Abstract
The treatment of persistent asthma has been aided by the recent approval of new medications. The combined inhaled corticosteroid (ICS)/long-acting β2 agonist (LABA) powder inhaler fluticasone furoate (FF)/vilanterol trifenatate (VI) is one of these new agents, which was recently approved as a maintenance therapy for persistent asthma. This once-daily ICS/LABA inhaler has previously been approved and used in chronic obstructive pulmonary disease as a maintenance therapy. Both FF and VI individually have been shown to have efficacy in the treatment of persistent asthma; the combination of FF/VI at the dose of 100/25 μg daily improves trough peak expiratory flows and forced expiratory volume in 1 s. It also reduces the frequency of asthma exacerbations in patients with persistent asthma. The once-daily dosing is well tolerated, with limited clinically significant adverse events; the once-daily inhaled dosing regimen should also improve medication adherence. The data supporting the use of the FF/VI inhaler in persistent asthma are reviewed. The dry powder inhaler of FF/VI (100/25 μg) is an effective and well tolerated once-daily maintenance treatment for patients with persistent asthma.
Keywords
Background
The syndrome of asthma is a variable clinical disorder associated with chronic airway inflammation, recurrent symptoms including wheezing, coughing, chest tightness, dyspnea and airway hyperresponsiveness (AHR). Asthma differs from the syndrome of chronic obstructive pulmonary disease (COPD) based on age of onset, tobacco/biomass exposure, variable presence of chronic bronchitis symptoms and the potential presence of emphysema with nonreversible or partially reversible airway obstruction. The asthma–COPD overlap syndrome (ACOS) recognizes a phenotype of patients with obstructive airway disease that overlaps the syndromes of asthma and COPD [Louie et al. 2013; GINA, 2015; GOLD, 2015]. The wide variety of asthma syndrome phenotypes has provided treatment challenges. Despite many drug categories available for the treatment of asthma, it often remains difficult for both the clinician and patient, and expensive, with an incremental direct cost of $3259 (in 2009 US dollars) per person per year [Barnett and Nurmagambetov, 2011]. The 2015 updated Global Initiative for Asthma (GINA) provides a comprehensive guideline for the recognition and treatment of the various phenotypes of asthma [GINA, 2015]. This paper reviews the literature supporting the use of the once-daily, combined dry powder inhaler fluticasone furoate (FF) and vilanterol trifenatate (VI) in the maintenance treatment of asthma.
Medication adherence in chronic diseases
Medication ‘adherence’ has been defined as the extent to which a patient follows medical plans, including taking medications and lifestyle modifications [Shams and Fineman, 2014]. This implies a mutual consent between patient and provider as opposed to medication ‘compliance’ which is more about the patient following the physician’s ‘instructions, prescriptions and proscriptions’ [Shams and Fineman, 2014]. There are many reasons for medication nonadherence and these are summarized in Table 1.
Factors contributing to nonadherence of asthma medications.

Rates of medication nonadherence have ranged from 30% to 70% among patients with asthma, and nonadherence has been associated with poor health outcomes [Lindsay and Heaney, 2013a]. A dosing frequency and medication adherence study in patients with chronic diseases (excluding human immunodeficiency virus, psychiatric illness, cancer and organ transplant) showed that the percentage of adherence rates for all regimens dosed more frequently than once daily were significantly lower compared with once-daily regimens [Coleman et al. 2012]. Compared with once-daily medication adherence, the rates for twice daily, three times daily and four times daily for actual taking of the drug were reduced respectively by −6.7%, −13.5% and −19.2%, and for regimen adherence were reduced respectively by −13.1%, −24.9% and −23.1% [Coleman et al. 2012]. A review of the literature evaluating medication adherence in chronic diseases also found that dose frequency and regimen complexity emerged as important predictors of medication adherence [Ingersoll and Cohen, 2008]. In two studies of patients with asthma, oral montelukast once daily demonstrated better overall medication adherence than twice-daily inhaled corticosteroids (ICS). In the first trial ICS were used less than prescribed on 49.5% of the days in the double-blind (DB) phase and 57.5% less during the open-label (OL) phase compared with 22.5% less during the DB phase and 28.6% less during the OL