Abstract
Membrane depolarization is critical to pulmonary arterial (PA) contraction. Both L-type Ca2+ channels (CaL) and Rho-kinase are important signaling components of this process and mitochondrial and non-mitochondrial generated superoxides can be part of the signaling process. Maturation and long-term hypoxia (LTH) each can modify depolarization-dependent contraction and the role of superoxides. By the use of wire myography, we tested the hypothesis that maturation and LTH increase pulmonary arterial reactivity to high-K+-induced membrane depolarization through enhancements in the importance of CaL and Rho-kinase-dependent pathways. The data show that maturation, but not LTH, increases contraction to 125 mM KCl (high-K+) without altering the EC50. High-K+-dependent contraction was inhibited to a similar extent in fetal and adult PA by multiple CaL blockers, including 10 μM diltiazem, 10 μM verapamil, and 10 μM nifedipine. Postnatal maturation increased the role for 10 μM nifedipine-sensitive CaL, and decreased that for 10 μM Y-27632-sensitive Rho-kinase. In all groups, the combination of nifedipine and Y-27632 effectively inhibited high-K+ contraction. Tempol (3 mM) but not 100 μM apocynin slightly reduced contraction in arteries from fetal hypoxic and adult normoxic and hypoxic sheep, indicating a limited role for non-mitochondrial derived superoxide to high-K+-induced contraction. Western immunoblot for alpha smooth muscle actin indicated small increases in relative abundance in the adult. The data suggest that while CaL therapies more effectively vasodilate PA in adults and rho-kinase therapies are more effective in newborns, combination therapies would provide greater efficacy in both young and mature patients regardless of normoxic or hypoxic conditions.
Keywords
INTRODUCTION
Depolarization of the vascular smooth muscle plasma membrane results in contraction through a combination of mechanisms, central to which is voltage-dependent Ca2+ influx.[1,2] This is exemplified by the inhibition of vessel contraction by L-type Ca2+ channel (CaL) blockers.[3–5] The depolarization-induced increase in cytosolic Ca2+ due to activation of CaL and other pathways, leads primarily to myosin light chain phosphorylation with resultant myocyte contraction. Such studies also demonstrate that membrane depolarization can cause arterial contraction through activation of the small GTPase Rho-kinase, which inhibits the myosin light chain phosphatase.[6,7] The consequential increase in Ca2+ sensitivity of the contractile filaments augments vasoconstriction when cytosolic Ca2+ increases following myocyte stimulation.[8,9]
Long-term hypoxia (LTH) can augment potassium-dependent contraction of pulmonary arteries. In rats, this is mediated by changes in CaL and Rho-kinase activity. More specifically, LTH suppresses CaL activity,[10] while it enhances Rho-kinase activity in rat pulmonary vasculature.[8] This contrasts with the newborn pig where LTH increases CaL activity.[11] These inter-species differences with regards to the effect of LTH on CaL activity may represent developmental age variability or simply species-specific differences. Although it is unknown whether age affects depolarization-dependent contraction in the pulmonary vasculature, previous studies illustrate that following birth potassium-dependent contraction in sheep middle cerebral arteries increases significantly.[12] Nonetheless, in the same vessels, CaL expression is more prominent in the fetus relative to that of the adult.[13]
These findings illustrate that both maturation and LTH alter membrane depolarization and arterial reactivity, and that Rho-kinase dependent responses are important to the resultant reactivity changes. Therefore, we designed a series of experiments to test the hypothesis that both maturation and LTH increase pulmonary arterial reactivity in response to potassium-induced membrane depolarization, through enhanced activities of CaL and Rho-kinase.
MATERIALS AND METHODS
Experimental animals
All experimental procedures were performed within the regulations of the Animal Welfare Act, the National Institutes of Health Guide for the Care and Use of Laboratory Animals, “The Guiding Principles in the Care and Use of Animals,” approved by the Council of the American Physiological Society, and the Animal Care and Use Committee of the university. We conducted these studies on fourth- and fifth-order pulmonary arteries with internal diameters of about 500–700 μm, from fetuses or adult ewes that lived at Nebeker Ranch (Lancaster, Calif., USA; 720 m) and were brought to the university animal care facility (353 m; arterial PaO2=95±5 Torr) for experimental study, or were acclimatized to high altitude (3,801m, PaO2=60±5 Torr) at the Barcroft Laboratory, White Mountain Research Station (Bishop, Calif., USA) for ~110 days.[14] Animals maintained at high altitude were transported to the university from the field station and shortly after arrival, a tracheal catheter was placed in the ewe, through which N2 flowed at a rate adjusted to maintain PaO2 at ~60 Torr, a value that is similar to that measured at the Barcroft laboratory.[15] This PaO2 was maintained until the time of the experimental study. Within 1 to 5 days after arriving at the university, sheep were euthanized with an overdose of the proprietary euthanasia solution, Euthasol (pentobarbital sodium 100 mg/kg and phenytoin sodium 10 mg/kg; Virbac, Ft. Worth, Tex., USA). Lungs with their vessels were removed and used immediately for contractility experiments. To avoid complications from endothelium-mediated effects, the endothelium was removed by carefully inserting a small roughened hypodermic needle or rotating the artery on the mounting wire.[3–5]
Tissue preparation
Pulmonary arteries for contractility were dissected free of parenchyma and cut into 5 mm long rings in ice-cold phosphate-free balanced salt solution (BSS) of the following composition (mM): 126 NaCl; 5 KCl; 10 HEPES; 1 MgCl2; 2 CaCl2; 10 glucose; at a pH of 7.4 (adjusted with NaOH). All contraction studies were performed with a modified Krebs–Henseleit (K-H) solution containing (mM): 120 NaCl; 4.8 KCl; 1.2 K2HPO4; 25 NaHCO3; 1.2 MgCl2; 2.5 CaCl2; and 10 glucose.
Contraction studies
Pulmonary arterial rings were suspended in organ baths (Radnoti Glass Instruments, Inc. Monrovia, Calif., USA) that contained 5 or 10 ml of modified K-H solution maintained at 37°C and aerated with 95% O2–5% CO2 (pH=7.4) as performed previously.[3–5] Each ring was suspended between two tungsten wires passed through the lumen. One wire was anchored to the glass hook at the bottom of the organ chamber; the other was connected to a tissue hook attached to a low compliance force transducer (Radnoti Glass Instruments Inc.) for the measurement of isometric force.[3–5] The transducers were connected to an analogue to digital data interface (Powerlab 16/30 AD Instruments, Colorado Springs, Colo., USA; or MP100, Biopac Systems Inc., Goleta, Calif., USA) attached to a computer. The changes in tension were recorded using Chart 5.5 (AD Instruments), or AcqKnowledge 3.9 (Biopac Systems, Inc.), and the data were stored on magnetic media for later analysis. At the beginning of each experiment, vessels were equilibrated without tension for a minimum of thirty minutes. By stretching the vessels progressively, as previously described,[3–5] vessel rings were tensioned to 447±13 dynes in 108 vessels from 12 fetal normoxic sheep, to 429±21 dynes in 120 vessels from 12 adult normoxic sheep, to 418±14 dynes in 52 vessels from 6 fetal hypoxic sheep, and to 316±16 dynes in 76 vessels from 7 adult hypoxic sheep. Isolated pulmonary arterial rings were stimulated with 125 mM KCl (high-K+) to depolarize the plasma membrane and activate CaL.[1] In most experiments, the tension was normalized to a control response obtained with high-K+ (%TK(control)). To evaluate dose-response characteristics, arteries were stimulated by applying cumulatively 4 mM to 125 mM K+ without washing in-between each K+-concentration increase.
Western immunoblot assay
Pulmonary arteries from normoxic and hypoxic fetal and adult sheep were isolated, cleaned in BSS, and frozen rapidly in liquid nitrogen. SDS gel and Western blot analyses were performed using protocols based on previous studies.[12,16] In particular, samples were homogenized and sonicated in RIPA buffer on ice containing 150 mM NaCl, 50 mM Tris, 1% Triton X-100, 0.5% deoxycholic acid, 1% SDS, 5 mM EDTA and Halt protease inhibitor (Thermo-Fisher Scientific, Rockford, Ill., USA). Nuclei and debris were pelleted by centrifugation at 1,000 g for 10 minutes at 4°C.
