Abstract
Background
Even though many types of stents have been tested in superficial femoral artery (SFA) and popliteal artery (PA), most of these devices have provided an unsatisfactory outcome, probably due their unsuitable anatomical and physiological characteristics. The Supera peripheral stent (Abbott Vascular, Santa Rosa, CA, USA) is a braided interwoven nitinol device specifically designed for treating atherosclerotic lesions of the femoro-popliteal segment. The aim of this multicenter retrospective study was to describe the effectiveness of Supera stents in the management of femoral-popliteal atherosclerotic lesions and to critically analyze our findings in the context of current and past literature.
Design and methods
In this study we enrolled only patients who satisfied the inclusion criteria: i) patients affected by chronic obstructive arterial disease (COAD) grade II, as per Rutherford classification; ii) patients treated with endovascular revascularization and Supera stent implantation in the femoro-popliteal axis. We retrospectively analyzed the Doppler Ultra-sound (US) follow- up at 12-24 and 36 months to detect the vascular occlusions. The primary patency, primary patency assisted and TLR were described statistically analyzed by survival analysis and the demographic data, clinical data, device safety following stenting were described as frequency and mean value.
Results
105 endovascular procedures on 99 patients for femoro-popliteal stenting with Supera were performed in four Italian hospitals. The median follow-up was 39 months (range 6-72), with primary patency rate of 83.1%, 74.3% and 69.5% at 12, 24 and 36 months after the procedure. The primary patency assisted was 89.9%, 76.8% and 73.4% in the same period, while the freedom from TLR values were 92.7%, 91.5% and 89.5% at 12, 24 and 36 months after the procedure, respectively. The mortality rate recorded at 12 months from the Supera implantation was 2.8% (3 out of 99 patients enrolled).
Conclusions
Our data were in agreement with the current literature, showing the non-inferiority Supera stent in relation to the other stent available. Supera stent showed an excellent safety, effectiveness profile and high durability for the treatment of PAD patients with femoro-popliteal artery disease.
Introduction
Peripheral artery disease (PAD) is a multifactorial condition associated with high morbidity and mortality, with an increasing incidence in Western countries 1 due to the widespread diffusion of chronic pathologies, such as type II diabetes. Among the various vascular districts affected by PAD, the femoro-popliteal vascular axis is one of the most challenging regions where to perform surgical or interventional radiology (IR) procedures due to the constant presence of strong biodynamic forces, i.e., longitudinal and radial compression, flexion, extension and torsion, causing stress to the vessel walls.1,2 From an interventional radiology perspective, the short-term durability of balloon angioplasty - the recommended first-line treatment for PAD - has tipped the balance in favor of femoro-popliteal artery stenting, which better preserves vessel patency. 3 The sustained benefit of stenting vs angioplasty in treating femoro-popliteal obstructions has indeed been reported in multiple studies.1,4-9 Even though many types of stents have been tested in superficial femoral artery (SFA) and popliteal artery (PA), the vast majority of these devices have provided an unsatisfactory outcome, probably due their unsuitable anatomical and physiological characteristics. For instance, laser cut stents have shown several limitations caused by possible rupture, kinking and/or induction of a significant inflammatory response on the vessel wall. Nonetheless, the continuous evolution of materials has led to the development of biomimetic stents able to reduce the amount of stress on the vessel walls and, at the same time, provide greater radial strength and fracture resistance.6,10-12 The Supera peripheral stent (Abbott Vascular, Santa Rosa, CA, USA) is a braided interwoven nitinol device specifically designed for treating atherosclerotic lesions of the femoro-popliteal segment.
In the present study, we report the results of a multicenter retrospective study on the effectiveness of Supera stents in the management of femoral-popliteal atherosclerotic lesions and critically analyze our findings in the context of current and past literature.
Significance for public health
Peripheral artery disease (PAD) is a benign multifactorial condition associated with high morbidity and mortality; the femoral-popliteal axis is one of the most critical vascular districts due to its complex biodynamic. Although balloon angioplasty represents the first line treatment, it is not uncommon the use of stents in order to preserve patency vessel. Among different stents available, however, Supera peripheral stent (Abbott Vascular, Santa Rosa, CA, USA) has the ability to fit better to the femoro-popliteal compartment which is subject to biomechanical stress. The aim of this paper is to retrospectively analyze a multicentric experience with the use of a specific device (Supera peripheral stent) for treatment of PAD in femoral-popliteal disease, comparing results with Literature. Through this paper, every interventional radiologist interested in PAD endovascular treatment could have an important reference in order to perform the best management in this chronic and complex benign pathology.