phase for once-daily oral montelukast (p < 0.0001) [Rand et al. 2007]. The second trial compared twice-daily ICS with once-daily oral montelukast in economically disadvantaged African American youths with asthma [McNally et al. 2009]. Medication adherence was, on average, higher (p < 0.01) with oral once-daily montelukast (40.9%) compared with twice-daily fluticasone propionate (FP) (33.9%). However, after a year of study, significantly low medication adherence rates were seen with both drugs, with only 31% adherence to prescribed doses of once-daily oral montelukast and 23% adherence to twice-daily ICS FP being found [McNally et al. 2009]. Nonadherence to asthma treatment is associated with poor baseline asthma control, increased frequency of exacerbations and asthma-related hospitalizations [Lindsay and Heaney, 2013]. When frequencies of ICS dosing are directly compared in patients with asthma, the use of once-daily mometasone furoate dry powder inhaler measured by an automatic dose counter was significantly better (p < 0.001) than the same drug given twice daily (93.3% versus 89.5%) in a 12-week OL trial in 1233 patients with asthma [Price et al. 2010]. Higher rates of medication adherence were self reported, and health-related quality of life improved by 20% in patients receiving once-daily ICS compared with a 14% improvement in those receiving twice-daily ICS [Price et al. 2010]. Frequency of dosing appears to be an important variable in determining asthma medication adherence. Asthma medication adherence is greater with ICS combined with a long-acting β2 agonist (LABA) than it is with ICS alone [Feehan et al. 2015].
The use of LABAs for asthma
Controversy surrounding the use of LABAs for asthma has existed for some time now. It centers on the role of the use of inhaled LABAs in the treatment of patients with asthma. As early as 1989, the results of a case controlled trial from New Zealand suggested an increase in mortality when the short-acting β2 agonist (SABA) fenoterol was used in patients with asthma [Crane et al. 1989; Spitzer et al., 1992]. Other large studies failed to find increased mortality in patients with asthma over 16 weeks with the LABA salmeterol (SAL) or SABA salbutamol [Castle et al. 1993]. Tolerance and lack of toxicity to the LABA SAL was reported in a small study of patients with mild asthma [Cheung et al. 1992]. In contrast, the SMART Trial (the Salmeterol Multicenter Asthma Research Trial) evaluated the use of the LABA SAL metered-dose inhaler (MDI) twice daily versus placebo MDI in 26,355 patients with asthma [Nelson et al. 2006]. Combined asthma-related deaths or life-threatening experiences showed a small but significant increase in total numbers, 37 with SAL versus 22 with placebo, giving a relative risk (RR) of 1.71 [95% confidence interval (CI) 1.01–2.89] for SAL versus placebo. The majority of the imbalance or negative effect was mostly associated with the African-American subjects compared with the white subjects [Nelson et al. 2006]. This and other study results have generated a ‘black-box’ warning for the use of all LABA agents in the treatment of patients with asthma by the US Food and Drug Administration (FDA). Despite this small increased risk, the use of LABA agents in combination with ICS is included in the current international GINA guidelines for the treatment of adults with persistent asthma symptoms [GINA, 2015]. The concomitant use of an inhaled LABA with an ICS may mitigate the increased risk of LABA use in patients with asthma. It is unclear whether this finding applies to all subpopulations with asthma [Weatherall et al. 2010]. A review of the addition of inhaled LABA to ICS as first-line therapy for persistent asthma in steroid-naive adults and children did not find that combination therapy (LABA/ICS) reduced the risk of asthma exacerbations requiring oral corticosteroids over that with just ICS [Ni Chroinin et al. 2009]. However, the combination of LABA/ICS significantly improved lung function and reduced symptoms and the need for rescue SABA use compared with ICS alone [Ni Chroinin et al. 2009]. A recent large study of up to 78 weeks in 2019 patients with asthma aged 12 years or over who had had at least one asthma exacerbation in the previous year showed a reduced risk of exacerbations and improved lung function with a combined ICS/LABA compared with just an ICS [Bateman et al. 2013]. There were only two deaths in the ICS alone group (pneumonia and cancer) and one in the ICS/LABA combination inhaler group (automobile accident). Patient-tailored, guideline-informed use of LABAs in asthma can significantly improve outcomes when combined with ICS use.