Protein concentrations were determined using a modification of the Bradford method.[12,17] A 8% polyacrylamide gel was loaded with 10 μg of protein mixed with an equal volume of electrophoresis sample buffer per lane and then electrophoresed at 90 V for 90 min. We used a Mini Trans-Blot Electrophoretic Transfer Cell system (Bio-Rad Laboratories, Hercules, Calif., USA) to transfer proteins from the gel to a nitrocellulose membrane at 100 V for 3 hours.
The membranes were blocked for nonspecific binding by incubating the membrane overnight in blotting solution (5% nonfat milk in tris-buffered saline with 0.1% Tween-20–TTBS) at 4°C. Then membranes were incubated in a 1:1000 dilution of rabbit polyclonal anti-smooth muscle specific α-actin (ab5694, Abcam, Cambridge, Mass.) in blotting solution for 1 hour at room temperature (22°C), or were incubated overnight (16 hours) at 4°C. The differences in incubation conditions did not cause any systematic differences in the measured optical density. Membranes were then washed three times with TTBS, and incubated with goat antirabbit IgG horseradish perioxidase-conjugated secondary antibody (Cell Signaling Technology, Beverly, Mass., USA) for 45 minutes at a 1:2000 dilution at room temperature. Following the secondary antibody incubation, the membrane was washed three times in TTBS, for 5 minutes each time. The membrane was then incubated with a chemiluminescent reagent (Santa Cruz Biotechnology, Santa Cruz, Calif., USA) for 1 minute, and the protein band of a predicted weight of 42 KDa was detected using a chemiluminescent Imager (Alpha Innotech, San Leandro, Calif., USA). The densities of each band were determined using the gel analysis tools distributed with ImageJ.[18] The expression levels are expressed in optical density units and varied by ±9–11% (s.e.m.) in all four groups.
Chemicals and drugs
Most reagents and chemicals were purchased from Sigma-Aldrich (St. Louis, Mo., USA). Transfer, running, and wash buffers for Western Immunoblot were purchased from Bio-Rad Laboratories. Y-27632 was purchased from Tocris (Ellisville, Mo., USA). Sources for other specialized reagents are noted in the text.
Statistical methods
All time-series recordings were graphed with IGOR Pro 6.0 (Wavemetrics, Lake Oswego, Oreg., USA), and the data presented as mean±S.E.M. Statistical analyses were made using GraphPad Prism 5.0 (La Jolla, Calif., USA). Data were evaluated for normality prior to any comparative statistical analysis using a D'Agostino-Pearson normality test. Between groups that were distributed normally, statistical differences were determined with a two-tailed unpaired Student's t-test. A Mann-Whitney U test was used for comparisons of non-normal data. Dose-response curves were fitted in Prism 5.0 using a Hill equation.[3–5] The N values reported reflect the total number of arterial segments and total number of sheep tested. P<0.05 was accepted as statistically significant.
RESULTS
Stimulation of arterial segments with 125 mM KCl results in depolarization-dependent contraction, and is a common method to evaluate the amount of active smooth muscle.[1] Figure 1 shows the contraction induced with high-K+ in arteries isolated from fetal and adult sheep under normoxic or LTH conditions. Figures 1A and B show representative isometric tension recordings from normoxic and LTH vessels, respectively, and illustrate the substantially greater contraction in vessels isolated from adults. This is emphasized in the summary data presented in Figure 1C, in which the high-K+-induced contraction was two- to threefold greater in adult vessels, regardless of the influence of hypoxia. The number of replicates and sheep used for these series of experiments are provided in the contraction studies methods section.
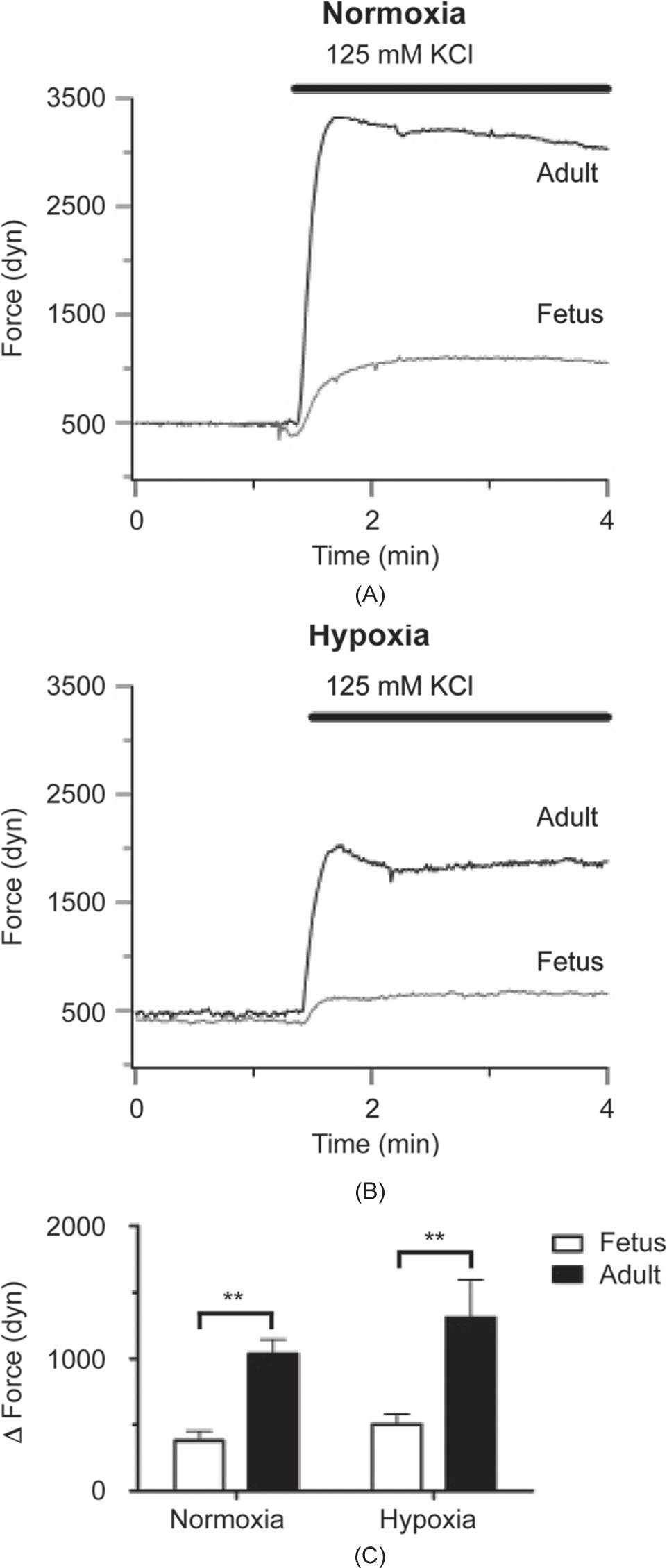
High-K+ elicits greater force in pulmonary arteries from adult relative to fetal sheep. Isometric tension recording of pulmonary arterial rings constricted with 125 mM KCl in fetus (gray) and adult (black) for animals housed in normoxic (A) or long-term hypoxic (B) conditions. (C) Bars (open: Fetus; solid: Adult) indicate mean±S.E.M. of the force generated by 125 mM KCl. **(P<0.01) denotes significant difference by two-way ANOVA relative to fetus.
Next, we performed experiments to determine the importance of CaL to high-K+-induced contraction, and the influence of maturation and LTH on the role of this channel. This was achieved by stimulating arteries with 125 mM KCl in the absence and presence of three major classes of CaL blockers, including dihydropyridines (10 μM nifedipine), phenylalkylamines (10 μM verapamil), and benzothiazepines (10 μM diltiazem). Figures 2A and B show normalized isometric tension recordings from normoxic fetal vessels, while Figures 2D and E show recordings from normoxic adult vessels. In the fetus, vehicle (0.1% DMSO) did not reduce contraction, while 10 μM nifedipine, verapamil or nifedipine (Fig. 2A and B) suppressed contraction by approximately one-third. Moreover, in the adult, nifedipine, verapamil, or diltiazem reduced high-K+-dependent vascular contraction about twice as much as compared to the fetus (Fig. 2D and E). The summarized data in Figures 2C and F exemplify this and show that nifedipine (fetal N=18/6; adult N=12/4), verapamil (fetal N=12/4; adult N=16/6), or diltiazem (fetal N=13/5; adult N=13/5) each reduced high-K+-induced contraction by roughly 35% in the fetus and 70% in the adult, with respect to control (fetal N=20/6; adult N=25/8).
