similar to Busse, 67 the SMART treatment versus a fixed higher-dose BUD/FF plus as-need terbutaline reliever (FHDBF) and fluticasone/salmeterol plus-as needed terbutaline reliever (FDFS) was evaluated in a large patient population of (>3300) adults and adolescents with asthma. 68 The SMART strategy, proved to be more effective clinically and less expensive over the alternative strategies of the fixed higher dose (by a factor of 2) BUD/FF plus as-needed terbutaline or fixed dose fluticasone/salmeterol plus as-needed terbutaline. Further, in another similar study, 1800 patients with persistent asthma, showed direct cost savings with BUD/FF maintenance and reliever therapy (SMART) compared with the conventional treatment regimens with at least equivalent efficacy. 69
Onset of effect
The onset of bronchodialation has been evaluated for formoterol pMDI 70 and the evidence suggests that ICSs may enhance the bronchodialation with LABA. 71 However, the onset of bronchodialation had not been evaluated for the particular formoterol formulation in the budesonide/formoterol pMDI. It was unclear whether the onset of bronchodialation, as measured by pulmonary function testing corresponds to the patient-perceived onset of effect (OE). To this effect, Kaiser et al 72 reported the results of a secondary end point; the time to onset of clinically significant bronchodialation (defined as a ≥15% improvement in FEV1 after medication administration) and patient-perceived OE of budesonide/formoterol pMDI. In the study, patients were randomized and treated for 12 weeks with one of the following therapies; study-1: budesonide/formoterol pMDI 160/4.5 μg, budesonide pMDI 160 μg, formoterol DPI 4.5 μg, budesonide pMDI 160 μg + formoterol DPI 4.5 μg, each 2 inhalations twice daily and placebo. Study-2: budesonide/formoterol pMDI 80/4.5 μg, budesonide pMDI 80 μg, formoterol DPI 4.5 μg, each 2 inhalation twice daily and placebo. The study data showed that rapid onset of clinically significant bronchodialation and percent of patients perceived an OE were similar for budesonide/formoterol pMDI and formoterol DPI. However, more rapid onset of bronchodialtion and greater percentage of patients who perceived an OE were observed for budesonide/formoterol pMDI vs. budesonide pMDI or placebo in patients with mild to moderate persistent asthma and in patients with moderate to severe persistent asthma. In a separate study by Palmqvist et al, 73 the median time to onset of clinically significant bronchodialation was reported to be related to the FF dose when delivered as a monoproduct via DPI. A shorter time to onset was reported for a 24-μg metered-dose (18-μg delivered dose) of formoterol DPI at 3.6 minute vs. a 12-μg metered-dose (9-μg delivered dose) of formoterol DPI at 12.4 minutes. Compared with this study, the time to onset of clinically significant bronchodialation with budesonide/formoterol pMDI using 9-μg delivered dose for FF at 13 minutes was similar to that observed using the 12-μg metered dose of formoterol DPI, 73 regardless of whether FF was delivered in combination with budesonide or via DPI or pMDI. In short, patients treated with budesonide/formoterol pMDI experienced faster onset of effect and more number of patients perceived their medication working right away compared to patients receiving budesonide pMDI or placebo. Furthermore, time to onset of clinically significant brochodialation and patient-perceived OE of budesonide/formoterol pMDI were comparable with those of formoterol DPI. These results may have clinical implications for optimizing maintenance of asthma control and improving treatment adherence.