Design and methods
Study design
This study was designed as a multicenter retrospective study aimed to determine the effectiveness of Supera stents in the treatment of complex femoro-popliteal lesions. Primary outcomes included patency (i.e., primary, primary assisted and secondary patency), target lesion revascularization (TLR) and safety of the device. Moreover, we performed a review of the literature, comparing the results obtained in our retrospective study with previously published data. Ethics committee approval was not necessary due to the retrospective nature of this study and of the data collected. Data collection does not violate patient privacy. The study has been conducted in accordance with the Helsinki Declaration. All study participants signed a written informed consent before receiving endovascular treatment.
Inclusion criteria were patients affected by chronic obstructive arterial disease (COAD) belonging category 3, 4 and 5, as per Rutherford classification, treated with endovascular revascularization and Supera stent implantation in the femoro-popliteal axis.
Study population
Between April 2015 and December 2019, 105 endovascular procedures for femoro-popliteal stenting with Supera were performed in four Italian hospitals.
Demographics, risk factors, ongoing therapies, comorbidities (i.e., chronic heart failure, coronary disease, stroke history and diabetes), postoperative outcomes and follow-up data were recorded. Clinical symptoms such as intermittent claudication, critical ischemia with rest pain and tissue loss were ranked according to Rutherford classification prior to endovascular treatment.
In order to analyze and classify the cohort of patients subjected to implantation, the authors segmented SFA in three regions: F1 (proximal third), F2 (middle third) and F3 (distal third). Similarly, PA was divided in three areas: P1 (from the intercondylar fossa to the proximal edge of patella), P2 (from the proximal part of the patella to the center of the knee joint space) and P3 (from the center of the knee joint space to the origin of the anterior tibial artery).
Supera stent implantation
All patients underwent initial diagnostic angiography of their lower limbs with standard contrast intra-arterial digital subtraction angiogram to outline the vascular anatomy and define the lesion characteristics. The endovascular stenting was performed by experienced interventional radiologists, with more than 10 years of interventional radiology practice, in a dedicated endovascular suite alongside a vascular surgeon. Before the beginning of the study, each participating center had to implant at least 10 Supera stents. These previous implants were not included in our analysis in order to reduce the operator's learning curve bias. The interwoven nitinol Supera stent, with his particular chronic outward force and conformability (i.e., high resistance to torsion and high radial stiffness), is a unique endovascular device specifically designed for districts subject to flexion and torsion, such as the femoralpopliteal district. 13 The Supera peripheral stent is a woven selfexpanding stent constructed from nitinol. Six pairs of closed-ended interwoven nitinol wires are arranged in a helical pattern designed to be both flexible and resistant to fracture and to reduce the risk of re-coiling, especially in vessels affected by atheroma and calcification. 14
The stent comes pre-mounted on an over-the-wire delivery system (6F or 7F) compatible with 0.018 stent is a woven selfexpanding stent constructed from nitinol. The vessel must be predilated before the stenting of 1mm more that the stent size. Only standard balloon angioplasty was performed to obtain this. When more than one stent was needed, stents were overlapped for at least 10 mm. Digital subtraction angiography (DSA) after each procedure recorded the balloon dilatation site and runoff vessels to assess diameter improvement and potential complications such as vasospasm, dissection, thrombosis or distal embolism (Figure 1). Before implantation, patients underwent 1-month dual antiplatelet therapy consisting of acetylsalicylic acid 100 mg/die and clopidogrel 75 mg/die. After the procedure, acetylsalicylic acid 100 mg/die was prescribed indefinitely.

DSA before- and post- Supera stent placement. The stent allowed vessel caliber restoration.
In all case of occlusion, the vessel revascularization of the arterial axis and stent was obtained using a intraluminal technique (never subintimal).
Follow-up
Any peri-procedural complication (e.g., hematoma, bleeding, pseudoaneurysm and early stent occlusion) arising during the 24-h post-implantation period was recorded. Clinical and ultrasound follow-up of the treated limb was performed in each patient at 1, 6, 12, and 24 months after the procedure.
Results
The aim of this retrospective study was to conduct a clinical and radiological follow-up to evaluate stent patency (i.e., primary, primary assisted and secondary patency), TLR and device safety following stenting. Between April 2015 and January 2020, we analyzed 112 patients, of whom 7 (6.3%) were excluded due to incomplete data. The 99 study participants (69 men and 30 women) had a mean age of 74.5 ± 9.4 years. A total of 105 lower limbs were implanted with a Supera stent.
Stratification of the patients according to Rutherford classification indicated that 35 patients (35.4%) suffered from severe claudication (walking < 100 m) on admission, 17 patients (17.2%) had pain at rest and 47 patients (47.4%) presented with tissue loss. Further details are shown in Table 1.
Study population.

FA, superficial femoral artery; PA, popliteal artery; ATK, above the knee; BTK, below the knee; TASC, Trans-Atlantic Inter-Society Consensus.
