Abstract
Schistosomiasis-associated pulmonary arterial hypertension (PAH) is one of the most common causes of pulmonary hypertension worldwide. A potential contributing mechanism to the pathogenesis of this disease is a localized immune reaction to retained and persistent parasite-derived antigens. We sought to identify Schistosoma-derived egg antigens present in the lungs of individuals who died of the disease. We obtained 18 lung samples collected at autopsy from individuals who died of schistosomiasis-associated PAH in Brazil. A rabbit polyclonal antibody was created to known
INTRODUCTION
Schistosomiasis affects over 200 million people in 74 countries, where it causes more than 250,000 deaths and up to 4.5 million disability-adjusted years lost annually.[1] Approximately 10% of those chronically infected with
The pathogenic mechanism by which this parasitic infection results in pulmonary vascular remodeling is unclear. Potential contributing factors include portal hypertension with resulting portopulmonary hypertension and/or egg embolism, and a host immune responses that may be systemic and/or locally directed at parasite antigens in the lung. Such a localized inflammatory response in particular could be directed at persistent antigens or, once initiated, could continue despite clearance of the inciting antigenic material.
Histologic examination of lung tissue from individuals with schistosomiasis-associated PAH reveals a dark pigment that is often located adjacent to sites of vascular remodeling, the nature of which is unclear, and which has historically been variously speculated to be derived from red blood cells,[6] “bile pigment,”[7] a component of scar tissue[7,8] or remnants of the parasite.[6,8–10] To clarify the nature of this pigment and potentially identify antigens that could be the target of a localized host inflammatory response, we sought to detect parasite egg antigens in the lung tissue from individuals who had died of schistosomiasis-associated PAH.
MATERIALS AND METHODS
Sources of human tissue
Tissue from patients who died of schistosomiasis-associated PAH was obtained from two centers in Brazil: Memorial S. Jose Hospital, Universidade de Pernambuco in Recife, Pernambuco; and Hospital Prof. Edgard Santos, Universidade Federal da Bahia, Salvador. This tissue had been previously collected at autopsy and was formalin fixed and paraffin embedded. As the material was derived from deceased persons, no Institutional Review Board approval was required.
Sources of mouse tissue
We developed an experimental mouse model of schistosomiasis-associated pulmonary hypertension.[11] Briefly, wild-type C57Bl6/J mice (Taconic) receive 5,000
Polyclonal antibody production and immunoblotting
A rabbit polyclonal antibody was prepared to hold
The ability of the generated antibody to detect proteins in SEA was tested by probing a Western blot of purified SEA. Pre- and post-immunization serum was applied to the Western blot membrane at a concentration of 0.1 ug/mL overnight at 4°C. The immunoblot secondary antibody was HRP-labeled goat anti-rabbit (Vector, Burlingame, CA, PI-1000), used at a concentration of 1:5,000 for 1 h at room temperature, and detected using enhanced chemiluminescence (GE Healthcare, Little Chalfont, UK, RPN2106, RPN2106). Mouse whole lung lysates prepared by macerating and sonicating samples of the frozen right lung tissue in buffer containing antiproteases were also probed using the anti-SEA antibody.
Tissue immunostaining
Sections of large intestine and lung from individuals with schistosomiasis-associated PAH and sections of lung from mice infected with
Sections of infected mouse lung tissue were stained with both the rabbit anti-SEA antibody and a rat anti-Mac-3 antibody to identify macrophage lysosomes.[14] The sections were heated at 100°C in Borg buffer for 20 min (Biocare #BD1000G1); blocked with a mixture of 10% horse serum, 10% goat serum, 40% Superblock (ScyTek AAA5000) and 40% of 5% bovine serum albumin reconstituted in PBS for 1 h; the combination of anti-SEA antibody (either preimmunization serum as a negative control or the postimmunization antibody) at a concentration of 5 μg/mL and anti-Mac-3 antibody (BD Pharmingen #550292) or rat IgG (negative control) at a dilution of 1:50 applied for 1 h at room temperature; and a combination of secondary antibodies of AF594-labeled donkey anti-rabbit (Invitrogen A21207) and AF488-labeled goat anti-rat (Invitrogen A11006) each diluted 1:200 applied for 1 h.
