Abstract
Secondary, radiation-induced neoplasms represent a significant long-term risk after radiation treatment, and radiation-induced sarcomas (RAS) have an especially poor prognosis. These have rarely been reported after irradiation for pituitary adenomas.
Case Report
A 62 year old male presented with confusion, lethargy, diffuse weakness, worsening vision, and a 32 kg unintentional weight loss over a one-year period. Physical exam findings included a complete left ophthalmoplegia with proptosis, as well as a right sixth nerve palsy and left trigeminal sensory disturbance. He had previously undergone a left frontotemporal craniotomy for resection of pituitary adenoma 25 years ago, with adjuvant external beam radiation treatment estimated to be between 45–54 Gy. After surgery and radiation therapy, the patient experienced panhypopituitarism and was treated with stable doses of prednisone, levothyroxine, and topical testosterone until the current presentation. He was admitted to a general medical service for his complaints, and was found to be hypovolemic. With volume repletion, the patient was found to be hypernatremic, with 3–7 L of urine output per day. Although he had not previously required treatment with desmopressin, laboratory findings supported the diagnosis of central diabetes insipidus requiring ongoing desmopressin use.
Because of this new presentation and his previous history, cranial magnetic resonance imaging (MRI) was obtained. This demonstrated a destructive soft tissue mass extending from the sella into the suprasellar cistern (Figure 1A), extending medially from the sphenoid sinus through the cavernous sinus into the medial anterior cranial fossa with associated bony erosion (Figure 1F) and vasogenic edema in the underlying temporal lobe (Figure 1B). Initial MRI was done without contrast, due to patients inability to continue, with the imaging appearance concerning for a skull base neoplasm which was thought to possibly be secondarily infected due to the central region of restricted diffusion adjacent to the temporal lobe (Figure 1D,E). Follow up gadolinium-enhanced images (Figure 1C) showed the lesion to have solid heterogenous enhancement in the sphenoid sinus but thick peripheral enhancement in the region of restricted diffusion in the middle cranial fossa. The imaging was most consistent with a recurrent tumor, possibly a high-grade, radiation-induced tumor, with possible secondary abscess formation. Neurosurgical consultation was obtained, and a re-do left frontotemporal craniotomy was performed for partial resection and for biopsy to obtain definitive diagnosis. A transsphenoidal procedure was contemplated and would have allowed access to the tumor in the central skull base but would not have allowed