Abstract
The Rare Cancer Network (RCN), founded in 1993, performs research involving rare tumors that are not common enough to be the focus of prospective study. Over 55 studies have either been completed or are in progress.
The aim of the paper is to present an overview of the 30 studies done through the RCN to date, organized by disease site. Five studies focus on breast pathology, including sarcoma, lymphoma, phyllodes tumor, adenoid cystic carcinoma, and ductal carcinoma
Keywords
Introduction
The Rare Cancer Network (RCN), which was founded in 1993 by Professor René Mirimanoff in Lausanne, started as an informal, well-organized group of radiation oncologists, from all over the world. The RCN was made possible by the revolution in electronic communication that occurred in the 1990's. Rapid communication through e-mail and a dedicated web site (www.rarecancer.net) permitted researchers across the globe to come together in an informal network dedicated to the study of rare cancers.
The RCN has typically studied malignancies that are too rare to the be the focus of prospective clinical trials. Although the results generated by RCN's pooled studies reflect their retrospective and multi-institutional nature, with all the biases that these limitations imply, they may represent the highest level of evidence available in the hierarchy of the evidence base, nonetheless.
Herein, we report a summary of the past studies of the RCN that have undergone peer review and publication.
Materials and Methods
The Rare Cancer Network consists of 130 investigators in 24 countries (Supplemen-tary Table 1).
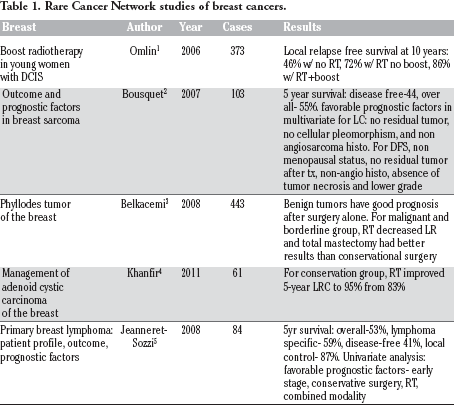
Rare Cancer Network studies of breast cancers.
Membership is available for any clinical investigator interesting in collaboration with the network. Members can propose research topics of rare cancers to the RCN coordinating office to survey the rest of the membership for level of interest.
All studies undergo required review by ethics or institutional review boards at each participating institution. Data is then extracted from existing cases locally at each department according to flow sheets designed by the primary investigator. This data is then pooled through electronic means for analysis. Authorship is a function of case contribution, with author rank determined by the relative number of patients contributed to each individual study.
The studies reported in this article were identified from the RCN web site (www.rarecancer.net) and through online searches of PubMed.
Results
Currently there are 55 studies either completed or in progress, including data from over 3500 patients, resulting in 28 peer reviewed publications. The median study size was 81 (ranging from 9 to 443). All studies were multi-center retrospective reviews. The studies focused on rare cancers or rare presentations of common cancers. The malignancies covered were of a wide variety with fair distribution among anatomic location and histology. Figure 1 illustrates the geographic location of study primary investigators.
Location of Rare Cancer Network Primary Investigators in Europe, Asia (A) and North America (B) for studies published 1993 to 2011.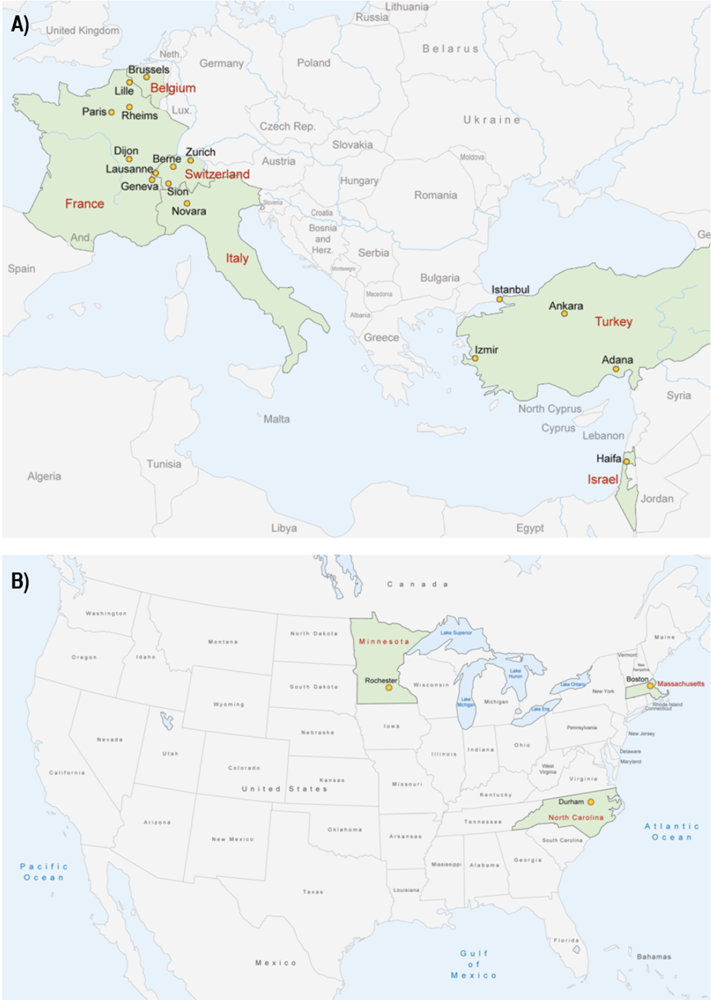
Studies
Breast
Five studies have focused on rare breast pathologies as are outlined in Table 1.1,5
The cancer studies include breast sarcomas, lymphoma, phyllodes tumor, ductal carcinoma
Prostate
There have been three multi-center retrospective studies that have focused on prostate cancers as outlined in Table 2.6–8
Rare Cancer Network studies of prostate cancer.