The median follow-up was 39 months (range 6-72). Seventynine patients (79.8%) were followed up for two years. In the first year post-implantation, 3 patients died (3.0%) due to causes unrelated to the endovascular procedure - cardiac failure (2/3) and pneumonia (1/3). Of note, patients with less than 6 months of follow- up were excluded from the study. Any case of major bleeding due to ongoing antiplatelet therapy was recorded at follow-up as well. Kaplan-Meier curves were calculated using statistical analysis. Primary patency (PP) rates at 12, 24 and 36 months after the procedure were 83.1%, 74.3% and 69.5% respectively (Figure 2). Mean PP rate was 75.6%. Similar analyses were performed by setting as independent variables the total length of the implanted stent (Figure 3), the stent position in SFA only vs treatment of the PA (Figure 4) or the size of the stent (Figure 5). We also assessed the relationship between PP rate and total length of the Supera stent placed in each treated leg (Figure 2). To this end, we stratified the patients in three subgroups: <10 cm (L1), between 10 and 18 cm (L2), >18 cm (L3). In L1 patients, PP rates were 85.7%, 78.5 % and 57.1% at12, 24 and 36 months post-implantation, respectively, whereas those in L2 and L3 were 71.4%, 65.7 %, 51.4% and 87.5%, 72.4%, 68.9%, respectively.
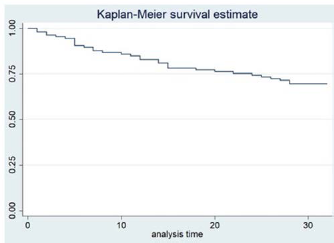
Primary patency rate of Supera stent during follow-up; 36 months after procedure, the primary patency rate was 69.5% of all cases.

Relationship between primary patency rate and total length of the Supera stent implanted. Kaplan-Meyer analysis did not reveal any statistical difference between the three groups (L1, <10 cm; L2:, between 10 and 18 cm; L3, >18 cm).

Position of Supera stent (superficial femoral artery only or combination with popliteal artery) as a function of its primary patency. Treatments of the popliteal alone were not stratified due to their small number (4/105).

Relationship between stent patency and its size. Small stents reported a lower patency value at 36 months.
Next, we analyzed the relationship between PP rates and treated vessels (SFA alone or together with PA) (Figure 3). On the one hand, patients implanted with Supera stent in SFA alone reported PP rates of 85.5%, 71.1 % and 69.5% at 12, 24 and 36 months postimplantation. On the other hand, the patients implanted with a Supera stent in both SFA and PA displayed PP rates of 86.4%, 75.2 % and 69.5% at 12, 24 and 36 months after the procedure, values not significantly different from those observed in the SFA alone group. Finally, we analyzed the relationship between PP rate and Supera stent size. For this purpose, three cohorts of patients were analyzed, each implanted with Supera stents with different diameters (4, 5 or 6 mm). The PP rates in patients treated with 4-mm diameter Supera stents were 86.2%, 75.5 % and 64.5% at 12, 24 and 36 months post-implantation, respectively. In contrast, patients implanted with larger diameter Supera stents showed an overall improved PP, with rates of 86.2%, 83.8 % and 72.3% (5 mm) and 100%, 83.1 % and 72.3% (6 mm) at the 12, 24 and 36 month time points. Table 2 summarizes the follow-up results according to the independent variables mentioned above.
Results and follow up.

TLR, target lesion revascularization; SFA, superficial femoral artery; PA, popliteal artery.
Freedom from TLR values were 92.7%, 91.5% and 89.5% at 12, 24 and 36 months after the procedure, respectively.