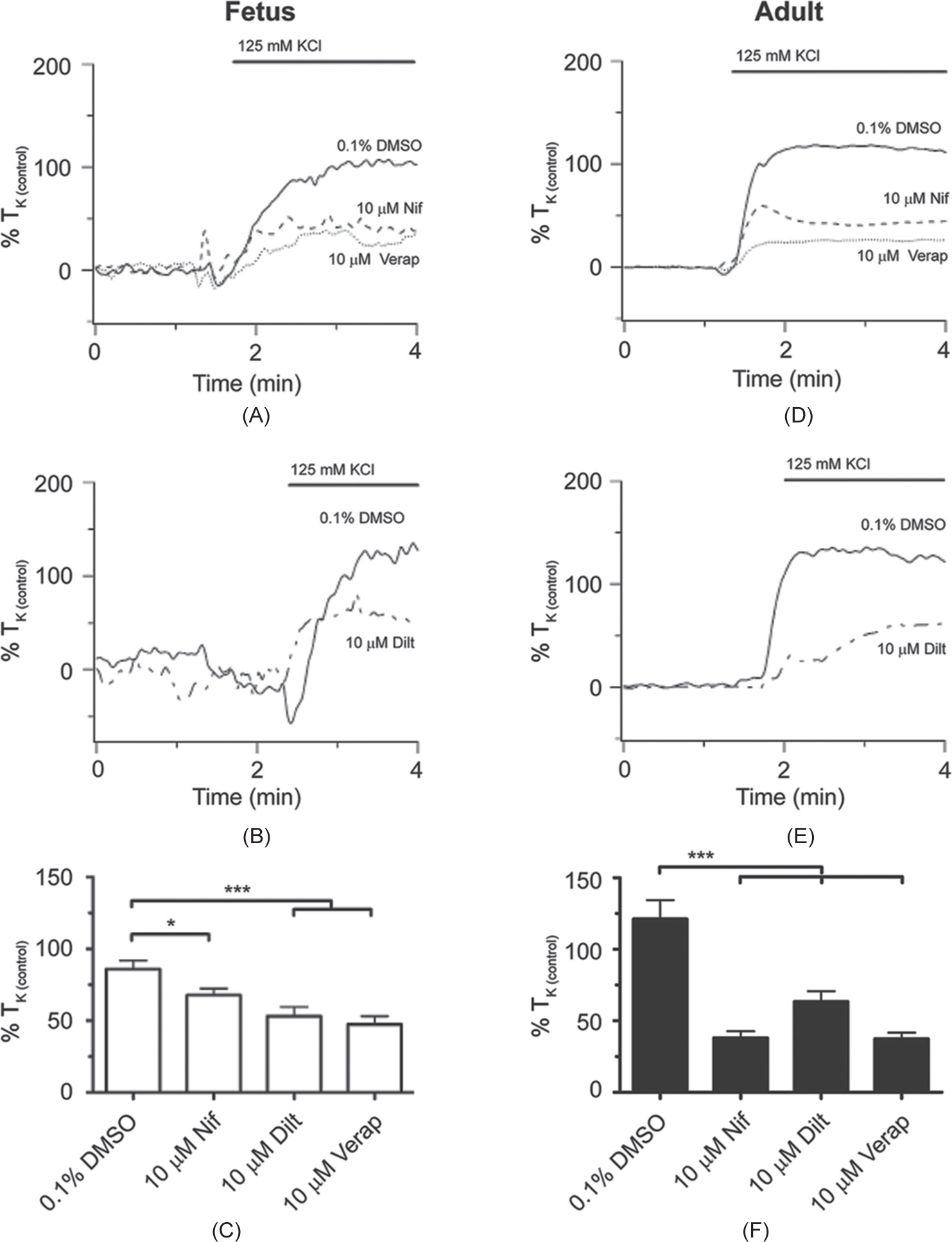
The L-type Ca2+ channel is important for high-K+-elicited pulmonary arterial contractility in fetal and adult sheep housed in normoxic conditions. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 10 μM nifedipine (Nif; dashed), 10 μM verapamil (verap; dotted), or 10 μM diltiazem (dilt; dotted and dashed) from fetus (A and B) or adult (D and E). Tracings are plotted in relation to maximal contraction from initial high-K+ stimulation (% TK (control)). (C and F) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or L-type Ca2+ channel antagonists for pulmonary arteries from fetal (C; open) and adult (F; solid) sheep. *(P<0.05), ***(P<0.001) denotes significant difference by one-way ANOVA.
We then performed parallel studies to assess the extent to which high-K+-induced contraction of pulmonary arteries from LTH sheep is dependent on CaL. This was done in the setting of prior conflicting studies, reporting that LTH-reduced CaL activity during ET-1 dependent Ca2+ responses in adult rats,[10] but increased CaL activity in newborn piglets.[11] Figures 3A and B show normalized isometric tension recordings from hypoxic fetal vessels while Figures 3D and E show those from hypoxic adult vessels. In the fetus, 10 μM nifedipine, verapamil or diltiazem mildly reduced high-K+-induced arterial contraction (Fig. 3A and B). The adult contrasts the fetus with 10 μM nifedipine, verapamil, or diltiazem reducing high-K+-dependent contraction to a greater extent (Fig. 3D and E). The summarized data in Figures 3C and F further illustrate the magnitude of these differences where nifedipine (fetal N=9/3; adult N=14/3), verapamil (fetal N=11/4; adult N=9/3), or diltiazem (fetal N=11/4; adult N=9/3) reduced high-K+-induced contraction to a greater extent in adult compared to fetus with respect to controls (fetal N=10/4; adult N=17/6). Interestingly, LTH did not substantially impact the influence of CaL blockers on high-K+-induced contraction per se (Figs. 2 and 3).
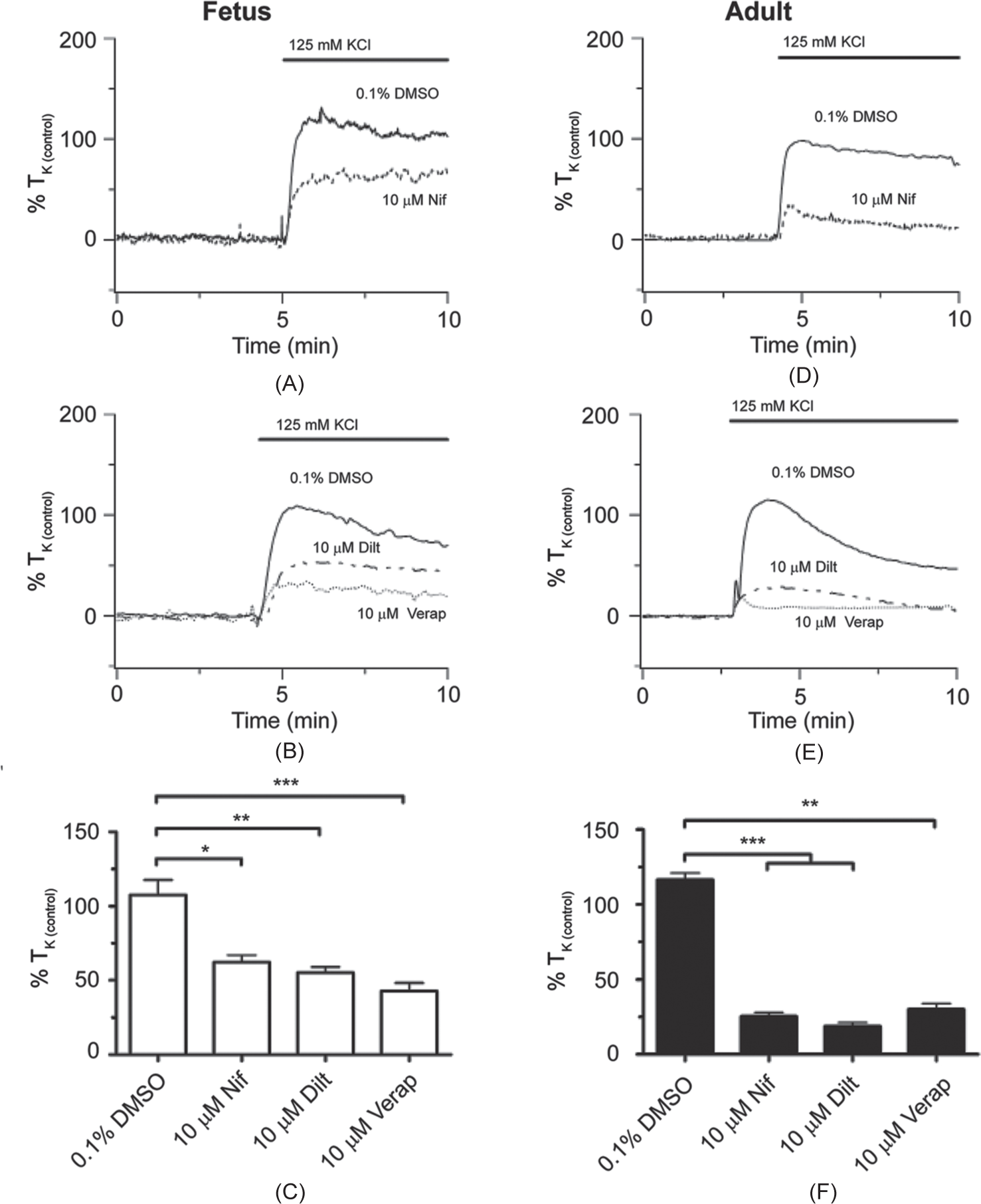
The L-type Ca2+ channel is important for high-K+ -elicited pulmonary arterial contractility in LTH fetal and adult sheep. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 10 μM nifedipine (Nif; dashed), 10 μM verapamil (verap; dotted), or 10 μM diltiazem (dilt; dotted and dashed) from fetus (A and B) or adult (D and E). Tracings are plotted in relation to maximal contraction from initial high-K+ stimulation (% TK (control)). (C and F) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or L-type Ca2+ channel antagonists for pulmonary arteries from fetus (C; open) and adult (F; solid) sheep. *(P<0.05), **(P<0.01), ***(P<0.001) denotes significant difference by one-way ANOVA.