Inhaled FF in the treatment of asthma
Inhaled FF was shown to have longer lung retention time than FP [Allen et al. 2013a]. The efficacy of once-daily inhaled FF in asthma has been demonstrated in several randomized and placebo-controlled trials (Table 2). The threshold of effect for inhaled FF appears to be around 50 μg once daily when given in the evening [O’Byrne et al. 2014b]. Doses of inhaled once-daily FF at 800 μg appear to be associated with an increase in frequency of adverse events [Busse et al. 2014]. In addition to pairing FF with a LABA, which will be discussed further later, FF combined with the long-acting muscarinic antagonist (LAMA) umeclidinium (UMEC) has been studied in asthma. In a dose-ranging study, FF (100 μg) combined with UMEC (31.25, 62.5 and 250 μg) once daily improved trough forced expiratory volume in 1 s (FEV1) compared with FF (100 μg) alone given once daily in adults with asthma [Lee et al. 2015]. A separate study by the same group suggested that a combined once-daily inhaler of FF (100 μg)/UMEC (250 μg) was associated with a 10% increase in trough FEV1 compared with FF (100 μg) alone [Yang et al. 2015]. Once-daily use of inhaled FF
Major clinical trials using inhaled fluticasone furoate in asthma.

AE, adverse event; DB, double blind; DD, double dummy; FEV1, forced expiratory volume 1 s; FF, fluticasone furoate; FP, fluticasone propionate; MC, multicenter; P, placebo; PC, placebo controlled; PEF, peak expiratory flow; PG, parallel group; R, randomized; X, crossover.
Inhaled VI in the treatment of asthma
The use of the inhaled LABA VI has been shown to have a bronchodilator effect in patients with asthma that lasts at least 24 h, allowing once-daily dosing (Table 3). Most trials in patients with asthma with inhaled VI have been done with patients on stable doses of ICS before the addition of VI, probably because of previously discussed safety concerns regarding LABAs in the treatment of asthma [Lotvall et al. 2012, 2014a; Sterling et al. 2012]. The GINA guidelines recommend the use of LABA only with an ICS usually as a fixed dose combination product [GINA, 2015]. Two of the three major outcome trials in patients on ICS medications demonstrated significant improvement in trough FEV1 after inhaled once-daily VI was added [Lotvall et al. 2012; Sterling et al. 2012] but one trial failed to show an improvement in trough FEV1 after adding inhaled VI to stable ICS treatment in patients with persistent asthma [Lotvall et al. 2014a]. Doses greater than or equal to 12.5 μg of inhaled VI daily appeared to be needed to achieve a bronchodilator effect in patients with asthma [Lotvall et al. 2012]. The pharmacokinetics of VI 25 μg daily was consistent in children aged 5–11 years regardless of age or sex [Oliver et al. 2014b]. No significant safety issues were uncovered with inhaled VI in these trials.
