Abstract
Secretory breast carcinoma is an exceptionally rare mammary gland neoplasia described mainly in adult females and children of both sexes, and very rarely in the elderly. It has particular histopathological and immunohistochemical features and a favorable prognosis. We report the case of a 79-year-old Hispanic woman with a palpable breast mass. Currently, the patient is disease free after a followup period of 6 years without local recurrence or axillary lymph-nodes nor distant metastases.
Introduction
Secretory breast carcinoma (SBC), also known as juvenile carcinoma, was first described in 1966 by McDivitt and Stewart. It is an infrequently encountered malignant neoplasia that may present from infancy in both sexes, but is most common in women in their third decade. It is less commonly observed from the fourth to seventh decades and extremely rare in the eighth decade. It makes up less than 0.15% of all breast cancer and is an entity with low aggressiveness, indolent clinical course and little capacity to metastasize to regional lymph nodes.1–3 It is macroscopically well defined, has its own histological and immunohistochemical features.
Case Report
A 79-year-old hypertensive woman presented with a bloody discharge from the left nipple that she had had for three months. Physical examination showed large mammary glands with no skin changes. The nipple of the left breast was retracted and was secreting a bloody fluid. Palpation revealed a hard, mobile subareolar nodule 1.5 cm in diameter. There were no palpable axillary adenopathies.
Ultrasound examination showed an encapsulated, solid, 1.7 × 1.5 × 1.1 cm nodule, and mammography revealed a well-defined nodule 1.5 cm in greatest diameter causing nipple retraction (BIRADS IV, corresponding to a lesion with low to moderate probability of malignancy; biopsy is recommended. The patient underwent a retroareolar lumpectomy (left breast).
Macroscopic showed an irregular 7 × 4 × 1 cm piece of fatty tissue. Incision showed a firm, yellowish-brown, 1.5 × 1 × 0.6 cm lesion with spiculated margins; there was an adjacent cystic formation 1.5 cm in diameter with a smooth internal wall and with soft, granular, yellowishbrown tissue inside (Figure 1A).

Left breast tumor. A) The arrow points to dilated ducts with brownish tissue inside. B) Secretory carcinoma with a solid growth pattern and microcystic structures containing amorphous eosinophilic material. The neoplastic cells are polygonal, with an oval nucleus and ample, pale granular cytoplasm with small vacuoles containing secretion (inset 40 ×). C) Presence of extracellular secretory material positive for PAS (20 ×). D) Extracellular and intracellular secretory material positive for Alcian Blue, similar to that found in the contents of the lumens (20x).
Microscopic findings showed a malignant neoplastic proliferation consisting of tubular structures, and solid areas with microcystic structures with a dense eosinophilic secretion reminiscent of thyroid colloid. The epithelial tumor cells had round, uniform nuclei with mild pleomorphism, no mitotic index and ample eosinophilic and vacuolated cytoplasm (Figure 1B). The tumor was surrounded by abundant hyalinized fibrous tissue and invaded the adjacent fatty tissue. There was no evidence of necrosis, microcalcifications, and perineural or vascular invasion.
Extracellular and intracellular secretory material was positive for periodic acid-Schiff (PAS) (Figure 1C), diastase-PAS and alcian blue (Figure 1D), similar to that found in the contents of the lumens. Immunohistochemical staining showed tumor cell positivity for polyclonal CEA, estrogen (5%) and progesterone (5%) receptors (Figure 2). HER2, p53
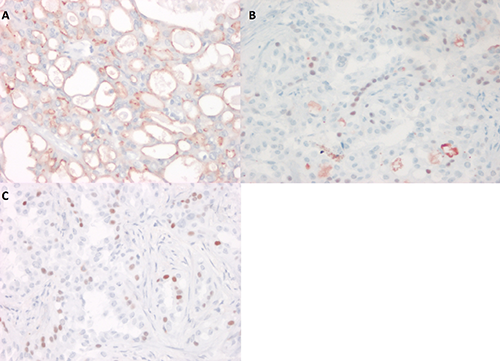
Immunohistochemical study of secretory carcinoma. A) Polyclonal CEA luminal staining in the microcystic structures (20 ×). B and C) Estrogen and progesterone receptors, nuclear staining in a few neoplastic cells (40 ×).
and GCDFP15 were negative, and the proliferation index (Ki-67) was low (results not shown).
Considering our patient's age and the fact that SBC is a low-grade tumor with very good prognosis, complementary axillary node dissection or total mastectomy were not done. There was no indication for adjuvant chemotherapy or radiation. Currently, the patient is disease free after a follow-up period of 6 years without local recurrence or axillary lymph-nodes nor distant metastases.
Discussion
Secretory carcinoma is a rare tumor first described in 1966 by McDivitt and Stewart that makes up less than 0.15% of all breast cancers. Around 100 cases have been published to date. This entity has been reported in both sexes and occurs preferentially during the third decade, with a median age at presentation of 25 years (range, 3-87 years).1–3 It is commonly seen in children and adolescents, but its peak incidence is in young adults, and less than 2.2% of cases appear in the eighth decade of life. In contrast to other breast carcinomas, SBC has not been proven to be associated with hormonal changes, family history, endocrinopathy or any underlying genetic abnormality; however, it has been described as related to gynecomastia or juvenile papillomatosis.3–5 It has a remarkably indolent clinical course.