To delineate further the extent of the changes in high-K+-mediated contraction by maturation and LTH, we applied cumulative doses of KCl to pulmonary arterial segments from normoxic and LTH animals. The dose-response data shown in Figure 4 for fetal normoxic (Fig. 4A; N=11/3), adult normoxic (Fig. 4B; N=12/4), fetal hypoxic (Fig. 4C; N=18/6), and adult hypoxic (Fig. 4D; N=16/6) show that pulmonary arteries constrict to low concentrations of KCl, with a maximal response by 30 to 40 mM. The summarized data in Figure 4E show that the EC50 was approximately 25 mM in all four animal groups, and was unaffected by either maturation or LTH.
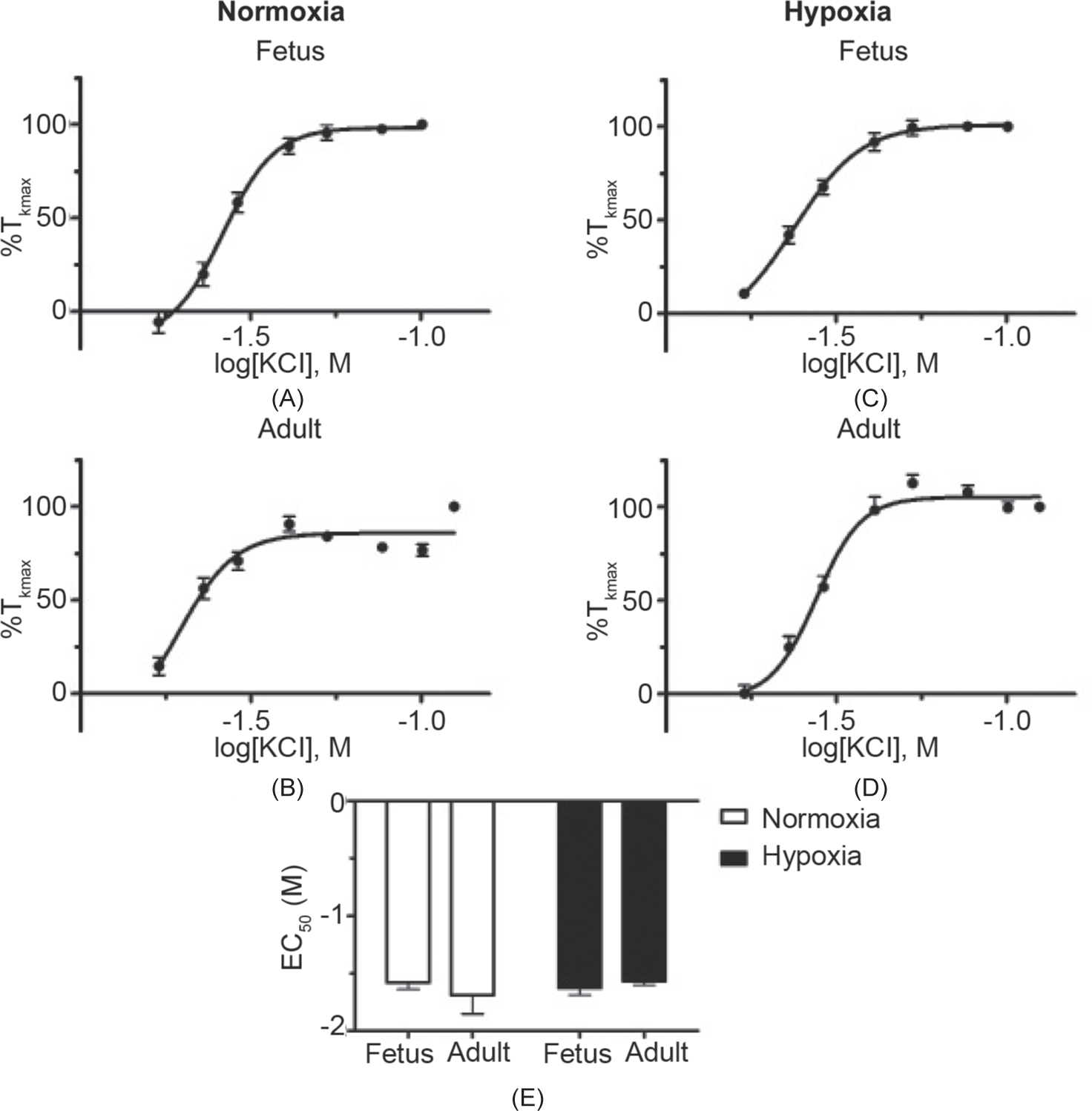
The potency for high-K+ -induced contraction is unaffected by maturation or LTH. Dose response relationships based on isometric recordings of pulmonary arterial rings stimulated with 4 mM to 125 mM potassium in a cumulative manner for fetal and adult sheep exposed to normoxic (A and B) or long-term hypoxic conditions (C and D). Data were fitted with a Hill equation to the mean values normalized to %TKmax (percent contraction compared to the maximal high-K+ contraction). (E) Comparison of mean±S.E.M. for EC50 values between normoxic (open) and LTH (solid) in pulmonary arteries of fetal and adult sheep. No significant differences were observed between the EC50 based on a two-way ANOVA.
The next series of experiments examined the role of Rho-kinase in high-K+-induced contraction, and determined the interaction between Rho-kinase and CaL. These studies were based on experiments performed on rodents in which Rho-kinase contributed to potassium-induced contraction, and LTH augmented the contribution of Rho-kinase.[8] In these experiments, Rho-kinase was inhibited with 10 μM Y-27632 while CaL was blocked with 10 μM nifedipine. Figures 5A and B show normalized isometric tension recordings from normoxic fetal vessels while Figures 5D and E show representative recordings from adult vessels. In the fetus, 10 μM Y-27632 and the combination of 10 μM nifedipine and 10 μM Y-27632 reduced high-K+-induced pulmonary artery contraction to a much greater extent than 10 μM nifedipine alone (Fig. 5A and B).