access to the area in the temporal lobe concerning for infection. Upon extradural dissection, the anterior clinoid was found to be eroded by tumor, with multiple foci of extradural tumor extension obviously visible along the middle cranial fossa. Intradural dissection revealed complete encasement of the left optic nerve and cavernous carotid artery by tumor. Multiple biopsies were taken for pathology and culture analysis.
Radiographic images of a radiation-induced sarcoma. A) Sagittal T1 demonstrates a heterogeneous sella mass extending from the sphenoid sinus (white arrowhead) into the hypothalamus and suprasellar cistern (white arrow); B) axial FSE T2 demonstrates the sphenoid component to be heterogenously hypointense (white arrowhead) extending into the cavernous sinus encasing the carotid (white arrow). The intracranial component is extraaxial and hyperintense with a rim of hypointensity (black arrowhead). Associated vasogenic edema in the underlying temporal lobe (black arrow). C) Axial T1 with gadolinium demonstrates solid enhancement (white arrowhead) in the extracranial portion with thick rim enhancement intracranially (black arrow); D) axial DWI (B1500) and E) ADC map both demonstrate restricted diffusion within the peripherally enhancing component; F) axial computed tomography bone window demonstrates bony erosion through the sphenoid (white arrow)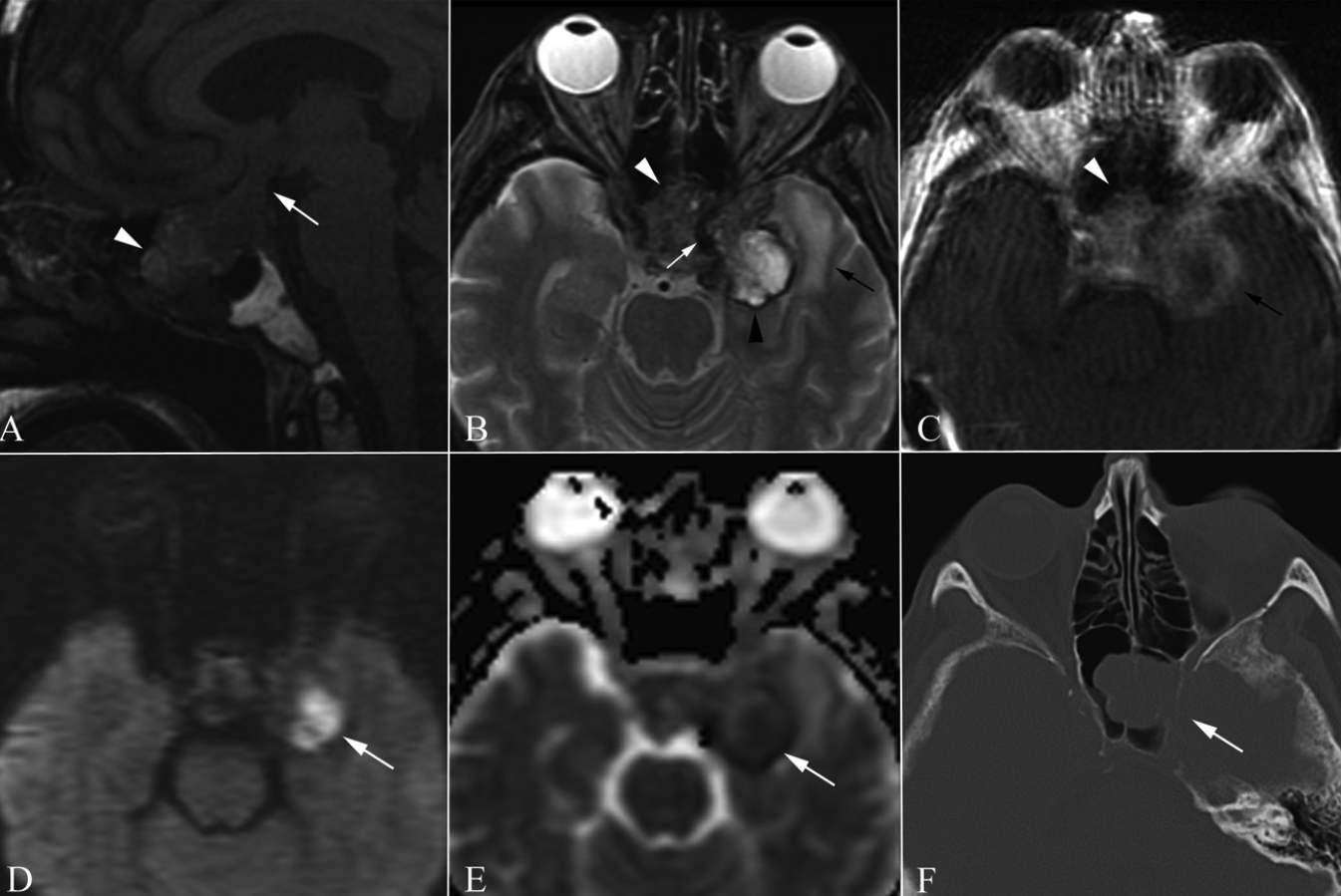
No evidence of abscess cavity was identified and all cultures obtained were negative for bacterial or fungal growth. Pathology confirmed grade 4 (of 4) spindle cell sarcoma from all surgical specimens. Immunohistochemical studies confirmed the diagnosis, demonstrating CD34 positive staining, while S-100, SMA and desmin were negative. MIB-1 demonstrated a moderately brisk proliferation rate. Reticulin stained positive (Figure 2A,D).
Histopathology of resected tumor demonstrates a high grade sarcoma. A) The tumor is a highly cellular, spindle cell sarcoma (hematoxylin and eosin magnification ×200) and is highly mitotically active (B, hematoxylin and eosin, magnification ×400). The tumor cells stain with antibodies to CD34 (C, magnification ×400), and show a peri-cellular reticulin staining pattern (D, magnification ×400). Immunostains for S100 protein and desmin were negative (not shown).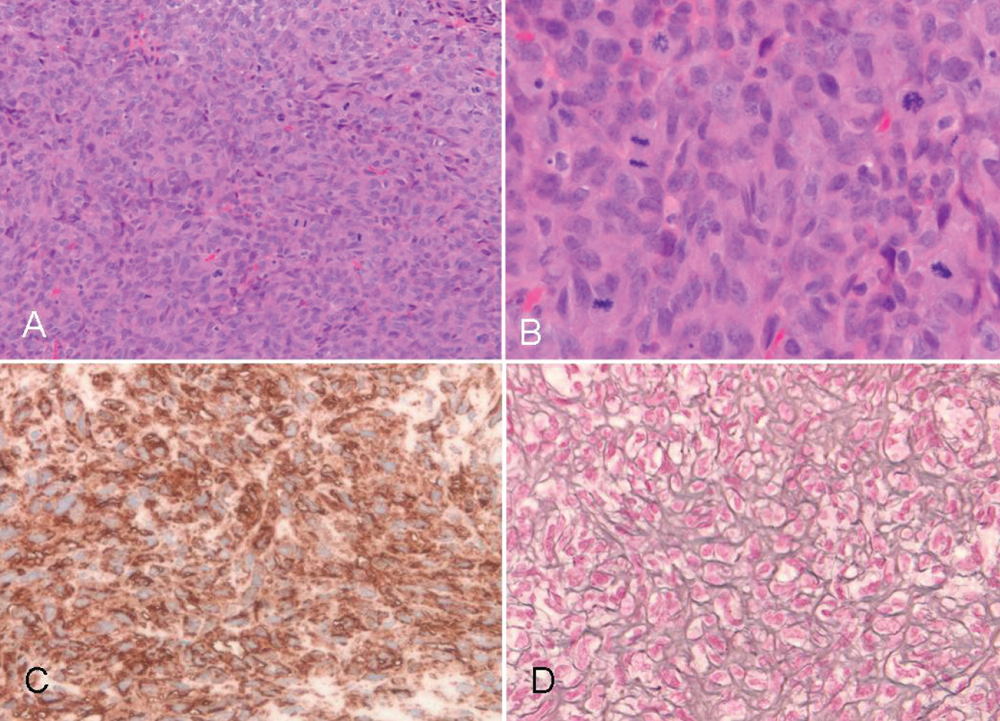
The patient awoke post-operatively to his preoperative neurologic baseline; moving all extremities and alert but confused. Nevertheless, his clinical course quickly deteriorated during the post-operative period. After compassionate discussion regarding the poor prognosis, the patient's family elected to withdraw aggressive care in favor of maximal comfort measures, and he expired on post-operative day 15.