Major clinical trials using inhaled vilanterol trifenatate in asthma.
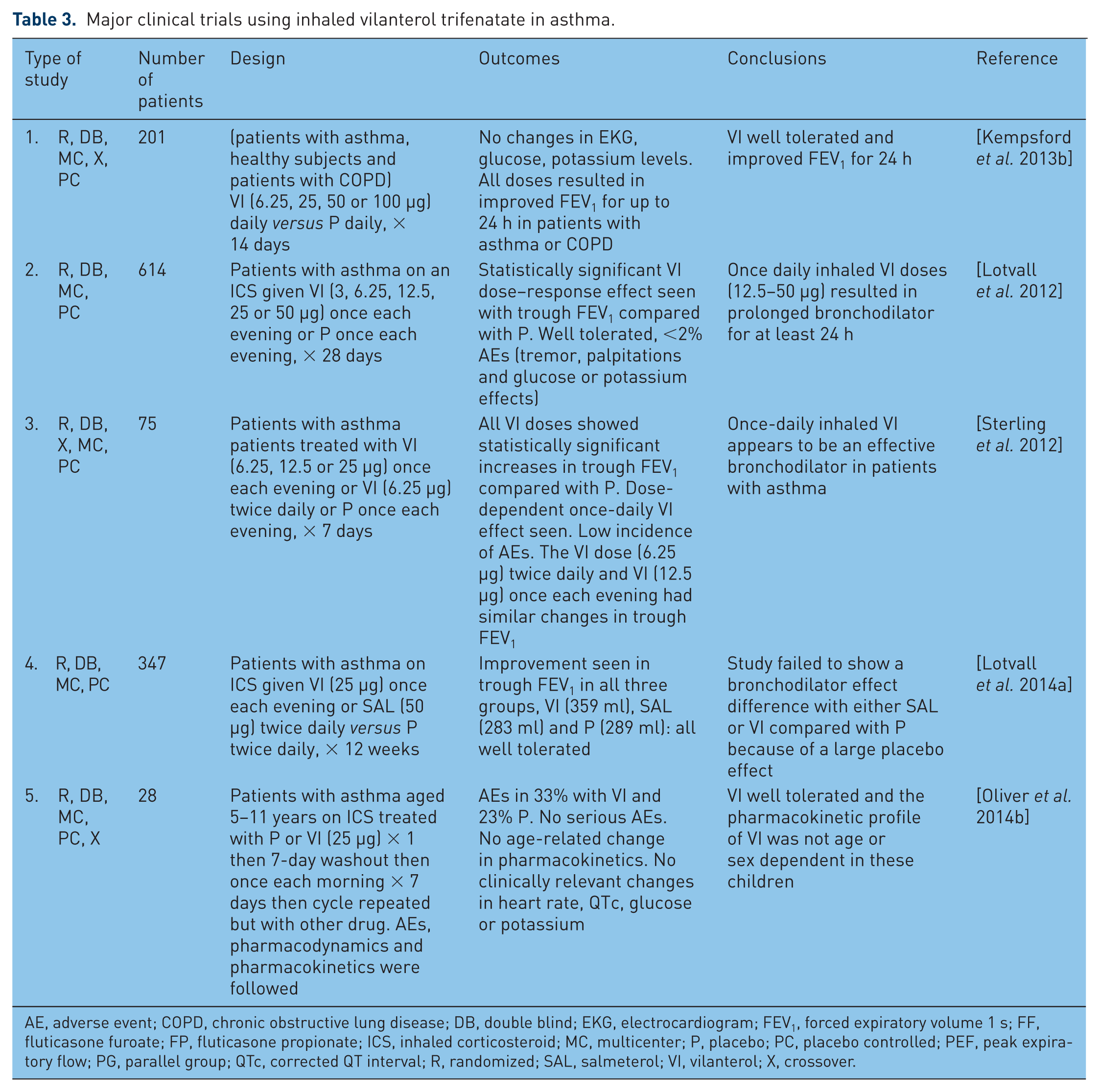
AE, adverse event; COPD, chronic obstructive lung disease; DB, double blind; EKG, electrocardiogram; FEV1, forced expiratory volume 1 s; FF, fluticasone furoate; FP, fluticasone propionate; ICS, inhaled corticosteroid; MC, multicenter; P, placebo; PC, placebo controlled; PEF, peak expiratory flow; PG, parallel group; QTc, corrected QT interval; R, randomized; SAL, salmeterol; VI, vilanterol; X, crossover.
Inhaled FF/VI combination in treatment of asthma
Inhaled FF/VI was FDA approved at the dose of 100/25 μg in May 2013 for the treatment of COPD based on several studies demonstrating efficacy and was not significantly different than inhaled twice daily 500/50 μg or 250/50 μg FP/SAL [Agusti et al. 2014; Dransfield et al. 2014]. In April 2015 the FDA approved powdered inhaled FF/VI (100/25 μg) for the once-daily maintenance treatment of asthma in patients 18 years of age and older [FDA, 2015].
A summary of published clinical trials of FF/VI in the treatment of asthma can be found in Table 4. Twenty-seven patients with asthma were randomized to once-daily FF (100 μg), VI (25 μg), FF/VI (100/25 μg) or placebo for 21 days and then were allergen challenged [Oliver et al. 2013]. The late and early asthmatic response (LAR and EAR) after allergen challenges were evaluated using weighted mean (wm) FEV1. The combination therapy provided significant protection against EAR after allergen challenge compared with placebo or the individual components of VI or FF alone [Oliver et al. 2013]. Similar findings were found with LAR, except the improvement with combined FF/VI was statistically significant only when compared with placebo and with VI alone. A similar larger study (52 patients with mild asthma) compared FF/VI (100/25 μg) with placebo or FF (100 μg). In this study, all drugs were given once daily in the evening for 28 days. EAR was evaluated after inhaled allergen challenge using the wm FEV1 as the outcome measure [Oliver et al. 2012]. Both FF alone and the combination of FF/VI significantly protected against EAR as measured by change in FEV1 after allergen challenge compared with placebo at 23 h after the last dose. Similarly, LAR was significantly modified by FF/VI compared with placebo or FF alone. FF/VI has been shown in small studies to protect against inhaled allergen challenge.