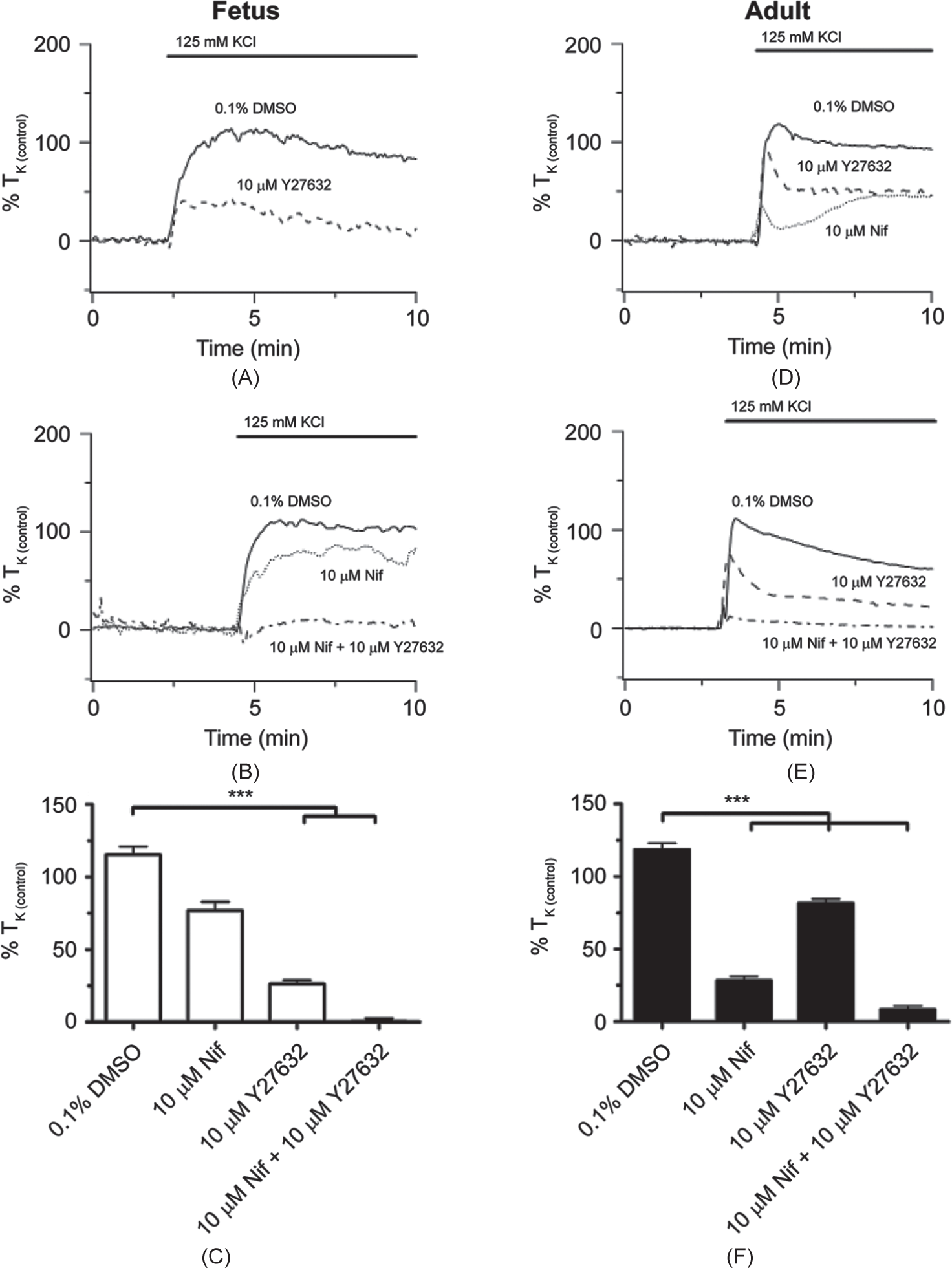
Maturation increases the role for Rho-kinase during high-K+-elicited pulmonary arterial contractility in sheep housed in normoxic conditions. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 10 μM Y-27632 (dashed), 10 μM nifedipine (Nif; dotted), or 10 μM Y-27632 + 10 μM nifedipine (Y-27632 + Nif; dotted and dashed) from fetus (A and B) or adult (D and E). Tracings are plotted in relation to maximal contraction from initial high-K+ stimulation (% TK (control)). (C and F) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or antagonists for pulmonary arteries from fetus (C; open) and adult (F; solid) sheep. ***(P<0.001) denotes significant difference by one-way ANOVA.
The findings in pulmonary arteries from normoxic adult sheep were distinct from those of fetuses. Figure 5D shows that 10 μM Y-27632 decreased arterial contraction modestly while 10 μM nifedipine reduced contraction somewhat more. Figure 5E then shows the combination of 10 μM nifedipine and 10 μM Y-27632 reduced contraction more substantially as compared to either 10 μM nifedipine or 10 μM Y-27632 alone. The summary data shown in Figures 5C and Fillustrate the differences in the actions of nifedipine and Y-27632 on high-K+ -induced contraction in pulmonary arteries from fetal as compared to adult. Even though the combination of 10 μM Y-27632 and 10 μM nifedipine (fetus N=9/3; adult N=16/3) reduced high-K+-induced contraction relative to control (fetus N=16/6; adult N=19/6) in both fetuses and adults, these summary data illustrate that 10 μM Y-27632 was more effective in arteries from fetus (fetus N=15/3; adult N=11/3) while 10 μM nifedipine was more effective in arteries from adult (fetus N=9/3; adult N=11/3) sheep.
Parallel studies were performed in pulmonary arteries from hypoxic sheep with equivalent findings. Results of these experiments are shown in Figure 6. Figures 6A and B show normalized isometric tension recordings from hypoxic fetal vessels, and Figures 6D and E show representative recordings from hypoxic adult vessels. In the fetus, the representative traces and summarized data (Fig. 6C) illustrate that 10 μM Y-27632 (N=12/4) alone as well as the combination of 10 μM Y-27632 and 10 μM nifedipine (N=9/4) reduced high-K+-induced arterial tension substantially, whereas 10 μM nifedipine alone (N=12/4) reduced contraction modestly with respect to control (N=9/4). In the adult, the representative and summary data (Fig. 6E) demonstrate that 10 μM Y-27632 (N=13/4) and 10 μM nifedipine (N=11/4) alone reduced high-K+-induced arterial tension by a similar, modest amount, while the combination of both antagonists (N=10/4) decreased it extensively compared to control (N=10/4).