Two studies done by Nguyen
A third article by Stein
Head and Neck
Six studies reviewed multi-center data for several different head and neck cancers as detailed in Table 3.9–14
Rare Cancer Network studies of head and neck cancers.

This diverse group includes a pediatric study analyzing nasopharyngeal carcinoma, a rare disease for which an optimal treatment has not been found. 12 The study, the largest of its kind, finds that patients had received combined therapy with chemotherapy and radiation had the longest survival.
The study also found evidence suggesting that a lower radiotherapy dose could be considered in patients with a good response to chemotherapy, although no definitive conclusions could be made given the retrospective nature of the study. Anacak
Treatment with both radiotherapy and surgery was associated with an improved outcome, both in a review of patients with olfactory neuroblastoma by Ozsahin. 13 A similar relationship was found for mucosal melanoma patients by Krengli and Mirimanoff. 11
Central Nervous System
The RCN has completed four series on rare cancers of the central nervous system (Supplementary Table 2).
A study by Weber on cerebellar glioblastoma multiforme included 45 patients and found that all patients had local progression.
15
Pasquier
On univariate analysis, the authors found survival was negatively affected by age >60, poor performance status, and a high mitotic rate. Fifty-two patients with spinal epidural lymphomas were evaluated in a study by Mirimanoff and were found to have good response to chemoradiation
In patients with spinal myxopapillary ependymoma, post-operative radiotherapy had a significant increase in 5-year progression free survival (82%) when compared to surgery alone (50%). 18
Other
There are various other studies done by the RCN as listed in Table 4.19–30
Rare Cancer Network studies of other tumors.

These studies vary from thyroid lymphomas to uterine papillary sarcomas (Supplementary Table 3).
Discussion
Rare cancers present a challenge to the scientific investigator. 31 Their rarity typically prevents the conduct of adequately powered clinical trials or definitive study by any single individual or institution. Multi-institutional efforts can increase the volume of clinical data available for rare cancer studies, but such efforts must compete with more common malignant entities for funding.
Although studying rare cancers through prospective clinical trials is possible, as evidenced by the work of the Children's Oncology Group, most adult rare cancers have not been the focus of a therapeutic clinical trial.
Population based registries such as the Surveillance Epidemiology and End Results database (www.seer.cancer.gov) can provide descriptive data on prevalence and outcomes, but they are not designed to address specific treatment related hypotheses in the manner of clinical trials. Continued evolution of health care informatics and population based care delivery may provide increasing levels of outcomes data on rare malignancies.
Recently, the International Rare Cancer Initiative was formed through a collaboration of EORTC, Cancer Research UK, the National Institute for Health Research Cancer Research Network (NCRN), and the United States National Cancer Institute (NCI). This organization will focus on designing clinical trials of treatments for rare cancers and should improve upon the quality of evidence in the future. By pooling resources and patient cohorts internationally through the infrastructure of existing national cancer research programs, it will provide valuable prospective data on a limited number of rare tumor entities such as small bowel cancer, rare head and neck cancers, gynecological sarcoma, fibromellar hepatocellular carcinomas, and penile carcinoma. (https://wiki.nci.nih.gov/display/ICWG/US-UK-EORTC+Rare+Cancer+Initiative).
Additionally, patient advocacy organization such as the National Organization for Rare Disorders (www.rarediseases.org/) and Rare Disease Europe (www.eurordis.org/) aid in the fight against rare malignancies.
The Rare Cancer Network was founded in 1993 to provide a multi-institutional framework to facilitate rare cancer research, with a focus on the specific role of radiation therapy, through pooled data analysis by participating academic medical centers across the world.
In the last 19 years, the RCN has studied many different rare cancers that previously only had small series or case reports. With pooling of data from around the world, the network was able to produce a number of studies for which meaningful clinical data is lacking on treatment outcomes.
Although such studies cannot rank with randomized controlled trials in terms of impact, they do fill an important niche in between anecdotal evidence and clinical trials
The multicenter and retrospective nature of the studies can be seen as a significant limitation on the conclusions of the studies due to the variability of treatment, technique, and population in different departments around the world. However, as noted previously, for many rare cancers, it would be difficult if not impossible to study such tumors in a cost-efficient fashion prospectively. The data and results from the RCN studies serve to shed sufficient light to advance the care for the unfortunate patients that are diagnosed with these rare diseases.
With further collaboration and growing members, the Rare Cancer Network can improve upon not only the variety of data available to physicians on rare malignancies, but also potentially outcomes for patients around the world.