Another ICS/LABA combination product that delivers both components in one dry powder inhaler (DPI) is fluticasone/salmeterol (Advair Diskus®, GSK, Research Triangle Park, NC). The early bronchodialotory effects of budesonide/formoterol pMDI and fluticasone/salmeterol DPI and albuterol pMDI were compared by Hampel et al. 74 The two identically designed studies were conducted in patients aged 18 years and older with persistent mild to moderate asthma that were previously treated with ICS. After 2 weeks on twice-daily pMDI 160 μg, patients received a randomized sequence of BUD/FF pMDI 80/4.5 μg x 2 inhalations, fluticasone/salmeterol DPI 250/50 μg x 1 inhalation, albuterol pMDI 90 μg x 2 inhalations, and placebo pMDI (3 to 14-day washout periods). Results showed improvement in FEV1 within 3 minutes and were significantly greater (p < 0.001) after treatment with BUD/FF pMDI compared with fluticasone/salmeterol DPI and was similar to that of abluterol pMDI. Additionally, significantly more number of patients (p < 0.001) treated with BUD/FF achieved a 15% improvement in FEV1 within 15 minutes compared with patients treated with fluticasone/salmetrol and placebo. It was concluded that the early bronchodilatory effects of BUD/FF pMDI were greater compared with fluticasone/salmetrol DPI.
A rapid effect is important to asthma patients, particularly during exacerbations. To compare the onset of bronchodilation and patient-perceived relief from dyspnoea following single-inhaler BUD/FF or salmeterol/fluticasone a model of acute bronchoconstriction was studied by Van der Woude. 75 In this randomized, single-dose, crossover, 27 outpatients with asthma (mean FEV1 90% of predicted normal) were induced bronchoconstriction by methacholine that caused a fall in FEV1 ≥ 30%. Immediately following this induction, patients inhaled BUD/FF (160/4.5 μg, 1 or 2 inhalations; Symbicort® Turbohaler®), salmeterol/fluticasone (50/250 μg; Seretide™ Diskus) or placebo on 4 study days. The lung function and Borg score were assessed for 30 min. During the methacholine-induced provocation (final mean FEV1 62.5% of baseline), the mean Borg score increased by 10-folds (from 0.3 to 3.0 units). Hereafter, the mean FEV1 at 3 min improved significantly more after BUD/FF 1 and 2 inhalations (37 and 38%, respectively) than after salmeterol/fluticasone (23%; p < 0.001) (Fig. 2). The median recovery time to 85% of baseline FEV1 were shorter for BUD/FF (1 or 2 inhalations: 3.3 and 2.8 min, respectively) than salmeterol/fluticasone (8.9 min, p < 0.001) and placebo (>30 min) (Fig. 3). One minute after BUD/FF administration, dyspnoea was significantly reduced (Borg score -0.86 units, both doses) compared to salmeterol/fluticasone (-0.55 units; P < 0.05) and placebo (-0.23 units; p < 0.001) (Fig. 4). It was shown that BUD/FF in single inhaler had a rapid onset of effect, providing effective bronchodialtion within 1 min of inhalation and return to at least 85% of baseline FEV1 within 4 min of inhalation (Fig. 3). Importantly, patients felt the rapid bronchodialation provided by BUD/FF as an immediate relief from dyspnoea as soon as 1 min after inhalation (Fig. 4), concluding the onset of effect was significantly faster with BUD/FF compared with the salmeterol/fluticasone.
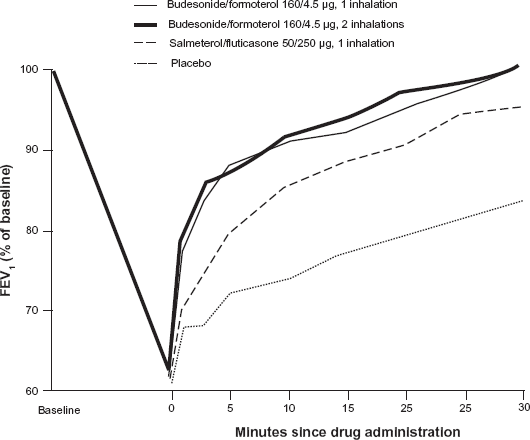
Mean FEV1 as percentage of baseline before and during the first 30 min following inhalation of study drugs after methacholine-induced severe bronchoconstriction.