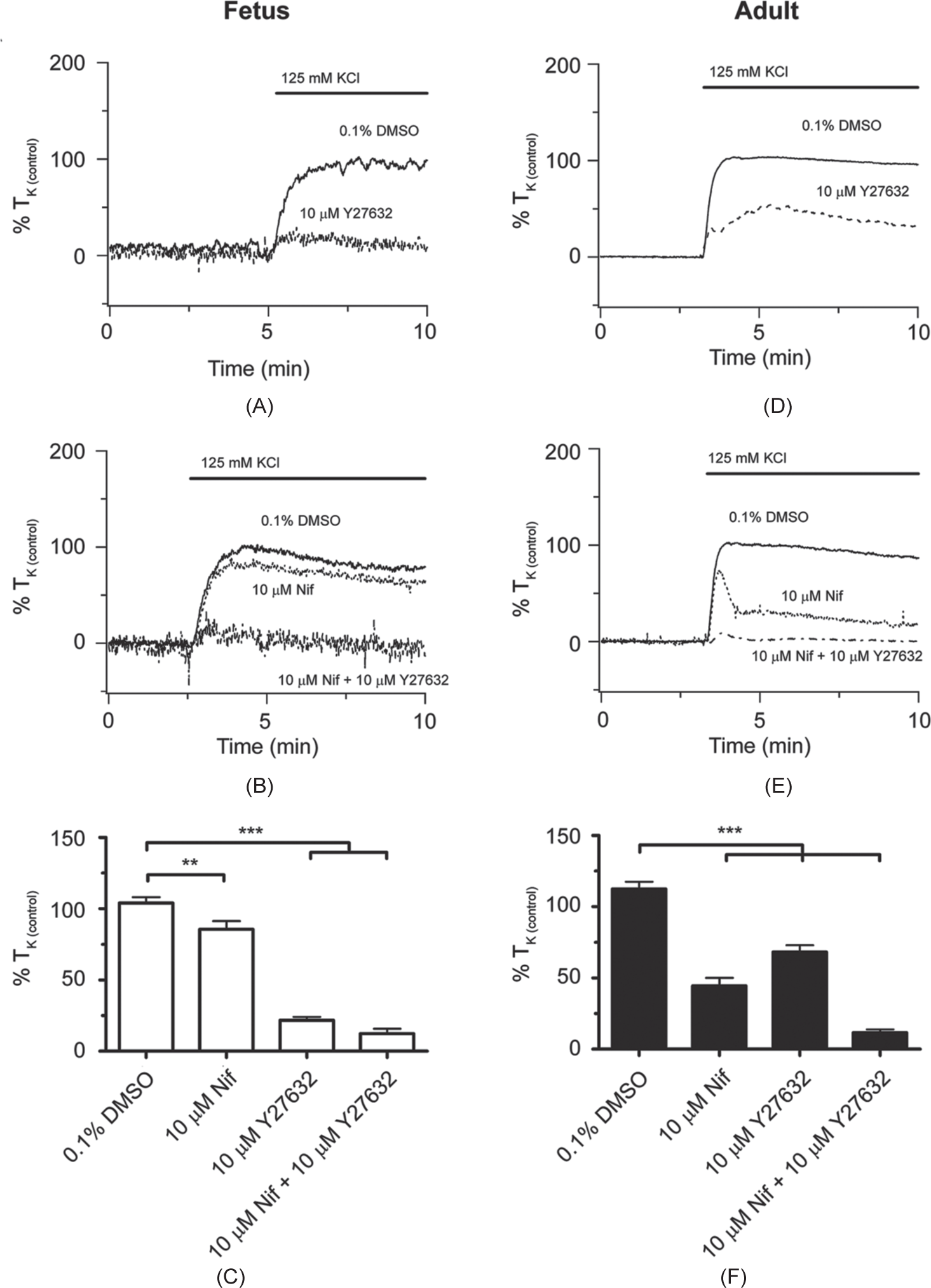
LTH does not greatly affect the role of Rho-kinase and the L-type Ca2+ channel during high-K+-elicited pulmonary arterial contractility in sheep. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 10 μM Y-27632 (dashed), 10 μM nifedipine (NIF; dotted), or 10 μM Y-27632 + 10 μM NIF (Y-27632 + NIF; dotted and dashed) from fetus (A and B) or adult (D and E). Tracings are plotted in relation to maximal contraction from initial high-K+ stimulation (% TK (control)). (C and F) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or antagonists for pulmonary arteries from fetus (C; open) and adult (F; solid) sheep. ***(P<0.001), **(P<0.01) denotes significant difference by one-way ANOVA.
In the next series of studies, we determined the extent to which high-K+-induced contraction is dependent on superoxide. These experiments were performed based on recent publications indicating that membrane depolarization can generate superoxides, which in turn activate Rho-kinase-dependent contraction.[8,19] The role of superoxides was addressed by using two inhibitors. NADPH-induced superoxide generation was inhibited with apocynin (100 μM), which has been used to examine the role of superoxides to newborn piglet pulmonary arterial reactivity.[20,21] Superoxide degradation to peroxide was increased by application of the superoxide dismutase mimetic tempol (3 mM), which has been used in contractility studies of pulmonary arteries.[22] Figures 7A and C show normalized isometric tension recordings from normoxic fetal and adult vessels, respectively. For fetal arteries, the summary data in Figure 7B show that superoxide was not critical to high-K+-induced contraction, as neither apocynin (N=14/4), or tempol (N=14/4) altered contraction relative to control (N=12/4). However, Figure 7D shows that, in the adult, tempol (N=14/4) but not apocynin (N=15/4) reduced high-K+-induced pulmonary arterial contraction by approximately 20% with respect to the control (N=12/4).
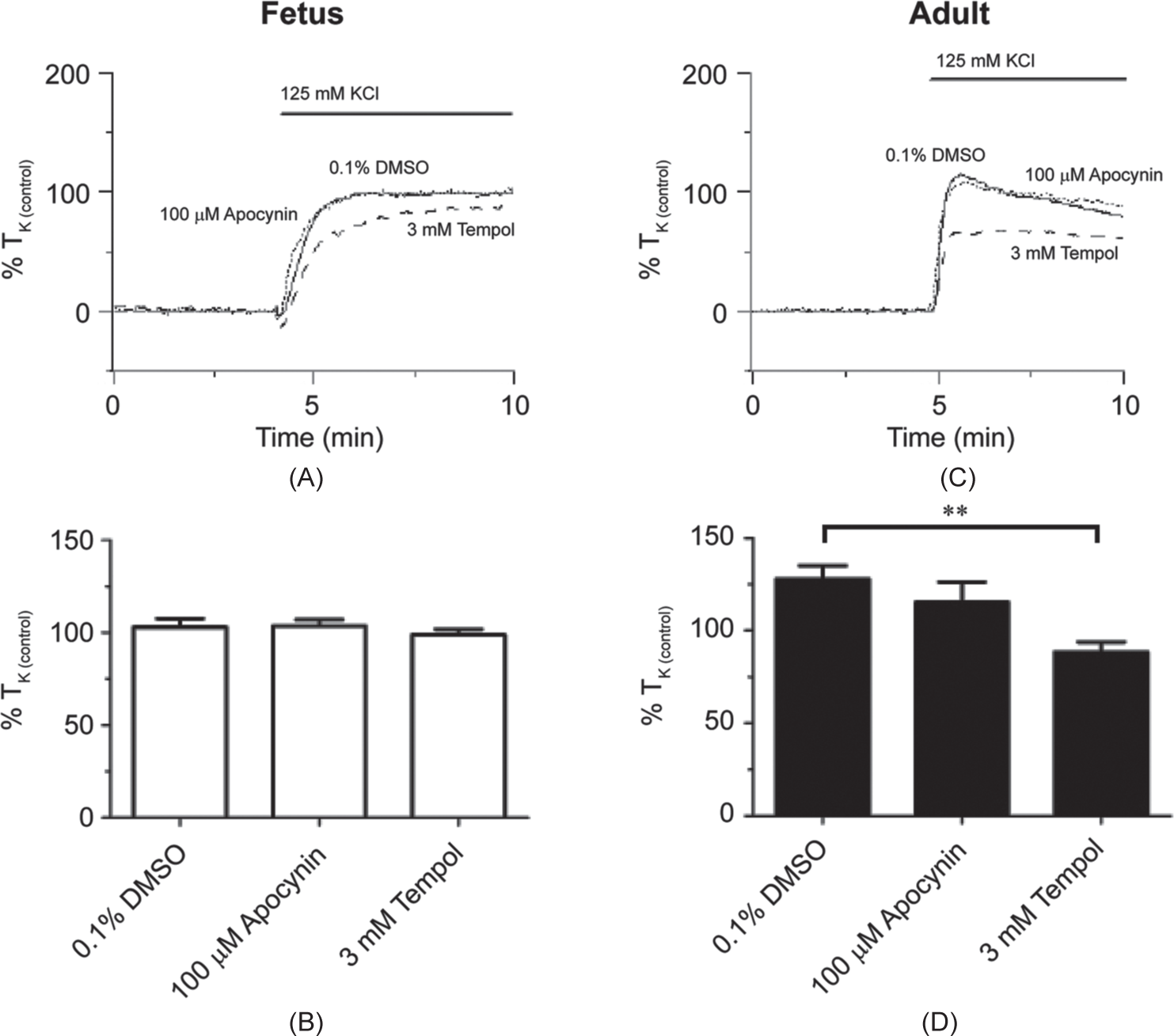
Maturation alters the role for superoxide during high-K+-elicited pulmonary arterial contractility in sheep housed in normoxic conditions. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 100 μM apocynin (dotted), 3 mM tempol (dashed) from fetus (A) or adult (C). Tracings are plotted in relation to maximal contraction from initial high-K stimulation (% TK (control)). (B and D) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or antagonists for pulmonary arteries from fetus (B; open) and adult (D; solid) sheep. **(P<0.01) denotes significant difference by one-way ANOVA.
The results of similar experiments performed in hypoxic sheep are shown in Figure 8. Figures 8A and C show normalized isometric tension recordings from hypoxic fetal and adult arteries, respectively. The summary data of Figures 8B and D show that in both immature and mature arteries tempol (fetus N=16/4; adult N=16/4) reduced contraction relative to control (fetus N=16/4; adult N=12/4) while apocynin failed to do so (fetus N=17/4; adult N=15/4).