Discussion
Radiation-induced tumors were originally described by Cahan in 1948, who also outlined four prerequisites for a tumor to be classified as such. 1 These include tumor location in a previously radiated area, pathologically-proven difference between the new tumor and the primary lesion, time lapse between radiation and tumor development, and lastly no other predisposing conditions to tumor development. 1
RAS are well-known to arise after ionizing radiation and have been characterized after radiation treatment for a number of primary neoplasms. In a retrospective review of 160 patients diagnosed with radiation-associated sarcomas, 26% were following treatment for breast cancer, 25% after lymphoma, and 14% after treatment for carcinoma of the cervix. 2 RAS can demonstrate a variety of histologic subtypes; in a previous series, 87% of tumors were high grade, and histologic variations included osteogenic, fibrous histiocytoma, and angiosarcoma. 2 Others have found that RAS frequently demonstrate rearrangements of chromosome 3p, 3 but regardless of histologic subtype, a key prognostic factor has been extent of surgical resection. 2
RAS in the sellar region after irradiation for pituitary adenoma is a known entity which often demonstrates rapid growth and an aggressive clinical course similar to RAS in other locations. A previously published, retrospective review of all reported cases only uncovered 52 such neoplasms reported in the literature, including those occurring after radiation for craniopharyngiomas in 2 patients. 4 The most common histologic types in the sellar region have been fibrosarcoma, spindle cell tumor, and high-grade or undifferentiated fibrous sarcoma. 4 Latency from radiation to time of diagnosis ranged from 5–20 years, 4 a somewhat shorter timeframe than that experienced by our patient. That previous review also noted a steep rise in incidence of these neoplasms occurred 5–13 years after radiation, and a mortality of approximately 60% at 6 months. 4 Symptom presentation was generally visual or oculomotor complaints, 4 which were noted in our patient; hormonal or electrolyte disturbances found in our patient are less commonly reported.
Because of the rarity of this lesion, its appearance on radiographic imaging has not been well-characterized and differentiating factors of its radiographic appearance have not been previously described. Of the 52 previous case reports present in the literature regarding RAS after treatment for pituitary adenomas, approximately twenty of these were reported prior to the computed tomography (CT) era. Those which did report CT findings, found positive contrast enhancement and bony erosion of the skull base.5–8 Of the more recent case reports during which MRI was available, only six report imaging characteristics in this modality, describing an appearance characteristic of other high-grade lesions such as contrast enhancement and invasion of surrounding structures.4,9–13 One series of two patients presents magnetic resonance spectroscopy findings of increased metabolic activity within the lesion, 9 which is also non-specific for high grade lesions. In the case of our patient, imaging included a diffusion weighted imaging sequence which was notable for showing restricted diffusion of the tumor, which led to initial concern about the possible presence of an abscess. Other features that were worrisome for high-grade tumor included: enhancement, marked increase in size over a short interval, and a new increased T1 signal, which could have been associated with subacute blood or proteinaceous fluid. These findings could have been seen in radiation necrosis, but as restriction of diffusion weighted images develops around the peripheral zone of enhancement, this is more suggestive of increase cellularity as the tumor is growing peripherally. With this initial uncertainty, this prompted more urgent surgical exploration, although no evidence of infection was found to be present.
RAS after sellar radiation for pituitary adenoma is a disease with a dismal prognosis due to its rapid growth and aggressive invasion of surrounding critical structures. No reported current treatment modalities have been effective. En bloc resection, a central tenet of sarcoma management, is generally impossible due to invasion of structures such as the optic nerve, cavernous sinus, and internal carotid artery. These lesions may be under-diagnosed because of their propensity to invade the structures of the skull base and cause sudden death, with several patients in the literature presenting with fatal hemispheric stroke or intratumoral hemorrhage. A diagnostic clue may be the late development of diabetes insipidus, which is rare with pituitary adenoma recurrence and may suggest a more aggressive lesion invading the posterior pituitary, prompting earlier investigation.
Although rare, the incidence of RAS is likely to increase with increasing use of radiation therapy for pituitary adenomas, as well as other histologically benign intracranial lesions such as schwannomas and skull base meningiomas. Physicians treating patients with a history of sellar radiation for pituitary adenomas or other sellar tumors must be cognizant of this late post-radiation complication.