Human and mouse immunofluorescence-stained sections were counterstained with DAPI and imaged with a Nikon Eclipse E800 microscope with either a color camera (Nikon) or a black and white CCD camera (Photometrics). The double immunofluorescence stain of anti-SEA and anti-Mac-3 was imaged using a Zeiss LSM 510 META confocal microscope system with a 100x oil-immersion objective.
RESULTS
We obtained and analyzed pulmonary tissue from 18 patients who had died of schistosom iasis-associated PAH. Histologically, all lung samples had evidence of pulmonary vascular remodeling (Fig. 1). All the samples (100%) contained plexiform lesions, and 16 of 18 (89%) had evidence of arterial medial thickening. Only four of the 18 (22%) samples had granulomas. Although described by others,[15,16] we did not observe any intact
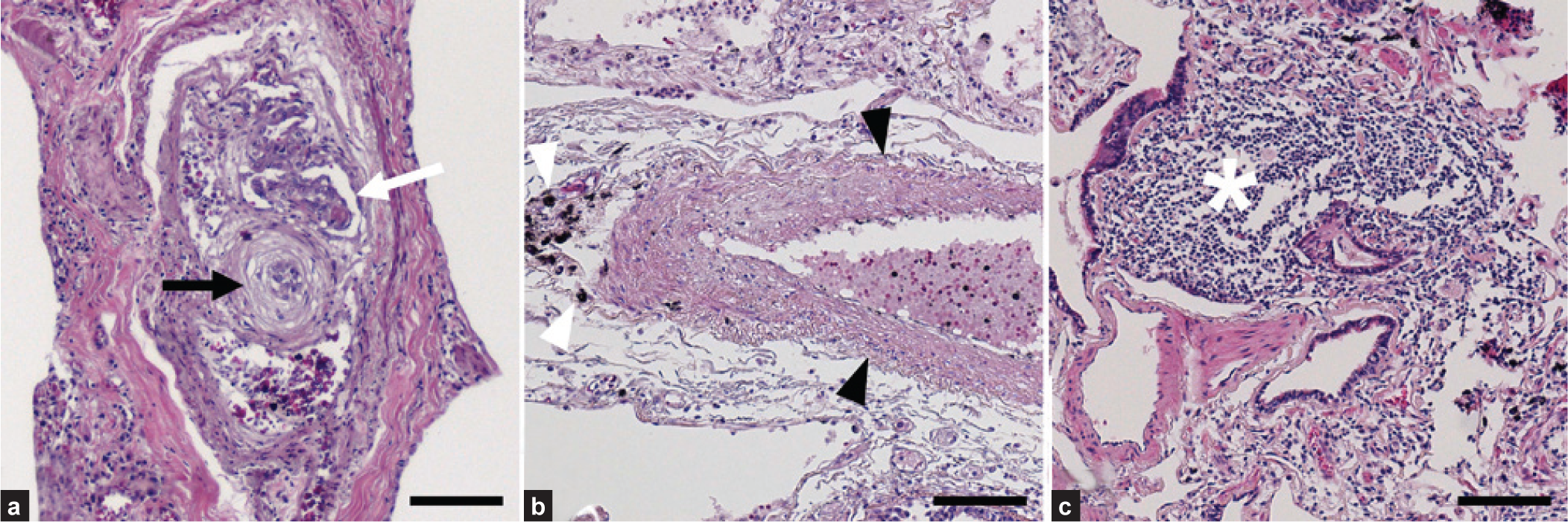
Representative pulmonary pathology of schistosomiasis-associated pulmonary arterial hypertension. (a) Plexiform lesion (white arrow) in close proximity to concentric (onion-skinned) intimal thickening (black arrow). (b) Increased medial thickness (black arrowheads) adjacent to dark pigment (white arrowheads). (c) A perivascular granuloma (white star). All stains are hematoxylin and eosin. Scale bars are 100 μm.