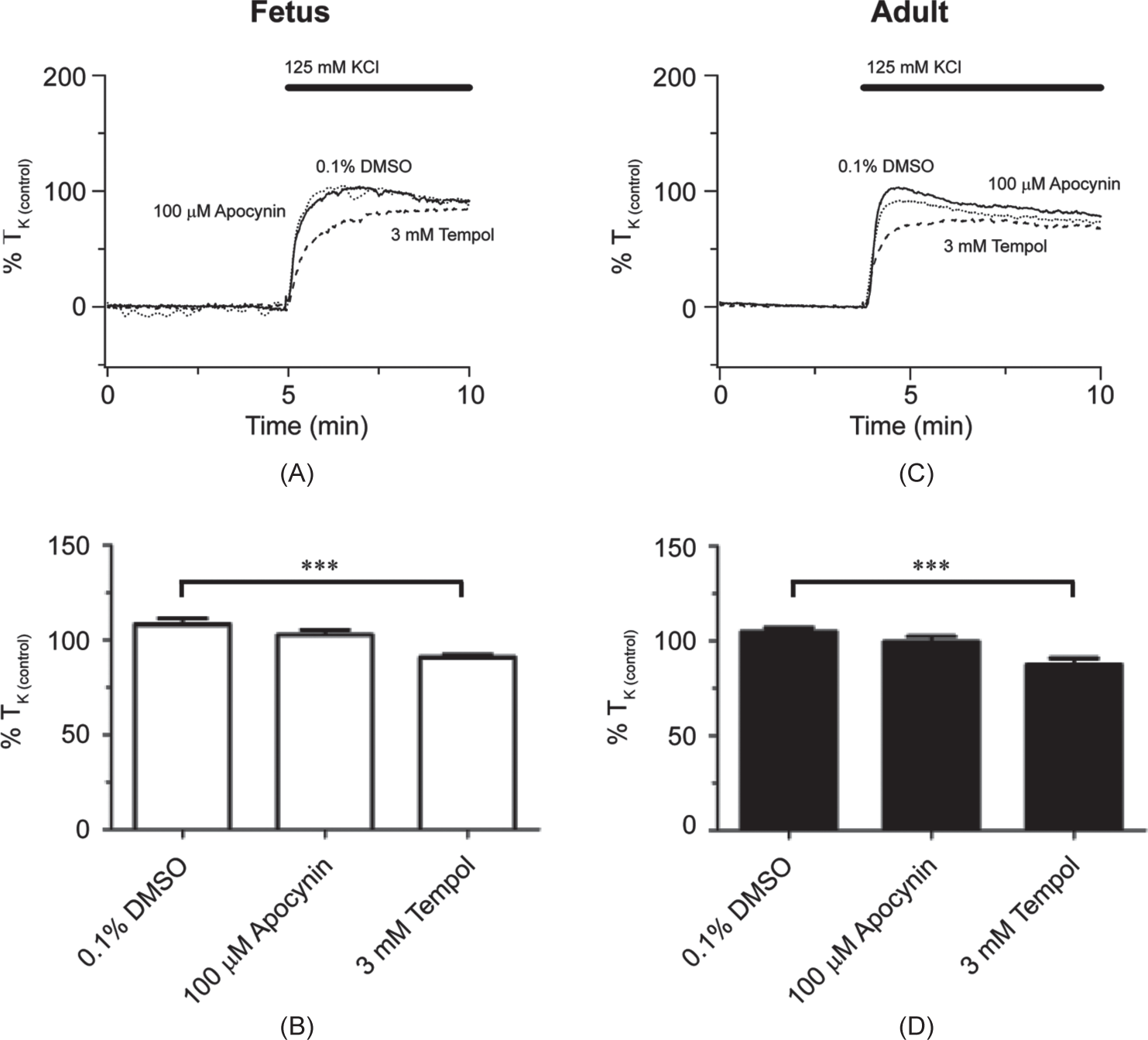
LTH increases the role for mitochondrial-derived superoxide during high-K+-elicited pulmonary arterial contractility in fetal sheep. Isometric tension recordings of pulmonary arterial rings constricted in the presence of 0.1% DMSO (solid), 100 μM apocynin (dotted), or 3 mM tempol (dashed) from fetus (A) or adult (C). Tracings are plotted in relation to maximal contraction from initial high-K+ stimulation (% TK (control)). (B and D) Bars indicate mean±S.E.M. of high-K+-induced contraction expressed as % TK (control) in the presence of vehicle control or antagonists for pulmonary arteries from fetus (B; open) and adult (D; solid) sheep. ***(P<0.001) denotes significant difference by one-way ANOVA.
Because arterial tension is based on the contraction of myocytes in the arterial wall, we performed a series of studies to determine if there might be differences in the proportion of the active smooth muscle between fetal and adult tissues. We quantified the active smooth muscle by measuring the proportion of alpha smooth muscle actin relative to the total protein content. Figure 9 shows the results of these Western immunoblot studies. Figure 9A shows a representative immunoblot for alpha smooth muscle actin from individual arteries, where each lane was loaded with 10 μg of sample. The density of the fetal bands was not dramatically different as compared to the bands from adult arteries. Figure 9B summarizes results from experiments performed in 22 replicates from a total of 6 animals for each experimental group, where replicates were run on separate gels. The figure demonstrates that alpha smooth muscle actin expression was ~10–15% lower in the fetus but expression was not systematically influenced by LTH.
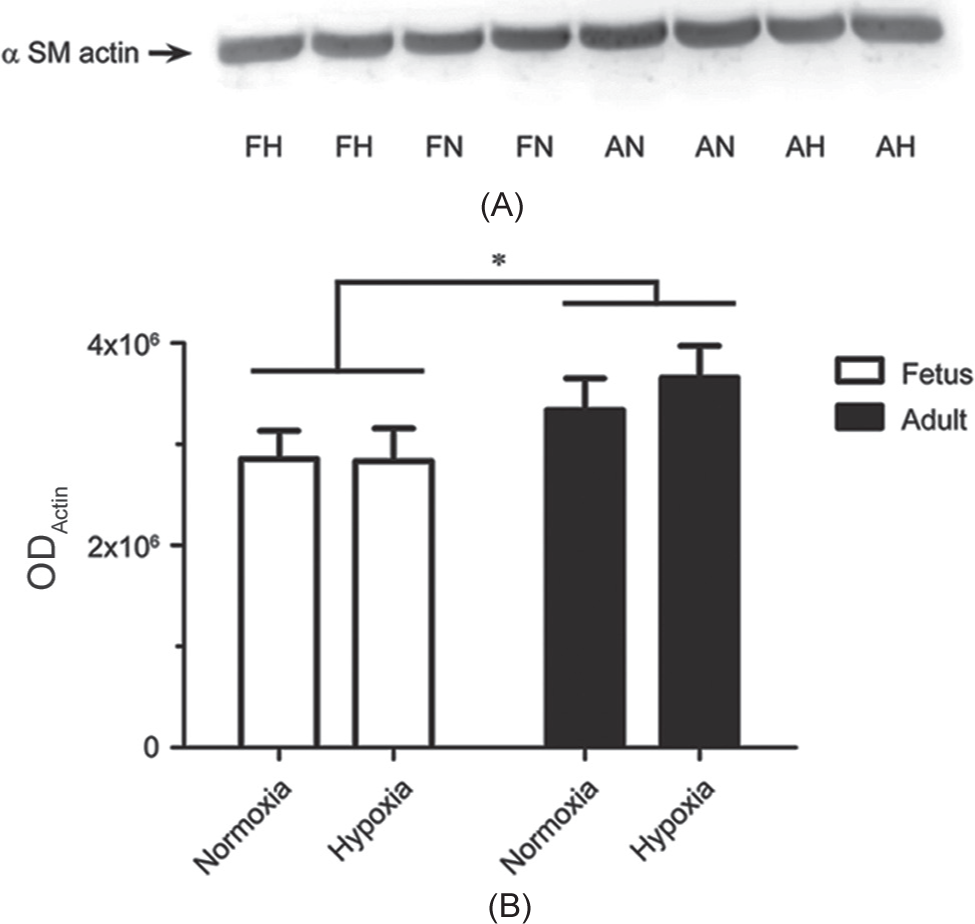
Maturation modestly increases a smooth muscle (SM) actin expression. (A) Western blot result for a SM actin abundance in pulmonary artery homogenates. Equivalent masses of total protein (10 μg) were loaded on each lane (FN: Fetal normoxic; FH: Fetal LTH; AN: Adult normoxic; AH: Adult LTH). (B) Bars indicate mean±S.E.M. based on densitometric quantification of alpha smooth muscle actin abundance in pulmonary arterial preparations (ODactin: Optical density of alpha smooth muscle actin). *(P<0.05) denotes significant difference based on cumulative fetal versus adult densitometries based on a two-way ANOVA. No significant differences were noted based on level of oxygenation or between individual fetal and adult groups for each level of oxygenation.