Secretory carcinoma may appear in any part of the breast. Subareolar lesions have been associated with nipple secretion and are of variable size, from 3 to 5 cm in greatest diameter. There is a case reported located in the axillary skin appendage glands. These tumors are mobile, slow-growing, well-defined and odorless, and can macroscopically mimic a fibroadenoma.1–5
A study of sonographic characteristics concluded that SBC appears as a small nodule or several nodules with a benign appearance, sometimes as a low clinical stage intraductal lesion. 8 Mammography usually reveals a single discrete tumor with irregular borders.5–9 Tavasoli and Norris 8 reported that lesions over 2 cm in diameter are more likely to metastasize. Histopathologically, these tumors are circumscribed with areas of invasion into the fatty tissue and with different patterns: honeycombed, tubular, microcystic, solid and follicular, the latter mimicking thyroid gland morphology (the epithelial neoplasia of our patient showed a mixed pattern of tubular, microcystic and solid areas). Neoplastic cells are polygonal of two types (A and B), with ample, pale granular cytoplasm with small vacuoles containing secretion. The nucleus is ovoid and contains a small nucleolus; pleomorphism is mild and mitoses are infrequent.1–4,6–8 The luminal secretory material is positive for PAS and PASAlcian blue stains and is diastase-resistant, indicating that it does not contain glycogen and revealing its sulfated mucopolysaccharide content. The stroma surrounding the tumor is hyalinized, fibrous and may contain microcalcifications.10–12
Most SBC usually lack expression of estrogen (ER) and progesterone receptors (PR) and do not overexpress HER2; therefore they are triple-negative. In rare cases, low expression for ER. The tumor cells express the cytokeratins 8/18 and 19 at least focally, while E-cadherin, CD117, and alpha-smooth-muscle actin can be expressed. One study reported strong membrane E-cadherin reactivity in the tumor cells, supporting the hypothesis regarding the ductal origin of SBC, which may be considered a variant of ductal carcinoma.1,5,11–15 Other markers consistently expressed include epithelial membrane antigen (EMA), S-100 protein (strong and diffuse), lactalbumin and less frequently polyclonal CEA.15,16 GCDFP-15 an apocrine differentiation marker has also been reported in SBC. 1
The nests of tumor cells do not show myoepithelial cells; alpha smooth muscle actin and p63 are thus negative. SBC has been associated with p53 mutation in 10% of cases. Positivity for S-100 and CD10 and negativity for ER and PR suggests a possible origin in the basal cells of the mammary ducts.1,2,10,12,15
In our case, the tumor cells showed mild positivity for ER and PR. The Rosen and Cranor study of women from 5 to 87 years of age included five tumors that were negative for estrogens and three that were positive for progesterone. 16 Another study showed the expression of estrogens and progesterone to be extremely variable. SBC pathogenesis is probably not associated with these hormones. Thus, the clinical relevance of the immunohistochemical findings in the abovementioned studies is uncertain.10,15
Cytogenetically, SBC has been associated with t(12;15) and with ETV6-NTRK3 gene fusion as the dominant active oncogene and the most common alteration. It is important to mention that this translocation has also been reported in congenital fibrosarcoma, cellular mesoblastic nephroma and acute myeloid leukemia.1–3,9,12,17,18
The differential diagnosis of SBC includes acinar cell carcinoma; it is low-grade variant of breast cancer. However, they do differ in their clinical presentation (no cases of acinic cell carcinoma in prepubertal or male patients), citologic aspect and molecular features. In contrast with SBC, acinic cell carcinoma not to harbor ETV6-NTRK3 translocation. In addition to histological and immunohistochemistry criteria, genomic analysis to investigate the presence of an ETV6 gene rearrangement may be useful to rule out an apocrine carcinoma, lipidor glucogen-rich carcinoma, mucinous carcinoma, or lactating adenoma.1,2,15–19
SBC has a better prognosis than the usual ductal carcinoma. Moreover, children and young adults under 30 years of age have a better prognosis than older adults, and age may be an important factor in disease progression and recurrence.8,17,18
Treatment is excision of the carcinoma with wide surgical margins or lumpectomy with lymph node dissection.14,15 Local recurrence may occur many (20) years after treatment and is predominantly due to incomplete excision of the tumor rather than its inherent aggressiveness, which is why continued monitoring of patients is recommended. Sentinel node biopsy without radiotherapy is recommended in children.16–18
Conclusions
SBC is an exceptionally rare neoplasm and it is even rarer for it to occur in a 79-year-old woman. It has distinctive histological features, with mild expression of estrogen receptors and progesterone, absence of GCDFP-15 and low aggressiveness.
Footnotes
Acknowledgements
The authors wish to thank Cesar Vaca and Fernando Ortiz (technologists) for their valuable technical support.
