Abstract
Background and Objectives:
In patients with chronic liver disease and ascites, spontaneous bacterial peritonitis (SBP) is a common complication. This can progress gradually and subtly or go undiagnosed until signs start to show. Early diagnosis is essential for effective treatment and the prevention of subsequent episodes because the mortality rate following a single episode varies from 20% to 40%. The goal was to study the incidence, clinical profile, and microbiological characteristics of SBP and its variants in people with liver cirrhosis and ascites.
Materials and Methods:
This observational study was done on 104 cirrhotic patients with ascites admitted at a tertiary care hospital. Basic demographics, symptoms, and clinical signs of patients were recorded. Diagnostic paracentesis was done aseptically and immediately sent for ascitic fluid cytology, biochemistry, and culture. Bacterial examination and antibiotic sensitivity tests were carried out by standard microbiological techniques.
Results:
Out of a total of 104 cirrhotic patients, 28 (26.9%) were diagnosed with SBP. Alcohol was the most common etiological factor, followed by hepatitis C infection. Among the SBP patients, the most common presentation was culture-negative neutrocytic ascites (71.4%), followed by classical SBP (17.8%), then mono bacterial non neutrocytic bacterascites MNB (10.7%). Among the symptoms, the most common was abdominal distension, followed by icterus, pedal edema, and abdominal pain. Majority of the patients belonged to Child–Pugh’s Grade C. Of 28 cases of SBP, 8 were culture positive. Gram-negative isolates were predominant (75%) and Escherichia coli were the most common isolate.
Interpretation and Conclusion:
The prevalence of SBP in cirrhotic patients was 26.9%, and Gram-negative isolates were predominant. Ascitic fluid culture and susceptibility testing can lead to accurate diagnosis of SBP and can guide for treatment as resistance to antibiotics is common.
Keywords
INTRODUCTION
The most common reason for liver-related deaths worldwide is cirrhosis. Cirrhosis-related fatalities made up 24% (23%–26%) of all deaths worldwide in 2017, up from 1.9% (1.8%–2.0%) in 1990.[1]
Anatomically, cirrhosis is defined as a diffuse process with fibrosis and nodule formation of the liver. The most frequent reasons are excessive alcohol consumption, viral hepatitis, nonalcoholic steatohepatitis, and autoimmune diseases. Clinically, patients may have jaundice, ascites, hepatic encephalopathy, bleeding varices (decompensated), or they may be asymptomatic (compensated).[2]
Ascites is free fluid within the peritoneal cavity. It develops as a result of peritoneum-related conditions (infection and cancer), as well as conditions caused by external illnesses (liver disease, heart failure, and hypoproteinemia). In the Western world, cirrhosis is the most frequent cause of ascites (75%), followed by peritoneal cancer (12%), cardiac failure (5%), and peritoneal tuberculosis (2%). Ascites is by far the most common initial decompensating occurrence in cirrhotic patients, indicating the change from compensated to decompensated cirrhosis. Its pathophysiology is primarily explained by portal (sinusoidal) hypertension and sodium retention as a result of vasodilation and subsequent stimulation of sodium retaining systems. The most frequent infection in cirrhosis is termed spontaneous bacterial peritonitis (SBP) because it develops without a contiguous source of infection or an intra-abdominal inflammatory focus.[3]
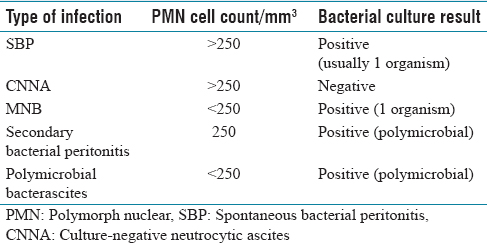
Based on the findings of ascitic cultures, the number of polymorph nuclear (PMN) cells, and the presence or absence of an infectious surgical source, ascitic fluid infection can be divided into five types as shown in Table 1. The prototype of the three forms of spontaneous ascitic fluid infection is SBP.[4]
Classification of ascitic fluid infection
Bacterial translocation, systemic, and local immune dysfunction, particularly a decreased opsonization activity in ascitic fluid, are the main elements in the pathogenesis of SBP. As a result, common causal organisms in the gut microflora include Escherichia coli, Klebsiella spp., Enterobacter spp., Enterococci, and Streptococci.[5]
A sudden onset of fever, chills, and abdominal discomfort with rebound tenderness over the abdomen are the hallmarks of SBP. In most cases, leukocytosis and absent bowel sounds are observed. PMN cells are the most prevalent white blood cells found in the ascitic fluid upon paracentesis.
