Abstract
Glomus tumors are rare neoplasms that typically occur within the dermis or subcutis of the subungual space. Primary glomus tumors of the thorax are exceedingly uncommon, thus standard-of-care management is lacking. In this report we describe the management of a patient presenting with a symptomatic glomus tumor of the posterior trachea, and provide a comprehensive review including all documented tracheal glomus tumor reports.
Keywords
Introduction
Glomus tumors, which account for less than 2% of all soft tissue tumors, are benign neoplasms originating from glomus cells surrounding an arteriovenous anastomosis. 1 These tumors typically occur in the skin corium, and are extremely uncommon in the thorax where glomus bodies are thought to be absent. To date, fewer than 40 tracheal glomus tumors have been reported. In this report we describe the staged endobronchoscopic and surgical management of 51-yearold female who presented with dysphagia, hemoptysis and dyspnea as a result of mass effect from a glomus tumor of the distal trachea.
Case Report
A 51-year-old female was admitted after several weeks of dysphagia, hemoptysis and increasing dyspnea. On admission, she was hemodynamically stable with normal oxygenation saturation on room air. Her laboratory values were unremarkable. A CT-angiogram of the chest revealed a nearly occlusive 2.6×2.3×1.7 cm intraluminal tracheal mass, just proximal to the carina (Figure 1a). 18FDG-PET visualized a hypermetabolic 2.6 cm retrotracheal lesion (Figure 1b). There was no evidence of FDG-avid distant metastasis of lymphadenopathy. Pharyngoesophagram revealed a smooth-surfaced extrinsic mass effect on the right lateral wall of the proximal thoracic esophagus with an associated esophageal diameter of 1.1 cm (Figure 1c).
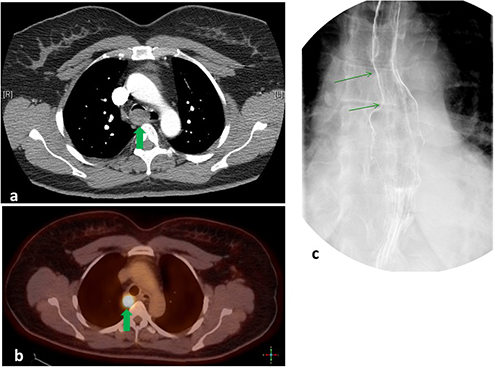
a) Computed tomography angiogram of the chest demonstrating a 2.6×2.3×1 cm intraluminal tracheal mass arising from the posterior wall of the trachea just above the carina. Significant narrowing of the tracheal lumen was noted. b) 18FDG-Positron Emission Tomography revealing a solitary hypermetabolic, 2.2×1.8 cm mass arising from the posterior wall of the trachea and just above the level of the azygous vein. No lesions suspicious metastases were noted. c) Pharyngoesophagram demonstrating smooth extrinsic compression on the right lateral aspect of the proximal thoracic esophagus suggestive of a mediastinal mass.
The patient underwent bronchoscopy with endoscopic ultrasound. The distal trachea was 80% obstructed by extrinsic compression from the posterior membrane (Figure 2a-c). The right and left bronchial tree appeared to be normal. Complete endobronchial ultrasound (EBUS) scanning of mediastinum localized the posterior tracheal mass (Figure 2d). Transbronchial needle aspiration (TBNA) of the mass, Level 7 and Level 11 lymph node stations were obtained. For symptomatic relief, a silastic Y-stent was placed (Figure 2e-g).

Bronchoscopy revealing clinically significant narrowing of the trachea with views from the (a) proximal trachea, (b) distal trachea and (c) carina. d) Endoscopic ultrasound revealing a 2.3 cm retrotracheal mass. Silastic Y-stent was placed covering the lesion with (e) overlap in the proximal trachea, (f) overlap in left mainstem bronchus and (g) fenestration to the right mainstem bronchus.
Cytologic evaluation of TBNA samples revealed polygonal and round cells closely packed, with round nuclei, dispersed chromatin, and moderate amount of clear and eosinophilic cytoplasm, supported by delicate stroma with dilated blood vessels. Immunohistochemical (IHC) analysis demonstrated neoplastic cells which were vimentin and SMA positive, suggestive of a glomus tumor. Level 7 and Level 11L lymph node aspirates were negative for neoplastic cells.
The patient was referred to Thoracic Surgery, and resection was recommended. At the time of right thoracotomy, the tumor was discovered to be located deep to the azygous vein, embedded between the esophagus and trachea (Figure 3). After further dissection it was noted that the tumor was adherent to the posterior membranous wall of the trachea. Therefore, the posterior tracheal wall was resected en bloc with the tumor. The posterior wall of the trachea was reconstructed with bovine pericardium (Figure 4) and reinforced with an intercostal muscle flap.