Range of histopathologic finding in 18 autopsy specimens from patients who died of schistosomiasis-associated PAH

Severity scores: (+++) severe, (++) moderate, (+) mild, (0) none
To identify antigenic material derived from
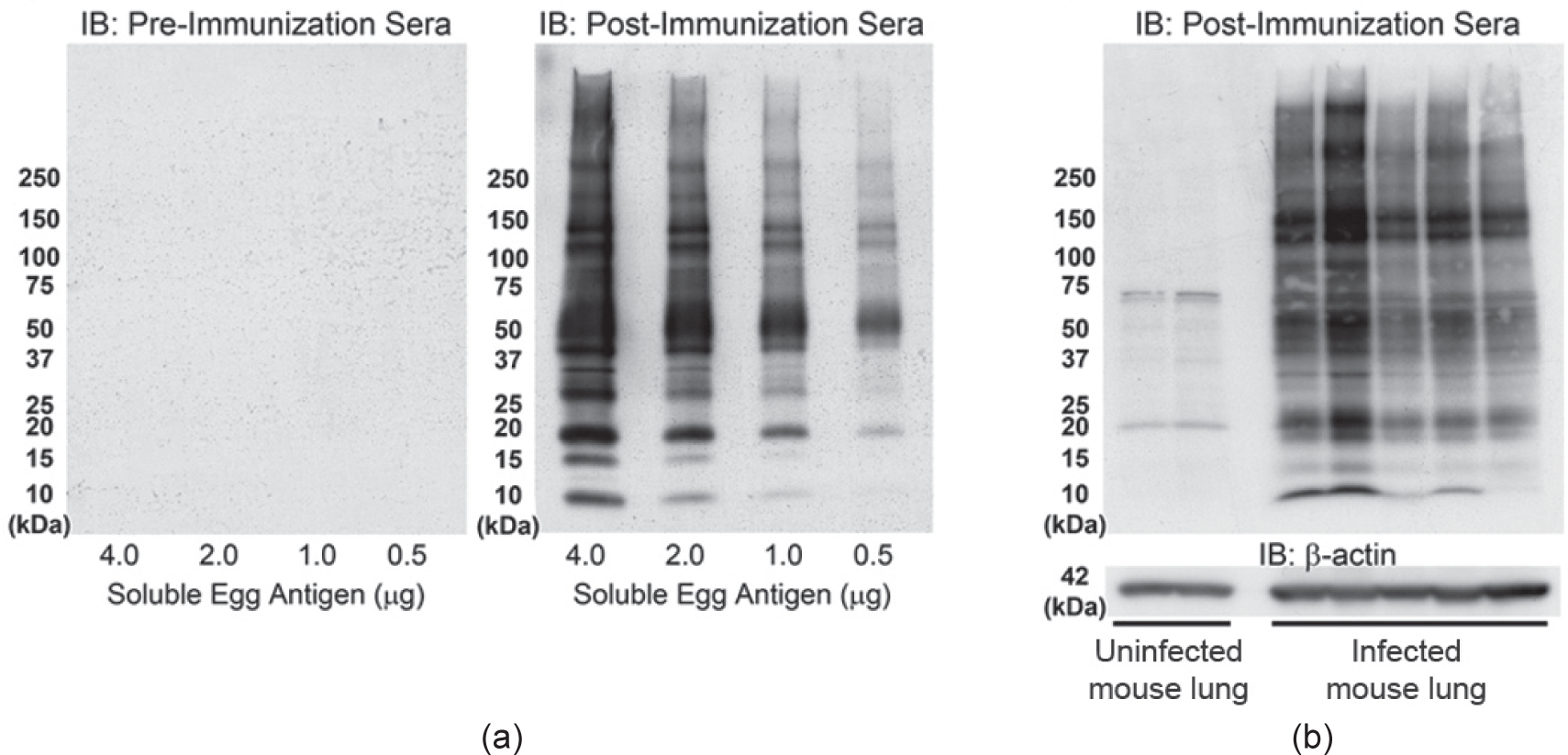
Immunoblots using
We had previously developed an experimental mouse model of schistosomiasis-associated pulmonary hypertension, which utilizes intraperitoneal sensitization with
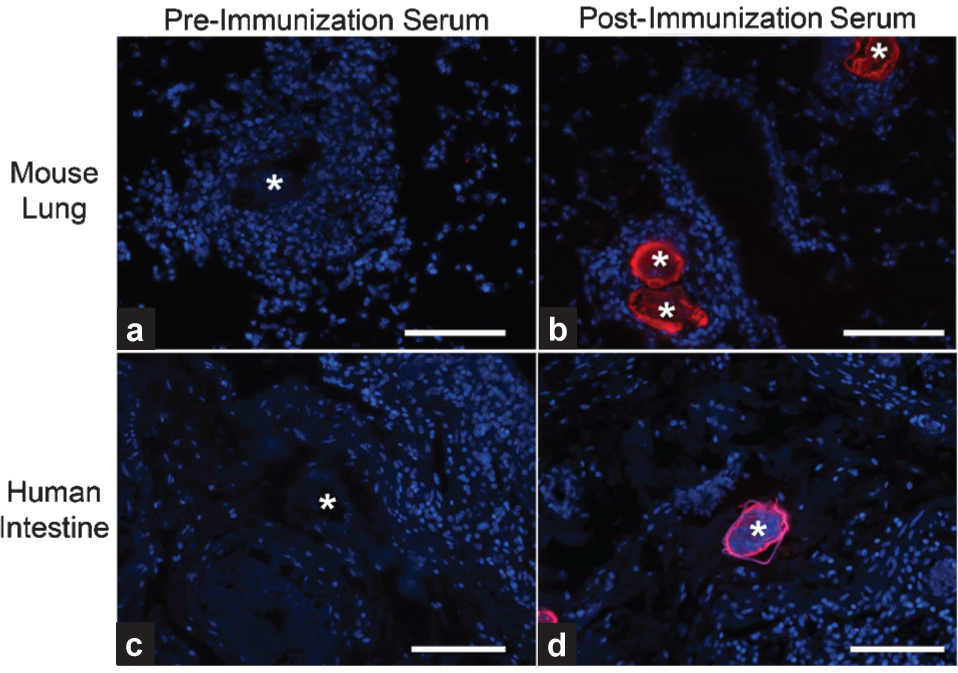
Anti-
We performed anti-SEA immunostaining of human tissue to detect
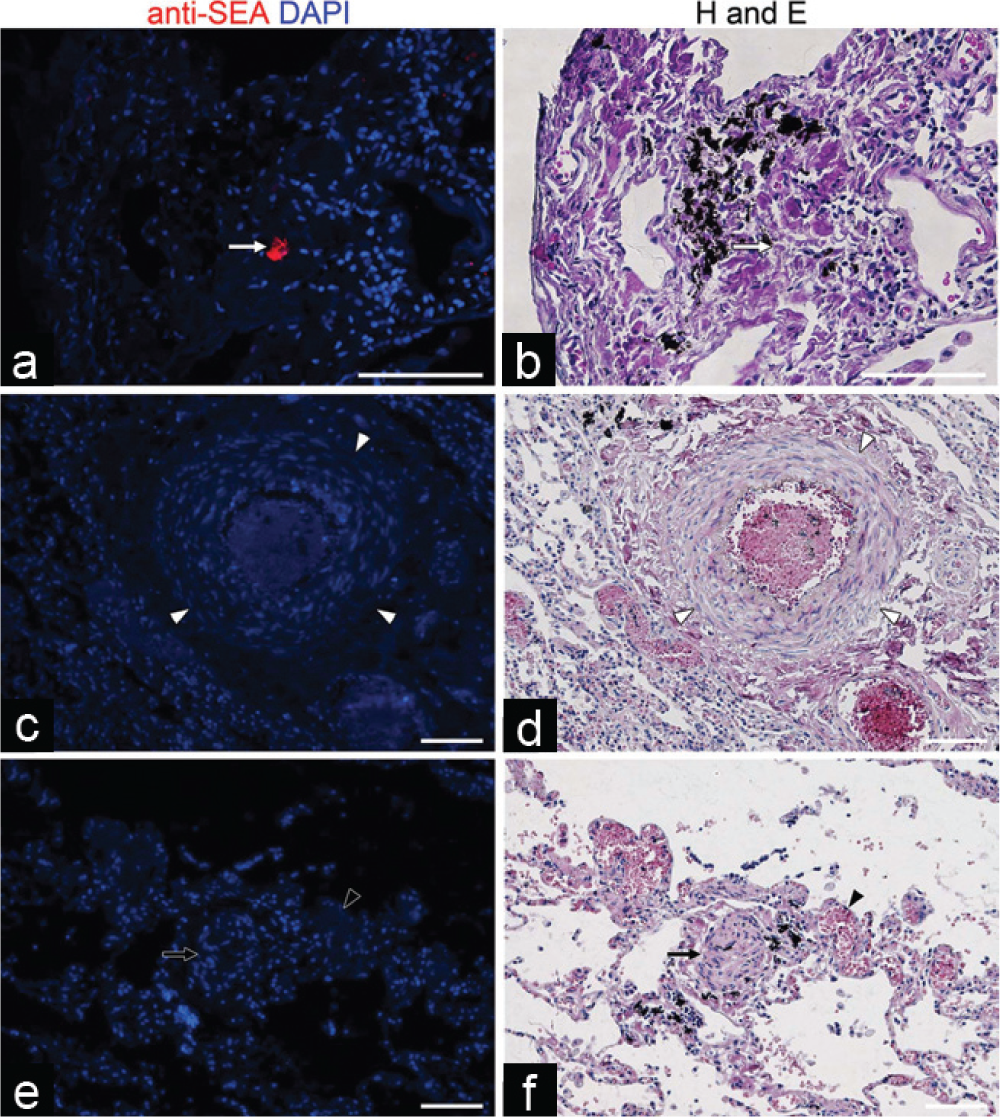
Anti-
We sought to localize the degraded egg fragments within granuloma cells. Costaining the mouse lung tissue for Mac-3/CD107b (a marker of macrophage plasma membranes and lysosomes[14]) and anti-SEA revealed that in granulomas encompassing degraded eggs, there were macrophage lysosomes containing intracellular egg antigens (Fig. 5).
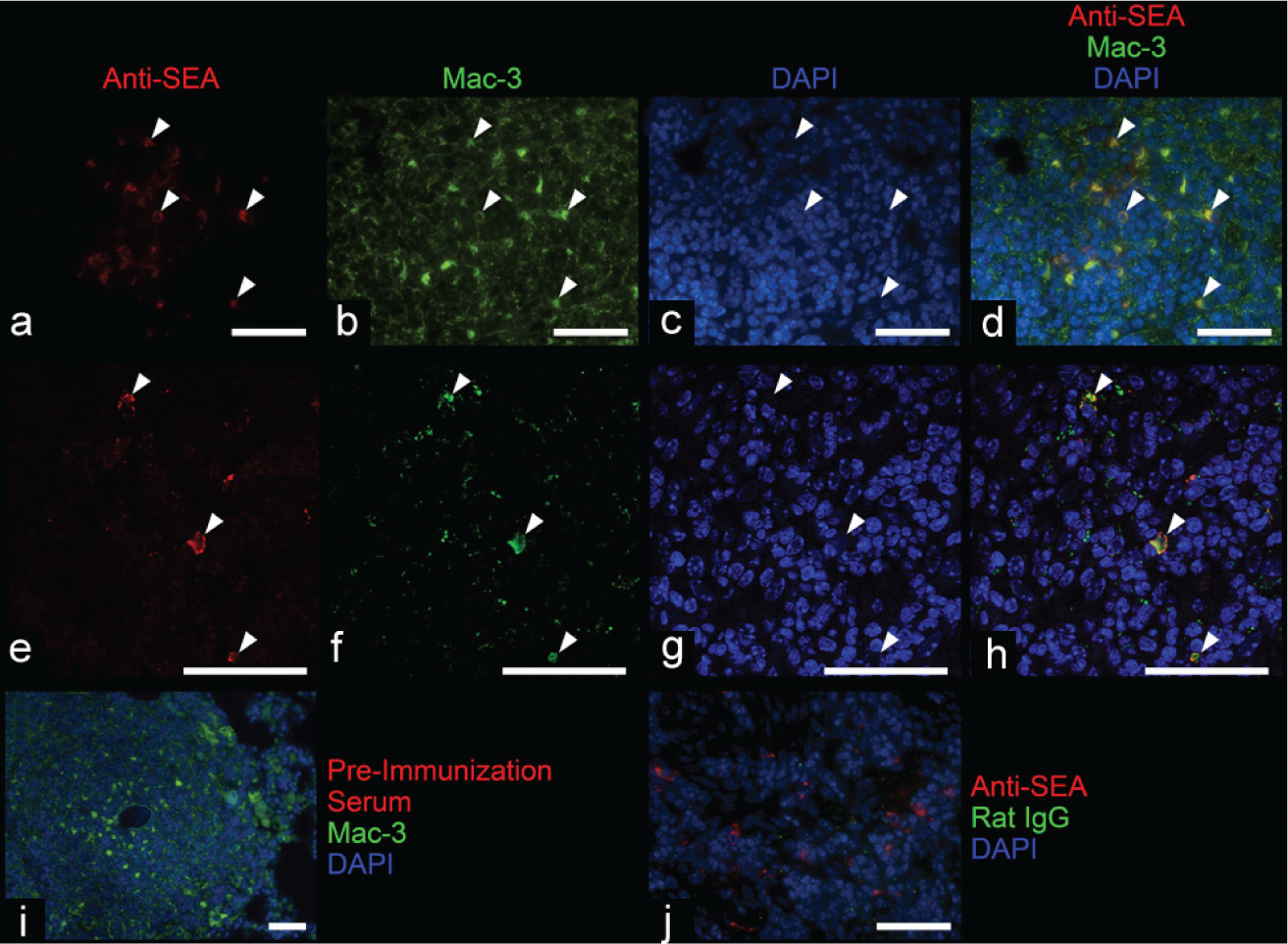
Immunofluorescence staining demonstrates colocalization of an anti-
DISCUSSION