SBP being the problem in cirrhosis with ascites, all cirrhotic patients should be screened for SBP using ascitic fluid analysis, PMN cell count, and ascitic fluid culture if they have ascites at the time of admission, as well as in the event of gastrointestinal bleeding, shock, signs of inflammation, hepatic encephalopathy, and worsening liver or renal function.
Aims and objectives
To estimate the prevalence of SBP and its variants in patients of cirrhosis of the liver with ascites
To study the clinical and microbiological profile of SBP and its variants
To study SBP with the severity of cirrhosis according to the Child–Pugh Score.
MATERIALS AND METHODS
This prospective observational study approved by the institutional ethical review committee was conducted on 104 cirrhotic patients with ascites admitted to Sri Guru Ramdas Medical College and Hospital Amritsar, Punjab, India. For this purpose, all newly diagnosed and already diagnosed patients with cirrhosis of the liver with ascites coming to hospital outpatient department and indoor patients department were enrolled after taking written and informed consent.
Inclusion criteria
Patients with a confirmed diagnosis of cirrhosis of the liver with ascites
Patient age >18 years.
Diagnostic criteria
Cirrhosis of liver was diagnosed by the following diagnostic tools:
History: Patients with gastroesophageal varices, ascites, or hepatic encephalopathy are likely to have cirrhosis if they have a history of chronic liver disease Physical findings: In patients with a diagnosis of chronic liver disease, physical findings of an enlarged left hepatic lobe with splenomegaly, along with the cutaneous stigmata of liver disease (palmar erythema, leukonychia, clubbing of fingernails, gynecomastia, spider telangiectasia’s, asterixis, dilated abdominal veins, and parotid enlargement) suggest cirrhosis Biochemical features (elevated liver enzymes, hypoalbuminemia, reversal of albumin-to-globulin ratio, and prolonged prothrombin time) Imaging studies: Imaging investigations (ultrasound [USG], computed tomography [CT]/contrast-enhanced CT) can aid in the diagnosis of cirrhosis when neither physical nor laboratory results are suggestive of the condition. USG abdomen showing ascites features of cirrhosis of the liver and portal hypertension.
Spontaneous bacterial peritonitis was diagnosed by the following criteria
An ascitic fluid neutrophil count > 250 cells/mm3 OR
A positive ascitic fluid culture, and
An absence of a primary source of infection in the abdomen.
METHODS
Ascitic fluid for analysis was aspirated as soon as the patient admitted and diagnosed to be suffering from cirrhosis of the liver, before giving any antibiotics. All patients underwent paracentesis within 24 h of admission. About 30 mL of ascitic fluid was tapped in each patient with aseptic precautions.
10 mL of ascitic fluid was immediately inoculated into blood culture bottles at the bedside for microbiological analysis
10 mL of ascitic fluid was sent to the laboratory in sterile test tubes for conventional culture
10 mL of ascitic fluid was utilized for biochemical and cytological examination.
Ascitic fluid of all patients was analyzed for the type of cells and cell count. Ascitic fluid was cultured to know the presence of pathogenic organisms.
Exclusion criteria
Those who were suspected to have secondary peritonitis such as perforated colon and ruptured appendix
Ascites due to renal, cardiac, tubercular, and malignant cause
The patient who had undergone abdominal surgery within 3 months before admission.