DISCUSSION
The present study is the first comparison of depolarization-induced contraction in pulmonary arteries from high-altitude acclimatized fetal and adult sheep, compared to normoxic controls. Key findings of these studies include these seven. (1) High-K+-induced pulmonary arterial contraction is considerably blunted in fetal, as compared to adult sheep. (2) Maturation-related increase in the expression of contractile proteins may account for a portion of this difference. (3) The role of CaL in high-K+-mediated contraction is blunted in fetal relative to adult sheep. (4) The role of Rho-kinase is significantly enhanced in fetal relative to adult sheep. (5) High-K+-dependent contraction and the role of CaL and Rho-kinase are preserved following LTH in both fetal and adult sheep. And (6) superoxide plays a minor role to high-K+-induced contraction of pulmonary arteries in sheep; however (7), its role is modestly increased after birth and by LTH in the fetus.
The finding that pulmonary arterial contractility is highly sensitive to low-K+-concentrations with a maximal response at ~30 mM was somewhat surprising. Based on the extracellular and estimated intracellular potassium concentrations, the Nernst equation predicts that 30 mM K+ would depolarize the plasma membrane to ~40 mV, which is below the predicted membrane potential that activates CaL in pulmonary arterial myocytes.[11,23] Secondarily, because the EC50 for K+-induced contraction was not altered by either maturation or LTH, the relevant process or processes are likely developed before birth and unaffected by LTH. One possible explanation for the substantial contraction at relatively negative membrane potentials is that membrane depolarization activates low-threshold T-type Ca2+ channels, which could contribute to depolarization dependent contraction and activation of CaL.[24,26] Alternatively, other channels may be activated even at relatively negative membrane potentials. In particular, voltage-gated Na+ channels could be activated by depolarization, which then further depolarize the membrane leading to the opening of CaL.[27,28]
The pharmacological studies comparing different inhibitors of CaL expand on the details provided by the potassium dose-response studies. These studies also illustrate CaL and other signaling pathways, are important to high-K+-induced pulmonary arterial contraction. Secondarily, given that dihydropyridines, benzothiazepines, and phenylalkylamines bind to different amino acid residues in the pore region of CaL, our data indicate that the permeation pathway for these channels in fetuses and adults is structurally similar and unaffected by LTH.[29] In support of this supposition, there is evidence that in rat hearts the expression of CaL increases after birth although their structure appears similar.[30]
The finding that adult arteries are far more reactive to high-K+ than those of the fetus was not surprising. Recent work has shown a similar result in ovine middle cerebral arteries.[12] As a litmus test for the amount of active smooth muscle in the arterial wall, and in an attempt to resolve the source of developmental change in arterial reactivity, we compared the ratio of the total protein content to that of alpha-smooth muscle actin. These measurements are indicative of small, generalized increases in the expression of smooth muscle after birth, which may contribute to the increased arterial reactivity.[12] The mild increase of alpha smooth muscle actin may also reflect a potential increase in expression of other contractile machinery components. As indicated by the varied roles for CaL versus Rho-kinase, the majority of the increases in contraction due to maturation are likely mediated through more complex alterations in cellular signaling.
The finding that Rho-kinase is important to K+-induced contraction was expected, as membrane depolarization and concomitant Ca2+ influx can activate Rho-kinase,[7,31] which subsequently causes pulmonary arterial contraction.[8,32–34] We did not anticipate, however, the substantial difference in the role of Rho-kinase to high-K+-dependent contraction between fetal and adult pulmonary arteries. We were also surprised that Y-27632 did not suppress contraction to a greater extent in arteries from hypoxic fetal and adult sheep, as LTH increases Rho-kinase II expression, myosin light-chain phosphatase phosphorylation and role to Endothelin-1 (ET-1) dependent contraction in pulmonary arteries of fetal sheep.[35] LTH also increases the expression of RhoA and the role of Rho-kinase to KCl contraction in adult rats.[8] In our studies, Y-27632 inhibited contraction to a similar extent in both normoxic and hypoxic animals. One notable difference between our experiments and the previous work in pulmonary arteries of LTH fetal sheep[35] is that we contracted pulmonary arteries with high-K+ while the previous study contracted them with ET-1. Furthermore, another recent study by Gao and co-workers performed on fetal sheep pulmonary veins, shows LTH reduced Rho-kinase II expression, cGMP-dependent relaxation via Rho-kinase pathways, and arachidonic acid induced activity.[36] Possibly, the augmented importance of Rho-kinase in pulmonary arteries of LTH fetal sheep[35] versus the lack of an increase in the present experiments may, in part, reflect differential Ca2+-dependent activation of Rho-kinase.[37,38] In particular, Rho-kinase activation can be either reliant or independent of increases in cytosolic Ca2+, where G-protein receptor activation may couple to Rho-kinase activation to a different extent as compared to K+-depolarization dependent elevations in cytosolic Ca2+. For example, Rho-kinase activation can occur independently of changes in cytosolic Ca2+ in renal tubules as well as in renal and femoral arteries.[39,40] Recent evidence indicates that such Ca2+-independent activation mechanisms also exist in rat pulmonary arteries.[8,9] With respect to studies in adult rats that show LTH increases the importance of Rho-kinase to K+-dependent contraction, explaining the differences from our studies is less clear. Conceivably, sheep are more resilient to LTH than rats. Whereas pulmonary arteries from adult rats have an increased role for Rho-kinase and enhanced contraction,[8,19] those of sheep are possibly maintained. Previous studies in sheep support this proposition, as long-term high-altitude hypoxia did not alter the maximum tension developed by ET-1 or serotonin in PA from sheep fetuses[3,35] or adults.[4]
The small role of superoxides to high-K+-induced contraction in the sheep, relative to the rat, also was unexpected. Published evidence shows that mitochondria-generated superoxides are an important component to K+-elicited contraction in rat pulmonary arteries.[8] In fetal sheep, we found no role for apocynin-dependent NADPH-mitochondrial-generated superoxides in high-K+-dependent contraction, while in the adult and fetal hypoxic arteries tempol-superoxide dismutase related non-NADPH-generated superoxide accounted for a small proportion of the contraction. This is indicative of species variability in terms of the acclimatization response to LTH.[3,4,8] As such, it will be of interest to determine the extent to which blunted superoxide responses in the sheep mitigate the deleterious influences of LTH on pulmonary vascular function.
There are several potential impacts of the increase in K+-dependent contraction after birth, and the shift from Rho-kinase to CaL-dependent contraction. In particular, the reduced pulmonary arterial contraction in the fetus may reflect the reduced pre-birth lung blood flow due to a patent and diverting ductus arteriosus. Moreover, the fetal pulmonary vasculature is characterized by a decreased ability for sustained vasodilation.[41,42] Nevertheless, Rho-kinase inhibitors achieved sustained vasodilation in late-gestation fetal lambs, suggesting that high Rho-kinase activity contributes substantially to maintenance of increased pulmonary vascular resistance in the fetal lung.[33,43] Secondarily, the increased activity of Rho-kinase in fetal arteries also may contribute to sustained contraction by preventing myosin dephosphorylation while Ca2+ influx is limited. This could conserve energy for other tasks, such as cell growth, as the rates of phosphorylation and dephosphorylation are potentially slowed in the fetal vessels relative to those of the adult. The end result would be a relatively stable and sustained arterial contraction, albeit with potentially reduced force generation. Lastly, the data suggest that while CaL therapies more effectively vasodilate pulmonary arteries in adults, and Rho-kinase therapies are more effective vasodilators in newborns, combination therapies would provide greater efficacy in both young and mature patients.
Footnotes
ACKNOWLEDGMENTS
We would like to acknowledge Srilakshmi Vemulakonda, Rachael Wilson, Nathan Matei, and Nina Chu for expert technical assistance.