Major clinical trials using combined inhaled fluticasone furoate/vilanterol trifenatate in patients with asthma.
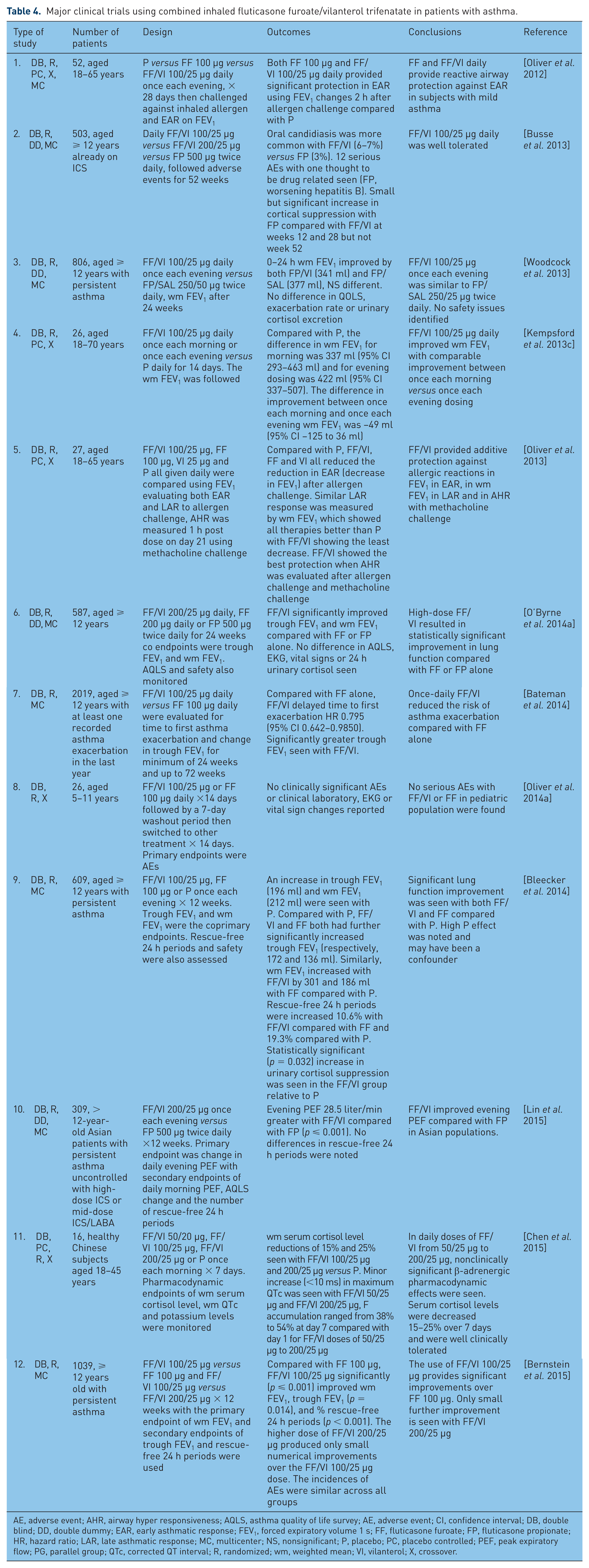
AE, adverse event; AHR, airway hyper responsiveness; AQLS, asthma quality of life survey; AE, adverse event; CI, confidence interval; DB, double blind; DD, double dummy; EAR, early asthmatic response; FEV1, forced expiratory volume 1 s; FF, fluticasone furoate; FP, fluticasone propionate; HR, hazard ratio; LAR, late asthmatic response; MC, multicenter; NS, nonsignificant; P, placebo; PC, placebo controlled; PEF, peak expiratory flow; PG, parallel group; QTc, corrected QT interval; R, randomized; wm, weighted mean; VI, vilanterol; X, crossover.