Via right thoracotomy, the pleura overlying the tumor was excised. The glomus (Gl) tumor was firmly adherent to the trachea (Tr) and vagus (V), and it was displacing the esophagus (Eso) by mass effect. The right main bronchus (RMB) was distal and uninvolved.
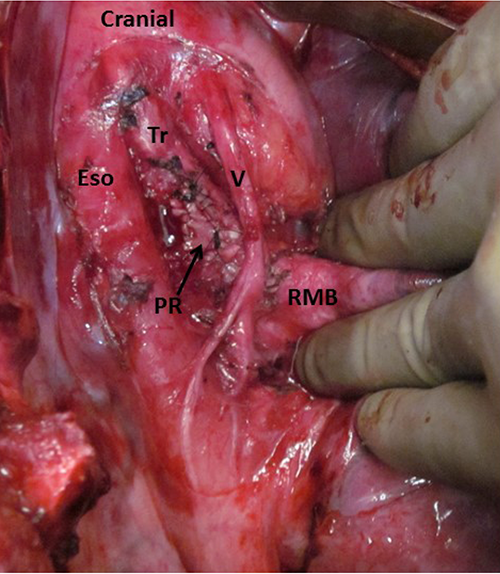
Following resection, the patch repair (PR) of the trachea (TR) could be visualized. The vagus (V) nerve was displaced anteriorly, and the esophagus (Eso) was located posteriorly. The right main bronchus (RMB) was located distally and could easily be visualized because the azygous vein was divided during the course of the operation.
Gross pathologic evaluation of the tumor revealed a 1.9×1.9×1.5 cm nodule. Histologic analysis revealed neoplastic cells with appreciable mitotic activity, but lacking nuclear pleomorphism (Figure 5a). The specimen was diffusely immunoreactive for smooth muscle actin, consistent with a glomus tumor (Figure 5b).
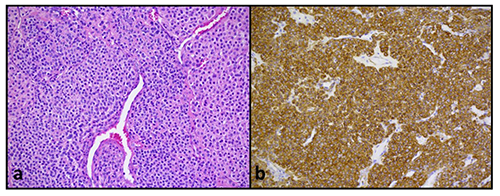
a) Hematoxylin and eosin staining of mediastinal mass revealing nests of clear epithelioid cells intimately associated with branching vascular channels. The neoplastic cells lack nuclear pleomorphism and appreciable mitotic activity. No necrosis, vascular, lymphatic or perineural invasion was identified. b) Immunohistochemical staining demonstrating diffuse immunoreactivity for smooth muscle actin.
Postoperatively the patient recovered uneventfully and was discharged on postoperative day 5. The patient is now 17 months out from her resection, and surveillance bronchoscopy at 6 months and 12 months have demonstrated an intact repair with no evidence of recurrent disease.
Discussion
Glomus tumors are exceedingly rare mesenchymal neoplasms which originate from perivascular smooth muscles cells responsible for thermoregulation. These tumors may arise in any part of the body, with the most common location being the subungueal area. In our report, we describe a patient presenting with a glomus tumor originating from the posterior tracheal wall. In addition to the trachea, extracutaneous glomus tumors have been identified within intrathoracic locations including the esophagus and lung.2–4 As seen in the presented patient, glomus tumors are histologically marked by a characteristic pattern of nests of clear epithelioid cells intimately associated with branching vascular channels, all within a connective tissue stroma with variable hyaline and myxoid changes. Each cell is surrounded by a sharply-defined basal lamina. Glomus tumors are positive for vimentin and smooth muscle actin, show variable expression of desmin, caldesmon, calponin and CD34 and are negative for keratins, neuroendocrine markers, CD31, S100 and CKIT.
Only 40 glomus tumors of the trachea have been documented since being first described by Hussarek and Reider in 1950. 5 In all reported cases, tumors arise from the membranous wall of the trachea where there is an increased density of mucous glands and vessels. 6 As seen in Table 1, the mean age at presentation is 51 (range 10-83), with males more likely to be affected in comparison to females (60% versus 40%). The most common location is the distal trachea (54%), followed by the proximal trachea (24%), then the middle trachea (22%). The mean tumor size was found to be 2.0 cm (range 1.2-3.0 cm).
Characteristics of all reported patients presenting with tracheal glomus tumor.

Tumor size describes lesion's greatest diameter
Resections described in Table 2.
Of the 40 reported tracheal glomus tumors, most are benign with only 2 (5.0%) displaying malignant histology.1,4 Given the potential for occult malignancy and the commonly symptomatic nature of the glomus tumor, resection is the standard of care. Most reports (67.5%) describe open resection; however, endoscopic resection has been reported (Tables 1 and 2).5,7–39 The utilization of endoscopic resection has become more common with endoscopic resections accounting for 12.5% of resections prior to 2005 versus 42.8% after 2005; P=0.04. In the proper patient in which malignancy has been ruled out, an endoscopic approach appears to be reasonable as survivals are similar between the two approaches (median survival of 18.0 months in open resection group vs. 18.5 months in the endoscopic groups; P=0.94). Despite similarities in survival, definitive conclusion based on published retrospective reports is impossible given the lack of long-term follow-up data reported. With regard to the patient we report, an open resection was ultimately recommended based on the location, which would have resulted in a challenging resection via endoscopic approaches.
Reported cases of tracheal glomus tumor.

F, Female; M, Male; N/D, not documented; mth, months; NR, no recurrence; Trach. Res., tracheal resection; End. Res., endoscopic resection; Nd-YAG, neodymium-yttrium-aluminum-garnet laser; Modified and updated from (17) and (6).
Conclusions
In summary, glomus tumors of the trachea are rare neoplasms, with 40 reported cases to date. In this report, we describe our management of a stereotypical patient presenting with a symptomatic glomus tumor of the distal trachea. We further summarize descriptive and survival data obtained from all documented reports. From these data we conclude that most patients present with symptomatic lesions, and thus require resection. There is an increasing trend to resect these lesions endoscopically with similar long-term results as compared to open resection. Despite this data, when considering endoscopic versus open resection, we urge careful consideration for tumor location and an estimated 5% risk of malignancy.
Footnotes
Acknowledgements
This work was support by an Association for Academic Surgery Foundation Research Grant to J.D.P.