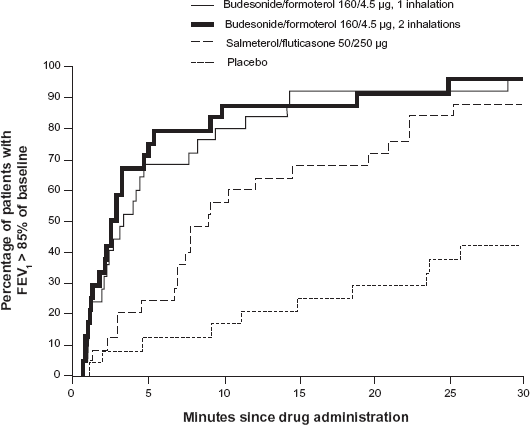
Proportion of patients showing a recovery in FEV1 (a return to 85% of baseline) in the first 30 min after inhalation of study drugs after methacholine-induced severe bronchoconstriction.
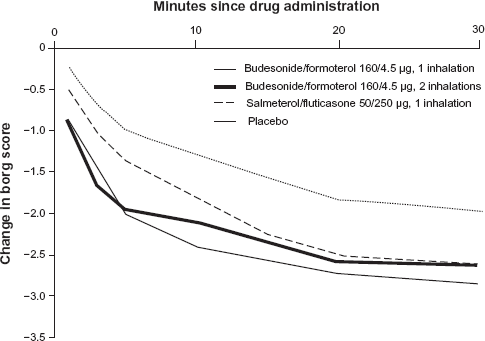
Patient's perception of relief from dyspnoea after inhalation of study drugs. Borg scores represent the change from methacholine challenge peak values.
In a similar study, Palmqvist et al 76 achieved similar results to Van der Woude's study, 75 when they compared the onset of action using combination of BUD/FF (Symbicort® Turbuhaler®) 160/4.5 μg, 2 x 160/4.5 μg and salmeterol/fluticasone (Seretide® Diskus®) 50/250 μg and placebo on patients with mean FEV1 2.541 (range: 1.48–4.28). Figure 5 shows the percent change in FEV1 up to 15 minutes and 3 hours with the combination of BUD/FF (one or two inhalations) (160/4.5 μg) compared with the combination salmeterol/fluticasone (50/250 μg) and placebo. The combination of 1 or 2 inhalations of BUD/FF showed a faster onset of action than salmeterol/fluticasone, both evaluated as the mean FEV1 at 3 min or as average FEV1 from 0 to 15 min. Furthermore, BUD/FF at both doses resulted in a higher FEV1 than salmeterol/fluticasone at 3 h. Concluding, the combination of BUD/FF has a faster onset of action compared with salmeterol/fluticasone.
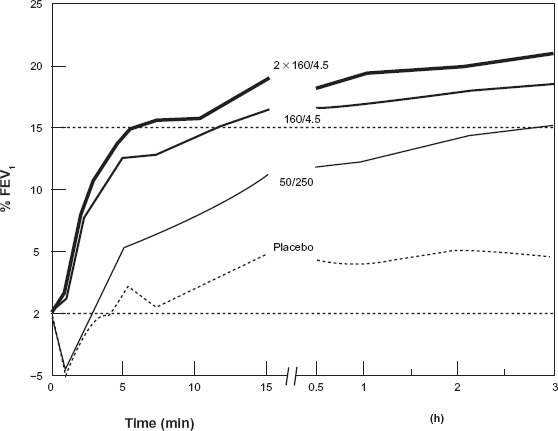
Percent change in FEV1 up to 15 minutes and 3 hours with the combination of budesonide formoterol (one or two inhalations) (160/4.5 μg) compared with the combination salmeterol/fluticasone (50/250 μg) and placebo.
BUD/FF in a single inhaler as rescue medication
The first study to compare the use of BUD/FF for relief of acute asthma brochoconstriction with that of salbutamol, which is widely used for day-to-day reliever medication and in hospital/emergency room management of exacerbations, was performed by Balanag in 2006. 77 The study showed that BUD/FF was well tolerated and provided similar rapid and effective bronchodilation as salbutamol in adult and adolescents in patients seeking medical attention for acute asthma.