In a randomized, DB repeated-dose, two-way crossover study in 26 children aged 5–11 years with asthma, treatment included once-daily FF/VI (100/25 μg) or FF (100 μg) by inhalation for 14 days followed by a 7-day washout period before crossover. The trial lasted a total of 11 weeks [Oliver et al. 2014a]. The primary endpoints were peak expiratory flow rates (PEFs), blood pressure, clinical laboratory measurements, maximum heart rate and electrocardiogram (EKG) parameters. Secondary endpoints included pharmacokinetic and pharmacodynamic properties. No clinically significant difference was found in the primary endpoints between FF/VI and FF. The combination dosing of FF/VI was well tolerated and no significant differences were seen in the pharmacokinetic or pharmacodynamics of FF. No adverse events were judged to be serious or treatment related [Oliver et al. 2014a] in this pediatric population.
A series of efficacy trials with inhaled once-daily FF/VI were reported in patients aged 12 years and over with persistent asthma. A moderately large randomized, double-blind (DB), double-dummy, parallel group study of 806 patients demonstrated FF/VI (100/25 μg) once daily in the evening was similar to FP/SAL (250/50 μg) given twice daily in improving lung function [Woodcock et al. 2013] and no safety issues were noted. A similar comparative study evaluated FF/VI (200/25 μg) once daily for 24 weeks in 587 patients with asthma aged 12 years and over [O’Byrne et al. 2014a]. This study used change from baseline in trough wm FEV1 as the primary endpoint. The secondary endpoints included percentage change in rescue inhaler use (rescue-free 24 h periods), symptom-free 24 h periods, and Asthma Quality of Life Survey (AQLS) scores. In this study, once-daily FF/VI significantly improved trough FEV1 and wm FEV1 versus once-daily FF or twice-daily FP. Significantly more 24 h periods without use of rescue medication and without symptoms were reported with FF/VI compared with FF alone [O’Byrne et al. 2014a]. No difference in AQLS scores or adverse events were noted. Using the endpoints of severe exacerbations in asthma, a study of 2019 patients greater than 12 years old who had at least one recorded exacerbation within the prior year found that once-daily FF/VI (100/25 μg) significantly delayed the time to the first asthma exacerbation and reduced the rate of annualized severe exacerbations compared with FF (100 μg) once daily [Bateman et al. 2014]. The study also found improved trough FEV1 with FF/VI compared with FF alone with no difference in adverse event rates. Similar findings were reported in a study of 609 patients with asthma aged 12 years and over when FF/VI (100/25 μg) once daily was compared with placebo and FF (100 μg) once daily [Bleecker et al. 2014]. FF/VI and FF both showed significant improvement in both trough FEV1 and wm FEV1 compared with placebo. In a recent trial, 1039 patients aged 12 years and over with persistent asthma were randomized to once-daily FF/VI (100/25 μg), FF (100 μg) or FF/VI (200/25 μg) for over 12 weeks [Bernstein et al. 2015]. Compared with FF (100 μg) alone, FF/VI (100/25 μg) significantly improved wm FEV1, trough FEV1, % time rescue inhalers used, % time symptom free, and morning and evening PEFs. Little further improvement was seen at the higher dose of FF/VI (200/25 μg) [Bernstein et al. 2015]. Examining 309 Asian patients with asthma aged 12 years and over, Lin and colleagues found that FF/VI (200/25 μg) once daily provided statistical improvements in morning and evening PEFs, symptom-free days and AQLS scores compared with twice-daily FP (500 μg) [Lin et al. 2015]. Together, these studies provide strong evidence of the efficacy of FF/VI in the maintenance treatment of asthma.
Tolerability of inhaled FF/VI
When FF/VI (100/25 μg) was dosed once daily in the morning and compared with once daily in the evening, comparable improvement in lung function was seen in patients with persistent asthma [Kempsford et al. 2013c]. A 52-week trial focusing on safety and tolerability was performed in patients with asthma aged 12 years and over who were treated with either once-daily FF/VI (100/25 or 200/25 μg) or FP (500 μg) twice daily [Busse et al. 2013]. Statistically significant greater cortisol suppression was seen with FP compared with both doses of FF/VI at weeks 12 and 28, but not at week 52. No clinically important changes in nonfasting glucose, potassium, corrected QT intervals on EKG or ophthalmic assessments were reported at week 52. The lack of significant changes in corrected QT intervals was also seen with FF/VI (200/25 μg) in a study of 85 healthy volunteers [Kempsford et al. 2014]. In these same patients, moxifloxacin alone, given orally, increased corrected QT intervals greater than 10 ms 1–8 h after ingestion. Supratherapeutic doses of FF/VI (800/100 μg) in the same study also demonstrated a small transient increase in the corrected QT interval. Small increases in heart rate 10 min after the last dose during week 52 were seen with FF/VI (100/25 μg) (3.4 bpm, 95% CI 1.3–5.6, p = 0.002) and with FF/VI (200/25 μg) (3.4 bpm, 95% CI 1.2–5.6, p = 0.003) both compared with FP alone. Overall, daily mean heart rates decreased in all groups when 24 h Holter Monitor results were compared with baseline rates [Busse et al. 2013].