Unpaired Student’s t-test and Chi-square tests were used to statistically analyze the data for independent and categorical variables, respectively. The statistical significance threshold was set at P = 0.05.
OBSERVATION AND RESULTS
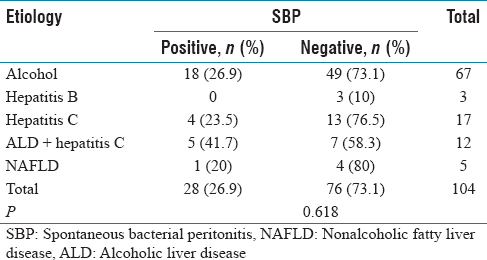
A total of 104 cirrhotic patients with ascites included 86 (82.7%) males and 18 (17.3%) females. The majority of the patients were in the age group of 41–50 years, followed by patients between 51 and 60 years. The mean age was 51.5 ± 12.29 years. In the study group, the most common etiology of cirrhosis was alcohol intake (64.4%), followed by hepatitis C infection [16.3%; Figure 1 and Table 2]. The most common clinical features in patients with SBP and those without SBP are depicted in Figure 2 and Table 3. A maximum number of patients with SBP presented with abdominal distention followed by icterus and pedal edema, and the result was statistically nonsignificant (P > 0.005) shown in Table 3. In both patients with SBP and those without SBP, the majority of patients (68) belonged to Child–Pugh’s Grade C (29.4% with SBP; 70.6% without SBP), followed by (35) Child–Pugh’s Grade B (22.9% with SBP; 77.1% without SBP). Of 104 cirrhotic patients with ascites, 28 (26.9%) had SBP, with 20 (19.2%) of them presenting with culture-negative neutrocytic ascites (CNNA), 8 (7.7%) were culture positive, 5 (4.8%) had classical SBP, and 3 (2.9%) had MNB. Among the culture-positive SBP, Gram-negative isolates were more common (75%) than Gram-positive isolates (25%). The most common isolate was E. coli (37.5%), followed by Acinetobacter haemolyticus, Providencia rettgeri, Pseudomonas aeruginosa, Staphylococcus epidermidis, and Staphylococcus haemolyticus [12.5% each; Figures 3 and 4 and Table 4]. Among the biochemical parameters, ascitic fluid cell count, ascitic fluid absolute neutrophil count, and INR showed a significant difference between patients with SBP and patients without SBP [Table 5].

Etiology of cirrhosis in the study population
Etiology of cirrhosis in the study groups
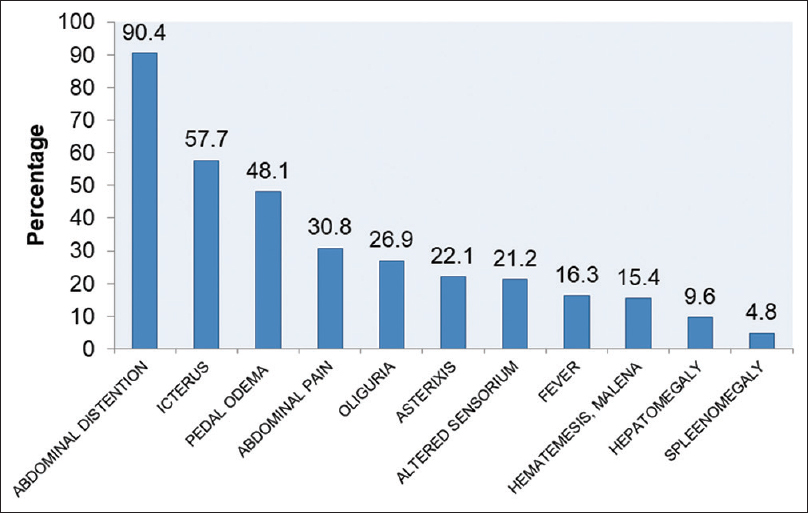
Clinical presentation of cirrhosis patients in the study group
Clinical presentation of cirrhotic patients


Spectrum of organisms in SBP. SBP: Spontaneous bacterial peritonitis
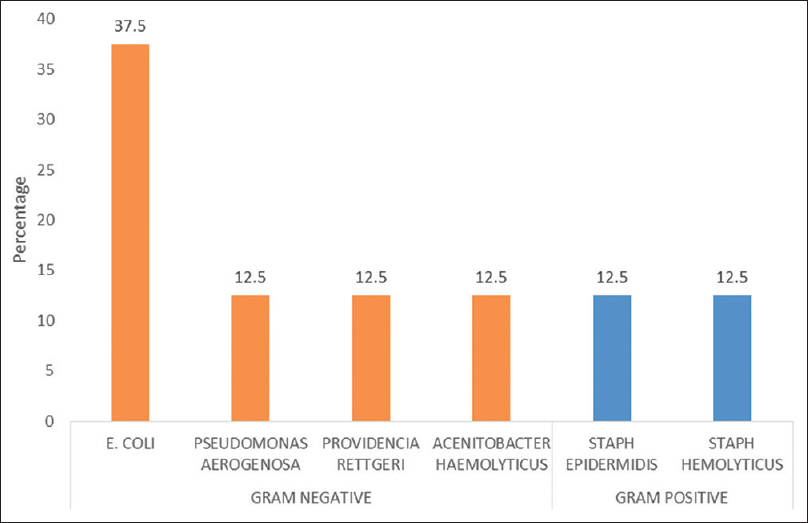
Microbiological classification of culture-positive SBP according to Gram staining. SBP: Spontaneous bacterial peritonitis
Classification of culture-positive spontaneous bacterial peritonitis according to Gram stain and microorganisms

Hematological and biochemical parameters in cirrhotic patients

Table 2 shows no significant correlation between etiology and SBP (P > 0.005).
Table 4 showed that majority of culture positive SBP patients had gram negative bacteria (75%). Overall E. coli is the most common organism (37.5%).
Table 5 shows that, among the hematological and biochemical parameters, ascitic fluid cell count, ascitic fluid absolute neutrophil count, and INR had a significant correlation between patients with SBP and patients without SBP (P < 0.005).
DISCUSSION