In a large study performed in an emergency room setting, Boonsawat et al 78 showed that high-dose formoterol was as rapid and effective as high-dose salbutamol in reversing the bronchoconstriction in patients with severe asthma, but formoterol produced greater improvement in lung function over 4 hours. This prompted him to suggest asthma patients experiencing acute symptoms may use their β2-agonist reliever medication repeatedly. However, there are evidences indicating that this can result in downregulation of β2-receptors and the consequent relative refractoriness to the bronchodilatory effects of this class of drugs.79,80 On the other hand, high doses of ICS have reported to upregulate these receptors and restore β2-agonist responsiveness. 81 Regarding this argument, Bateman and coworkers 82 showed that BUD/FF (Symbicort®) and formoterol (Oxis®) provided similarly rapid relief of acute brochoconstriction in patients with acute asthma that were specifically showing refractoriness (mean reversibility 2% of predicted normal after inhalation of 400 μg) to short acting β2-agonists such as salbutamol.
Safety and tolerability with high-dose combination therapy
The acute tolerability of high dose of BUD/FF was assessed and compared with placebo, was studied for the first time in asthma patients. 83 It was a randomized placebo-controlled study where patients, initially received maintenance treatment for up to 8 weeks with two inhalation of BUD/FF (Symbicort® Turbohaler®) 160/4.5 μg twice daily (total daily dose 640/18 μg). There were three tolerability test days during the study: the first was 2–4 weeks after start of maintenance treatment, and the remaining two days were each 1-4 weeks after the previous test. Administration of a high dose test treatment was started immediately after the morning maintenance dose of BUD/FF as following: single inhaler BUD/FF 1600/45 μg; FF 45 μg (Oxis® Turbohaler®, 4.5 μg per inhalation); placebo. Test treatment was given as three dose increments over 1 hour with 30 min in between increments (2 + 4 + 4 inhalations). Thus the total doses on tolerability test days were: BUD/FF test day 1920 μg BUD, 54 μg FF; formoterol test day 320 μg BUD, 54 μg FF; placebo test day 320 μg BUD, 9 μg FF. The overall result of this assessment was that the tolerability of a high dose of BUD/FF in a single inhaler was well tolerated without any clinically important effects on serum K, pulse rate, systolic and diastolic blood pressure, heart rate, QT-interval, QTc, blood glucose levels and plasma lactate levels. In brief, the authors concluded that BUD/FF in a single dose inhaler is well tolerated at high doses and can be used by asthma patients for relief of their asthma symptoms. 83
Quality of patients’ health and satisfaction
In typical studies of treatment of asthma conditions, the changes in clinical parameters may not necessarily correlate with the changes in how patients feel or function. Studies have shown that there is poor correlation between clinically meaningful improvements in lung function with improvements in patients’ health-related quality of life (HRQL) when treating asthma or COPD.84,85 In 2008, Murphy et al 86 published an article based on a large study conducted in patients with mild-to-moderate persistent asthma and demonstrated there is significantly better pulmonary function with twice-daily treatment with budesonide/formoterol pMDI compared with its monocomponenets. 87 It reported the effects of BUD and FF delivered via one pressurized metered-dose inhaler (pMDI) on patient-reported outcome (PRO) that included health-related quality of life (HRQL), i.e. quality of sleep, satisfaction with treatment and asthma control compared with placebo in patients previously treated with low-to-medium doses of ICS. It described, also, the relative contributions of BUD and FF to budesonide/formoterol pMDI combination therapy. An important conclusion was that the budesonide/formoterol pMDI lead to significant improvements from baseline in HRQL and sleep quality and greater patient satisfaction with asthma medication at the end of treatment when compared with placebo.