Healthy Chinese subjects were enrolled to study the pharmacodynamics and pharmacokinetics of various doses of FF/VI (50/25, 100/25 or 200/25 μg) or placebo [Chen et al. 2015]. Minimal and nonclinically significant β-adrenergic pharmacodynamic effects were seen with FF/VI. Inhaled daily doses of FF (200 or 800 μg) for 7 days or a single intravenous dose of FF (250 μg) showed only modestly higher serum levels in healthy East Asian subjects compared with white subjects without significant effect on cortisol levels [Allen et al. 2014]. Severe renal impairment had no significant effect on FF/VI pharmacokinetic or pharmacodynamic properties, while liver impairment increased FF levels and decreased serum cortisol levels [Allen et al. 2012].
A 12-week study of FF/VI (100/25μg or 200/25 μg) once-daily or placebo was performed in 185 patients with a history of asthma [Allen et al. 2013b]. The doses of FF/VI were found to be ‘noninferior’ to placebo in affecting the hypothalamic–pituitary–adrenal axis with no significant suppression of cortisol after 42 days. A meta-analysis on FF systemic exposure and cortisol suppression demonstrated the absorbed dose of FF correlated with reductions in serum and urine cortisol levels [Allen, 2013]. When computer modelling was used, it was suggested that clinical doses of FF less than 200 μg daily are unlikely to provide significant cortisol suppression and that supratherapeutic range doses would be required to consistently suppress cortisol levels [Allen, 2013]. A study of 48 healthy Japanese male subjects exposed to inhaled single doses of FF (200, 400 or 800 μg) and VI (25 or 12.5 μg) for 7 days found that repeat dosing of FF (200, 400 or 800 μg) reduced serum cortisol levels by 32%, 38% and 97%, respectively [Nakahara et al. 2013]. No significant changes in heart rate were seen in the repeated dose or single dose arm of the study. Serum cortisol levels decreased by 27% when 400 mg of oral ketoconazole was added to daily inhaled FF/VI (200/25 μg) for 11 days [Kempsford et al. 2013a]. Table 5 summarizes the major adverse effects associated with the use of inhaled FF/VI.
Adverse effects of fluticasone furoate/vilanterol trifenatate inhalation.

Similar rates tended to be seen in adverse events for FF/VI as in FF, VI and FP alone. Based in part on Busse et al. [2013] and Syed [2015].
FF, fluticasone furoate; FP, fluticasone propionate; VI, vilanterol.
The delivery system used for the inhaled dry powder FF/VI is the ELLIPTA (GlaxoSmithKline, England) system. Using qualitative interviews, this system was associated with high patient satisfaction and was preferred over other systems in 75 patients with COPD and asthma aged 18 years and over [Svedsater et al. 2013]. At the time of survey, 44 of the 75 patients were on DISKUS (GlaxoSmithKline, England) inhalers or the DISKUS and Handi-Haler (Boehringer Ingelheim, Germany) systems. The others were on metered dose inhalers or other dry-powdered inhalation systems. From these limited data, acceptance of the FF/VI dry powder once-daily inhaler delivery system appears high.
Conclusion
The dry powder inhaler of FF/VI is an effective and well tolerated once-daily maintenance treatment for patients with persistent asthma. Both FF and VI have demonstrated efficacy in asthma. The combination of FF/VI has also proven efficacy in the treatment of COPD. The once-daily dosing of FF/VI offers efficacy and increased convenience, and possible increased adherence for patients with asthma.
Footnotes
Funding
The authors received no financial support for the research, authorship, or publication of this article.
Conflict of interest statement
JR and AZ declared no potential conflicts of interest with respect to the research, authorship, or publication of this article. TEA reports speaker honoraria from GSK and BI on the topic of COPD.