One of the most frequent bacterial infections in cirrhosis with ascites patients is SBP. In individuals with liver cirrhosis, a failure to diagnose SBP on time frequently results in a fatal outcome. The majority of patients in the current study were men. The male-to-female ratio was 4.7:1, which was similar to that reported by Nadagouda et al.[6] and Paul et al.,[7] The increased frequency of alcoholic cirrhosis in the investigated male participants may account for the study’s male predominance. The mean age of cirrhotic patients was found to be 51.5 ± 12.29 years. The findings were consistent with a study reported by Syed et al.,[8] where the mean age was 51.1 ± 11.7 years. The majority of patients had a history of alcoholism as the underlying cause of cirrhosis, consistent with that reported by Nadagouda et al.[6] and Chawla et al.[9] Most of the patients (68) were in Child–Pugh’s Grade C (29.4% in the SBP group and 70.6% in the non-SBP group), similar to a study done by Choubey et al.[10] in a tertiary care center where 42% of patients of SBP were in Child–Pugh’s Grade C. In the current study, 26.9% of cirrhotic patients had SBP. Results similar to this study (27%) were cited in a study done by Bhat[11] and Bibi et al.,[12] among patients with chronic liver disease in Karachi, 25% were diagnosed with SBP. Various authors reported the overall frequency of SBP as 38%,[13] 47.5%,[14] and 56%[15] in chronic liver disease patients. In the current study, patients with SBP presented with a variety of symptoms, the most prevalent of which was abdominal distension. Similar results were reported by Paul et al.[7] In the current study, icterus was the second most common presentation (57.7%) and none of sign and symptoms were statistically significant. However, this was not observed in earlier studies.[6,7] Among the biochemical and hematological parameters, ascitic fluid cell count, ascitic fluid absolute neutrophil count, and INR shows a significant correlation between SBP-positive and SBP-negative patients. Similar results were reported by Gharabawy et al.[16] in 2018. Of 28 cases of SBP, CNNA was the most common (71.4%), followed by classical SBP (17.8%) and MNB (10.7%). The results were comparable with the studies done at Ayub Medical College, Abbottabad, Pakistan[17] and Jinnah Postgraduate Medical Centre, Karachi, Pakistan[12] in chronic liver disease patients with ascites where CNNA was the most common variant of SBP. The patients received an ascitic tap at a very early stage, when the bacterial inoculum was still minimal, which may have contributed to the unusually high prevalence of CNNA. Another factor could be the proficiency of the culturing methods used by the various institutions, which varied widely. The ascitic fluid culture was positive in 28.5% of SBP cases. Similar results were observed in various studies with culture positivity of 31%,[14] but some studies have reported much lower rates of culture positivity, i.e., <25%.[15,18] This difference could be attributed to the different cultural techniques. International literature suggests a culture positivity rate of 31%–71%.