Budesonide/formoterol pMDI and DPI formulations
In comparing the two formulations of budesonide/formoterol combination, the long-term safety and efficacy profile of BUD/FF in pressurized metered-dose inhaler (pMDI, hydrofluoroalcane, HFA) formulation was compared with that of BUD/FF in dry powder inhaler (DPI, Turbohaler®). 88 It was a 52-week, randomized, open, parallel-group study including patients aged 12 years and above with asthma with FEV1 ≥ 50% of normal. All these patients used ICS (400–1200 μg/day) and needed additional short-acting β2-agonist therapy. Patients received BUD/FF pMDI or DPI 160/4.5 μg, two inhalations twice daily. Results indicated there was no clinically significant difference between treatments, incidence or severity of adverse events or laboratory parameters. The number of patients that experienced adverse events was comparable in the pMDI [332/446 (70%)] and DPI [175/227 (77%)] groups. The percentage of patients that discontinued as a result of adverse events was low in both groups [pMDI 12/446 (3%),DPI 2/227 (1%)]. Lung function was improved to similar extent in both groups and there was no detectable difference in time to first severe asthma exacerbation. The novel HFA pMDI formulation of BUD/FF proved to be equally well tolerated and effective treatment for adults and adolescents with asthma as the budesonide/formoterol DPI. In a different study, Busse and et al 67 had showed similar comparability between the two BUD/FF formulations.
Conclusion
The long term use of budesonide/formoterol in single inhaler (pMDI) (Symbicort®) as maintenance in adults and adolescents provided better lung function compared with high dose budesonide (pMDI) alone. The long-term use of BUD/FF compared to concurrent use of individual BUD and FF has shown comparable results in lung function improvement, asthma control and health-related quality of life with significantly fewer patients withdrawing in the BUD/FF group and at a lower cost. Similar studies have indicated that BUD/FF in a single inhaler to be more effective in treating asthma patients compared with concurrent individual drugs used. Furthermore, the budesonide/formoterol combination in single inhaler used as maintenance and reliever (SMART) has proven to be more effective than high dose BUD alone or in combination with a SABA as relief medication in improving the lung function, system control, prolonging the time to first severe exacerbation and reducing the frequency of severe exacerbations resulting in less exposure to systemic corticosteroids or as needed medication and less overall financial cost to the patients. These results strongly suggest that the as-needed BUD/FF may prevent asthma worsening into an exacerbation compared to the use of traditional reliever medications such as salbutamol. Comparing the SMART approach with other combination drugs in single inhaler such as fluticasone/salmeterol + a SABA, provided significantly better results in the time to first severe exacerbation and the frequency of the exacerbation episodes. Eventhough, both treatments provided improvement in the symptoms, as needed-use and FEV1, but the difference was in favor of the SMART group and less number of SMART patients withdrew due to asthma.
The BUD/FF onset of effect (OE) and percent of patient perceived OE were compared with FF or albutereol, while the observed and perceived OE was significantly faster in BUD/FF compared with BUD or placebo. This OE of BUD/FF has been shown to considerably faster compare with other combinations such as fluticasone/salmeterol (Advair®). Similarly, other studies have shown the comparable rapid bronchodialation capability of BUD/FF to other fast acting β-agonist such a salbutemol for relief in adult and adolescent patients with acute asthma, supporting the appropriateness of its use as a rescue medication.
Many of these studies have shown that budesonide/formoterol in single dose inhaler is well tolerated and can safely be used by asthma patients for relief and maintenance of their asthma symptoms, even at high doses. Studies have shown that budesonide/formoterol pMDI (pressurized metered-dose inhaler) formulation and budesonide/formoterol DPI (dry powder inhalation) formulation are equally well tolerated and effective treatment for adults and adolescents with asthma. Pharmacokinetically, there has been no evidence of interaction between budesonide and formoterol when used in single inhaler compared with the individual components. Considering the efficacy, rapid onset of effect, simplicity of using one medication both as maintenance and reliever, effective and sustained improvement of quality of patient life, less or comparable adverse effects and cost effectiveness, one can consider a greater tendency for asthma patients’ preference in using budesonide and formoterol in a single inhaler.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgement
Author would like to thank Dr. Samuel Akapo for his referral and valuable inputs during the preparation of the review.