We sought to identify parasite proteins within the lungs of individuals who had died of schistosomiasis-associated PAH using a polyclonal antibody generated to known soluble SEA. The antibody was validated by identifying egg antigens in vitro and in mouse lung tissue and human intestinal tissue. We did not find evidence of egg antigens in the human lung tissue except for a very small amount within visible granulomas, which are presumably eggs being degraded by the host immune system. In particular, the commonly seen black granular pigment did not contain apparent schistosomal antigen, and given its identical appearance to anthracotic pigment, may represent carbon fragments from environmental smoke inhalation.
Although unlikely, we cannot entirely exclude the possibility that there may be a more extensive distribution of parasite-derived antigens, such as entirely insoluble proteins (not included in the soluble egg antigen inoculation of the rabbits) or nonegg parasite proteins left behind during the initial passage of the schistosomula through the lungs in acute schistosomiasis infection (Katayama syndrome).[17] Similarly, the dark pigment frequently observed adjacent to pulmonary vascular lesions in schistosomiasis-associated PAH, and which has been speculated to be derived from the parasite,[6,8–10] is unlikely to be parasite derived.
Numerous investigators have postulated that an underlying cause of pulmonary vascular remodeling is inflammation.[18,19] Historically, Schistosoma ova were frequently seen in the lungs of individuals who died of schistosomiasis-associated PAH. For example, in 1954, de Faria reported histologically visible eggs present in 18 of 18 (100%) autopsy cases, with the number of eggs seen in a single 2.5 cm2 section ranging from four to approximately 250.[16] The report by Crosby et al. that mice chronically infected with schistosomiasis develop PH only when eggs are present in the lung, and that treatment with the anti-helmenthic praziquantel prevents or reverses this PH,[20] suggests that the localized parasite antigens trigger inflammation-mediated vascular remodeling, which stops once the antigenic material is eliminated.
In comparison with the historical reports, it is striking that we saw no intact intrapulmonary ova in similarly sized sections from any of our 18 samples. A potential explanation for this difference is the introduction of antihelmenthic therapies such as praziquantel, which was developed in the late 1970s.[21] However, although praziquantel is effective at killing the parasite (and thereby preventing further eggs from being laid), clinically, praziquantel is largely ineffective at treating the vascular remodeling in those who have developed PAH after chronic and repeated infection with
Although alternatively activated (M2) macrophages have been described to be present in the inflammatory granulomas surrounding Schistosoma eggs,[25] to our knowledge, it has not been previously directly demonstrated that some of the macrophages contain degraded
Overall, there is unlikely to be a significant amount of persistent parasite-derived antigens within the lungs of individuals who die of schistosomiasis-associated PAH. This suggests that retained and persistent parasite proteins are not driving a localized immune response contributory to the pathogenesis of pulmonary vascular remodeling.