[18] In this study, Gram-negative organisms were found to be a more common cause of SBP (75%), similar to that reported by Chawla et al.[9] and Bibi et al.[12] E. coli was the most prevalent isolate among the Gram-negative isolates. Similar results were cited in different studies conducted by Syed et al.,[8] Chawla et al.,[9] Bibi et al.,[12] and Zaman et al.[17] Bacterial translocation from the gut is the primary contributor of SBP.[5] Therefore, enteric Gram-negative rods are often pathogens that are frequently isolated in SBP. Gram-positive organisms have also been observed to predominate in some investigations; however, this is highly uncommon and frequently the result of prophylaxis or a prior intervention.[19] Therefore, all cirrhotic patients with ascites should have diagnostic paracentesis and ascitic fluid analysis for early identification and better antibiotic selection to minimize antimicrobial resistance, which leads to better outcomes for cirrhotic patients.
CONCLUSION
SBP and its variations are a frequent side effect of liver cirrhosis with ascites. The absence of conventional symptoms and indicators in these patients meant that only ascitic fluid investigation and culture could provide a conclusive diagnosis. SBP being the problem in cirrhosis with ascites, all cirrhotic should be screened for SBP. Diagnostic paracentesis is carried out during hospitalization to detect infections early and treat them quickly because they have a bad prognosis and a high death rate if not treated early. CNNA was the most common variant of SBP. Due to the prevalence of antibiotic resistance, ascitic fluid culture and susceptibility testing can provide an accurate diagnosis of SBP and serve as a therapeutic guide. E. coli was the most often observed organism among several isolated organisms. Therefore, understanding the SBP’s microbiological spectrum is crucial for choosing the right antibiotic. As a result, every cirrhotic patient should have their SBP checked because a delayed diagnosis can have catastrophic consequences.
Footnotes
Conflicts of interest
There are no conflicts of interest.
Ethical approval
The study was approved by the institutional ethics committee, Sri Guru Ramdas Institute of Medical Sciences & Research (SGRDIMSR), Amritsar, Punjab.
Funding
Nil.
Author’s contribution
Pardeep Kumar prepared the first draft, data collection, data analysis, manuscript review and editing. Gurinder Mohan & Parminder Singh done supervision, provided overall guidance, finalized the manuscript on the basis of comments from other authors and reviewer feedback. All authors read and gave final approval of the version to be submitted.
Acknowledgements
First of all, I thank God Almighty, my professor Dr. Gurinder Mohan and my parents Mr. Santosh Kumar and Mrs. Lovely Rani, my sister Priyanka Goyal, and my brother Sunny Mittal who supported me with love and understanding. Without them, I could never have reached this current level of success. I owe a special thanks to my colleagues Dr. Kanupriya Bector and Dr. Rajandeep Kaur. Their encouragement and moral support have been my driving force.