Discussion
PAD is a growing global problem 5 due to the worldwide increased incidence of type II diabetes and other factors, such as smoking, which augment the risk of atheroma development, mostly affecting the lower limbs and the femoral-popliteal district, with a worldwide prevalence between 3% to 12%. 15 Severe ischemia of the lower limbs tends to affect more women than men (about 3:1), with an increasing incidence among individuals older than 40 years of age.16,17
The treatment of this pathology involves numerous surgical and/or radiological interventional options that are even more challenging when carried out in the femoro-popliteal district due to its peculiar characteristics. Indeed, the vessels in this anatomically complex district are constantly exposed to strong biodynamic forces during daily activities, which makes the vessel wall more prone to elongation, kinking and rotation. 18 Even though percutaneous angioplasty is the therapeutic gold standard for the treatment of patients with moderate stenosis in the femoro-popliteal district, stenting is only recommended in cases of severe stenosis and/or dissection (iatrogenic or not). 19 However, this approach does not always have a positive outcome due to the massive presence of the aforementioned mechanical forces. For example, during knee flexion, stented vessels tend to get stiff and kinked, which may lead to stent thrombosis and restenosis, 20 especially when the stent is positioned in the intra-articular PA. Indeed, calcific lesions very frequently result in restenosis, patency loss or amputation. 21 In cadaver models, the Supera stent was shown to be the only type of stent that would not be crushed or pinched during hip/knee flexion deformation, 22 ensuring optimal luminal flow. This aspect is particularly important given that reduced stent kinking may increase stent durability over a 3-year period. 23
Here, we retrospectively report the experience of four Italian centers with Supera stenting of the femoro-popliteal district. In particular, we enrolled primary stenting-treated patients with symptomatic atheromasclerotic lesions in SFA and PA and evaluated the outcomes at 3-year follow-up. In terms of population characteristics, our results are in good agreement with those reported by San Norberto et al. 12 Specifically, the mean age and the prevalence of TASC-II D and critical limb ischemia recorded in our study are quite similar to those reported by San Norberto et al. (74.5 vs 74.4 years; 41.9% vs 44%; and 64.4 vs 64%, respectively). In contrast, the lesions treated in this study were on average much longer (150.4 vs 114.3 cm) and more often localized in PA (46.5% vs 28%) compared to those described by San Norberto et al. We evaluated the follow-up data performed in the centers enrolled three years after the procedure. Although multiple stents were positioned with their extremities at the P2 portion of PA, the PP rates observed in our study were 83.1%, 74.3% and 69.5% at 12, 24 and 36 months, respectively, compared to 89.6%, 72.3% and 70.2% reported by San Norberto et al. at the same time points. 12 The mean PP rate was 75.6%, in good agreement with previous 3-year follow- up studies, reporting values ranging from 74.9 to 77.4%.3,12
Importantly, while the PP rates of Supera stents that we observed at 1 year are very similar to those reported by Sibé et al. 24 using Bail-out nitinol stents (83.1% vs 81.5%, respectively), the 2- year Supera stent PP rates are substantially higher than those of Bail-out nitinol stents (74.3 vs 67.2%, respectively). The assisted PP rate at 1 year observed in our study (89.4%) is in line with those reported by other studies, with values ranging from 80.5% to 94%.3,9, 10,12,23,25-31 Considering that the use of the Supera stent requires a greater learning curves then traditional bare stents, the inter-operator variability could represent a study bias. 22
Chan et al. have recently published one of the longest followup studies in patients treated with Supera stents. From their results, it emerges that 4-mm stents tend to have a PP < 5 mm for the first 46 months (p=0.131). Our results at 32-month follow-up up indicate a lower PP in 4-mm stents compared to that observed in 5- or 6-mm stents (p=0.028). This result may be affected by a bias due to the positioning of the stents. In fact, the 4-mm Supera were more frequently placed in PA, where the PP rate is generally lower than that of SFA-positioned stents. Further studies stratifying the results according to the position of the stent along the arterial axis may resolve this discrepancy. The TLR of Supera stent has been evaluated in numerous studies. Currently, the value of freedom from TLR at 3 years is between 70% and 94% (cited articles in the bulleted list). Our multicenter study reported a freedom from TLR at 12, 24 and 36 months of 92.7%, 91.5% and 89.5%, respectively, which can be superimposed on the current scientific literature. 9,28,29,32
Recently, Gouëffic et al. have compared polymer-free selfexpanding paclitaxel-eluting stents with bare-metal stents in femoropopliteal lesion treatment. The authors reported a PP for the polymer-free stent of 85.1% at 1-year follow-up and 78.8% at 2- year follow-up, while the freedom from TLR was 93.6% and 87.2% at the same follow-ups. Bare-metal stents were found not to be superior to paclitaxel-eluting stents at both 1- and 2-year follow- ups (PP: 84.8% and 74.6%; freedom from TLR: 93.3% and 87.6%, respectively). Similarly, our results seem to indicate that Supera stents are not superior to polymer-free self-expanding paclitaxel-eluting stents with bare-metal stents in femoropopliteal lesion treatment. Future studies on more homogeneous patient cohorts are clearly needed to further test this hypothesis.
Lastly, we report a mortality rate at 12 months of 3% (3 patients out of 99 enrolled). This is similar to what reported by Armstrong et al. in a comparative study of Supera stents vs bare nitinol stents (BNS), where the 1-year mortality rate of Supera vs BNS in the matched groups was 2.5% vs 5.1%. 33
Conclusions
This study highlights the excellent safety and effectiveness profile and high durability of Supera stents for the treatment of PAD patients with femoro-popliteal artery disease. Despite treating long and severe stenosis, we managed to obtained good values of freedom from TLR at 3 years post-implantation. Thus, the Supera stent should be considered a valid option for primary stenting in this anatomically complex vascular district.
Footnotes
The authors have no conflicts of interest to declare that are relevant to the content of this article.
